Abstract
The relation between obesity and stroke outcome has been disputed. This study was aimed to determine the association of body mass index (BMI) with mortality and functional outcome in patients with acute ischemic stroke. Data were from a national, multi-centre, prospective, hospital-based register: the ChinaQUEST (Quality Evaluation of Stroke Care and Treatment) study. Of 4782 acute ischemic stroke patients, 282 were underweight (BMI < 18.5 kg/m2), 2306 were normal-weight (BMI 18.5 to < 24 kg/m2), 1677 were overweight (BMI 24 to <28 kg/m2) and 517 were obese (BMI ≥ 28 kg/m2). The risks of death at 12 months and death or high dependency at 3 and 12 months in overweight (HR: 0.97, 95% CI: 0.78–1.20; OR: 0.93, 95% CI: 0.80–1.09; OR: 0.95, 95% CI: 0.81–1.12) and obese patients (HR: 1.07, 95% CI: 0.78–1.48; OR: 0.96, 95% CI: 0.75–1.22; OR: 1.06, 95% CI: 0.83–1.35) did not differ from normal-weight patients significantly after adjusting for baseline characteristics. Underweight patients had significantly increased risks of these three outcomes. In ischemic stroke patients, being overweight or obese was not associated with decreased mortality or better functional recovery but being underweight predicted unfavourable outcomes.
Similar content being viewed by others
Introduction
Obesity is an established risk factor for cardiovascular disease and poses a serious public health burden worldwide1,2,3,4. While epidemiological investigations have concluded that obesity is strongly associated with an increased risk of stroke in the general population5, 6, controversy exists regarding the relationship between obesity and outcome in patients who have had an established stroke7,8,9,10,11,12,13,14,15,16,17,18. Several articles report that increased body mass index (BMI) independently predicts a lower mortality after stroke or transient ischemic attack (TIA)9,10,11,12,13,14,15, but others have found no survival advantage in being overweight or obese after stroke16,17,18. Although Doehner et al. have found that obese patients have a better functional outcome than normal-weight patients following acute stroke15, the relation between BMI and functional disability is not well established. Such controversy raises doubts about the appropriateness of recommending weight loss for the secondary prevention of stroke in overweight and obese patients16, 19. Moreover, most investigations have been restricted to Western countries8,9,10,11,12,13,14,15,16, raising further uncertainty about the importance of BMI in Asian stroke patients who have relatively low BMI but high stroke incidence than in other populations6, 20.
The aim of our study was to investigate the association between BMI and stroke outcome, including mortality and functional measures, in a broad range of hospitalized patients with ischemic stroke in China.
Methods
Patients and study protocol
This study was on data from the ChinaQUEST (Quality Evaluation of Stroke Care and Treatment), a multicentre, prospective, hospital-registry (n = 62) study in 37 cities in China. The design and conduct of the ChinaQUEST has been described previously21, 22. In brief, consecutive patients (aged ≥ 15 years) admitted to a hospital within 30 days from onset of acute stroke due to ischemic stroke or intracerebral hemorrhage were included during a 5-month period in 200622. Patients with subarachnoid hemorrhage were excluded.
Patients were assessed 4 times during the course of the study, at admission, discharge, and approximately 3 and 12 months post-stroke22. Baseline information such as socio-demographic variables, medical history and clinical features of the index stroke, was obtained predominantly by face-to-face interviews. In-hospital details on diagnostic and treatment strategies were obtained through medical records (46%) and interviews with patients or their proxies (54%). Follow-up information about functional outcome as measured by the modified Rankin scale (mRS) was collected mainly by telephone interview (>80%) at 3 and 12 months. Death and cause of death were ascertained by family members (64.7%), a health professional (18.4%), records (e.g. medical, police [11.8%]), death certificate (3.5%) or other sources (1.5%).
Written informed consent was obtained from all patients or an appropriate family member (if the patient was unable to give it) to participate. The study was approved by the ethics committees of Peking University First Hospital (Beijing), Ruijin Hospital (Shanghai), Prince of Wales Hospital (Hong Kong), and The University of Sydney. Good Clinical Practice guidelines in accordance with the Declaration of Helsinki were used and the privacy of patients was strictly protected.
Variables of interest and outcomes
BMI was calculated from height and weight (kilograms per square metre) measured by nurses or obtained from the patient or the accompanying relatives on admission. The patient population was sub-grouped into the following categories according to the Chinese Guidelines on Prevention and Control of Obesity in Adults23, 24: underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5 to <24 kg/m2), overweight (BMI 24 to <28 kg/m2) and obese (BMI ≥ 28 kg/m2).
The outcomes included all-cause mortality at 12 months, and a composite of death or high dependency at 3 and 12 months post-stroke. High dependency was defined as a mRS score of 3–5.
Covariates considered included socio-demographic variables, medical history and clinical features of the index stroke, including: age, sex, partnership (living alone or living with a partner), education level (‘low level’, being illiterate or having only primary education; ‘high level’, having secondary education or higher) and annual household income (<20000 Chinese Yuan [CNY], approximately $2857 US; ≥20000 CNY; or declined to respond/unknown); medical history of hypertension, diabetes, hyperlipidaemia, atrial fibrillation, prior stroke/TIA, coronary artery disease (any history of heart attack and/or myocardial infarction, angina or coronary heart disease) either self-reported or diagnosed in hospital post-stroke; prior dependency, current cigarette smoking, and regular alcohol consumption within the 3 months before stroke onset; time of presentation at the hospital (<6 hour; ≥6 hour; or unknown), Glasgow coma scale (GCS) score on admission (‘mild’ 13–15; ‘moderate’ 8–12; or ‘severe’ 3–7) and Oxfordshire Community Stroke Project (OCSP) classification (total anterior circulation infarct [TACI]; partial anterior circulation infarct [PACI]; lacunar infarct [LACI]; posterior circulation infarct [POCI]; or unknown).
Statistical analysis
Baseline characteristics were compared among different BMI subgroups using the one-way analysis of variance (ANOVA) and Chi-square test for continuous and for categorical variables as appropriate. The Cox proportional hazard model was performed to assess the association between BMI and all-cause mortality at 12 months. Similarly, logistic regression models were performed to evaluate the association between BMI and the composite outcome of death or high dependency at 3 and 12 months post-stroke. The association of BMI and high dependency was also evaluated in the survivors at 3 and 12 months after stroke. The distribution of causes of death in those who had died during the follow-up period was compared with Chi-square test among the BMI subgroups. We performed four additional analyses to assess the robustness of our main findings. First, we performed a sensitivity analysis using the World Health Organization (WHO) BMI classification criteria25, which defined underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5 to <25 kg/m2), overweight (BMI 25 to <30 kg/m2) and obese (BMI ≥ 30 kg/m2). Second, we restricted the study population to those who had not received thrombolysis treatment. Third, another definition of high dependency as a mRS score of 4–5 was used to validate the association between BMI and functional outcome15. Fourth, we performed a subgroup analysis according to smoking status (current smoker and current non-smoker). All P values were two-sided, with P < 0.05 considered statistically significant. All statistical analyses were performed using SPSS version 13.0 (SPSS Inc., Chicago, IL, USA).
Results
Baseline characteristics
There were 6508 acute stroke patients enrolled in the ChinaQUEST study. After excluding 1572 patients with intracerebral hemorrhage and 154 with uncertain diagnosis and missing data, a total of 4782 patients with acute ischemic stroke were included in our analysis. The mean age of those with ischemic stroke was 64.6 years (range 15–102 years) and 37.9% were female. Their average BMI was 23.8 ± 3.5 kg/m2. Of them, 282 patients (5.9%) were underweight, 2306 (48.2%) had normal weight, 1677 (35.1%) were overweight, and 517 (10.8%) were obese.
Baseline characteristics between the BMI subgroups are shown in Table 1. Underweight patients were older and more likely to have had little education, a low income, atrial fibrillation, prior dependency and low level of consciousness at baseline. Compared with normal-weight patients, obese patients were younger and more often had a medical history of hypertension, diabetes, hyperlipidaemia, previous stroke/TIA, and coronary artery disease. Overweight group had the highest proportions of male, current smoker and regular alcohol user.
Body mass index and stroke outcomes
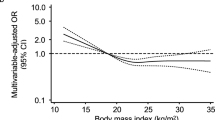
There were 484 patients who had died within 12 months after stroke. High dependency was found in 1221 patients at 3 months and in 942 patients at 12 months. Unadjusted analysis showed obese patients did not differ from patients of normal BMI significantly in the risks for death and death or high dependency after stroke. Overweight patients had a slightly lower proportion of death or high dependency at 12 months than normal-weight patients. Those who were underweight had the highest risks of the three endpoints (Table 2).
After adjustment of all covariates, neither overweight (hazard ratio [HR]: 0.97, 95% confidence interval [CI]: 0.78–1.20; odds ratio [OR]: 0.93, 95% CI: 0.80–1.09; OR: 0.95, 95% CI: 0.81–1.12; compared with normal-weight group) nor obese (HR: 1.07, 95% CI: 0.78–1.48; OR: 0.96, 95% CI: 0.75–1.22; OR: 1.06, 95% CI: 0.83–1.35; compared with normal-weight group) was associated with death at 12 months, and death or high dependency at 3 and 12 months significantly. Underweight patients remained at significantly highest risk of poor outcome (Table 2).
In the surviving patients, there was no significant difference in the risk of high dependency in overweight and obese patients compared with normal-weight group, either (Table 3). In those who had died during the follow-up period, the distribution of causes of death did not differ significantly among the BMI subgroups (p = 0.200; Table 4).
Sensitivity analyses
The relation between BMI and the outcomes were consistent in sensitivity analyses (Table 5). The multivariable analyses showed the risks for death and death or high dependency in overweight and obese patients did not differ from normal-weight patients significantly when using the WHO BMI classification criteria, or excluding those patients who had received thrombolysis treatment. Using another definition of high dependency as a mRS score of 4–5 did not change the association between BMI and functional disability. The stratified analysis by smoking status showed no significant differences in these outcomes among normal-weight, overweight and obese patients in both subgroups.
Discussion
The present study did not find any outcome advantage associated with being overweight or obese at the time of experiencing an acute ischemic stroke. The risks of death and functional disability in overweight and obese patients did not differ significantly from normal-weight patients. However, underweight patients had consistently the worst outcomes after ischemic stroke.
Several studies reported a lower mortality in obese patients than normal-weight patients after stroke9,10,11,12,13,14,15. Doehner et al. also found obesity predicted a decreased risk of severe disability15. While in our study, the risks of death and death or high dependency after ischemic stroke in overweight and obese patients did not differ significantly from that of normal-weight patients. In the surviving patients, overweight and obesity was not associated with better functional recovery, either. The discrepancy of the results may be due to several reasons. First, our study included only subjects after ischemic stroke, whereas some previous studies included also subjects with hemorrhagic stroke10, 12, 14, 15. Taking into account the significant differences in pathogenesis and prognostic pattern, combining ischemic and hemorrhagic stroke was not appropriate for the evaluation of the relationship between obesity and ischemic stroke outcome. Second, our analysis controlled for a wide range of covariates, including stroke severity and socioeconomic status, which were the important confounders of the association of obesity and stroke outcome but often absent in previous studies10,11,12,13,14, 16,17,18. In a cohort of Korean patients with ischemic stroke, the favorable effects of overweight and obesity disappeared after adjustment of initial stroke severity18. Third, the outcomes were evaluated at different time point. Those studies which found a benefit of obesity often assessed the mortality and functional outcome after a relatively long follow-up period, ranging from 30 months to 10 years9,10,11,12, 15, 26. Considering that many other diseases might influence the survival and functional status of stroke patients within a longer follow-up period and limited information of these co-morbidities was collected and adjusted in previous analyses9,10,11,12, 15, 16, 26, their findings were more vulnerable to confounding bias. In contrast, our study selected the mortality within 12 months and the proportion of death or high dependency at 3 and 12 months after stroke as the primary outcomes, which were well-recognized measures of stroke outcome and used as study endpoints in many clinical trials on stroke27, 28. So our result was a more reliable estimate of the independent impact of obesity on stroke prognosis. Another study using stroke-related mortality at 1 week and 1 month post-stroke as the outcomes also found no survival advantage in obese patients16, which was consistent with our findings. Additionally, the sensitivity analysis with a different definition of high dependency validated our result on functional outcome.
Although we found no significant differences of survival and functional disability in overweight and obese patients compared with normal-weight patients, our results still support the recommendation of current guidelines to strive for normal weight after stroke, given the association between weight loss and improvements in major cardiovascular risk factors, including dyslipidaemia, diabetes and hypertension19, 29, 30.
Our study showed underweight patients had the highest all-cause mortality and the worst functional recovery after stroke, which was consistent with previous studies8, 9, 11, 12, 26. Although underweight individuals may more often have serious systemic diseases or cancer16, our analysis found the distribution of causes of death in underweight group did not differ from that of other BMI subgroups significantly, which indicated the increased mortality associated with underweight could not be attributed to other co-morbidities.
Given the BMI distribution in Asian population is left-shifted as compared with that of Western population26, 31, our study utilized the obesity criteria according to the Chinese Guidelines on Prevention and Control of Obesity in Adults23, 24. Additionally, the current WHO obesity definition was used in the sensitivity analysis and did not change our results25.
Stroke severity is closely associated with the prognosis after stroke16,17,18, 32. Although the National Institutes of Health Stroke Scale (NIHSS) score was not collected in the ChinaQUEST database, we included GCS score and OCSP classification as confounders in our analyses, which had been used as measures of stroke severity in previous studies and had high inter-observer reliability33,34,35. Additionally, GCS score and OCSP classification had been validated as the independent predictors of stroke outcome in the ChinaQUEST cohort22.
Some studies indicated smoking was an important confounder of the relation between BMI and mortality3. To avoid the potential influence of smoking, we performed a subgroup analysis to assess the association of BMI and stroke outcome in current smokers and current non-smokers, respectively. The analysis showed there was no advantage of survival or functional recovery associated with overweight and obese in both subgroups, which supported our main findings.
There are several limitations to the present study that should be addressed. First, our study is an observational investigation that suffers from the typical limitations of such studies. We adjusted the outcome analyses for a range of identified confounding variables; however, the findings may be influenced by unknown confounders. Second, we could not exclude the possibility of bias due to other treatment and intervention that might influence stroke outcome, although the sensitivity analysis showed that the intravenous thrombolysis treatment did not affect the results of our main analysis. Third, the definition of ‘overweight’ and ‘obesity’ was based on BMI in our study. However, BMI does not reflect body fat distribution, which is also important to the evaluation of obesity36. Further studies with measures of body fat distribution, such as waist circumference or waist-to-hip ratio, may provide a more accurate assessment of the association between obesity and stroke outcome.
In conclusion, the present study shows that there is no survival advantage of overweight or obesity after ischemic stroke. The risk of death and functional disability is not significantly different in overweight and obese patients as compared with normal-weight patients. Underweight is associated with the highest risk of death and high dependency after stroke. Our results support the rationality of weight control for overweight and obese patients recommended by current guidelines on secondary prevention of stroke. However, the observational nature of our study warrants further validation.
References
Wilson, P. W., D’Agostino, R. B., Sullivan, L., Parise, H. & Kannel, W. B. Overweight and obesity as determinants of cardiovascular risk: the Framingham experience. Arch Intern Med. 162, 1867–1872, doi:10.1001/archinte.162.16.1867 (2002).
Bibbins-Domingo, K., Coxson, P., Pletcher, M. J., Lightwood, J. & Goldman, L. Adolescent overweight and future adult coronary heart disease. N Engl J Med. 357, 2371–2379, doi:10.1056/NEJMsa073166 (2007).
Hu, F. B. et al. Adiposity as compared with physical activity in predicting mortality among women. N Engl J Med. 351, 2694–2703, doi:10.1056/NEJMoa042135 (2004).
Murphy, N. F. et al. Long-term cardiovascular consequences of obesity: 20-year follow-up of more than 15000 middle-age men and women (the Renfrew-Paisley study). Eur Heart J. 27, 96–106, doi:10.1093/eurheartj/ehi506 (2006).
Strazzullo, P. et al. Excess body weight and incidence of stroke: meta-analysis of prospective studies with 2 million participants. Stroke. 41, e418–e426, doi:10.1161/STROKEAHA.109.576967 (2010).
Bazzano, L. A. et al. Body mass index and risk of stroke among Chinese men and women. Ann Neurol. 67, 11–20, doi:10.1002/ana.v67:1 (2010).
Scherbakov, N., Dirnagl, U. & Doehner, W. Body weight after stroke: lessons from the obesity paradox. Stroke. 42, 3646–3650, doi:10.1161/STROKEAHA.111.619163 (2011).
FOOD, T. Collaboration. Poor nutritional status on admission predicts poor outcomes after stroke. Stroke 34, 1450–1456 (2003).
Olsen, T. S., Dehlendorff, C., Petersen, H. G. & Andersen, K. K. Body mass index and poststroke mortality. Neuroepidemiology. 30, 93–100, doi:10.1159/000118945 (2008).
Vemmos, K. et al. Association between obesity and mortality after acute first-ever stroke: the obesity-stroke paradox. Stroke. 42, 30–36, doi:10.1161/STROKEAHA.110.593434 (2011).
Skolarus, L. E. et al. The association of body mass index and mortality after acute ischemic stroke. Circ Cardiovasc Qual Outcomes. 7, 64–69, doi:10.1161/CIRCOUTCOMES.113.000129 (2014).
Andersen, K. K. & Olsen, T. S. The obesity paradox in stroke: lower mortality and lower risk of readmission for recurrent stroke in obese stroke patients. Int J Stroke 10, 99–104, doi:10.1111/ijs.12016 (2015).
Wohlfahrt, P. et al. The obesity paradox and survivors of ischemic stroke. J Stroke Cerebrovasc Dis. 24, 1443–1450, doi:10.1016/j.jstrokecerebrovasdis.2015.03.008 (2015).
Barba, R. et al. The obesity paradoxin stroke: impact on mortality and short-term readmission. J Stroke Cerebrovasc Dis. 24, 766–770, doi:10.1016/j.jstrokecerebrovasdis.2014.11.002 (2015).
Doehner, W., Schenkel, J., Anker, S. D., Springer, J. & Audebert, H. J. Overweight and obesity are associated with improved survival, functional outcome, and stroke recurrence after stroke or transient ischaemic attack: observations from the TEMPiS trial. Eur Heart J. 34, 268–277, doi:10.1093/eurheartj/ehs340 (2013).
Dehlendorff, C., Andersen, K. K. & Olsen, T. S. Body mass index and death by stroke: no obesity paradox. JAMA Neurol. 71, 978–984, doi:10.1001/jamaneurol.2014.1017 (2014).
Ryu, W. S., Lee, S. H., Kim, C. K., Kim, B. J. & Yoon, B. W. Body mass index, initial neurological severity and long-term mortality in ischemic stroke. Cerebrovasc Dis. 32, 170–176, doi:10.1159/000328250 (2011).
Kim, Y., Kim, C. K., Jung, S., Yoon, B. W. & Lee, S. H. Obesity-stroke paradox and initial neurological severity. J Neurol Neurosurg Psychiatry. 86, 743–747, doi:10.1136/jnnp-2014-308664 (2015).
Kernan, W. N. et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline for healthcare professionals from the American Heart Association/ American Stroke Association. Stroke. 45, 2160–2236, doi:10.1161/STR.0000000000000024 (2014).
Ni Mhurchu, C., Rodgers, A., Pan, W. H., Gu, D. F. & Woodward, M. Body mass index and cardiovascular disease in the Asia-Pacific region: an overview of 33 cohorts involving 310000 participants. Int J Epidemiol. 33, 751–758, doi:10.1093/ije/dyh163 (2004).
Heeley, E. L. et al. Role of health insurance in averting economic hardship in families after acute stroke in China. Stroke. 40, 2149–2156, doi:10.1161/STROKEAHA.108.540054 (2009).
Wei, J. W. et al. Comparison of recovery patterns and prognostic indicators for ischemic and hemorrhagic stroke in China: the ChinaQUEST(QUality Evaluation of Stroke Care and Treatment) Registry study. Stroke. 41, 1877–1883, doi:10.1161/STROKEAHA.110.586909 (2010).
Zhou, B. F. Predictive values of body mass index and waist circumference for risk factors of certain related diseases in Chinese adults-study on optimal cut-off points of body mass index and waist circumference in Chinese adults. Biomed Environ Sci. 15, 83–96 (2002).
Zhang, X. et al. Association of obesity and atrial fibrillation among middle-aged and elderly Chinese. Int J Obes 33, 1318–1325, doi:10.1038/ijo.2009.157 (2009).
WHO. Obesity: preventing and managing the global epidemic. Report of a WHO Consultation. WHO Technical Report Series 894. Geneva: World Health Organization (2000).
Kim, B. J. et al. Dynamics of obesity paradox after stroke, related to time from onset, age, and causes of death. Neurology. 79, 856–863, doi:10.1212/WNL.0b013e318266fad1 (2012).
Duncan, P. W., Jorgensen, H. S. & Wade, D. T. Outcome measures in acute stroke trials: a systematic review and some recommendations to improve practice. Stroke. 31, 1429–1438, doi:10.1161/01.STR.31.6.1429 (2000).
Prabhakaran, S., Ruff, I. & Bernstein, R. A. Acute stroke intervention: a systematic review. JAMA. 313, 1451–1462, doi:10.1001/jama.2015.3058 (2015).
Caterson, I. D. et al. Maintained intentional weight loss reduces cardiovascular outcomes: results from the Sibutramine Cardiovascular OUTcomes (SCOUT) trial. Diabetes Obes Metab 14, 523–530, doi:10.1111/j.1463-1326.2011.01554.x (2012).
Neter, J. E., Stam, B. E., Kok, F. J., Grobbee, D. E. & Geleijnse, J. M. Influence of weight reduction on blood pressure: a meta-analysis of randomized controlled trials. Hypertension. 42, 878–884, doi:10.1161/01.HYP.0000094221.86888.AE (2003).
WHO expert consultation. Appropriate body–mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 363, 157–163, doi:10.1016/S0140-6736(03)15268-3 (2004).
Andersen, K. K., Andersen, Z. J. & Olsen, T. S. Predictors of early and late case-fatality in a nationwide Danish study of 26,818 patients with first-ever ischemic stroke. Stroke. 42, 2806–2812, doi:10.1161/STROKEAHA.111.619049 (2011).
Myint, P. K. et al. The SOAR (Stroke subtype, Oxford Community Stroke Project classification, Age, prestroke modified Rankin) score strongly predicts early outcomes in acute stroke. Int J Stroke. 9, 278–283, doi:10.1111/ijs.12088 (2014).
Counsell, C., Dennis, M., McDowall, M. & Warlow, C. Predicting outcome after acute and subacute stroke: development and validation of new prognostic models. Stroke. 33, 1041–1047, doi:10.1161/hs0402.105909 (2002).
Gill, M. R., Reiley, D. G. & Green, S. M. Interrater reliability of Glasgow Coma Scale scores in the emergency department. Ann Emerg Med. 43, 215–222, doi:10.1016/S019606440300814X (2004).
Frankenfield, D. C., Rowe, W. A., Cooney, R. N., Smith, J. S. & Becker, D. Limits of body mass index to detect obesity and predict body composition. Nutrition. 17, 26–30, doi:10.1016/S0899-9007(00)00471-8 (2001).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (No. 81400944). The ChinaQUEST study was supported by grants from the Macquarie Bank Foundation, The George Foundation, and AstraZeneca Pharmaceutical China. We thank all study participants and staffs in the China QUEST (Quality Evaluation of Stroke Care and Treatment) study.
Author information
Authors and Affiliations
Contributions
W.S. and Y.H. conceived and designed the study; Y.H., Z.J., J.W.W., J.G.W., M.L. and C.S.A. contributed in data collection; S.Z., R.L. and F.L. performed the statistical analysis; W.S. drafted the manuscript; Y.H., Y.X. and C.S.A. contributed to the critical revision of the manuscript. All authors reviewed and approved the submitted manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sun, W., Huang, Y., Xian, Y. et al. Association of body mass index with mortality and functional outcome after acute ischemic stroke. Sci Rep 7, 2507 (2017). https://doi.org/10.1038/s41598-017-02551-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-02551-0
This article is cited by
-
The Influence of Body Mass Index on Outcomes in Patients Undergoing Mechanical Thrombectomy for Anterior Circulation Large Vessel Occlusion: Institutional Experience and Meta-analysis
Neurocritical Care (2024)
-
Effects of body mass index on the immune response within the first days after major stroke in humans
Neurological Research and Practice (2023)
-
Serum NT-proBNP level for predicting functional outcomes after acute ischemic stroke
Scientific Reports (2023)
-
Non-linear association between body weight and functional outcome after acute ischemic stroke
Scientific Reports (2023)
-
Differential effects of body mass index on domain-specific cognitive outcomes after stroke
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.