Abstract
Differentiated thyroid cancer (DTC) patients with negative serum thyroglobulin (Tg), negative 131I whole–body scintigraphy (131I-WBS) at first post-ablation and progressively increased TgAb level are a relatively rare entity in the follow-up after total thyroidectomy and radioactive iodine therapy. The value of 18F-FDG PET/CT in detecting the recurrence of disease in these patients has only been reported in a small case series. The goal of this study was to investigate the diagnostic accuracy of 18F-FDG PET/CT in detecting recurrent disease in these specific PTC patients and to identify risk factors for patients with positive 18F-FDG PET/CT results. Eighty-two PTC patients who had 18F-FDG PET/CT scans with negative Tg, negative 131I-WBS at first post-ablation and progressively increased TgAb levels were included. We found that the sensitivity, specificity, positive predictive value, negative predictive value, and accuracy of 18F-FDG PET/CT in this patient group were determined as 84%, 72%, 92%, 57% and 82%, respectively. 18F-FDG PET/CT scan had a good diagnostic performance and should be performed routinely in PTC patients with negative Tg, negative 131I-WBS at first postablation and progressively increased TgAb level, especially when span for progressively increased TgAb level ≥ 3 years and/or progressively increased TgAb value up to 150 IU/mL.
Similar content being viewed by others
Introduction
Differentiated thyroid cancer (DTC) is becoming more and more common in the United States between 1975 and 2012, with an estimated 62,450 new cases in 20151. Despite the high overall survival rate and good prognosis, the recurrence rate of DTC is not negligible and ranges from 14% to 23%2, 3. Therefore, detection of persistent or recurrent disease is very important in DTC management and follow-up. 131I whole–body scintigraphy (131I-WBS), measurement of serum thyroglobulin (Tg) level and neck ultrasonography are mainstream approaches for detecting persistent or recurrent disease after total or near-total thyroidectomy and radioiodine remnant ablation4. Serum Tg level is the most sensitive and reliable marker indicating persistent or recurrent disease in the follow-up of DTC because serum Tg only originated from differentiated thyroid cancer cells5.
According to the current American Thyroid Association (ATA) guideline, negative serum Tg, defined as the low serum Tg levels during TSH suppression (Tg < 0.2 ng/mL) or after stimulation (Tg < 1 ng/mL) after total or near-total thyroidectomy and radioiodine remnant ablation, suggests the disease-free status for DTC patients in the follow-up6. However, antithyroglobulin antibody (TgAb) can interfere with the measurement of Tg and reduce the accuracy of Tg as a predictor of DTC activity. In a previous study, TgAb was present in 10–25% of patients with PTC7, 8. Therefore, negative Tg with the presence of positive TgAb could lead to a clinical dilemma in terms of therapeutic decision and follow-up.
Whether or not elevated serum TgAb concentrations can be used as a surrogate marker of persistent or recurrent disease remains controversial9, 10. Some argued that TgAb levels did not predict disease status in DTC because TgAb production primarily arises in coexisting lymphocytic thyroiditis or Graves’ disease in DTC patients11. It has been reported that only the progressively increased TgAb level was useful for predicting clinical recurrence or persistence of Tg-negative patients with PTC12, 13.
18F-fluorodeoxy-D-glucose positron emission tomography/computed tomography (18F-FDG PET/CT) is routinely performed to search for the recurrent or persistent disease in patients with DTC14. However, only a few studies including a small case series evaluated the value of 18F-FDG PET/CT in DTC patients who have negative 131I-WBS, negative Serum Tg, and increased TgAb titer15, 16.
In the present study, we aimed to investigate the diagnostic accuracy of 18F-FDG PET/CT, performed over one year after their first remnant ablation, in detecting recurrent disease of PTC in a relatively large clinical samples of patients with negative Tg, negative 131I-WBS at first postablation and progressively increased TgAb level. Moreover, we also identified the correlation of clinical and pathological factors with positive 18F-FDG PET/CT findings in this specific cohort.
Results
Patient’s characteristics
According to the inclusion and exclusion criteria, eighty-two PTC patients who underwent 18F-FDG PET/CT scans with negative Tg, negative 131I-WBS at first post-ablation and progressively increased TgAb level were confirmed and included. Of them, 58 (71%) PTC patients with serum Tg levels < 0.2 ng/mL (TSH suppression) and 24 (29%) PTC patients with serum Tg levels < 1 ng/mL (TSH stimulation >30 IU/mL) at 6 months after first remnant ablation. Serum Tg levels were always <0.2 ng/mL under TSH suppression for all these patients in the follow-up. The characteristics of the study cohort at diagnosis of recurrent PTC were shown in Table 1.
18F-FDG PET/CT finding
Of 82 patients with 18F-FDG PET/CT findings, 59 (72%) patients had results interpreted as positive and 23 (28%) patients as negative. In 59 cases with positive 18F-FDG PET/CT findings, 54 (91.5%) patients were classified as true-positive confirmed pathologically by surgical specimens (Fig. 1). Neck US was performed in all patients before 18F-FDG PET/CT scan. 39 patients with 51 lymph node metastases were found in the neck, among which 34 patients with 42 lesions were detected by neck US. Two cases with 2 lymph nodes metastases, one case with 1 lymph node metastasis and 6 cases with 7 lymph node metastases were found in pharyngeal space, parotid, and mediastinum, respectively, all of which were detected by contrast-enhanced CT after 18F-FDG PET/CT scanning. 4 patients with 18F-FDG PET lung metastases were diagnosed, all of which were detected and confirmed by chest CT (Table 2). 7 false-positive lesions were found on 18F-FDG PET/CT scan in 5 patients in the neck, 5 FDG-avid lesions in 4 patients and 2 FDG-avid lesions in 1 patient were diagnosed as reactive infection of lymph node and hyperplasia of lymph nodes (Fig. 2). All these patients had suspicious lymph nodes for recurrence on the neck US.
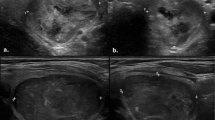
A true-positive lesion on the left neck region was detected by 18F-FDG PET/CT. A 35-year old woman underwent total thyroidectomy with central lymph node dissection on the right neck region and radioiodine ablation for remnant PTC and thyroid tissue. 131I-WBS obtained 5 days after an oral administration of 3.7 GBq of 131I showed negative finding. Six months after ablation, the patient had negative Tg (<0.1 ng/mL) but abnormal TgAb of 108 IU/mL at TSH suppression status. Subsequently, during the follow-up 3.5 years later, TgAb level progressively increased from 108 IU/mL to 623 IU/mL. 18F-FDG PET/CT revealed 18F-FDG-avid nodal lesion with SUVmax of 4.7 in the left neck (a,b and c, crossing line). Surgical pathology confirmed the metastatic nodal lesion from PTC after left neck dissection. The patient had markedly decreased TgAb level afterwards.
A false-positive lesion on the left neck region was revealed by 18F-FDG PET/CT. A 62-year-old woman underwent total thyroidectomy with radical left neck dissection for PTC followed by radioiodine therapy with 3.7 GBq of 131I. Three days later, post-therapy 131I WBS was performed and showed negative results. Six months after ablation. The serum Tg level was 0.18 ng/mL and TgAb level was 46 IU/mL at TSH suppression status. TgAb level was stable for 4.2 yr after 131I therapy. But subsequently, TgAb gradually increased at TSH suppression status. TgAb level elevated from 51 IU/mL to 137 IU/mL in the next follow-up of 2.2 years. 18F-FDG PET/CT demonstrated increased foci radiotracer uptake in the left submandibular region (a,b, crossing line), which was localized by CT image to the left submandibular lymph nodes with SUVmax of 2.6 (c, crossing line). However, infectious lymph node was diagnosed on histopathology after surgery.
In 23 cases with negative 18F-FDG PET/CT findings, 10 patients were interpreted as false-negative, among which 13 lymph node metastases from 8 patients were detected in the neck and 2 lymph node metastases from the remaining 2 patients were found in the mediastinum. All the 10 patients with false-negative results received 131I treatment once again. 2 cases with 2 neck lymph node metastases and one case with 1 mediastinal lymph node metastasis were diagnosed by 131I-WBS combined with 131I-SPECT/CT after 131I treatment. The remaining 7 patients with 10 lesions who had negative 131I-WBS results were confirmed by surgical pathology. All these patients had suspicious lymph node recurrence on the neck US or contrast-enhanced CT of chest (Table 3).
Of 13 true-negative patients, neck US finding was suspicious for recurrence in 7 patients with 12 lesions, but disease recurrence was not detected by surgical excision. The remaining 6 patients underwent 131I treatment once again and negative findings were shown on the 131I-WBS, and recurrent diseases were not be detected by US neck and chest contrast-enhanced CT in these 6 patients. In addition, bone metastases from PTC weren’t detected on the 99mTc-bone scan, but TgAb levels were gradually rising in the follow-up. These 6 patients were also classified as true-negative.
Factors influencing positive 18F-FDG PET/CT results
Patient age, sex, subtypes of PTC, tumor size, extrathyroid extension, bilateral tumor, multifocal tumor, whether patient with neck lateral dissection at initial surgery, N stage, whether pathology with lymphocytic thyroiditis not significantly associated with positive 18F-FDG PET/CT results (P > 0.05). TgAb level at diagnosis and span for progressively increased TgAb level were statistically significant in predicting positive 18F-FDG PET/CT findings (P < 0.05). Compared with TgAb level <150 IU/mL at diagnosis and span for progressively increased TgAb level less than 3 years, univariate regression analysis showed that OR value of TgAb level ≥ 150 IU/mL at diagnosis and span for progressively increased TgAb level longer than 3 years were as much as 4.18 [CI:1.52–11.54] and 3.60 [CI:1.24–10.41] times for progressively increased TgAb level (Table 4).
Diagnostic accuracy of 18F-FDG PET/CT scans
In all these patients, the true-positive, false-positive and false-negative, true-negative cases of 18F-FDG PET/CT findings were 54, 5, 10, and 13, respectively. The sensitivity, specificity, positive predictive value, negative predictive value, and accuracy of 18F-FDG PET/CT in this patient group were determined as 84%, 72%, 92%, 57% and 82%, respectively (Table 5).
When comparing different TgAb levels at diagnosis, the sensitivity, specificity, positive predictive value, negative predictive value, and accuracy of 18F-FDG PET/CT for patients whose TgAb levels ≥ 150 IU/mL at diagnosis is higher than for those whose TgAb levels < 150 IU/mL at diagnosis. The sensitivity, specificity, positive predictive value, negative predictive value, and accuracy increased from 62% to 95%, from 67% to 78%, from 81% to 95%, from 43% to 78% and from 63% to 94% respectively (Table 5). When comparing different span for progressively increased TgAb level, the sensitivity, specificity, positive predictive value, and accuracy of 18F-FDG PET/CT for span for progressively increased TgAb level longer than 3 years at diagnosis were superior to that less than 3 years at diagnosis, the sensitivity, specificity, positive predictive value, and accuracy of 18F-FDG PET/CT scan increased from 76% to 91%, from 71% to 75%, from 85% to 97%, from 74% to 90%. However, the negative predictive value of 18F-FDG PET/CT for patients whose span for progressively increased TgAb level ≥ 3 years at diagnosis was inferior to that <3 years at diagnosis. The negative predictive value decreases from 59% to 50% (Table 5).
Discussion
Our study demonstrated that 18F-FDG PET/CT was a useful method for detecting recurrent disease in PTC patients with negative Tg, negative 131I-WBS at first post ablation and progressively increased TgAb level. In the current study, the sensitivity, specificity, positive predictive value, negative predictive value, and accuracy of 18F-FDG PET/CT for this patient group were confirmed as 84%, 72%, 92%, 57% and 82%, respectively. The diagnostic performance of 18F-FDG PET/CT scanning for detecting the recurrent thyroid cancer with negative Tg, negative 131I-WBS and increased TgAb has been reported several retrospective studies16,17,18,19,20,21 (Table 6). The sensitivity, specificity, positive predictive value, negative predictive value, and accuracy of 18F-FDG PET/CT examination for these patients ranged from 75% to 100%, from 50% to 100%, from 50% to 100%, from 50% to 100%, from 25% to 100% and from 72.7% to 88.4%. The difference within these several studied may reflect the heterogeneity of the number and patients included, definition of Tg negativity, selection criteria for TgAb level, follow-up time, specific 18F-FDG PET/CT technique used, or the reference standard against which the accuracy of 18F-FDG PET/CT scan were analyzed. Among them, five articles have a small number of cases (16, 18–21), so the results were very easy to produce deviation. The largest 18F-FDG PET/CT series to date was the retrospective study by Asa et al. and included 40 DTC patients, the sensitivity, specificity, positive predictive value, negative predictive value, and accuracy in this large study were 78.5%, 50%, 91.6%, 25% and 75%21, all of which lower than those reported by ours. This difference may reflect possibly definition of Tg negativity, selection criteria bias of TgAb and follow-up time. In our study, Negative serum Tg was defined as Tg < 0.2 ng/mL (TSH suppression) or Tg < 1 ng/mL (after stimulation) at 6 months after the first 131I remnant ablation, while Asa et al. considered that the PTC patients had negative serum Tg level as Tg ≤ 1 ng/mL (TSH suppression) or Tg ≤ 2 ng/mL (after stimulation) in the follow-up period after total-near total thyroidectomy and 131I ablation21. If DTC patients with coexistent clinical Hashimoto thyroiditis Graves’ disease, or focal autoimmune thyroiditis, all TgAb disappeared more slowly and the median disappearance time was 3 years for TgAb after total thyroidectomy and radioiodine ablation22. Therefore, increased TgAb level without upward trend in a short follow-up time might not be viewed as persistent or recurrent diseases of DTC. In our cases, increased TgAb level without upward trend has been excluded. Although Asa et al. selected the TgAb standard for persistently/progressive increased TgAb, whether the content of the article including increased TgAb level without upward trend was unclear21. Otherwise, it was reported that the TgAb levels measured 6–12 months after ablation therapy were significantly rising in the DTC patients with residual disease compared to those with no residual disease23. Whether this situation for DTC patients ruled out was indeterminate for the study by Asa et al. because DTC patients in their groups have relatively short follow-up times (9–36 months).
In our study, of 59 cases with positive 18F-FDG PET/CT finding, 54 (91.5%) patients were classified as true-positive confirmed pathologically by surgical resection and 5 patients were diagnosed for false-positive on 18F-FDG PET/CT scans. False-positive 18F-FDG uptake in the neck was often caused by several sources including muscle, brown fat, salivary glands, vocal cords, tonsils, and other lymphoid tissues. Moreover, reactive hyperplasia lesions, inflammatory lesions and benign tumors can also lead to FDG uptake24. All false-positive uptakes were located in the neck, which was similar to what Ozkan et al. reported16. Ozkan et al. considered that it was difficult to distinguish recurrent lesions from false-positive lesions using SUVmax ≥ 2.5 t in the neck region because of overlapping SUVs between them16. Therefore, the criterion for positive lesion was accepted as 18F-FDG uptake greater than that of the normal surrounding tissue or when the SUVmax was ≥ 2.5 in our study.
With respect to distant metastases from included PTC patients, only 4 cases with lung metastases were detected by 18F-FDG PET/CT scan, all of which showed that PTC patients with negative Tg, negative 131I-WBS and progressively increased TgAb level weren’t prone to distant metastases. It was very possible because distant metastases of DTC produced more serum Tg which couldn’t be completely interfere by TgAb25. All of them received additional 131I therapy and showed negative post therapy 131I-WBS scan results.
Of 23 cases with negative 18F-FDG PET/CT finding, 10 patients were interpreted as false-negative. Suspicious recurrent lymph node metastases were detected on the neck, which may suggest that negative 18F-FDG PET/CT finding may represent a small or well-differentiated metastatic lesions26. Subsequently, these patients underwent empirical 131I therapy using 150 mCi. After 3–5 days, 3 patients with metastatic lesions were detected and confirmed by 131I-WBS combined with 131I-SPECT/CT. The remaining 7 patients showed negative 131I-WBS and recurrent lesions were confirmed pathologically by surgical resection. In the current study, there are also 13 true-negative patients with progressively increased TgAb level, while 7 out of these patients were surgically confirmed as disease free, and the remaining 6 cases were confirmed by follow-up.
With regard to true-negative and false-positive finding in these patients, it was not uncertain whether a few other reasons made serum TgAb rise continuously except for the recurrent diseases of PTC, for that a few other diseases could lead to the increased TgAb, such as type 1 diabetes, rheumatoid arthritis, pernicious anemia, collagen vascular diseases, scleroderma, chronic urticarial, autoimmune hyperthyroidism and increasing age in healthy women has been reported22, 27,28,29,30.
In our series, univariate analysis revealed that different TgAb level at diagnosis and span for progressively increased TgAb level was related to the positive 18F-FDG PET/CT finding. The results showed that the sensitivity, specificity of 18F-FDG PET/CT when TgAb level ≥ 150 IU/mL at diagnosis and span for progressively increased TgAb level ≥ 3 years were clearly higher than that when TgAb level < 150 IU/mL at diagnosis and span for progressively increased TgAb level 3 years, respectively. In previous studies, a varying prevalence of TgAb value for predicting persistent or recurrent of DTC after total thyroidectomy has been reported. Chung et al. considered a serum TgAb level below 100 U/mL as negative, and found elevated TgAb levels in 22.6% of DTC patients after 131I ablation17. Seo et al. reported that recurrence for DTC was more frequent in patients who showed a persistently elevated TgAb level over 140 U/mL31. While other studied have defined TgAb level of 6–100 U/mL as positive32, 33. In our study, different TgAb levels were used as cutoff values to evaluate the diagnostic performances of 18F-FDG PET/CT scan in the detection of recurrent PTC. 18F-FDG PET/CT results could be affected by lesion size, false-positive finding and degree of differentiation of PTC and so on, therefore, above research results for TgAb level may be not related to the positive 18F-FDG PET/CT finding. Our study also showed that longer span for progressively increased TgAb level (≥3 years) rather than shorter pan for progressively increased TgAb level (<3 years) indicated a higher sensitivity, specificity of 18F-FDG PET/CT scanning in detecting recurrent PTC, suggesting the recurrent diseases of these PTC patients developed more slowly and have a relatively good prognosis.
However, several limitations of this study should be discussed. First, of 13 true negative patients, 6 patients weren’t confirmed having metastases by pathology. These cases were classified as true negative through follow-up. In the course of follow-up, these 6 patients showed negative findings on the post-therapy 131I-WBS, neck US and contrast-enhanced chest CT, 99mTc-MDP bone scan, but some occult lesions may still not be found. Second, Of 10 false-negative patients, 3 patients were confirmed by 131I-WBS combined with 131I-SPCET/CT rather than the results of pathology, because 131I-SPECT/CT effectively excluded residual noncancerous thyroid tissue located outside the thyroid bed (substernal goiter or ectopic foci along the thyroglossal duct), physiologic uptake in non-thyroidal tissues, and contamination34, 35. The retrospective nature of the data in the present study may be another limitation.
Conclusions
Our study demonstrated that the 18F-FDG PET/CT scanning had a good diagnostic performance in the selected PTC patients with negative Tg, negative 131I-WBS at first postablation and progressively increased TgAb level. Span for progressively increasing TgAb level and TgAb level at diagnosis were closely associated with positive 18F-FDG PET/CT findings. Therefore, 18F-FDG PET/CT scanning could be performed routinely for PTC patients with negative Tg, negative 131I-WBS at first postablation ablation and progressively increased TgAb level, especially for those whose span for progressively increased TgAb level ≥3 years and/or progressively increased TgAb value up to 150 IU/mL.
Patients and Methods
Patients
This retrospective study was approved by our institutional review board. Informed consents have been waived for most patients except for two patients, whose SPECT/CT images were used in the current study. All methods were performed in accordance with the relevant guidelines and regulations. Files of consecutive 7843 patients treated with 131I between January 2005 and January 2014 were reviewed. Clinical follow-up data of 1257 patients with DTC who underwent 18F-FDG PET/CT scanning were evaluated retrospectively. The inclusion criteria were as follows: (1) patients with histologically proven PTC. (2) patients with PTC treated with total or near-total thyroidectomy and postoperative 131I ablation. (3) postablation negative 131I-WBS defined by the absence of non-physiological 131I uptake outside the thyroid bed or abnormal 131I uptake confirmed for physiological uptake or contamination by 131I single photon emission computed tomography/computed tomography (131I-SPECT/CT) outside the thyroid bed. (4) negative Tg defined as Tg < 0.2 ng/mL (TSH suppression) or Tg < 1 ng/mL (after stimulation) 6 months after the first remnant ablation. (5) progressively increased TgAb level including TgAb which persistently rose or TgAb which kept stable/decreased for some time but subsequently rose after remnant ablation. (6) 18F-FDG PET/CT was performed more than 1 year after the first remnant ablation. The exclusion criteria were as follows: (1) Tg ≥ 0.2 ng/mL (TSH suppression) or ≥1 ng/mL (after stimulation) in the follow-up. (2) persistently high TgAb but had no rising trend (3) a temporarily increased TgAb at 6–12 months after ablation therapy.
131I empiric treatment
After surgery, each patient received an ablative dose of 131I and was put on a low iodine diet for 3–4 weeks before 131I therapy (TSH reached 30 mIU/L). Subsequently, the patients were subjected to oral administration of 131I after the following conventional measurements, including FT3, FT4, TSH, Tg, TgAb, neck ultrasonography (US), and CT scans. The dose of oral standard administration of 3.7GBq (100 mCi) of 131I was used to ablate the thyroid remnants. 131I-WBS and/or 131I-SPECT/CT fusion imaging was performed 3–5 days after 131I oral administration. 131I-WBS was performed in both anterior and posterior projections using a dual-head SPECT with High-energy collimators and a 364-keV photo peak. 131I-SPECT/CT images were acquired immediately after planar imaging for PTC patients who presented suspicious finding on 131I-WBS.
18F-FDG PET/CT Scan
Patients were instructed to fast for at least 6 hours before the injection of 18F-FDG. Blood glucose level was measured before injection and 18F-FDG was administered at glucose levels < 150 mg/dL. 18F-FDG PET/CT scanning was performed after an i.v. injection of 3–4MBq/Kg 18F-FDG, followed by a one hour uptake phase. No intravenous contrast agent was administered. 18F-FDG PET/CT images were performed using a dedicated GE Discovery PET/CT scanner including 64 slice CT scanners with a dedicated PET (BGO plus crystal). 18F-FDG images were acquired for 4 minutes at each bed position from the skull base to the superior mediastinum with patients’ arms along the chest and from the neck to the mid-thigh with patients’ arms above the head. No specific breathing instructions were given. The CT scan was obtained from the orbitomeatal line and progressed to the mid-thigh with the use of a standardized protocol involving 140 kV, 110 mA, 0.8 seconds/rotation, pitch of 1.75:1, length of scan: 1.0 to 1.6 m, 0.625 spatial resolution, and slice thickness of 3.75 mm. Attenuation correction of PET images was performed using attenuation data from CT and images reconstruction was done using a standard reconstruction algorithm with ordered subset expectation maximization (OSEM). Image fusion was performed using coordinate based fusion software and subsequently reviewed at a workstation (Xeleris) that provided multi-planar reformatted images and displayed PET, CT, and PET/CT fusion images.
Neck US
US were performed on the day of 131I administration and every 3–6 months after 131I ablation on a high-resolution ultrasound system equipped with a high-energy 14 MHz linear probe, allowing to work in fundamental B-mode and in power Doppler mode. The thyroid bed, central and lateral neck compartments were included for neck US examination. Suspicion of lymph node metastases of PTC was based on the following criteria: hyperechoic punctuations, cystic appearance, hypervascularization, round shape node without hyperechoic hilum and a short axis greater than 7 mm36.
Tg and TgAb measurement
Serum Tg and TgAb levels were measured by electrochemiluminescence immunoassay (ECLIA) methods on the Cobas analyzer (Roche Diagnostics GmbH). The analytical sensitivity was <0.1 μg/L with reference range 1.4–78 μg/L. The analytical sensitivity of TgAb is < 10 IU/mL with a reference range of 10–4000 IU/mL.
Follow-up
Serum Tg was performed at TSH suppression or at after TSH suppression 6 months after first remnant ablation. Subsequently, FT3, FT4, TSH, Tg, TgAb at TSH suppression and neck US were measured and performed every 3–6 months in the follow-up of the period, respectively. Progressively increased TgAb levels were measured no less than three times. All 18F-FDG PET/CT scan were performed at TSH suppression. Contrast-enhanced CT was performed for a few patients after 18F-FDG PET/CT scan. The last neck US at diagnosis, contrast-enhanced CT and 18F-FDG PET/CT scan were performed at a maximum interval of less than 30 days. The follow-up period was 2–9 yr with a median follow-up of 5.1 yr.
Image analysis
18F-FDG PET/CT images were reviewed and interpreted by 2 experienced nuclear medicine physicians (Z-L Qiu and W-J Wei). All 18F-FDG PET/CT were considered as negative or positive. The criterion for positive lesion was accepted as 18F-FDG uptake greater than that of the normal surrounding tissue or when the SUVmax was ≥ 2.5. The anatomical confirmation with a lesion was detected with matched CT scan. The criterion for negative lesion was that there was no 18F-FDG uptake and no corresponding identifiable lesion on matched CT scans.
Evaluation of 18F-FDG PET/CT findings
18F-FDG PET/CT results were correlated with surgical and histopathological findings, 131I-WBS combined with 131I-SPECT/CT after 131I treatment once again, other imaging modalities including neck US, chest CT, 99mTc-MDP bone scan and follow-up. A true-positive finding was confirmed when a lesion was detected as positive by 18F-FDG PET/CT and the patient was found to have recurrent disease by surgical pathology. A false-positive finding was confirmed when a lesion was excluded by surgical pathology in the patients with positive lesions on 18F-FDG PET/CT. A false-negative finding was confirmed when a lesion couldn’t be detected on 18F-FDG PET/CT, but it could be found to be recurrent disease by surgical pathology or by 131I-WBS combined with 131I-SPECT/CT after 131I treatment once again. A true-negative finding were confirmed when a lesion was detected as negative by 18F-FDG PET/CT and the patient was found to have benign disease by surgical pathology or it could be wasn’t found to have recurrent disease on other imaging modalities including neck US, chest CT and 131I-WBS and 99mTc-MDP bone scan in the follow-up period.
Statistical analysis
Statistical analyses were performed with the SPSS v.17.0 statistical package (SPSS, Inc., Chicago, IL, USA). Descriptive statistics were represented as frequency and percentage. Categorical variables were compared by Pearson Chi-square. The categorical variables for positive 18F-FDG PET/CT were analyzed by univariate logistic regression. The sensitivity, specificity, positive and negative predictive values, and accuracy of 18F-FDG PET/CT for the detection of recurrent thyroid cancer were calculated. Together with their 95% confidence intervals (CIs), the odds ratios (OR) for 18F-FDG PET/CT findings were calculated by univariate logistic regression. A P value of <0.05 was considered to be statistically significant and all reported P values are two-side.
References
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2015. CA Cancer J Clin 65, 5–29, doi:10.3322/caac.21254 (2015).
Brierley, J., Tsang, R., Panzarella, T. & Bana, N. Prognostic factors and the effect of treatment with radioactive iodine and external beam radiation on patients with differentiated thyroid cancer seen at a single institution over 40 years. Clin Endocrinol (Oxf) 63, 418–427, doi:10.1111/cen.2005.63.issue-4 (2005).
Lin, J. D., Chao, T. C., Hsueh, C. & Kuo, S. F. High recurrent rate of multicentric papillary thyroid carcinoma. Ann Surg Oncol 16, 2609–2616, doi:10.1245/s10434-009-0565-7 (2009).
Filesi, M., Signore, A., Ventroni, G., Melacrinis, F. F. & Ronga, G. Role of initial iodine-131 whole-body scan and serum thyroglobulin in differentiated thyroid carcinoma metastases. J Nucl Med 39, 1542–1546 (1998).
Francis, Z. & Schlumberger, M. Serum thyroglobulin determination in thyroid cancer patients. Best Pract Res Clin Endocrinol Metab 22, 1039–1046, doi:10.1016/j.beem.2008.09.015 (2008).
Haugen, B. R. et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26, 1–133, doi:10.1089/thy.2015.0020 (2016).
Rosario, P. W., Mineiro Filho, A. F., Prates, B. S., Silva, L. C. & Calsolari, M. R. Postoperative stimulated thyroglobulin of less than 1 ng/mL as a criterion to spare low-risk patients with papillary thyroid cancer from radioactive iodine ablation. Thyroid 22, 1140–1143, doi:10.1089/thy.2012.0190 (2012).
Pacini, F. et al. Thyroid autoantibodies in thyroid cancer: incidence and relationship with tumour outcome. Acta Endocrinol (Copenh) 119, 373–380, doi:10.1530/acta.0.1190373 (1988).
Spencer, C. A. Clinical review: Clinical utility of thyroglobulin antibody (TgAb) measurements for patients with differentiated thyroid cancers (DTC). J Clin Endocrinol Metab 96, 3615–3627, doi:10.1210/jc.2011-1740 (2011).
Souza, S. L., Montalli Da Assumpcao, L. V. & Ward, L. S. Impact of previous thyroid autoimmune diseases on prognosis of patients with well-differentiated thyroid cancer. Thyroid 13, 491–495, doi:10.1089/105072503322021160 (2003).
Smooke-Praw, S. et al. Thyroglobulin antibody levels do not predict disease status in papillary thyroid cancer. Clin Endocrinol (Oxf) 81, 271–275, doi:10.1111/cen.2014.81.issue-2 (2014).
Hsieh, C. J. & Wang, P. W. Sequential changes of serum antithyroglobulin antibody levels are a good predictor of disease activity in thyroglobulin-negative patients with papillary thyroid carcinoma. Thyroid 24, 488–493, doi:10.1089/thy.2012.0611 (2014).
Kim, W. G. et al. Change of serum antithyroglobulin antibody levels is useful for prediction of clinical recurrence in thyroglobulin-negative patients with differentiated thyroid carcinoma. J Clin Endocrinol Metab 93, 4683–4689, doi:10.1210/jc.2008-0962 (2008).
Palmedo, H. et al. Integrated PET/CT in differentiated thyroid cancer: diagnostic accuracy and impact on patient management. J Nucl Med 47, 616–624 (2006).
Liu, Y. The role of 18F-FDG PET/CT in the follow-up of well-differentiated thyroid cancer with negative thyroglobulin but positive and/or elevated antithyroglobulin antibody. Nucl Med Commun 37, 577–582, doi:10.1097/MNM.0000000000000480 (2016).
Ozkan, E., Aras, G. & Kucuk, N. O. Correlation of 18F-FDG PET/CT findings with histopathological results in differentiated thyroid cancer patients who have increased thyroglobulin or antithyroglobulin antibody levels and negative 131I whole-body scan results. Clin Nucl Med 38, 326–331, doi:10.1097/RLU.0b013e318286827b (2013).
Chung, J. K. et al. Clinical significance of elevated level of serum antithyroglobulin antibody in patients with differentiated thyroid cancer after thyroid ablation. Clin Endocrinol (Oxf) 57, 215–221, doi:10.1046/j.1365-2265.2002.01592.x (2002).
Sanz Viedma, S. et al. Use of 18F FDG-PET in patients with suspicion of recurrent differentiated thyroid cancer by elevated antithyroglobulin antibodies levels and negative 131I scan. Rev Esp Med Nucl 30, 77–82, doi:10.1016/j.remn.2010.10.012 (2011).
Bogsrud, T. V. et al. Prognostic value of 18F-fluorodeoxyglucose-positron emission tomography in patients with differentiated thyroid carcinoma and circulating antithyroglobulin autoantibodies. Nucl Med Commun 32, 245–251, doi:10.1097/MNM.0b013e328343a742 (2011).
Ozkan, E., Soydal, C., Araz, M., Aras, G. & Ibis, E. The additive clinical value of 18F-FDG PET/CT in defining the recurrence of disease in patients with differentiated thyroid cancer who have isolated increased antithyroglobulin antibody levels. Clin Nucl Med 37, 755–758, doi:10.1097/RLU.0b013e31825ae77b (2012).
Asa, S. et al. The role of FDG PET/CT in differentiated thyroid cancer patients with negative iodine-131 whole-body scan and elevated anti-Tg level. Ann Nucl Med 28, 970–979, doi:10.1007/s12149-014-0897-7 (2014).
Chiovato, L. et al. Disappearance of humoral thyroid autoimmunity after complete removal of thyroid antigens. Ann Intern Med 139, 346–351 (2003).
Nam, H. Y. et al. Monitoring differentiated thyroid cancer patients with negative serum thyroglobulin. Diagnostic implication of TSH-stimulated antithyroglobulin antibody. Nuklearmedizin 53, 32–38, doi:10.3413/Nukmed-0604-13-06 (2014).
Nakamoto, Y. et al. Normal FDG distribution patterns in the head and neck: PET/CT evaluation. Radiology 234, 879–885, doi:10.1148/radiol.2343030301 (2005).
Qiu, Z. L., Song, H. J., Xu, Y. H. & Luo, Q. Y. Efficacy and survival analysis of 131I therapy for bone metastases from differentiated thyroid cancer. J Clin Endocrinol Metab 96, 3078–3086, doi:10.1210/jc.2011-0093 (2011).
Na, S. J. et al. Diagnostic accuracy of 18F-fluorodeoxyglucose positron emission tomography/computed tomography in differentiated thyroid cancer patients with elevated thyroglobulin and negative 131I whole body scan: evaluation by thyroglobulin level. Ann Nucl Med 26, 26–34, doi:10.1007/s12149-011-0536-5 (2012).
Oh, K. Y., Kim, Y. H., Yang, E. M. & Kim, C. J. Frequency of Diabetes and Thyroid Autoantibodies in Patients with Type 1 Diabetes and Their Siblings. Chonnam Med J 52, 136–140, doi:10.4068/cmj.2016.52.2.136 (2016).
Brcic, L. et al. Association of established thyroid peroxidase autoantibody (TPOAb) genetic variants with Hashimoto’s thyroiditis. Autoimmunity 1–6 (2016).
Gangemi, S., Saitta, S., Lombardo, G., Patafi, M. & Benvenga, S. Serum thyroid autoantibodies in patients with idiopathic either acute or chronic urticaria. J Endocrinol Invest 32, 107–110, doi:10.1007/BF03345696 (2009).
Ottesen, M., Feldt-Rasmussen, U., Andersen, J., Hippe, E. & Schouboe, A. Thyroid function and autoimmunity in pernicious anemia before and during cyanocobalamin treatment. J Endocrinol Invest 18, 91–97, doi:10.1007/BF03349707 (1995).
Seo, J. H., Lee, S. W., Ahn, B. C. & Lee, J. Recurrence detection in differentiated thyroid cancer patients with elevated serum level of antithyroglobulin antibody: special emphasis on using 18F-FDG PET/CT. Clin Endocrinol (Oxf) 72, 558–563, doi:10.1111/j.1365-2265.2009.03693.x (2010).
Gorges, R. et al. Development and clinical impact of thyroglobulin antibodies in patients with differentiated thyroid carcinoma during the first 3 years after thyroidectomy. Eur J Endocrinol 153, 49–55, doi:10.1530/eje.1.01940 (2005).
Aras, G., Gultekin, S. S. & Kucuk, N. O. The additive clinical value of combined thyroglobulin and antithyroglobulin antibody measurements to define persistent and recurrent disease in patients with differentiated thyroid cancer. Nucl Med Commun 29, 880–884, doi:10.1097/MNM.0b013e328308e079 (2008).
Schmidt, D., Szikszai, A., Linke, R., Bautz, W. & Kuwert, T. Impact of 131I SPECT/spiral CT on nodal staging of differentiated thyroid carcinoma at the first radioablation. J Nucl Med 50, 18–23, doi:10.2967/jnumed.108.052746 (2009).
Blum, M., Tiu, S., Chu, M., Goel, S. & Friedman, K. I-131 SPECT/CT elucidates cryptic findings on planar whole-body scans and can reduce needless therapy with I-131 in post-thyroidectomy thyroid cancer patients. Thyroid 21, 1235–1247, doi:10.1089/thy.2011.0010 (2011).
Nascimento, C. et al. Persistent disease and recurrence in differentiated thyroid cancer patients with undetectable postoperative stimulated thyroglobulin level. Endocr Relat Cancer 18, R29–40, doi:10.1677/ERC-10-0292 (2011).
Author information
Authors and Affiliations
Contributions
Z.-L. Qiu designed the study. Z.-L. Qiu and W.-J. Wei analyzed and interpreted the data, wrote the majority of the manuscript. C.-T. Shen, H.-J. Song, X.-Y. Zhang prepared the figures and tables. Z.-K. Sun indexed all the relevant references while Q.-Y. Luo supervised and edited the paper. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Qiu, ZL., Wei, WJ., Shen, CT. et al. Diagnostic Performance of 18F-FDG PET/CT in Papillary Thyroid Carcinoma with Negative 131I-WBS at first Postablation, Negative Tg and Progressively Increased TgAb Level. Sci Rep 7, 2849 (2017). https://doi.org/10.1038/s41598-017-03001-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-03001-7
This article is cited by
-
Diagnostic performance of 2-[18F]FDG PET/CT in recurrent differentiated thyroid cancer and elevated antithyroglobulin antibodies: an updated systematic review and bivariate meta-analysis
Endocrine (2024)
-
Langerhans’ Cell Histiocytosis Masquerading as Metastatic Papillary Thyroid Cancer on F-18 FDG PET/CT: Diagnostic Dilemma Solved by PET/CT-Guided Biopsy
Nuclear Medicine and Molecular Imaging (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.