Abstract
Although habitat transformation is one of the main causes of biodiversity loss, there are many examples of species successfully occupying and even proliferating in highly human-modified habitats such are the cities. Thus, there is an increasing interest in understanding the drivers favoring urban life for some species. Here, we show how the low richness and abundance of predators in urban areas may explain changes in the habitat selection pattern of a grassland specialist species, the burrowing owl Athene cunicularia, toward urban habitats. Predation release improves the demographic parameters of urban individuals, thus favoring an increment in the breeding density of the species in urban areas that accounts for the apparent positive selection of this habitat in detriment of the more natural ones that are avoided. These results suggest that traditional habitat selection analyses do not necessarily describe habitat choice decisions actively taken by individuals but differences in their demographic prospects. Moreover, they also highlight that cites, as predator-free refuges, can become key conservation hotspots for some species dependent on threatened habitats such as the temperate grasslands of South America.
Similar content being viewed by others
Introduction
Humans have transformed ecosystem patterns and processes across the biosphere, with a critical terrestrial transition from mostly wild to mostly anthropogenic between 1,700 and 2,0001 that precipitated a major global biodiversity crisis2. However, not all species respond equally to habitat transformations, and there are many examples of species occupying and even proliferating in transformed environments3, 4. In this sense, there is an increasing interest in studying how species colonize and thrive in novel habitats5, 6 as well as understanding the drivers of change in habitat preferences shown by many species as a response to anthropogenic habitat changes.
Changes in habitat selection processes may be involved in the preferential use or avoidance of new anthropogenic habitats. Habitat selection is defined as the process by which an animal chooses between habitats and is usually measured by the use of a particular habitat relative to its availability7, 8. This last point is important, as habitat availability can affect the strength of habitat selection, so when a habitat becomes rare, selection could be stronger compared to when it is common9, 10. For species that may utilize a wide range of habitats, quantifying habitat selection is particularly challenging, spatial and temporal changes in resource abundance or ecological pressures can result in changes in selection preference affecting the robustness of habitat selection models. Thus, understanding the complex, dynamic interaction between species occurrence and habitat patterns is essential to predict and manage responses of species to natural or anthropogenic environmental changes.
Besides critical resources such as food and breeding sites7, 11, predation risk may be an important determinant during the habitat selection process12,13,14. Presence and abundance of predators can change across habitat types15 so that variability in predation risk creates heterogeneous “landscapes of fear” that could be important during the habitat selection process16. In this sense, because predators tend to be more sensitive than prey to habitat changes and human disturbances, some species might benefit (via release from predation) from habitat transformations, thus positively selecting human-modified environments17,18,19,20.
In this paper, we investigated breeding habitat selection in the burrowing owl (Athene cunicularia), a species typically associated to North and South American grasslands that can also breed in habitats with different degrees of transformation, including cities21,22,23. In particular, we were interested in understanding differences in the occupation of urban and rural habitats. Therefore, we assessed whether the abundance of breeding pairs in each habitat was proportional to its availability or, conversely, if habitats are differentially occupied, through a habitat selection approach. As a potential driver of habitat selection, we evaluated differences in predator richness and abundance between both habitat types. Our results show a positive selection of urban sites over the much more extended natural and rural habitats. The abundance and richness of predators was much lower in urban areas, which can explain the higher demographic parameters and breeding densities of burrowing owls in the city. These results suggest that predation pressure through top-down regulatory processes could be one important driver of the population bloom of some avian urban populations. Additionally, they highlight the role of cities as conservation hotspots for some species that are now thriving better in urban areas than in their increasingly threatened natural habitats.
Results
Distribution of breeding pairs between habitats and breeding densities
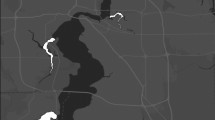
We recorded 1,749 active nests of burrowing owls distributed across the years (2008–2013) and habitats present in the study area (Fig. 1). Most of the rural nests were located in grazed habitat plots (grazed grasslands and regenerating grazed grasslands, n = 273 and 201, respectively), being scarcer in more natural areas (natural grasslands and regenerating grasslands, n = 94 and 86, respectively) and crops (n = 61). The rural area represented on average 81% of the total surveyed area (322.77 km2), and was mainly covered by natural grasslands (41%) and crops (21%), with less relevance of the other habitat types (<14%). On the other hand, 432 nests were located in the urban area during the study period (Fig. 1) over a surface of 74 km2 (29% of the total surveyed area).
Study area showing urban and rural nests (grey and white dots, respectively) of burrowing owls. Grey lines represent transects used to assess availability of rural habitats. Note that many dots overlap. Map was generated with Q-GIS 2.8.1 (http://qgis.osgeo.org).
Savage selectivity indexes indicated that burrowing owls negatively selected natural grasslands and crops and positively selected grazed grasslands, with other rural habitats showing less consistent patterns across years (Fig. 2). Overall, owls preferentially used urban over rural habitats (Savage selectivity indexes: Wurban = 2.46; Wrural = 0.67; Fig. 3A), a pattern that remained highly consistent along years (Figure S1). Accordingly, urban landscapes showed average densities 7 times higher than rural ones (rural density: 0.52 ± 0.05 breeding pairs/km2; urban density: 3.64 ± 0.83 breeding pairs/km2, Fig. 3B).
(A) Habitat availability and use, (B) breeding densities (number of breeding pairs/km2) of burrowing owls and (C) abundance (individuals/h) and richness (species/h) of potential predators in urban and rural habitats. Savage selectivity indexes are shown above the bars in A (***p < 0.0001). Dots represent mean values while bars depict SE.
Abundance and richness of predators
We recorded 1,137 potential predators of burrowing owls during 2,073 h of fieldwork, 966 predators during 1,158 h in the rural areas and 171 predators during 915 h in the urban areas. The abundance of predators differed highly between urban and rural landscapes (Table S1).
The average abundance of predators was near six times higher in rural areas (1.84 ± 0.37 indiv/h) compared to urban habitats (0.27 ± 0.05 indiv/h, F = 157.94, p < 0.0001; Fig. 3C). Predator richness was also higher in the rural areas (1.50 ± 0.07 species/h) than in the urban areas (0.46 ± 0.04 species/h; χ2 = 117.96, p < 0.001; Fig. 3C). In both cases, the duration of the fieldwork session and year of sampling were retained in models (all p < 0.0145). This pattern remained constant when both terrestrial and aerial predators were separately analyzed (Table S2).
Breeding performance
We monitored 3,161 breeding events from 2007 to 2014 in both rural (n = 1,813) and urban (n = 1,348) habitats. Breeding failure affected 27% of the nests, being higher in rural habitats (45%) than in urban ones (27%, Yate’s corrected χ2: p < 0.0001). Although some nests were lost after intense rain, house building or land management, predation was the main known cause of breeding failure both in urban (83%, n = 64) and rural nests (80%, n = 117). Extrapolating these proportions to the entire urban and rural unsuccessful nests, we estimated that 22% of urban and 36% of rural territories were predated (Yate’s corrected χ2: p < 0.0001).
Differential predation risk among habitats seemed to translate into higher reproductive parameters in urban rather than in rural pairs (breeding success: urban: 73%, rural: 55%; χ2 = 122.98, p < 0.0001; brood size: urban: 2.85 ± 0.04; rural: 2.76 ± 0.04; F = 4.70, p = 0.0303; productivity: urban: 2.08 ± 0.04; rural: 1.50 ± 0.04; χ2 = 79.56, p < 0.0001), after controlling for inter-annual variability in all breeding parameters (all p < 0.05).
Discussion
Burrowing owls are open area specialists, foraging and breeding in short, low-density vegetation, mainly grasslands. As this is one of the most endangered habitats of the world due to agricultural and urban development, many burrowing owl populations are declining, and the species is protected in USA, Canada and Mexico24, 25. Our results, however, suggest that some burrowing owl populations are able to exploit human-made habitats, as shown by the burrowing owl’s preference of urban over rural areas (natural grasslands and low-intensive agro-pastoral lands) consistently shown in our study area over years. Differences in predation risk, as measured by the abundance and richness of predators, may explain why breeding density was seven times higher in urban than in rural areas. A habitat selection pattern related to predator risk was also detected within the rural habitats, where owls consistently avoided the most natural habitats, selecting areas highly used for livestock grazing. These results are in agreement with other studies where owls select habitats with short, sparse vegetation, avoiding habitats with high, dense pastures26. Livestock grazing keeps the vegetation short, creating an open habitat that can allow for early detection of predators as traditionally did fossorial mammals such as prairie dogs (Cynomis sp) or plain viscachas (Lagostomus maximus) in North and South America, respectively. Overall, predation may act as the main ecological force driving an unexpected pattern of habitat selection, where burrowing owls seem to prefer the most human-modified habitats (urban > grazed grasslands > natural grasslands).
Habitat selection is usually considered as a consequence of animals actively selecting where to live, reflecting behaviours mechanistically linked to movement27. However, abiotic and biotic processes can create heterogeneity in the distribution of ecological conditions (e.g., microclimates, food, vegetation cover) that can translate into individual’s fitness28, 29, so apparent habitat selection patterns can passively arise as a consequence of differences in the ability of a species to persist or even proliferate in certain habitat types instead of in others. Predators can control prey populations by the lethal removal of individuals, creating a top-down regulatory mechanism commonly invoked to explain the composition and dynamics of natural populations30, 31. However, the actual impact of this consumptive effect on population abundances is controversial (e.g., refs 30,31,32,33,34,35), mainly because the heterogeneity in predation pressure across landscapes can affect its relative importance in front of other regulatory mechanisms such as bottom-up or parallel ones16. The “landscape-of-fear” model36, 37 has helped to understand these inconsistencies, showing that the proportion of risky to safe habitats across a landscape can affect the relative importance of regulatory mechanisms, both top-down but also bottom-up, in determining the absolute population density of a species16. Here, we show how predation can have direct impacts on rural birds by reducing their breeding success and, therefore, their productivity. Although also lower among rural birds, brood size was less affected, suggesting that the low reproductive performance in rural habitats is not because of differences in food resource availability, as seems to happen among some passerines38 but mainly as a consequence of predation that removes entire clutches or broods39. Previous work suggests that predation can also contribute to reducing survival rates of these individuals, as shown by the lower annual survival estimates obtained for rural compared with urban burrowing owls (φurban = 0.59; φrural = 0.2540). Altogether, higher survival and reproductive output in urban than in rural habitats paralleling differences in predation pressure suggest that the apparent switch of the species to prefer urban over natural habitats can arise as a passive consequence of the differential demography of urban and rural subpopulations rather than through active habitat selection processes at the individual level. This idea is supported by the short natal and breeding dispersal distances shown by these burrowing owls (medians 0.30 and 0.02 km respectively, Authors in prep.), so individuals living close to the city could prospect and freely choice among different rural and urban habitats, but the urban choice would be hard for individuals living farther (Fig. 1).
Although evidence is not conclusive41,42,43,44, urban areas have been considered as refuges from predators for wildlife39, 45. The “predator refuge hypothesis” states that urban species thrive because they are released from predation pressures that they would ordinarily face in the wild. However, people present in urban sites can be perceived as potential predators, regardless of whether or not they pose a genuine threat to animals46,47,48, thus contributing to increase the heterogeneity in the landscapes of fear49, 50. Fear of humans has been largely overlooked as a behavioural trait precluding the colonization of urban environments. However, recent work has shown that only tame individuals from species showing high intraspecific variability in fear of humans could colonize urban areas22. This results in significantly lower flight initiation distances among urban individuals compared with rural ones22, 51 without suffering the costs, measured as long-term stress, of living in close contact with humans40. Therefore, only species with individuals able to colonize and thrive in urban areas could benefit from the predator release effect through an improvement of their fitness parameters. More research is however needed to properly understand the ecological and evolutionary consequences of the different demographic dynamics between urban and rural populations of urban dwellers, and the roles played by dispersal as a potential force that homogenizes or, contrarily, reinforces the differences between them52.
Cities as conservation hotspots for native species
By 2030, approximately 5 billion of the world’s 8 billion residents will live in urbanized areas, with projections of nearly 6 million square kilometers of land converted to urban areas. As both the number of people and the area of developed land on Earth increase, so does the potential for negative effects on biodiversity2. Threats to biodiversity come from direct land conversion and from increased colonization by introduced species, that result in a reduction on the number of native species present in urban areas. Although random processes account for part of this species loss, urbanization causes the simplification and homogenization of avian communities through extinctions53, with endemic and threatened species being especially affected54. However, in many cases, urbanization also leads to a reduction of predators55. Cites can thus emerge as predator-free refuges where species able to colonize urbanized habitats by individuals with heritable tolerance to humans22, 40, 56 can thrive and flourish. Larger population densities or abundances in urban than in rural habitats have been also recorded for other species22, 45, suggesting a significant role of predation release in the success of urban dwellers39, 57.
Cities can thus become key conservation hotspots for species that, unexpectedly as it is our case, now perform better there than in their currently threatened natural habitats. For example, Australian cities actually support substantially more nationally threatened animal and plant species than non-urban areas58 and 22% of the occurrences of endangered US plant populations are located in the 40 largest metropolitan areas59. Although in both cases authors argue that cities are relatively young and may be carrying extinction debts, our results support that, at least in some cases, this urban life can actually be the consequence of a successful occupation of this new habitat. Therefore, while urbanization should be considered a threatening factor for many species, it should be also viewed as critical conservation areas for others. In this sense, wildlife conservation should be integrated into urban planning through different strategies, such as the creation of green corridors or the maintenance of open spaces, depending on the species considered. The potential value of gardens for enhancing biodiversity has long been recognised60, and public ‘gardenwatch’ initiatives, such as the Big Garden Birdwatch promoted by Royal Society for the Protection of Birds (http://www.rspb.org.uk/birdwatch/) and the British Trust for Ornithology Garden BirdWatch (http://www.bto.org/gbw/) in the UK, and Project FeederWatch in the USA and Canada (http://www.birds.cornell.edu/pfw/), underline the importance of gardens for raising awareness about biodiversity and the public understanding of science. This ‘citizen science’ movement has huge potential for enhancing urban environments by coordinating public management actions to produce cumulative positive impacts on biodiversity61. However, a more effective ‘citizen urban conservation science’ can be boosted by fuelling people with scientific information showing which species have major conservation problems in nature54, thus requiring focused conservation efforts in cities, and which are the drivers of successful urban life for some species, as shown here, to manage them.
The relative role of predator release as a driver of successful urban life may vary between and even within cities, to the point that the rarefaction of predators may favor some species in some urban areas while local increases in predator numbers can lead to decreases in other urban populations of the same species through impacts on their survivorship or reproduction62. In some areas, mesopredators such as coyotes, foxes, and hawks are abundant in the surrounding of urban habitats. Others, including introduced and domestic species such as cats, increase proportionally with human density. For example, free-ranging domestic cats can kill 1.3–4.0 billion birds and 6.3–22.3 billion mammals annually in USA, un-owned cats causing the majority of this mortality63. However, it has been shown that some prey populations actually have greater survival rates in urban areas, and hypotheses proposed to explain this paradox are (1) the large quantities of food often available in cities to wildlife, (2) the hyper-abundance of co-existing prey populations in urban habitats, thus causing predator satiation, (3) the high specialization of predators on particular prey species, reducing the predation pressure on other prey populations, (4) the different predator species living in cities (species present in urban areas have a lower predator pressure in both urban and rural sites compared with species not present), which exert less pressure on prey populations64, 65, and (5) the prey composition that also changes in urban landscapes (so most of the species that remain in urban habitats are well-adapted to urban predators)66. In any case, efforts to reduce the impact of some predators of no conservation concern, such as domestic cat, can help to improve the prospects of urban biodiversity.
Although predation release is an important determinant of successful city life for some species in certain areas, other factors should be also taken into account to understand and preserve urban biodiversity. Recently, Beninde et al.67 suggest that increasing the area of habitat patches and creating a network of corridors is the most important strategy to maintain high levels of urban biodiversity. However, authors recognize that this result does not take into account the potential increments on invasive species, which presence has been shown to be negative for some native species of conservation concern68. Moreover, habitat improvements can also increase the presence of predators in cities, thus potentially increasing predation risk for some species. Thus, future research should assess the relative role of all factors involved in allowing urban life in different taxa. Importantly, these studies should be performed not using traditional richness or diversity indexes but focusing in the conservation ‘quality’ of species if we really want to give cities a true role in the conservation of biodiversity.
Methods
Study area and habitats
The study was carried out in an area of ca. 6.200 km2, comprising the city of Bahía Blanca and its surrounding rural areas (38°43′S 62°16′W; Buenos Aires, Argentina; Fig. 1). This relatively young city (founded in 1828) was a small village until the middle of the twentieth century, reaching 300,000 inhabitants in 2010. The city is located in the semi-arid Pampas, a large area consisting of natural grasslands interspersed in a matrix of transformed lands dedicated to low-intensive agriculture and livestock grazing. This ecoregion is actually considered as endangered and is regarded as a high priority conservation area at the regional scale69.
Some burrowing owls occupy the residential areas surrounding the core of the city. There, urban pairs excavate their own nests in private and public gardens, spaces between houses, street curbs and along large avenues, usually within 10–100 m of inhabited buildings23. Rural pairs breed in the surrounding expanses of natural grasslands and pastures devoted to wide-ranging livestock and low-intensive cereal crops, where owls also excavate their own nests but can also occupy burrows from fossorial mammals70. The city is immediately surrounded by large and flat rural extensions, with no clear interface between urban and rural habitats51. Unpaved roads cross these rural areas with very low pedestrian and vehicle traffic22, which allow farmers and rangers to access their low-intensive managed lands. These private lands are fenced to avoid the free movement of livestock. Therefore the rural landscape is composed of a mosaic of squared fenced plots (averaging 1 km2) that are subject to different management regimes, namely: (1) natural grasslands (grasslands approaching the pristine ecosystem of the region), (2) grazed grasslands (natural grasslands exposed to livestock grazing), (3) active crops (mostly wheat), (4) regenerating grasslands (abandoned crops with advanced regeneration of grasslands), and (5) regenerating grazed grasslands (as above, but grazed by livestock). Management of these habitat plots rotates and thus their relative surface varies across years. Other natural habitats (dry forests and scrubs, and salt lakes) are very marginal and are never used for breeding by burrowing owls. Burrowing owls have small home-ranges, with most of the activity restricted to the surroundings of their nests along the annual cycle. Although we have no detailed data on movements obtained from radio-tagged individuals, our observations of hundreds of banded individuals across ten years23, 40, 51, 56 indicate that birds stay most of the time close to the nests during both the breeding season (from the mating to the chick fledgling stages), for mating, resting, hunting, guarding and raising young, moving away only for short periods of time that restrict the possibility of performing large movements, and the non-breeding season. Accordingly, radio-tracked burrowing owls in Florida, where the population of the species is also sedentary, show that most of the movements of the individuals took place in the immediate surroundings of the nests, typically within a radius of 50 m71. Therefore, even when we actually measured breeding habitat selection, our study model is not similar to the situation observed in other raptors daily moving up to tens of kilometres for hunting during the breeding season or thousands of kilometres for reaching their breeding quarters, and thus our approach can be considered as an integrate habitat selection study.
Location of breeding pairs
The study area was repeatedly visited –almost daily– across six breeding seasons (November-January of 2008–2013) to locate breeding pairs and geo-reference their active nests. Nests are easily located since breeders show diurnal activity and usually perch in the entrance of their burrows or on nearby small bushes and fences.
The discrete extension of urban areas allowed us to yearly conduct a complete census of the breeding pairs by driving a car through the streets and on foot. Censusing the whole surrounding rural area was however logistically impractical, so we relied on the survey of a number of unpaved roads, distributed across rural areas to cover the whole spatial variability (Fig. 1), to locate breeding pairs and estimate breeding densities, as well as habitat use and availability. We repeatedly drove a car at low speeds (20–30 km/h) through the selected roads, with a driver and at least one observer (e.g. ref. 15), and stopped every time we observed a new owl in order to locate its nest.
Habitat availability
Habitat availability slightly changes annually and thus was separately evaluated for each breeding season from 2008 to 2013, with the exception of 2011 due to logistic constraints. Habitat availability in rural areas was estimated by recording the number of each habitat patch on both sides of the unpaved roads. Thus, we measured habitat availability in real time and at the same spatial scale in which habitat use is estimated, assuming that, in large sample sizes, plot size approaches the mean size of all plots evaluated72, 73. An average plot length (i.e., the length of the plot in contact with the road) was estimated by measuring it in 60 randomly selected fenced plots across the study area using aerial photographs. Plot width (i.e., perpendicular distance from the road) was estimated using the distance D within which we located 99% of the nests, that varied among habitats depending on vegetation cover and height (natural grasslands: 628 m, grazed grasslands: 618 m, crops: 468 m, regenerating grasslands: 252 m, regenerating grazed grasslands: 664 m). The total availability of rural habitat was therefore obtained as \(\Sigma \bar{L}\ast {n}_{i}\ast {D}_{i}\), where ni was the number of plots of each habitat i, while Di and \(\bar{L}\) were their width and mean length, respectively.
The availability of urban habitat was estimated by measuring the surface of peripheral urbanized areas (i.e., residential neighborhoods with scattered houses and open gardens where the species can breed, excluding the fully paved core of the city; Fig. 1) in aerial photographs using Q-GIS. We assumed full detectability of breeding owls and nests in this habitat given the high visibility of individuals from the dense street network.
Breeding habitat selection and density estimation
We assessed whether birds used the different habitat types for breeding in proportion to their availability using the Savage selectivity index W = Ui/ pi, where Ui is the proportion of breeding pairs recorded in a given habitat and pi is the proportion of that habitat against total available habitat74. The ratio theoretically varies between 0 (full avoidance) and ∞ (full preference). A value of 1 indicates random habitat use, i.e. that the proportion used equals the proportion available74. The statistical significance of this index was obtained by comparing the statistic (wi − 1)2/sewi 2 with the corresponding critical value of a χ2 distribution with one degree of freedom, the null hypothesis being that birds use habitats in proportion to their availability. The standard error of the index (sewi) was calculated as √[(1 − pi)/(u+ *pi)], where u+ is the total number of breeding pairs recorded. Statistical significance was obtained after applying the Bonferroni correction for multiple tests. Breeding density in rural and urban habitats was annually calculated as the number of active nests divided by the measured surface of each habitat.
Predator surveys
Potential and confirmed predators of burrowing owls include mammals, reptiles and birds. While aerial predators can prey on adults and juveniles when they are in the surrounding of their burrows, terrestrial predators can get into burrows and prey on eggs, chicks and adults. Therefore, we separately considered the abundance and richness of avian and terrestrial predators given their possible differential predation effects.
Relative indexes of richness (number of species) and abundance of predators were obtained by recording all predators observed during a good proportion of our fieldwork sessions, accounting for the time invested on each one to obtain frequencies of observations per hour75. Although this method underestimates the abundance of nocturnal predators, the relative indexes obtained are valid for comparing different habitats or sites75. Fieldwork sessions usually lasted from one hour before dawn to midday, and from four hours before to one hour after dusk. Species not easily distinguishable in the field such as the large hairy armadillo (Chaetophractus villosus) and Southern long-nosed armadillo (Dasypus hybridus) were pooled as “armadillos”. Similarly, snakes were pooled for analyses. Domestic dogs were not considered as potential predators because they are fed by their owners and do not represent a true predation threat to birds (authors personal observations).
We employed Generalized Linear Models (GLM) for assessing differences in richness and abundance of predators (response variables; binomial negative error or Poisson error distribution, log link function) between urban and rural habitats (fixed factor), fitting time invested on each fieldwork session as a covariate, and year as a fixed effect.
Breeding performance
From 2007 to 2014, we repeatedly visited all located nests to record the number of young that reached fledging age. We assessed whether breeding pairs successfully produced at least one fledgling (breeding success), the brood size at fledging of successful pairs (brood size) and the number of young fledged per breeding attempt (productivity). We tried to ascertain the causes of breeding failure, also recording signs of predation such as the presence of corpses or plucked owl feathers at the entrance of the nests.
Differences in breeding parameters between urban and rural nests were assessed through GLMs (breeding success: binomial error distribution, logistic function; (log)brood size: normal error distribution, identity link function; productivity: negative binomial distribution, log-link function). Habitat (urban or rural) and year were fitted as fixed effects, thus controlling for inter-year variability in breeding performance.
Ethics statements
Field work and procedures were conducted under permits from the Argentinean wildlife agency (22500-4102/09), and the owners of private properties, in accordance with the approved guidelines of the Ethics Committee of CSIC (CEBA-EBD-11-28).
Change history
12 April 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41598-023-32913-w
References
Ellis, E. C., Goldewijk, K., Siebert, S., Lightman, D. & Ramankutty, N. Anthropogenic transformation of the biomes, 1700 to 2000. Global Ecology and Biogeography 19, 589–606 (2010).
Newbold, T. et al. Global effects of land use on local terrestrial biodiversity. Nature 520, 45–50 (2015).
Shochat, E., Warren, P. S., Faeth, S. H., McIntyre, N. E. & Hope, D. From patterns to emerging processes in mechanistic urban ecology. Trends in Ecology and Evolution 21, 186–91 (2006).
Burhans, D. E. & Thompson, F. R. Songbird abundance and parasitism differ between urban and rural shrublands. Ecological Applications 16, 394–405 (2006).
Møller, A. P. et al. Urbanized birds have superior establishment success in novel environments. Oecologia 178, 943–950 (2015).
Sol, D., Lapiedra, O. & González-Lagos, C. Behavioural adjustments for a life in the city. Animal Behavior 85, 1101–1112 (2013).
Clark, R. G. & Shutler, D. Avian Habitat Selection: Pattern from Process in Nest-Site Use by Ducks? Ecology 80, 272–287 (1999).
Jones, J. Habitat selection studies in avian ecology: a critical review habitat selection studies in avian ecology: A critical review. Auk 118, 557–562 (2001).
Osko, T. J., Hiltz, M. N., Hudson, R. J. & Wasel, S. M. Moose habitat preferences in response to changing availability. Journal of Wildlife Management 68, 576–584 (2004).
Godvik, I. M. R. et al. Temporal scales, trade-offs, and functional responses in red deer habitat selection. Ecology 90, 699–710 (2009).
Wolfe, J. D., Johnson, M. D. & Ralph, C. J. Do birds select habitat or food resources? Nearctic-neotropic migrants in northeastern Costa Rica. PLoS One 9, 1–10 (2014).
Fontaine, A. J. J. & Martin, T. E. Habitat selection responses of parents to offspring predation risk: an experimental test. American Naturalist 168, 811–818 (2006).
Forsman, J. T., Mönkkönen, M., Korpimäki, E. & Thomson, R. L. Mammalian nest predator feces as a cue in avian habitat selection decisions. Behavioral Ecology 24, 262–266 (2013).
Ahnesjö, J. & Forsman, A. Differential habitat selection by pygmy grasshopper color morphs; interactive effects of temperature and predator avoidance. Evolutionary Ecology 20, 235–257 (2006).
Carrete, M., Tella, J. L., Blanco, G. & Bertellotti, M. Effects of habitat degradation on the abundance, richness and diversity of raptors across Neotropical biomes. Biological Conservation 142, 2002–2011 (2009).
Laundré, J. W. et al. The landscape of fear: The missing link to understand top-down and bottom-up controls of prey abundance? Ecology 95, 1141–1152 (2014).
Berger, J. Fear, human shields and the redistribution of prey and predators in protected areas. Biology Letters 3, 620–623 (2007).
Leighton, P. A., Horrocks, J. A. & Kramer, D. L. Conservation and the scarecrow effect: Can human activity benefit threatened species by displacing predators? Biology Conservation 143, 2156–2163 (2010).
Muhly, T. B., Semeniuk, C., Massolo, A., Hickman, L. & Musiani, M. Human activity helps prey win the predator-prey space race. PLoS One 6, 1–8 (2011).
Shannon, G., Cordes, L. S., Hardy, A. R., Angeloni, L. M. & Crooks, K. R. Behavioral responses associated with a human-mediated predator shelter. PLoS One 9, e94630 (2014).
Clayton, K. M. & Schmutz, J. K. Is the decline of burrowing owls Speotyto cunicularia in prairie Canada linked to changes in Great Plains ecosystems? Bird Conservation International 9, 163–185 (1999).
Carrete, M. & Tella, J. L. Inter-individual variability in fear of humans and relative brain size of the species are related to contemporary urban invasion in birds. PLoS One 6, e18859 (2011).
Rodríguez-Martínez, S., Carrete, M., Roques, S., Rebolo-Ifrán, N. & Tella, J. L. High urban breeding densities do not disrupt genetic monogamy in a bird species. PLoS One 9, e91314 (2014).
Desmond, M. J., Savidge, J. A. & Eskridge, K. M. Correlations between burrowing owl and black-tailed prairie dog declines: a 7-year analysis. Journal of Wildlife Management 64, 1067–1075 (2000).
Conway, C. J., Garcia, V., Smith, M. D., Ellis, L. A. & Whitney, J. L. Comparative demography of Burrowing Owls in agricultural and urban landscapes in southeastern Washington. Journal of Field Ornithology 77, 280–290 (2006).
Green, G. A. & Anthony, R. G. Nesting success and habitat relationships of Burrowing Owls in the Columba Basin, Oregon. Condor 91, 347–354 (1989).
Moorcroft, P. R. & Barnett, A. Mechanistic home range models and resource selection analysis: a reconciliation and unification. Ecology 89, 1112–1119 (2008).
Sutcliffe, O. L., Thomas, C. D., Yates, T. J. & Greatorex-Davies, J. Correlated extinctions, colonizations and population fluctuations in a highly connected ringlet butterfly metapopulation. Oecologia 109, 235–241 (1997).
Thomas, D. W., Blondel, J., Perret, P., Lambrechts, M. M. & Speakman, J. R. Energetic and fitness costs of mismatching resource supply and demand in seasonally breeding birds. Science 291, 2598–2600 (2001).
Hanski, I. H., Henttonen, H., Korpimäki, E., Oksanen, L. & Turchin, P. Small-rodent dynamics and predation. Ecology 82, 1505–1520 (2001).
Terborgh, J. et al. Ecological Meltdown in Predator-Free Forest Fragments. Science 294, 1923–1926 (2001).
Ballard, W. B., Lutz, D., Keegan, T. W., Carpenter, L. H. & DeVos, J. C. Jr. Deer-predator relationships: a review of recent North American studies with emphasis on mule and black-tailed deer. Wildlife Society Bulletin 29, 99–115 (2001).
Meserve, P. L., Kelt, D. A., Milstead, W. B. & Gutierrez, J. R. Thirteen years of shifting top-down and bottom-up control. Bioscience 53, 633 (2003).
Previtali, M., Lima, M., Meserve, P., Kelt, D. & Gutierrez, J. Population dynamics of two sympatric rodents in a variable environment: rainfall, resource availability, and predation. Ecology 90, 1996–2006 (2009).
Erlinge, S. et al. Predation as a regulating factor on small rodent populations in Southern Sweden. Oikos 40, 36–52 (1983).
Laundré, J. W., Hernández, L. & Altendorf, K. B. Wolves, elk, and bison: reestablishing the “landscape of fear” in Yellowstone National Park, USA. Canadian Journal of Zoology 79, 1401–1409 (2001).
Laundré, J. W., Hernández, L. & Ripple, W. J. The landscape of fear: ecological implications of being afraid. Open Ecology Journal 3, 1–7 (2010).
Chamberlain, D. E. et al. Avian productivity in urban landscapes: a review and meta-analysis. Ibis 151, 1–18 (2009).
Stracey, C. M. & Robinson, S. K. Are urban habitats ecological traps for a native songbird? Season-long productivity, apparent survival, and site fidelity in urban and rural habitats. Journal of Avian Biology 43, 50–60 (2012).
Rebolo-Ifrán, N. et al. Links between fear of humans, stress and survival support a non-random distribution of birds among urban and rural habitats. Scientific Reports 5, 13723 (2015).
Jokimäki, J. & Huhta, E. Artificial nest predation and abundance of birds along an urban gradient. Condor 102, 838–847 (2000).
Thorington, K. K. & Bowman, R. Predation rate on artificial nests increases with human housing density in suburban habitats. Ecography 26, 188–196 (2003).
Gering, J. C. & Blair, R. B. Predation on artificial bird nests along an urban gradient: predatory risk or relaxation in urban environments? Ecography 22, 532–541 (1999).
Haskell, D. G., Knupp, A. M. & Schneider, M. C. Nest predator abundance and urbanization. Pages 243–258 in Marzluff, J.M., Bowman, R., Donnelly, R. editors Avian ecology and conservation in an urbanizing world Kluwer Academic Publishers, Boston (2001).
Møller, A. P. et al. High urban population density of birds reflects their timing of urbanization. Oecologia 170, 867–875 (2012).
Frid, A. & Dill, L. M. Human-caused disturbance stimuli as a form of predation risk. Conservation Ecology 6, 11 (2002).
Beale, C. M. & Monaghan, P. Human disturbance: People as predation-free predators? Journal of Applied Ecology 41, 335–343 (2004).
Valcarcel, A. & Fernández-Juricic, E. Antipredator strategies of house finches: are urban habitats safe spots from predators even when humans are around? Behavioural Ecology and Sociobiology 63, 673–685 (2009).
Ciuti, S. et al. Human selection of elk behavioural traits in a landscape of fear. Proceedings of the Royal Society B 1483, 4407–4416 (2012).
Støen, O. G. et al. Physiological evidence for a human-induced landscape of fear in brown bears (Ursus arctos). Physiology & Behavior 152, 244–248 (2015).
Carrete, M. & Tella, J. L. High individual consistency in fear of humans throughout the adult lifespan of rural and urban burrowing owls. Scientific Reports 3, 3524 (2013).
Edelaar, P. & Bolnick, D. I. Non-random gene flow: an underappreciated force in evolution and ecology. Trends in Ecology and Evolution 27, 659–665 (2012).
Sol, D., González-Lagos, C., Moreira, D., Maspons, J. & Lapiedra, O. Urbanisation tolerance and the loss of avian diversity. Ecology Letters 17, 942–950 (2014).
González-Oreja, J. A. Birds of different biogeographic origins respond in contrasting ways to urbanization. Biological Conservation 144, 234–242 (2010).
Díaz, M. et al. The geography of fear: a latitudinal gradient in anti-predator escape distances of birds across Europe. Plos One 8, e64634 (2013).
Carrete, M. et al. Heritability of fear of humans in urban and rural populations of a bird species. Scientific Reports (2016).
Tella, J. L., Canale, A., Carrete, M., Petracci, P. & Zalba, S. M. Anthropogenic nesting sites allow urban breeding in burrowing parrots Cyanoliseus patagonus. Ardeola 61, 311–321 (2014).
Ives, C. D. et al. Cities are hotspots for threatened species. Global Ecology and Biogeography 25, 117–126 (2016).
Shwartz, A., Turbé, A., Julliard, R., Simon, L. & Prévot, A. C. Outstanding challenges for urban conservation research and action. Global Environmental Change: Human and Policy Dimensions 28, 39–49 (2014).
Goddard, M. A., Dougill, A. J. & Benton, T. G. Scaling up from gardens: biodiversity conservation in urban environments. Trends in Ecology and Evolution 25, 90–98 (2010).
Cooper, C. B., Dickinson, J., Phillips, T. & Bonney, R. Citizen Science as a tool for conservation in residential ecosystems. Ecology and Society 12, 11 (2007).
Rivera-López, A. & MacGregor-Fors, I. Urban predation: a case study assessing artificial nest survival in a neotropical city. Urban Ecosystems 19, 649–655 (2016).
Loss, S. R., Will, T. & Marra, P. P. The impact of free-ranging domestic cats on wildlife of the United States. Nature Communications 4, 1396 (2013).
Stracey, C. M. Resolving the urban nest predator paradox: The role of alternative foods for nest predators. Biological Conservation 144, 1545–1552 (2011).
Sims, V., Evans, K. L., Newson, S. E., Tratalos, J. A. & Gaston, K. J. Avian assemblage structure and domestic cat densities in urban environments. Diversity and Distributions 14, 387–399 (2008).
Fischer, J. D., Cleeton, S. H., Lyons, T. P. & Miller, J. R. Urbanization and the predation paradox: the role of trophic dynamics in structuring vertebrate communities. BioScience 62, 809–818 (2012).
Beninde, J., Veith, M. & Hochkirch, A. Biodiversity in cities needs space: a meta-analysis of factors determining intra-urban biodiversity variation. Ecology Letters 18, 581–592 (2015).
Hernández-Brito, D., Carrete, M., Popa-Lisseanu, A. G., Ibáñez, C. & Tella, J. L. Crowding in the city: losing and winning competitors of an invasive bird. Plos One 9, e100593 (2014).
Dinerstein, E. et al. Conservation assessment of the terrestrial ecoregions of Latin America and the Caribbean The World Wildlife Fund and The World Bank, Washington, Columbia, USA (1995).
Carrete, M. & Tella, J. L. Individual consistency in flight initiation distances in burrowing owls: a new hypothesis on disturbance-induced habitat selection. Biology Letters 6, 167–170 (2010).
Mrykalo, R. The florida burrowing owl in a rural environment: Breeding habitat, dispersal, post-breeding habitat, behavior, and diet. M.S. University of South Florida (2005).
Ursúa, E., Serrano, D. & Tella, J. L. Does land irrigation actually reduce foraging habitat for breeding lesser kestrels? The role of crop types. Biological Conservation 122, 643–648 (2005).
Blanco, G., Tella, J. L. & Torre, I. Traditional farming and key foraging habitats for chough Pyrrhocorax pyrrhocorax in a Spanish pseudosteppe. Journal of Applied Ecology 35, 232–239 (1998).
Manly, B. F. J., McDonald, L. L. & Thomas, D. L. Resource selection by animals: statistical design and analysis for field studies. Chapman and Hall New York (1993).
Vögeli, M., Serrano, D., Pacios, F. & Tella, J. L. The relative importance of patch habitat quality and landscape attributes in a declining steppe-bird metapopulation. Biological Conservation 143, 1057–1067 (2010).
Acknowledgements
We thank N. Lois, S. Briones, M. Santillán, P. Laiolo, M. de la Riva, N. Tella-Carrete, and M. Vázquez for their help in capturing and monitoring owls over the years. Field work was conducted under permits from Argentinean wildlife agencies and the owners of private properties.
Author information
Authors and Affiliations
Contributions
M.C. and J.L.T. conceived the idea. N.R.I., J.L.T. and M.C. conducted field work. N.R.I., J.L.T. and M.C. analyzed the data. N.R.I., J.L.T. and M.C. wrote the paper. N.R.I., J.L.T. and M.C. discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rebolo-Ifrán, N., Tella, J.L. & Carrete, M. Urban conservation hotspots: predation release allows the grassland-specialist burrowing owl to perform better in the city. Sci Rep 7, 3527 (2017). https://doi.org/10.1038/s41598-017-03853-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-03853-z
This article is cited by
-
Predation of two species of Columbidae by the burrowing owl Athene cunicularia (Strigiformes, Strigidae) in urban environment
Ornithology Research (2024)
-
Anthropogenic nesting sites and density of Burrowing Parrot (Cyanoliseus patagonus) in northern Argentinian Patagonia
Revista Chilena de Historia Natural (2023)
-
Intraspecific competition and individual behaviour but not urbanization affect the dietary patterns of a generalist avian predator
Scientific Reports (2023)
-
Breeding performance is explained for coloniality and phenology but not for urbanization in a generalist raptor bird
Urban Ecosystems (2023)
-
Siblicide in the city: the urban heat island accelerates sibling cannibalism in the black widow spider (Latrodectus hesperus)
Urban Ecosystems (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.