Abstract
Whereas the protein composition and overall shape of several giant virus capsids have been described, the mechanism by which these large capsids assemble remains enigmatic. Here, we present a reconstruction of the capsid of Cafeteria roenbergensis virus (CroV), one of the largest viruses analyzed by cryo-electron microscopy (cryo-EM) to date. The CroV capsid has a diameter of 3,000 Å and a Triangulation number of 499. Unlike related mimiviruses, the CroV capsid is not decorated with glycosylated surface fibers, but features 30 Å-long surface protrusions that are formed by loops of the major capsid protein. Based on the orientation of capsomers in the cryo-EM reconstruction, we propose that the capsids of CroV and related giant viruses are assembled by a newly conceived assembly pathway that initiates at a five-fold vertex and continuously proceeds outwards in a spiraling fashion.
Similar content being viewed by others
Introduction
Viruses with long dsDNA genomes (>200 kilobases) and large particles (>0.2 µm) are a common occurrence in nature and several new families of giant viruses have been reported in recent years1,2,3,4,5,6, which inspired various discussions regarding their evolutionary origin7,8,9,10,11. However, whereas rapidly advancing DNA sequencing methods facilitate genomic analysis of giant viruses, structural studies of large viral capsids are lagging behind. In contrast to small and medium-sized capsids, the >200 nm isometric capsids of giant DNA viruses still pose a significant technical challenge for high-resolution methods such as X-ray crystallography and cryo-EM12. Other techniques, including atomic force microscopy, scanning electron microscopy, and X-ray free electron lasers, have been used to study giant virus structures, but are unable to provide near-atomic resolution13,14,15,16. During the last decade, cryo-EM has become an increasingly powerful tool to determine the structure of virus particles, circumventing the need for crystallization17,18,19,20. The resolution of cryo-EM reconstruction of viruses has gradually improved from sub-nanometer to near atomic levels21,22,23,24,25,26,27,28,29,30. In addition, combined with X-ray crystallography of purified capsid proteins, it is possible to fit the atomic structures of individual components into the cryo-EM reconstruction map and determine a pseudo-atomic structure31,32,33,34. Examples for the successful combination of X-ray crystallography and cryo-EM to determine large DNA virus structures are Paramecium bursaria Chlorella virus 1 (PBCV-1)35, 36 and Chilo iridescent virus (CIV)37.
Here, we push the limits of cryo-EM application to large virus particles by reconstructing the capsid of Cafeteria roenbergensis virus (CroV)38. The giant virus CroV infects the widespread marine zooplankter Cafeteria roenbergensis, a single-celled eukaryote and ecologically important bacterivore39. Phylogenetically, CroV is a distant relative of the giant Acanthamoeba polyphaga mimivirus (APMV)1 and the sole member of the genus Cafeteriavirus in the family Mimiviridae. With a diameter of 3000 Å, the CroV particle is smaller than the 5000 Å wide capsid of APMV40 or the 1.5 × 0.5 µm ovoid particles of Pithovirus sibericum 4, which renders it more easily accessible to cryo-EM. Nevertheless, obtaining a close-to-nanometer cryo-EM resolution for an intact virus capsid of these dimensions is still problematic. The thickness of the vitreous ice that embeds the viral capsid in cryo-EM is one of the most important factors limiting the resolution. Multiple and inelastic scattering of electrons increases as the ice becomes thicker, which reduces the signal-to-noise ratio. Unlike APMV, the CroV capsid is not covered by a dense layer of 125 nm-long surface fibers that would complicate cryo-EM analysis41. Thus, CroV is ideally suited to advance the limit of structural studies of giant viruses. Although crystallization is not required for cryo-EM, thousands of homogenous particles need to be imaged and analyzed to achieve a high resolution structure. In this study, we observed that the CroV particles were homogenous and could be averaged to high resolution. Such detailed structural information of an intact giant virus capsid may help to shed light on their assembly mechanism. Based on our cryo-EM reconstruction of the CroV capsid and by comparison with other giant viruses, we propose a new spiral assembly pathway for the formation of large icosahedral virus capsids.
Results
Cryo-EM reconstruction and T-number
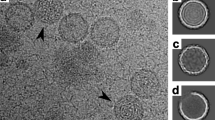
Purified from ≈40 L of infected Cafeteria roenbergensis culture, we obtained enough CroV sample for cryo-EM data collection (Fig. 1A). In total, 6698 particles were processed and 2471 particles were used in the final cryo-EM reconstruction. The refinement process required about three million CPU hours to reach the final reconstruction. The 21 Å resolution cryo-EM reconstruction of CroV reported here clearly shows individual capsomers on the virion surface (Fig. 2A and video in Supplementary s01). The major capsid protein (MCP) that forms the trimeric capsomers in most icosahedral giant viruses consists of a double “jelly-roll” fold12. Each jelly-roll is a wedge-shaped structure composed of eight anti-parallel β-strands42. The trimeric capsomer has a pseudo-hexagonal shape with six single jelly-rolls contained in three double jelly-roll MCPs. The vertices of the icosahedral particle are occupied by pentameric capsomers that probably consist of single jelly-roll proteins30. A multiple sequence alignment of several giant virus MCPs shows relatively high similarity in β-strand regions, whereas the inter-strand regions often contain insertions of varying length, such as the DE2 loop of the second jelly-roll (Fig. 3A)12. These insertions form protrusions on the exterior of the capsomers, conveying a truly trimeric look (magnified areas in Fig. 2A(a) and (b)). Capsomer arrangements of icosahedral viruses can be mathematically described by the triangulation number T, as defined by Caspar and Klug43. The T-number is a measurement of how many monomers (e.g. jelly-roll domains) exist in one icosahedral asymmetric unit. By tracing the capsomers in the hexagonal array from one 5-fold vertex to the neighboring one along axes h and k, which follow the center of the MCP (Fig. 2A), the T-number can be calculated using the equation: T = h2 + hk + k2. Based on the well resolved individual capsomers in our cryo-EM reconstruction, the T-number of the CroV capsid equals 499 (h = 7, k = 18) (Fig. 2B). Previously, the highest accurately determined T-number was that of Faustovirus (T = 277)44. The giant APMV capsid is estimated to have a T-number of ≈1000, but so far, technical barriers have prevented high-resolution reconstructions of APMV41.
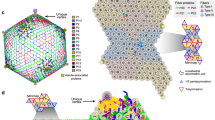
Cryo-EM reconstruction of the CroV virion and capsomer arrangements of other giant icosahedral viruses. (A) Reconstruction of the CroV capsid. The isosurface of the map was colored by pentasymmetrons (purple) and trisymmetrons (blue, red, green, cyan and orange). One of the 30 edges of the icosahedron is marked by a cyan line. Two surface areas (a,b) are magnified and selected capsomers are labeled by yellow triangles to show their orientations. (B–E) Isolated icosahedral faces of CroV, PBCV-1, CIV and PpV01 capsids are shown schematically. Their T-numbers, asymmetric unit capsomer numbers, and trisymmetron capsomer numbers are listed. 5-fold, 3-fold, and 2-fold symbols are indicated in red and ASUs are outlined in blue.
The double jelly-roll major capsid protein (MCP) of large icosahedral viruses. (A) Multiple sequence alignment of the MCPs of CroV, PBCV-1, PpV01 and APMV. Both CroV and APMV have large DE2 loops. (B) The CroV MCP (red) was homologously modeled and superposed with the X-ray structure of PBCV-1 (cyan) to show the longer DE2 loop of CroV. (C) Ribbon diagram of the trimeric homologously modeled CroV double jelly-roll MCP capsomer displaying a pseudo-hexagonal shape. Individual double jelly-roll MCPs are color-coded. (D) Central cross-section of the cryo-EM map of CroV superposed with that of PBCV-1. CroV not only has a larger virion diameter (3000 Å vs 1800 Å), its capsid layer is also thicker (105 Å vs 75 Å), which is consistent with the extended DE2 loop shown in panel (B).
Surface landscape
Giant virus capsids differ markedly in their surface architecture. APMV has a dense layer of fibers on its surface (Fig. 1C)45. The CIV virion has short fibers on most capsomers37, while PBCV-1 and Phaeocystis pouchetti virus (PpV01) capsids have fibers only on certain capsomers36, 46. In contrast, the CroV capsid is not decorated with fibers (Fig. 1A and B), but has surface protrusions that are almost 30 Å higher than those of PBCV-1 (Fig. 3D), owing to the 83 amino acids longer DE2 loop in the CroV MCP (Fig. 3A and B). This longer loop might play a role in virus-host interactions similar to the fibers of other giant viruses. The absence of external fibers, which convey great physical stability to APMV virions13, 41, leads to a higher signal-to-noise ratio in the CroV cryo-EM data. On the other hand, the exposed capsid surface renders CroV virions quite fragile, thus broken and deformed particles were frequently observed (Fig. 1A).
Unique Vertex and Genome-Containing Core
Many bacteriophages have unique portal structures for DNA packaging47 or genome delivery48. A so-called “star-gate” portal was discovered on the APMV capsid using transmission and scanning electron microscopy14, which was later confirmed by cryo-EM reconstruction with 5-fold averaging41. Using the same technique, the PBCV-1 capsid was found to have a modified vertex with a pocket underneath a needle-like spike structure, which may be involved in penetrating the host cell36. We also applied 5-fold averaging to the CroV reconstruction in order to detect potential unique vertex modifications. This resulted in densities at the outside of two opposing vertices along the 5-fold averaging axis, which might indicate a unique portal on the CroV capsid. However, due to noise in the data and the limited number of analyzable cryo-EM images, we were unable to confirm the existence of a CroV portal. If such a unique structure exists, it is probably quite small, as we did not observe any obvious portals in the original cryo-EM imagery.
The genome of APMV and related giant viruses is contained in a spherical inner compartment called the viral core, which is separated from the capsid layer by the inner viral membrane at a capsid – core distance of 300–500 Å41. The APMV core was found to have a concave depression beneath the unique vertex. Re-examination of the original APMV cryo-EM micrographs showed that the core depression is directly visible for most of the particles. For example, four of the seven particles published in the first cryo-EM study of APMV display this feature45. With CroV, we only observed very few particles that had a similarly deformed core (Fig. 1B). Most CroV cores seem to have a more or less spherical shape with a higher electron density in the center (Fig. 1A). In agreement with observations from other icosahedral giant viruses, the CroV cryo-EM reconstruction shows a possible membrane layer just beneath the protein capsid (Fig. 3D). No additional structural layers were observed, as seen in initial cryo-EM studies of APMV41, 45.
Discussion
Large, well-organized triangular arrays of capsomers were first discovered in samples of degraded Sericesthis iridescent virus49, which indicated that the capsid of large icosahedral viruses might be assembled from a pre-formed array of MCPs. The 5-fold and 3-fold arrays were named pentasymmetrons and trisymmetrons, respectively. In cryo-EM reconstructions of sufficiently high resolution, the symmetrons can be distinguished from each other by discontinuous lines separating capsomers of different orientations. This distinction is possible because the capsomers have only quasi-six-fold symmetry, but a true three-fold symmetric appearance12. The 231 capsomers of one CroV trisymmetron are rotated by 60° compared to the capsomers of a neighboring trisymmetron (Fig. 2A(b)), which clearly defines the trisymmetron boundary. In all available cryo-EM reconstructions of icosahedral giant viruses, including CroV, the pentasymmetrons consistently contain 31 capsomers with three concentric layers of pseudo-hexameric capsomers and one pentameric capsomer at the 5-fold vertex. This arrangement leads to a fixed h-number of 7 for all these giant viruses (Fig. 2B–E). The identical capsomer arrangement around the 5-fold vertices present in CroV, PBCV-1, PpV01, and CIV, leads us to conclude that they are likely assembled in a similar manner, starting with the pentameric capsomer. Analysis of the orientation of pentasymmetron capsomers shows that one of the six capsomers in the asymmetric unit has a different orientation, compared to the other five (Fig. 2A(a)). This unique capsomer is oriented the same way as the capsomer at the tip of a nearby trisymmetron, whereas the other five capsomers are rotated by 60°, creating an obvious boundary between pentasymmetron and trisymmetron. Coloring the capsomers according to their orientation reveals a spiral pattern around the 5-fold vertex that resembles five interlocked golf club heads surrounding the pentameric capsomer (Fig. 4). This is not only true for CroV, but also for PBCV-1 and CIV (Fig. 4B and C). Cryo-tomography studies have shown that the in vivo assembly of APMV capsids starts from the 5-fold vertex and proceeds gradually to complete the capsid shell14, 50, 51. No pre-assembled arrays or symmetrons have been observed inside the cell. These results challenge the idea that capsids assemble via pre-formed multicapsomeric units49. Therefore, we propose a continuous assembly pathway (Fig. 4D and animation in Supplementary s02) based on the common spiral pattern of capsomer orientations observed in icosahedral giant viruses (Fig. 4A–C). According to our model, capsomers first assemble in two layers around the pentameric capsomer, forming five triangles each with the same orientation (Fig. 4D step 1 and 2). For the third layer, instead of extending the triangle, one capsomer will “spiral” into the counterclockwise neighboring triangle (Fig. 4D step 3 and 4). This differently orientated capsomer will then seed the trisymmetron by recruiting more capsomers of the same orientation (Fig. 4D step 5, 6 and 7).
The proposed spiral assembly pathway of large icosahedral capsids. Isosurfaces of CroV (A), PBCV-1 (B), and CIV (C) cryo-EM maps were centered on the 5-fold axis. Capsomers in all three panels are colored based on their orientation in red, blue, green, cyan, and orange. The pentameric capsomers are depicted as purple stars. (D) Schematic diagram showing the right-handed spiral assembly pathway following arrows (1) to (6), with multiple extensions (7) of the trisymmetrons to form similar patterns as shown in (A–C). One set of capsomers (red) is labeled by Roman numerals if they are part of the pentasymmetron and by Arabic numerals if they are part of a trisymmetron.
In our model, we considered only the main structural components of the capsid, the MCP and the penton protein. However, the CroV genome encodes three additional paralogous capsid genes that could also influence the assembly process. A proteome analysis of purified CroV particles revealed that all four capsid proteins are present in the virion, although in different quantities52. Whereas the MCP was the most abundant protein, it is estimated that the CroV capsid contains only 60 copies of capsid protein 2, and 1–2 copies of the remaining two capsid proteins. Based on these copy numbers, it is possible that capsid protein 2 could be involved in the proposed spiral assembly process. As stated above, one capsomer in the pentasymmetron asymmetric unit is oriented differently (Fig. 2A(a) and capsomer VI in Fig. 4D) and “spirals” into the neighboring trisymmetron. Each pentasymmetron has five of those unique capsomers and there are 60 such unique positions per virion. Hence, the unique pentasymmetron capsomers could be heterotrimers consisting of one capsid protein 2 molecule and two copies of the MCP. The rare capsid proteins 3 and 4 could be involved in the aforementioned unique vertex structure that possibly decorates the CroV capsid. However, the resolution of our current reconstruction does not provide sufficiently detailed structural information to distinguish between different capsid proteins.
In summary, the 21 Å resolution cryo-EM reconstruction of the CroV virion allowed us to accurately determine the structure of a giant virus capsid, revealing the largest T-number reported to date (499), and the biggest trisymmetron consisting of 231 capsomers. Based on the orientation of capsomers around 5-fold axes, we propose that the capsids of CroV and related icosahedral giant viruses are assembled in a spiral mechanism, rather than from preformed MCP arrays, as has been generally accepted for giant icosahedral viruses. It is noteworthy that our assembly model is based on the observation that MCP capsomers are oriented in an identical fashion around the pentasymmetron in several giant icosahedral viruses. Although our model lacks direct experimental support, it is consistent with the continuous capsid assembly observed in cryo-tomography studies14, 50, 51. Furthermore, our model focuses only on the initial assembly steps, but does not address the question how the assembly continues to incorporate neighboring pentasymmetrons into the growing capsid, or how capsid size is regulated in giant icosahedral viruses. We hope that future studies will provide experimental data to test our spiral assembly hypothesis and clarify how trisymmetron size and spiral assembly at neighboring 5-fold vertices are controlled.
Methods
CroV was grown and purified as previously described52. CroV samples were loaded on Quantifoil S7/2 grids (Quantifoil Micro Tools GmbH, Germany), blotted manually and frozen using a guillotine style plunging device. The cryo-EM images were recorded on Kodak SO-163 film in a FEI CM300 field emission gun microscope at a calibrated magnification of 20,629 and a dose level of approximately 25 e-/Å2 at the Purdue University Cryo-EM facility. Micrographs were scanned on a Nikon Coolscan 9000 with a final pixel size of 6.156 Å. The cryo-EM reconstruction was calculated using the program FREALIGN53. Most computation was performed at Texas Advanced Computing Center (TACC). Resolution of the reconstruction was determined using Fourier Shell correlation (FSC) for the capsid part with threshold of 0.333.
Data Availability Statement
All data associated with the manuscript are available to readers on request.
References
Raoult, D. et al. The 1.2-megabase genome sequence of Mimivirus. Science 306, 1344–50 (2004).
Boyer, M. et al. Giant Marseillevirus highlights the role of amoebae as a melting pot in emergence of chimeric microorganisms. Proc Natl Acad Sci USA 106, 21848–53 (2009).
Philippe, N. et al. Pandoraviruses: Amoeba viruses with genomes up to 2.5 Mb reaching that of parasitic eukaryotes. Science 341, 281–286 (2013).
Legendre, M. et al. Thirty-thousand-year-old distant relative of giant icosahedral DNA viruses with a pandoravirus morphology. Proc Natl Acad Sci USA 111, 4274–9 (2014).
Legendre, M. et al. In-depth study of Mollivirus sibericum, a new 30,000-y-old giant virus infecting Acanthamoeba. Proc Natl Acad Sci USA 201510795 (2015).
Reteno, D. G. et al. Faustovirus, an Asfarvirus-related new lineage of giant viruses infecting amoebae. J Virol 89, 6585–6594 (2015).
Raoult, D. & Forterre, P. Redefining viruses: lessons from Mimivirus. Nat. Rev. Microbiol. 6, 315–319 (2008).
Forterre, P. The origin of viruses and their possible roles in major evolutionary transitions. Virus Res 117, 5–16 (2006).
Claverie, J. Viruses take center stage in cellular evolution. Genome Biol. 1–5 (2006).
Colson, P., de Lamballerie, X., Fournous, G. & Raoult, D. Reclassification of giant viruses composing a fourth domain of life in the new order Megavirales. Intervirology 55, 321–32 (2012).
Yutin, N., Wolf, Y. I. & Koonin, E. V. Origin of giant viruses from smaller DNA viruses not from a fourth domain of cellular life. Virology 466–467, 38–52 (2014).
Xiao, C. & Rossmann, M. G. Structures of giant icosahedral eukaryotic dsDNA viruses. Curr. Opin. Virol 1, 101–9 (2011).
Kuznetsov, Y. G. et al. Atomic force microscopy investigation of the giant mimivirus. Virology 404, 127–137 (2010).
Zauberman, N. et al. Distinct DNA exit and packaging portals in the virus Acanthamoeba polyphaga mimivirus. PLoS Biol. 6, e114 (2008).
Seibert, M. M. et al. Single mimivirus particles intercepted and imaged with an X-ray laser. Nature 470, 78–81 (2011).
Ekeberg, T. et al. Three-dimensional reconstruction of the giant Mimivirus particle with an X-Ray free-electron laser. Phys. Rev. Lett. 114, 1–6 (2015).
Baker, T. S., Olson, N. H. & Fuller, S. D. Adding the third dimension to virus life cycles: three-dimensional reconstruction of icosahedral viruses from cryo-electron micrographs. Microbiol. Mol. Biol. Rev. 63, 862–922 (1999).
Grimes, J. M., Fuller, S. D. & Stuart, D. I. Complementing crystallography: the role of cryo-electron microscopy in structural biology. Acta Crystallogr. D. Biol. Crystallogr 55, 1742–9 (1999).
Chang, J., Liu, X., Rochat, R. H., Baker, M. L. & Chiu, W. Reconstructing virus structures from nanometer to near-atomic resolutions with cryo-electron microscopy and tomography. Adv. Exp. Med. Biol 726, 49–90 (2012).
Chiu, W., McGough, A., Sherman, M. B. & Schmid, M. F. High-resolution electron cryomicroscopy of macromolecular assemblies. Trends Cell Biol 9, 154–9 (1999).
van Heel, M. et al. Single-particle electron cryo-microscopy: towards atomic resolution. Q. Rev. Biophys. 33, 307–69 (2000).
Zhang, X. et al. Near-atomic resolution using electron cryomicroscopy and single-particle reconstruction. Proc. Natl. Acad. Sci. USA 105, 1867–72 (2008).
Zhang, X., Jin, L., Fang, Q., Hui, W. H. & Zhou, Z. H. 3.3 Å cryo-EM structure of a nonenveloped virus reveals a priming mechanism for cell entry. Cell 141, 472–82 (2010).
Böttcher, B., Wynne, S. A. & Crowther, R. A. Determination of the fold of the core protein of hepatitis B virus by electron cryomicroscopy. Nature 386, 88–91 (1997).
Conway, J. F. et al. Visualization of a 4-helix bundle in the hepatitis B virus capsid by cryo-electron microscopy. Nature 386, 91–4 (1997).
Zhou, Z. H. et al. Seeing the herpesvirus capsid at 8.5 Å. Science 288, 877–80 (2000).
Zhou, Z. H. et al. Electron cryomicroscopy and bioinformatics suggest protein fold models for rice dwarf virus. Nat. Struct. Biol 8, 868–73 (2001).
Jiang, W. et al. Backbone structure of the infectious epsilon15 virus capsid revealed by electron cryomicroscopy. Nature 451, 1130–4 (2008).
Sirohi, D. et al. The 3.8 Å resolution cryo-EM structure of Zika virus. Science 352, 467–70 (2016).
Zhang, X. et al. Structure of Sputnik, a virophage, at 3.5-Å resolution. Proc. Natl. Acad. Sci. USA 109, 18431–6 (2012).
Rossmann, M. G., Morais, M. C., Leiman, P. G. & Zhang, W. Combining X-ray crystallography and electron microscopy. Structure 13, 355–62 (2005).
Chiu, W., Baker, M. L., Jiang, W., Dougherty, M. & Schmid, M. F. Electron cryomicroscopy of biological machines at subnanometer resolution. Structure 13, 363–72 (2005).
Rossmann, M. G., Bernal, R. & Pletnev, S. V. Combining electron microscopic with x-ray crystallographic structures. J. Struct. Biol. 136, 190–200 (2001).
Leiman, P. G., Kanamaru, S., Mesyanzhinov, V. V., Arisaka, F. & Rossmann, M. G. Structure and morphogenesis of bacteriophage T4. Cell. Mol. Life Sci. 60, 2356–70 (2003).
Nandhagopal, N. et al. The structure and evolution of the major capsid protein of a large, lipid-containing DNA virus. Proc. Natl Acad. Sci. USA 99, 14758–14763 (2002).
Zhang, X. et al. Three-dimensional structure and function of the Paramecium bursaria chlorella virus capsid. Proc Natl Acad Sci USA 108, 14837–14842 (2011).
Yan, X. et al. The capsid proteins of a large, icosahedral dsDNA virus. J Mol Biol 385, 1287–1299 (2009).
Fischer, M. G., Allen, M. J., Wilson, W. H. & Suttle, C. A. Giant virus with a remarkable complement of genes infects marine zooplankton. Proc. Natl. Acad. Sci 107, 19508–19513 (2010).
Fenchel, T. & Patterson, D. J. Cafeteria roenbergensis nov. gen., nov. sp., a heterotrophic microflagellate from marine plankton. Mar. Microb. Food Webs 3, 9–19 (1988).
Klose, T. et al. The Three-Dimensional Structure of Mimivirus. Intervirology 53, 268–273 (2010).
Xiao, C. et al. Structural studies of the giant mimivirus. PLoS Biol. 7, e92 (2009).
Rossmann, M. G. & Johnson, J. E. Icosahedral RNA virus structure. Annu. Rev. Biochem. 58, 533–73 (1989).
Caspar, D. L. & Klug, A. Physical principles in the construction of regular viruses. Cold Spring Harb. Symp. Quant. Biol. 27, 1–24 (1962).
Klose, T. et al. Structure of faustovirus, a large dsDNA virus. Proc. Natl. Acad. Sci 113, 6206–6211 (2016).
Xiao, C. et al. Cryo-electron microscopy of the giant Mimivirus. J. Mol. Biol. 353, 493–496 (2005).
Yan, X., Chipman, P. R., Castberg, T., Bratbak, G. & Baker, T. S. The marine algal virus PpV01 has an icosahedral capsid with T = 219 quasisymmetry. J Virol 79, 9236–43 (2005).
Simpson, A. A. et al. Structure of the bacteriophage phi29 DNA packaging motor. Nature 408, 745–50 (2000).
Rossmann, M. G., Mesyanzhinov, V. V., Arisaka, F. & Leiman, P. G. The bacteriophage T4 DNA injection machine. Curr. Opin. Struct. Biol. 14, 171–80 (2004).
Wrigley, N. G. An electron microscope study of the structure of Sericesthis iridescent virus. J Gen Virol 5, 123–34 (1969).
Suárez, C. et al. Open membranes are the precursors for assembly of large DNA viruses. Cell. Microbiol. 15, 1883–95 (2013).
Mutsafi, Y., Shimoni, E., Shimon, A. & Minsky, A. Membrane Assembly during the Infection Cycle of the Giant Mimivirus. PLoS Pathog. 9, e1003367 (2013).
Fischer, M. G., Kelly, I., Foster, L. J. & Suttle, C. A. The virion of Cafeteria roenbergensis virus (CroV) contains a complex suite of proteins for transcription and DNA repair. Virology 466–467, 82–94 (2014).
Grigorieff, N. FREALIGN: high-resolution refinement of single particle structures. J. Struct. Biol. 157, 117–25 (2007).
Acknowledgements
We thank the Purdue Cryo-EM facility for the use of their scopes to collect the data. Use of computer clusters at Texas Advanced Computing Center was supported by allocations from Texas (TG-MCB100154) and NSF TeraGrid (later renamed as XSEDE) (MCB110016). C.X. was supported by UTEP new faculty startup fund from Texas STARs Program. M.G.F. was supported by the Max Planck Society, as well as by fellowships from the Gottlieb Daimler- and Karl Benz-Foundation Germany, EMBO, and the University of British Columbia. Grants to C.A.S. from the Natural Sciences and Engineering Research of Canada, the Canadian Foundation for Innovation and the British Columbia Knowledge Development Fund enabled culturing and purification of viruses at UBC.
Author information
Authors and Affiliations
Contributions
C.X. performed the cryo-EM data collection, reconstruction and computation. C.X. and M.G.F. wrote the manuscript with input from C.A.S. M.G.F. purified the virus. D.M.B., N.U.-R. and G.A.A. helped in digitizing the micrographs and processing the raw data. C.X. and C.A.S. conceived the project.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Accession code: The 21 Å cryo-EM map of CroV with associated FSC curve has been deposited in the Electron Microscopy Data Bank with accession code EMD-8748.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xiao, C., Fischer, M.G., Bolotaulo, D.M. et al. Cryo-EM reconstruction of the Cafeteria roenbergensis virus capsid suggests novel assembly pathway for giant viruses. Sci Rep 7, 5484 (2017). https://doi.org/10.1038/s41598-017-05824-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-05824-w
This article is cited by
-
Near-atomic architecture of Singapore grouper iridovirus and implications for giant virus assembly
Nature Communications (2023)
-
A novel capsid protein network allows the characteristic internal membrane structure of Marseilleviridae giant viruses
Scientific Reports (2022)
-
Crystal structures of FNIP/FGxxFN motif-containing leucine-rich repeat proteins
Scientific Reports (2022)
-
Giant virus biology and diversity in the era of genome-resolved metagenomics
Nature Reviews Microbiology (2022)
-
Near-atomic, non-icosahedrally averaged structure of giant virus Paramecium bursaria chlorella virus 1
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.