Abstract
Single-cell RNA-seq (scRNA-seq) of pancreatic islets have reported on α- and β-cell gene expression in mice and subjects of predominantly European ancestry. We aimed to assess these findings in East-Asian islet-cells. 448 islet-cells were captured from three East-Asian non-diabetic subjects for scRNA-seq. Hierarchical clustering using pancreatic cell lineage genes was used to assign cells into cell-types. Differentially expressed transcripts between α- and β-cells were detected using ANOVA and in silico replications of mouse and human islet cell genes were performed. We identified 118 α, 105 β, 6 δ endocrine cells and 47 exocrine cells. Besides INS and GCG, 26 genes showed differential expression between α- and β-cells. 10 genes showed concordant expression as reported in rodents, while FAM46A was significantly discordant. Comparing our East-Asian data with data from primarily European subjects, we replicated several genes implicated in nuclear receptor activations, acute phase response pathway, glutaryl-CoA/tryptophan degradations and EIF2/AMPK/mTOR signaling. Additionally, we identified protein ubiquitination to be associated among East-Asian β-cells. We report on East-Asian α- and β-cell gene signatures and substantiate several genes/pathways. We identify expression signatures in East-Asian β-cells that perhaps reflects increased susceptibility to cell-death and warrants future validations to fully appreciate their role in East-Asian diabetes pathogenesis.
Similar content being viewed by others
Introduction
Data suggest that East-Asians may develop Type 2 diabetes (T2D) at a younger age and at lower BMI levels as compared to European ancestry populations1, 2. Worryingly, subjects with younger onset and lean diabetes tend to be less likely to achieve metabolic targets and have a higher prevalence of subsequent comorbidities2. Genome-wide association studies have successfully uncovered numerous common variants associated with T2D and highlight on inter-ethnic differences in frequency and effect size at these risk loci (for eg. at the TCF7L2 locus)3. Despite these accumulating genetic information, due to modest effect sizes conferred at these common T2D risk loci, major limitations still exists in clearly delineating the disease phenotype observed in East-Asians.
Islet cells are centrally involved in the etiology of diabetes. Ethnic differences in islet cell function may exist due to inherent genetics and epigenetic changes driven by varied lifestyles and is suggested to particularly predispose Asian subjects to T2D4, 5. Evaluation of gene expression in target tissues perhaps represents a combined reflection of pure genetic effects and lifestyle and environmental influences and may identify novel pathways associated with disease6. Advances in single-cell RNA-seq (scRNA-seq) techniques enable identification of novel transcripts and cellular heterogeneities and very recent studies in mice7 and human8,9,10,11 pancreatic islets have provided novel transcriptomic insights into islet cell-type biology. However, as most human islet scRNA-seq studies have been performed predominantly in subjects of European ancestry, it is unclear if reported gene signatures are transferrable across ethnicities. We performed scRNA-seq on islet cells captured from three non-diabetic Singaporean Chinese subjects and aimed to evaluate for common and unique expression signatures with recent studies7,8,9,10,11.
Methods
Human islets
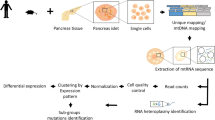
Pancreatic islets were obtained from three non-diabetic Singaporean Chinese subjects from the LKCMedicine Islet Isolation Facility that obtains human pancreata through the Singapore National Organ Transplant Unit (Supplementary Table 1). Informed consent was obtained from all subjects, all methods were carried out in accordance with relevant guidelines and regulations and the study was approved by the Institutional Review Board of the Singapore National Organ Transplant Unit (#IRB-2013-09-005). Islets were cultured for 3 days in complete CMRL-1066 media prior to being handpicked under a stereomicroscope for both functional assay (GSIS, Glucose Stimulated Insulin Secretion) and scRNA-seq studies. Islets with hypoxic cores were discarded. Subsequently, handpicked islets were dissociated into single-cells using Accutase® Cell Detachment Solution (Sigma Aldrich, St.Louis, MO, USA) and re-suspended in complete CMRL-1066 media. For GSIS, islets were incubated in 3 mmol/L glucose for one hour before being placed in a perfusion chamber and exposed to 3 mmol/L glucose (Low Glucose) for 10 minutes followed by 16.7 mmol/L glucose (High Glucose) for 10 minutes. These studies confirmed that islets used in this study exhibited normal insulin secretion profiles (Supplementary Table 1).
Single-cell RNA-seq (scRNA-seq)
Single human islet cells were quantified using an automated cell counter (Bio-Rad TC20™) and single-cell suspension concentrations were adjusted to approximately 200,000 cells/ml prior to cell capture, as recommended (Fluidigm). Dissociated islet cells had a consistent viability of about 95% and were observed with a size range of approximately 8 to 14 µm. Single human islet cells were captured using medium filter chips (10 to 17 µm) on the Fluidigm C1™ Auto-prep system, as previously performed7,8,9. Captured cells in each well of the C1 chip were visually inspected on a Nikon ECLIPSE Ti microscope, fitted with a 96-well C1 chip holder. Wells with no cell captured or with more than one cell captured were excluded (Supplementary Figure 1).
138, 84 and 226 single-cells from subject 1, 2 and 3, respectively (Supplementary Table 1) were processed for RNA-seq using Nextera XT kits (Illumina). Cell lysis, reverse transcription (SMARTer Ultra Low RNA kit, Clontech) and PCR amplification (Advantage® 2 kit, Clontech) were subsequently performed on the C1™ Auto-prep module. cDNA were aliquoted and quantified using picogreen serial dilutions. Approximately 0.15 ng of cDNA from each cell was processed for RNA-seq using Nextera XT DNA library preparation kits (Illumina). cDNA from 63 and 75 single cells from subject 1, 84 single cells from subject 2 and 82, 73 and 71 single cells were pooled into multiplex libraries and sequenced on 1 lane on Hiseq 4000 each.
scRNA-seq quality control (QC)
Six cells with low sequence reads (<1 million reads) were excluded from analysis. Mean reads in remaining cells was 3.68 million (1.02 million–8.21 million). Sequence reads were aligned to hg19 and mRNA quantified to RPKM values using Partek Genomics Suite (ver6.6). mRNA levels between-samples were normalized by quantile normalization before statistical analysis. 47 cells (22 from patient 1, 2 from patient 2 and 23 from patient 3) that showed low alignment to exonic regions (<30% of reads), perhaps due to contamination with genomic DNA, were excluded from analysis (Supplementary Figure 2). Mean alignment to exonic regions for remaining 395 cells was 55.3% (34.4–92.6%). Similar to previous studies7,8,9,10,11, we observed several cells to be bi-hormonal - expressing high levels of two hormones from INS, GCG or SST. Most bi-hormonal islet single-cells were previously shown to be false positives7,8,9,10 and it has been reported that the C1 may capture doublet cells (Fluidigm white-paper). We thus evaluated INS, GCG and SST transcripts expression individually in each cell and excluded 101 cells with >10% expression of secondary hormone compared to the primary hormone expressed.
Identification of islet cell-types
Hierarchical clustering of 294 cells that passed QC procedures were performed using Hierarchical Cluster Viewer (6.5) on Partek Genomics Suite (ver6.6). Hierarchical clustering of cells were initially performed using all transcripts with RPKM values ≥500 in ≥2% of cells (Supplementary Figure 3) and subsequently, to distinctly separate out various islet cell-types, using expressed transcripts from pancreatic cell lineage genes12 (Supplementary Figure 4). These clustering analyses further identified 18 possible doublet cells (Supplementary Figure 4) that were excluded before statistical analyses.
We excluded 47 exocrine cells (with high REG1A expression) and 18 doublet cells and performed additional hierarchical clustering and PCA analyses based on INS, GCG and SST transcript expression levels (Supplementary Figure 5). These analyses revealed that the 118 GCG expressing cells (33 from patient 1, 15 from patient 2 and 70 from patient 3), 105 INS expressing cells (32 from patient 1, 33 from patient 2 and 40 from patient 3) and 6 SST expressing cells (1 from patient 2 and 5 from patient 3) were predominantly mono-hormonal. Additional hierarchical clustering to identify possible within subtype groups of cells were performed using 118 GCG expressing cells (Supplementary Figure 6) and 105 INS expressing cells (Supplementary Figure 7).
Statistical analysis
ANOVA analyses were performed to identify transcripts that were differentially expressed between INS expressing cells and high GCG expressing cells. To identify gene expression signatures among the INS and GCG expressing cells in our study, we utilized cells from patient 1 and 2 as the discovery stage (65 high INS expressing cells and 48 high GCG expressing cells) and cells from patient 3 for validation 40 high INS expressing cells and 70 high GCG expressing cells). ANOVA analyses were restricted to more highly expressed [582 transcripts (412 genes) with RPKM >50 in at least 25% of cells). P-values were adjusted by Bonferroni correction for the number of tests performed (582 tests) and significant associations were defined as those with an adjusted p-value < 0.05 and fold-change >or <1.5.
All uniquely expressed genes in α- or β-cells in non-diabetic human and mouse single islet cell studies2,3,4,5,6 were selected for validation among our East-Asian islet cells. To improve power to validate these recent findings, all α- and β-cells from patients 1, 2 and 3 in our study were utilized (118 α-cells and 105 β-cells). ANOVA analyses, as indicated above, were performed to identify transcripts that were differentially expressed between our East-Asian α- and β-cells. P-values were adjusted by Bonferroni correction for the number of tests performed and significant associations were defined as those with an adjusted p-value < 0.05 and fold-change >or <1.5.
Ingenuity pathway analysis (IPA ver1-07) was used to evaluate for potential upstream regulators and over-representation in canonical pathways for identified genes in our study.
Immunohistochemistry validation
Human pancreatic samples were fixed in 4% paraformaldehyde followed by 30% sucrose overnight before embedding in tissue freezing medium (Leica, Germany). Sections were prepared on Superfrost Plus (Fisher Scientific, USA) at 10 um thickness. Thereafter slides were blocked for 1 hr at room temperature followed by incubation with primary antibodies (Guinea Pig Anti-insulin (Dako, Denmark), Mouse Anti-Glucagon (Sigma Aldrich, USA), Rabbit Anti-DNAJC3 (Abcam, USA), Rabbit Anti-PFKFB2 (Abcam, USA), and Mouse Anti-UCHL1 (Sigma Aldrich, USA) overnight in 1X TBS with 1% BSA at 4 °C. Slides were washed in 1X TBS with 0.025% Triton X-100 and incubated with secondary antibodies Alexa 647 Anti-Guinea Pig (Life Tech, USA), Alexa 568 Anti-Mouse (LifeTech, USA), Alexa 488 Anti-Rabbit (LifeTech, USA), Alexa 488 Anti-Mouse (Life tech, USA), DAPI (LifeTech, USA) at room temperature for 1 hr. Following incubation and washing, slides were mounted with Vectashield mounting media (Vectalabs, USA). Images were taken using a Zeiss LSM800 and analysed with ZEN Blue® software.
Results
Islet cell-types
Hierarchical clustering of human islet cells using all transcripts with RPKM values ≥500 in ≥2% of cells [252 transcripts (146 genes)] revealed three main clusters based on known islet cell transcripts at GCG, INS and REG1A, indicating that cells captured were mainly pancreatic endocrine (α- and β-cells) and exocrine cells (Supplementary Figure 3). Cells were subsequently clustered using highly expressed transcripts (RPKM values ≥500 in ≥2% of cells) from pancreatic cell lineage genes12 together with REG1A to distinctly separate out islet cell-types (Supplementary Figure 4). These analyses further identified and excluded 18 doublet cells due to dual expression of transcripts from REG1A, GCG, INS and SST. Remaining 276 cells were assigned as 47 REG1A expressing exocrine cells, 118 GCG expressing α-cells, 105 INS expressing β-cells and 6 SST expressing δ-cells (Supplementary Figures 4 and 5). Hierarchical clustering of assigned α-cells and β-cells did not reveal distinct sub-groups of cell-types (Supplementary Figures 6 and 7).
46,390 transcripts from 17,287 genes were detected among the 276 cells that passed QC filters. On average 14,796 transcripts (8,450–19,547) and 5,638 genes (3,136–7,601) were expressed per cell (Supplementary Figure 8). Similar to previous reports5, 6, a large proportion of transcripts were detected at relatively low levels – approximately 25% of transcripts with total RPKM <5.
Differentially expressed transcripts among β-cells and α-cells
To identify β- and α-cell gene expression patterns among our data, we utilized cells from patient 1 and 2 in a discovery stage. ANOVA analysis was performed among 65 β-cells and 48 α-cells in the discovery stage. Identified transcripts were subsequently validated in 40 β-cells and 70 α-cells from patient 3. We identified 12 transcripts (11 genes - INS, HADH, SEC11C, PFKB2, MAP1B, SCD, RNR2, OLMALINC, DNAJC3, UCHL1 and G6PC2) to be highly expressed in β-cells and 17 transcripts (17 genes - GCG, TTR, ALDH1A1, CLU, TMEM176B, GC, BEX3, HIGD1A, ELL2, MYL6, CYSTM1, H3F3B, LDHA, NAA20, FABP5, STMN1, RBX1) to be highly expressed among α-cells (Table 1). Ingenuity pathway analysis (IPA ver1-07) of replicating genes indicated involvement in multiple nuclear receptor and AMPK signaling pathways (Supplementary Figure 9) and revealed FOXA2, MAFA, HNF1A, NKX6-2 as top upstream transcriptional regulators (Supplementary Table 2). Differentially expressed β and α-cell genes with increased level of stringency, where highly expressed transcripts were cut-off based on proportion of cells in the smallest islet cell endocrine group in the discovery stage [232 transcripts (314 genes) with RPKM >50 in at least 42% of cells] and replication stage [202 transcripts (298 genes) with RPKM >50 in at least 47% of cells] is also provided in Supplementary Table 3.
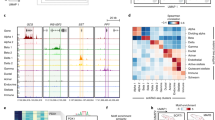
For three hits (DNAJC3, UCHL1 and PFKB2) that showed higher expression levels in our β-cells (as compared to α-cells) we further performed immunohistochemistry validations on islet sections of the three subjects used in the study. All three hits showed strong co-expression in cells producing INS and DNAJC3 and PFKB2 did not co-localize with GCG (Fig. 1, Supplementary Figures 10–12). UHCL1 with GCG co-stains were not carried out because of primary antibody incompatibility.
Representative immunofluorescence staining showing co-localization of transcripts with INS + cells and not GCG + cells from pancreas of three different subjects. Subject 1 (A,D and G), subject 2 (B,E and H) and subject 3 (C,F and I). (A,B and C) shows DNAJC3 (green), INS (red), GCG (blue) and nucleus (grey) stains. (D,E and F) shows PFKB2 (green), INS (red), GCG (blue) and nucleus (grey) stains. (G,H and I) shows UCHL1 (green), INS (red) and nucleus (grey) stains. Cell co-expressing INS and DNAJC or PFKB2 or UCHL1 are represented as yellow. Inserts show cells expressing insulin (red) and the respective gene of interest (green) at higher magnification and at individual channels. Scale bar represents 20 µm.
In silico replication of rodent and human α- and β-cells gene expression signatures
We next evaluated, in silico, unique gene expression signatures of non-diabetic mice and human α- and β-cells7,8,9,10,11 among our East-Asian scRNA-seq results utilizing cells from all three subjects (118 α-cells and 105 β-cells). ANOVA analyses, using 624 transcripts (584 genes) with RPKM >50 in at least 25% of cells, were performed to identify transcripts that were differentially expressed between our East-Asian α- and β-cells.
We replicated five upregulated mouse α-cell genes (GCG, TTR, HIGD1A, GPX3 and PDK4, adjusted P-value between 4.96 × 10−98 and 1.36 × 10−5) and five upregulated mouse β-cell genes (INS, HADH, G6PC2, RRAGD and IAPP, adjusted P-value between 3.93 × 10−82 and 3.25 × 10−5) among our scRNA-seq data (Supplementary Table 4). FAM46A that was reported to be upregulated in mouse β-cells7 however showed higher expression in our human α-cells (adjusted P-value = 4.21 × 10−4).
Validation of human non-diabetic islet gene expression8,9,10,11 showed concordantly high expression of transcripts in sixteen genes among our α-cells (GCG, TTR, GC, TMEM176B, HIGD1A, GPX3, TM4SF4, FABP5, LDHA, NAA20, CRYBA2, SERPINA1, PDK4, HLA-E, PALLD and C10orf10, adjusted P-value between 2.28 × 10−97 and 0.01) and 14 genes among our β-cells (INS, HADH, PFKB2, G6PC2, RPL3, EIF4A2, RRAGD, HNRNPC, IAPP, CANX, FAM162A, ACAT1, TVP23B and SERINC1, adjusted P-value between 2.81 × 10−81 and 0.04) (Tables 2 and 3). Association results of all 624 transcripts tested are indicated in Supplementary Table 5. The validated common (across European and East-Asian ancestry) α-cells genes were over-represented in nuclear receptor signaling as well as the acute phase response signaling pathway and the common β-cells genes were implicated in six pathways (Supplementary Figure 13).
Nine α-cell genes (ALDHA1, CLU, BEX3, ELL2, MYL6, CYSTM1, H3F3B, STMN1 and RBX1) and seven β-cell genes (OLMALINC, RNR2, SCD, MAP1B, DNAJC3, SEC11C and UCHL1) were significantly expressed in our East-Asian data (Table 1) but not reported as unique non-diabetic upregulated human α- or β-cell genes8,9,10,11. IPA analysis of these genes revealed a role for protein ubiquitination pathway among East-Asian β-cells (Supplementary Figures 14 and 15), while no pathway remained significant among the discordant α-cell genes after multiple test corrections.
Discussion
We utilized Fluidigm C1™ to capture single islet cells from non-diabetic East-Asian subjects and utilized these for RNA-seq analyses. Reassuringly the main genes expressed in our scRNA-seq data of human islets were INS and GCG, indicating that predominantly endocrine cells were captured. Besides these determinant gene classifiers, our data corroborated the role of several well-characterized as well as more novel α- and β-cell genes identified very recently7,8,9,10,11. Pathway-based analyses were used to evaluate the role of genes in aggregate and substantiated multiple known pathways such as nuclear receptor activations and AMPK signaling, in addition to more novel ones, such as the EIF2 signaling in β-cells7,8,9,10,11.
We report on specific mouse-human species difference in islet cell transcriptomic patterns, where mouse β-cell gene, FAM46A, showed higher expression in our human α-cells instead. Such species differences in expression patterns for other genes, such as GC and DLK1, have been identified from previous studies9, 10 and may reflect functional differences at the species level13.
Our East-Asian scRNA-seq data enabled for an ethnic-specific evaluation of transcriptomic signatures with recently published data. Besides the nuclear receptor activation pathways, identification of acute phase response signaling as a common pathway involved in α-cells reinforces the view that α-cells may be better protected, as compared to β-cells, against microorganism infections14. Similarly identification of glutaryl-coA and tryptophan degradations may indicate on the importance of adiposity or cholesterol levels in influencing β-cell functions15, 16.
Interestingly, we identified the protein ubiquitination pathway that included DNAJC3 and UCHL1 genes, as a pathway in East-Asian β-cells. These genes have been associated with T2D and perhaps indicate the vulnerability of pancreatic β-cells for protein misfolding, increased ER stress and proteotoxicity, hallmarks for islet cell defects in T2D17,18,19,20. Intriguingly, comparing our β-cell gene signatures in non-diabetic East-Asian individuals to genes highly expressed in β-cells of T2D subjects11 also revealed transcript at HSP90B1 that is involved in ER stress (data not shown). Our comparative analyses therefore indicate that pancreatic β-cells in non-diabetic East-Asians may already be predisposed to increased ER stress and subsequent cell death. Nevertheless, given that present human islet single-cell studies are still presently modest in the number of cells analyzed, whether this protein ubiquitination pathways render β-cells more susceptible to cell death and if these result in subsequent development of T2D disease risks, warrants further validations in large-scale East-Asian islet single-cells, including subjects at various disease states (i.e. pre-diabetes and type 2 diabetes) and specific functional evaluations.
Our study has limitations such as the high number of doublet cells and may indicate experimental contaminations. This however, is a common feature among islet single-cell studies that resulted in the exclusion of about 30% of captured cells7,8,9,10. We believe that our stringent QC procedures that excluded a similar proportion of cells, would have removed most contaminated data from analyses. Another limitation was that we had restricted our analyses to highly expressed transcripts to identify differential ones between α- and β-cells. This may have resulted in a reduced number of mouse and human islet transcripts tested during in silico validations. However, due to the relatively modest number of cells analyzed, we have reduced power to identify rare transcripts as well as to identify sub-groups of cells.
In conclusion, we present for the first time scRNA-seq data from non-diabetic East-Asian islets and corroborated multiple genes and pathways that may be commonly involved in α- and β-cells across ancestry groups. At the same time we identify signatures that may play a more prominent role in East-Asians, such as those involved in protein ubiquitination, and these require subsequent evaluations to fully appreciate their roles in diabetes etiology.
References
Kong, A. P. et al. Diabetes and its comorbidities–where East meets West. Nat Rev Endocrinol 9(9), 537–547 (2013).
Yeung, R. O. et al. Metabolic profiles and treatment gaps in young-onset type 2 diabetes in Asia (the JADE programme): a cross-sectional study of a prospective cohort. Lancet Diabetes Endocrinol 2(12), 935–943 (2014).
Cho, Y. S. et al. Genetics of type 2 diabetes in East Asian populations. Curr Diab Rep 12(6), 686–96 (2012).
Yabe, D. & Seino, Y. Type 2 diabetes via β-cell dysfunction in East Asian people. Lancet Diabetes Endocrinol 4(1), 2–3 (2016).
Misra, A. et al. South Asian diets and insulin resistance. Br J Nutr 101(4), 465–473 (2009).
Chen, B. H. et al. Peripheral Blood Transcriptomic Signatures of Fasting Glucose and Insulin Concentrations. Diabetes. 65(12), 3794–3804 (2016).
Xin, Y. et al. Use of the Fluidigm C1 platform for RNA sequencing of single mouse pancreatic islet cells. Proc Natl Acad Sci 113(12), 3293–3298 (2016).
Wang, Y. J. et al. Single cell transcriptomics of the human endocrine pancreas. Diabetes. 65(10), 3028–3038 (2016).
Li, J. et al. Single-cell transcriptomes reveal characteristic features of human pancreatic islet cell types. EMBO Rep. 17(2), 178–187 (2016).
Xin, Y. et al. RNA Sequencing of Single Human Islet Cells Reveals Type 2 Diabetes Genes. Cell Metab. 24(4), 608–615 (2016).
Segerstolpe, Å. et al. Single-Cell Transcriptome Profiling of Human Pancreatic Islets in Health and Type 2 Diabetes. Cell Metab. 24(4), 593–607 (2016).
Hrvatin, S. et al. Differentiated human stem cells resemble fetal, not adult, β cells. Proc Natl Acad Sci 111(8), 3038–3043 (2014).
Arrojo e Drigo, R. et al. New insights into the architecture of the islet of Langerhans: a focused cross-species assessment. Diabetologia. 58(10), 2218–2228 (2015).
de Beeck, A. O. & Eizirik, D. L. Viral infections in type 1 diabetes mellitus–why the β cells? Nat Rev Endocrinol 12(5), 263–273 (2016).
Cochran, B. J. et al. Impact of Perturbed Pancreatic β-Cell Cholesterol Homeostasis on Adipose Tissue and Skeletal Muscle Metabolism. Diabetes. 65(12), 3610–3620 (2016).
Oxenkrug, G. Insulin resistance and dysregulation of tryptophan-kynurenine and kynurenine-nicotinamide adenine dinucleotide metabolic pathways. Mol Neurobiol. 48(2), 294–301 (2013).
Costes, S., Gurlo, T., Rivera, J. F. & Butler, P. C. UCHL1 deficiency exacerbates human islet amyloid polypeptide toxicity in β-cells: evidence of interplay between the ubiquitin/proteasome system and autophagy. Autophagy. 10(6), 1004–1014 (2014).
Synofzik, M. et al. Absence of BiP co-chaperone DNAJC3 causes diabetes mellitus and multisystemic neurodegeneration. Am J Hum Genet. 95(6), 689–697 (2014).
Plaisance, V. et al. Endoplasmic Reticulum Stress Links Oxidative Stress to Impaired Pancreatic Beta-Cell Function Caused by Human Oxidized LDL. PLoS One. 11(9), e0163046 (2016).
Brackeva, B. et al. Potential of UCHL1 as biomarker for destruction of pancreatic beta cells. J Proteomics 117, 156–167 (2015).
Acknowledgements
Y.A. and B.O.B. were both supported by their respective Start-up-grant, Lee Kong Chian School of Medicine, Nanyang Technological University, Singapore. Y.A. is additionally supported by the Ministry of Education Singapore (1T1-02/04 and MOE2015-T2-2-087) and by the Lee Foundation Grant 2015. We thank the Human Pancreas Islet Isolation team, Lee Kong Chian School of Medicine NTU Singapore, for harvesting and subsequently characterizing islet function.
Author information
Authors and Affiliations
Contributions
B.O.B., L.J., Y.A. and R.D. were involved in designing the study, acquisition of data, experimental procedures, data analysis and interpretation and wrote and critically reviewed article. V.S.Y.T., J.K. and S.S. were involved in experimental procedures and critically reviewed article.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Accession code: GEO Accession No is GSE97655.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dorajoo, R., Ali, Y., Tay, V.S.Y. et al. Single-cell transcriptomics of East-Asian pancreatic islets cells. Sci Rep 7, 5024 (2017). https://doi.org/10.1038/s41598-017-05266-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-05266-4
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.