Abstract
Astrocytes are glial cells that support and protect neurons in the central nervous systems including the retina. Retinal ganglion cells (RGCs) are in contact with the astrocytes and our earlier findings showed the reduction of the number of cells in the ganglion cell layer in adult progranulin deficient mice. In the present study, we focused on the time of activation of the astrocytes and the alterations in the number of RGCs in the retina and optic nerve in progranulin deficient mice. Our findings showed that the number of Brn3a-positive cells was reduced and the expression of glial fibrillary acidic protein (GFAP) was increased in progranulin deficient mice. The progranulin deficient mice had a high expression of GFAP on postnatal day 9 (P9) but not on postnatal day 1. These mice also had a decrease in the number of the Brn3a-positive cells on P9. Taken together, these findings indicate that the absence of progranulin can affect the survival of RGCs subsequent the activation of astrocytes during retinal development.
Similar content being viewed by others
Introduction
Astrocytes are present in the retinal nerve fiber layer (RNFL) and they secrete different types of growth factors and cytokines especially when the retina is injured. Astrocytes originate from neural precursor cells and more specifically from astrocyte precursor cells (APCs)1, 2. The APCs express Pax2, vimentin, and A2B52, and they migrate into the retina from the optic nerve during embryogenesis and spread toward the periphery2. The astrocytes mature in the retina and they are mainly located in the central retina during the neonatal stage and even on postnatal day 02.
The retinal ganglion cells (RGCs) are located in the ganglion cell layer (GCL) among the astrocytes, and they send their axons toward the optic nerve and to the lateral geniculate nucleus and superior colliculus3. The astrocytes, microglia, and oligodendrocytes in the optic nerve support and nourish the axons of the RGCs4, 5. APCs in the retina guide the axons toward the optic nerve during embryonic development2, and they are required for the normal development of the synapses of the RGCs6, 7.
The RGCs interact with astrocytes. For example, growth factors such as PDGF-A and sonic hedgehog are synthesized by RGCs, and they promote the migration and proliferation of APCs2, 8, 9. It was recently reported that an excessive activation of astrocytes induce death in cultured striatal neurons on embryonic day 1810. Mature astrocytes play a key role in neurogenesis during development11. It is believed that a balance of the activation of astrocytes is important for the normal development of neurons.
Progranulin is a multipotent growth factor that is expressed in neurons and microglia. Progranulin has been shown to promote neuronal survival and the regulation of inflammation in the brain, retina, and spinal cord12,13,14,15,16. We have shown that the retinas of adult progranulin-deficient mice, Grn −/− mice were not normal17. More specifically, the thickness of the outer nuclear layer (ONL) and the level of rhodopsin were significantly reduced in Grn −/− mice. In addition, we showed that recombinant progranulin promoted photoreceptor differentiation from retinal precursor cells in neonatal retinal cells in culture. We concluded that progranulin regulated normal retinal development17. Moreover, we confirmed that the cell number in GCL was decreased in adult Grn −/− mice although there was no change in the thickness of the inner plexiform layer (IPL), inner nuclear layer (INL) and outer plexiform layer (OPL). The purpose of this study was to determine when the alterations of the RGC and other retinal structures occur in Grn −/− mice.
Results
Number of Brn3a-positive retinal ganglion cells is reduced and expression of GFAP is increased in adult Grn −/− mice
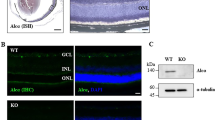
Our previous study found that the number of cells in the RNFL was reduced in Grn −/− mice17. To confirm whether the cell loss was due to a decrease of RGCs, we performed immunostaining for Brn3a, a specific marker for RGCs in the retina of WT and Grn −/− mice. Brn3a-positive (Brn3a+) RGCs and the overall number of cells in the GCL were reduced in Grn −/− mice (Fig. 1A,B).
Decrease of Brn3a+ retinal ganglion cells (RGCs) and high GFAP expression in adult Grn −/− mice. (A) Brn3a staining (green) shows the RGCs in the ganglion cell layer (GCL) of wild-type (WT) and progranulin-knockout (KO) mice. Nuclei are seen as cyan by Hoechst 33342 staining. (B) Number of Brn3a+ cells and the number of cells in the GCL. The number of Brn3a+ cells and the cells in the GCL are reduced in Grn −/− mice. (C,D) Representative images show the activated astrocyte (red by GFAP expression) in WT and Grn −/− mice. The fluorescent intensity is increased in Grn −/− mice. (E,F) Number of Iba-1+ microglia (green) is not changed in Grn −/− mice. (G,H) Progranulin expression (magenta) is observed around the S100β+ astrocytes (green) and co-localized with Iba-1+ microglia (green). Data are the means ± S.E.M. (n = 4 or 5). ## P < 0.01, # P < 0.05 vs. WT (Student’s t-test). WT: wild-type; KO: knock-out. Scale bar = 20 μm.
Next, we investigated whether the glial cells were activated during retinal degeneration by staining with markers of astrocytes and microglia. GFAP is a marker of activated astrocytes18, and a high expression of GFAP showed that the astrocytes were activated in the Grn −/− retina. The fluorescent intensity of GFAP was significantly increased in Grn −/− retina and was higher than that in the WT retina (Fig. 1C,D). The number of Iba-1 positive (Iba-1+) microglia in the Grn −/− mice did not differ significantly from that in WT mice (Fig. 1E,F). Moreover, an expression of progranulin was observed around S100β-positive (S100β+) astrocytes and it was co-localized with Iba-1+ microglia (Fig. 1G,H). S100β is a marker of mature astrocytes19. These results demonstrated that progranulin deficiency caused the decrease in the number of RGCs and the activation of the astrocytes. Moreover, there was no change in the expression of cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) between WT and Grn −/− retina (Supplementary Fig. 1).
Activation of astrocytes in optic nerve of Grn −/− mice
An expression of progranulin was detected in longitudinal retinal sections and cross sections of the optic nerve (Fig. 2A,B). In the cross sections of the optic nerve, phosphorylated neurofilament heavy (p-NFH) and NFH were used as markers of RGC axons5, 20. p-NFH and NFH were accumulated in Grn −/− mice although the fluorescent intensity was not changed (Fig. 2C,D). The fluorescent intensity of Iba-1 in Grn −/− mice was not different from that in WT mice (Fig. 2C,D). Astrocytes were activated and the fluorescent intensity of GFAP was increased in Grn −/− mice (Fig. 2C,D). The optic nerve sections of Grn −/− mice showed degenerating neuronal axons and activation of astrocytes, but not microglial activation. These results indicated that there was degeneration of the RGCs in the Grn −/− retinas (Fig. 1A,B), and the axonal degeneration may precede that of the RGCs as reported in a mouse model of glaucoma21.
Changes of optic nerve in adult Grn −/− mice. (A) Progranulin expression (magenta) in WT optic nerve. Neurofilament heavy (NFH) stains the optic nerve (green). Progranulin expression is not present in optic nerve of KO mice. (B) Progranulin expression (magenta) in cross sections of WT and KO optic nerve. (C) Representative image showing phosphorylated neurofilament heavy (p-NFH), NFH, myelin basic protein (MBP), Iba-1 and GFAP. p-NFH and NFH are accumulated in Grn −/− mice. Iba-1+ microglia (green) is not changed in Grn −/− mice. GFAP+ astrocytes (red) are activated in Grn −/− mice. (D) The fluorescent intensity of NFH and Iba-1 is not changed. GFAP expression is increased in Grn −/− mice. Data are the means ± S.E.M. (n = 3). ## P < 0.01 vs. WT (Student’s t-test). WT: wild-type; KO: knock-out. Scale bar = 20 μm.
An expression of progranulin was observed in Iba-1+ microglia, S100β+ astrocytes and CC-1-positive oligodendrocytes, but not in myelin basic protein (MBP)-positive myelin (Fig. 3A–D). We investigated the expression of GFAP in the hippocampus at CA1, CA3, and dentate gyrus (DG). There was no difference in the staining between the WT and Grn −/− hippocampus (Supplementary Fig. 2). We concluded that the astrocytes were activated selectively in the retina and optic nerve.
Astrocyte activation and retinal ganglion cell loss occur in Grn −/− retina on P9
To determine when the number of RGCs is decreased and astrocytes are activated, we first examined the expression of S100β and GFAP, respectively, in P9 retinas. The fluorescent intensity of S100β and GFAP expression was increased in Grn −/− RNFL and western blotting showed an increase of GFAP expression (Fig. 4A,C). Moreover, the expression of glutamine synthetase (GS), another glial marker, was increased in the Grn −/− RNFL and INL consisting of astrocytes and Müller glia respectively (Fig. 4B,C).
Retinal astrocyte activation and retinal ganglion cell loss in Grn −/− mice at P9. (A) S100β+ and GFAP+ astrocytes (green and red) in P9 mice retina. Fluorescent intensity of S100β and GFAP expression is increased in Grn −/− mice. Data are the means ± S.E.M. (n = 5 or 6). # P < 0.05 vs. WT (Student’s t-test). (B) GS (red) stained astrocytes and Müller glia. Astrocytes activation is observed in Grn −/− mice at P9. (C) Typical band showed GS, GFAP, p-MET, MET, and β-actin. The quantitative data show the relative expression of GS and GFAP per β-actin and p-MET per MET. GS and GFAP expressions are increased in Grn −/− mice retina and the phosphorylation of MET is decreased in Grn −/− mice retina at P9. Data are the means ± S.E.M. (n = 3 to 5). ## P < 0.01, # P < 0.05 vs. WT (Student’s t-test). (D) Brn3a staining (green) shows the RGCs. The nuclei are stained cyan by Hoechst 33342. The quantitative data show that the number of Brn3a+ cells and the cell numbers in the GCL were decreased in Grn −/− mice at P9. Data are the means ± S.E.M. (n = 5). # P < 0.05 vs. WT (Student’s t-test). WT: Wild-type; KO: Knock-out. Scale bar = 20 μm. The cropped blots are used in this Figure and the full-length blots are presented in Supplementary Fig. S4.
The results of our previous study showed that progranulin was associated with the hepatocyte growth factor (HGF) receptor which is also called MET15, 17. We confirmed that progranulin deficiency strongly inhibited the phosphorylation of the HGF receptor (Fig. 4C). At this stage, the number of Brn3a+ RGCs and the cell number in GCL were reduced in Grn −/− mice. These results demonstrated that astrocytes and RGCs were already affected at P9. Another finding suggested that progranulin deficiency inhibited the maturation of photoreceptors from the photoreceptor precursor cells at P9. More specifically, the expression of CRX was increased and the rhodopsin expression was decreased in the Grn −/− retina at P9 (Supplementary Fig. 3). These results are a complementary findings of the earlier report that progranulin promoted the differentiation of photoreceptors during development17.
Progranulin does not affect astrocytes in Grn −/− retina on P1
Finally, we investigated the changes of the astrocyte in the Grn −/− retina at P1 to determine whether progranulin deficiency affected the gliogenesis at this time. There was no change in the expression of S100β in the central retina of Grn −/− mice on P1 (Fig. 5A,B). An expression of GFAP was rarely observed at P1 in the retina of WT and Grn −/− mice (data not shown). The expression of progranulin was investigated at P1 and P9 by western blotting in the retina of WT and Grn −/− mice. Progranulin expression was not detected in Grn −/− mice at P1 or P9 (Fig. 5C). The WT retinas at P9 expressed progranulin higher than at P1 in WT retinas (Fig. 5C). Moreover, immunostaining showed that progranulin was localized in the retinas at P1 and P9 of WT mice. S100β+ astrocytes were partly co-localized with progranulin in P9 WT retinas, but not with P1 WT retinas (Fig. 5D). Iba-1+ microglia was almost fully co-localized with progranulin in P1 and P9 retina (Fig. 5D). In normal development, progranulin expression was increased from P1 to P9. RGCs and astrocytes strongly depended on the presence of progranulin during this stage (Fig. 5E). We suggest that progranulin is associated with the survival of RGCs during retinal development (P1 to P9).
No change in retinal astrocytes in Grn −/− mice at P1. (A,B) Representative image show the S100β+ astrocytes (green) in P1 mice retina. There is no change in the expression of S100β in Grn −/− mice at P1. Data are the means ± S.E.M. (n = 4, 5). Student’s t-test. (C) Progranulin expression in P1 and P9 mice retina by western blotting. Progranulin expression is not observed in Grn −/− mice at P1 and P9 but is expression in WT mice at both times. The expression of progranulin is higher at P9 than at P1 in WT mice retina. Data are the means ± S.E.M. (WT: n = 3 or 4, KO: n = 2). # P < 0.05 vs. P1 WT (Student’s t-test). (D) Progranulin expression (magenta) in P1 and P9 WT mice retina by immunostaining. S100β+ astrocyte (green) is partly co-localized with progranulin in P9 WT retina but not in P1 WT retina. Iba-1+ microglia (green) are almost fully co-localized with progranulin in P1 and P9 retina. (E) Retinal development with or without progranulin. Progranulin is mainly secreted by microglia. Progranulin deficiency leads to astrocyte activation and RGC loss at P9, but not at P1. WT: Wild-type; KO: Knock-out; N.S.: Not significance. Scale bar = 20 μm. The cropped blots are used in this Figure and the full-length blots are presented in Supplementary Fig. S4.
Discussion
The maturation of RGCs is completed soon after birth22. In the present study, the astrocytes have not been activated and the number of astrocytes in Grn −/− mice does not differ significantly from that in WT mice at P1 (Fig. 5A,B). This indicates that the astrocytes probably do not affect the development of the retinal neurons until at least P1 in Grn −/− mice. To support this hypothesis, the results of this study showed that the expression of the progranulin protein was upregulated in WT retina from P1 to P9 (Fig. 5C). Thus, progranulin may not be absolutely necessary for the normal development of the retina up to P1.
The phosphorylation of HGF receptors is reduced in Grn −/− mice at P9 (Fig. 4C). From this observation and previous reports, it is highly likely that progranulin is associated with the HGF receptor15, 17, 23. More specifically, a decrease of phosphorylation in the Grn −/− retina and an increase of the phosphorylation in primary retinal cells by recombinant progranulin were observed in the earlier and present studies. These changes should result from the activity of retinal neuronal cells and, not by only astrocytes considering the abundance of astrocytes in the retina. It has been reported that c-met, a HGF receptor gene, was present in RGCs and exogenous HGF protected RGCs through HGF receptor signaling24, 25. Therefore, progranulin may contribute to the direct protection of RGCs through HGF receptor signaling at immature stage followed by the activation of astrocytes.
While, it was recently reported that excessive activation of astrocytes induces the death of neuronal cells in culture10. In the present study, astrocyte markers, S100β, GFAP, and GS were up-regulated in Grn −/− mice at P9 (Fig. 4A–C). The expression of GFAP probably indicates that the astrocytes are activated18, 19, and the activation was occurred at P9 in Grn −/− mice (Fig. 4A). Recently, it was reported that recombinant progranulin directly suppresses the activation of astrocytes using murine primary astrocytes26. Therefore, the excessive activation of astrocytes in Grn −/− retina might have caused the loss of Brn3a+ RGCs (Fig. 4D). It was believed that astrocyte activation and RGCs degeneration are sustained at the adult stage, which then results in axonal degeneration (Figs 1A–D and 2C).
An expression of progranulin was observed in the microglia at P1, P9 and adult retinas and optic nerves (Figs 1H, 3A and 5D). This is reasonable considering previous reports that progranulin is mainly secreted by microglia and neurons in the brain during early and adult ages16, 27, 28.
Our immunostaining data showed that the S100β+ astrocytes were partly co-localized with progranulin positive cells (Figs 1G, 3B and 5D). This is generally not observed in the brain although it was reported that some of the astrocytes in the brain express progranulin28. This could be due to the differences in the astrocyte markers, S100β as we used GFAP as previously performed. Although it is not certain whether astrocytes secrete progranulin, we think the possibility that the progranulin surrounding the astrocytes may play a role on astrocytes at P9 and also of adult retinas and optic nerve (Figs 1C, 2C and 4A).
The expression of progranulin has been detected in the brain, spinal cord, and retinal ganglion cells during development29, 30. It was later reported that a mutation of the progranulin gene was associated with the frontotemporal lobar degeneration31. However, the role played by progranulin during the development of the CNS has not been fully determined. The results of this study showed that progranulin played a key role in the survival of RGCs. These findings indicate that progranulin plays important roles during development of the retina.
Materials and Methods
Animals
Adult C57BL/6 mice (Japan SLC, Hamamatsu, Japan) and neonatal mice were maintained under controlled lighting environment (12 h/12 h light/dark cycle). Grn −/− mice generated by Kayasuga et al.32 were obtained from Riken BioResource Center (Tsukuba, Japan) and were backcrossed with C57BL/6 mice. All experiments were performed in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research, and the procedures were approved and monitored by the Institutional Animal Care and Use Committee of Gifu Pharmaceutical University.
Preparation of brain, eye, and optic nerve tissues
Mice were anesthetized with an intraperitoneal injection of 50 mg/kg sodium pentobarbital (Nacalai Tesque, Inc., Kyoto, Japan), perfused with saline and then immediately perfused with 4% paraformaldehyde (PFA) for 8 min. The brain, eye, and optic nerve were removed and further fixed in 4% PFA for 24 h at 4 °C. The tissues were immersed in 25% sucrose solution for 24 h at 4 °C and frozen in optimal cutting temperature compound (Sakura Finetechnical Co., Ltd., Tokyo, Japan). Finally, the brain (12 μm) and eye and optic nerve (10 μm) were sectioned and mounted on glass slides (MAS COAT; Matsunami Glass Ind., Ltd., Osaka, Japan).
Immunostaining
Immunostaining was performed according to the methods described in detail17. Briefly, the sections were blocked by non-immune serum and incubated overnight with the primary antibody at 4 °C. The mouse-on-mouse (M.O.M) immunodetection kit (Vector Labs, Burlingame, CA, USA) was used for blocking and solvents. After an overnight incubation with the primary antibody, the sections were incubated with the secondary antibody for 1 h. They were then counterstained and mounted.
For the Brn3a staining, the retinal sections were pre-treated with 0.1% trypsin (Wako Pure Chemical Industries, Ltd., Osaka, Japan) at 37 °C for 10 min and incubated with the Brn3a antibody [1:50 dilution: SantaCruz (Dallas, Texas, USA)] for 1 h at 37 °C.
The following primary antibodies were used: mouse anti-rhodopsin (1:1000 dilution: Millipore, Bedford, MA, USA), mouse anti-GS (1:1000 dilution: Millipore), mouse anti-GFAP (1:1000 dilution: SantaCruz), mouse anti-NFH (1:1000 dilution: Millipore), mouse anti-p-NFH (1:1000 dilution: Millipore), mouse anti-MBP (1:200 dilution: Millipore), mouse anti-CC-1 (1:100 dilution: Millipore), rabbit anti-Iba-1 (1:50 dilution: Wako), rabbit anti-S100β [1:200 dilution: Abcam (Cambridge, MA, USA)], rabbit anti-NFH (1:1000 dilution: Millipore), rabbit anti-CRX (1:20 dilution: SantaCruz), sheep anti-progranulin (1:5 dilution: R&D Systems, Minneapolis, MN, USA), goat anti-TNF-α (1:20 dilution: SantaCruz), rabbit anti-IL-1β (1:100 dilution: Abcam), Alexa Fluor®488 goat anti-mouse IgG, Alexa Fluor®488 goat anti-rabbit IgG, Alexa Fluor®488 donkey anti-goat IgG, Alexa Fluor®546 goat anti-mouse IgG, Alexa Fluor®546 donkey anti-rabbit IgG, and Alexa Fluor®647 donkey anti-sheep IgG (Invitrogen, Carlsbad, CA, USA).
Images were acquired with a confocal microscope (FLUOVIEW FV10i; Olympus, Tokyo, Japan). For quantitative data, photographs were taken at 500 µm from the optic nerve head (P9 and adult retina) and at the central retina (P1 retina). The fluorescent intensity was measured by ImageJ (National Institutes of Health, Bethesda, MD). The Brn3a+ cells were counted within the area of the image (211.968 × 211.968 µm). The density is presented as the number/mm. The number of Iba-1+ cells were counted within whole retinal sections.
Western blotting analysis
Western blotting was performed according to our methods described in detail17. Briefly, mice retinas were lysed using a buffer containing inhibitors of protease and phosphatase. The tissue was homogenized and the cell lysate was centrifuged. The supernatant was used for the subsequent experiments. The protein concentration was measured using a protein assay kit (Thermo Scientific, Waltham, MA, USA). Samples were analyzed by SDS-PAGE using 5–20% gradient gels (Wako), and the proteins were transferred onto a membrane. After blocking for 30 min at room temperature, the membranes were washed and then incubated with the primary antibody overnight at 4 °C. The following primary antibodies were used: mouse anti-rhodopsin (1:1000 dilution: Millipore), mouse anti-GS (1:1000 dilution: Millipore), mouse anti-GFAP (1:1000 dilution: SantaCruz), mouse anti-MET [1: 500 dilution, Cell Signaling Technology (Danvers, MA, USA)], rabbit anti-CRX (1:100 dilution: SantaCruz), rabbit anti-p-MET (1: 1000 dilution, Cell Signaling Technology), sheep anti-progranulin (1:100 dilution: R&D systems), and mouse anti-β-actin (1:2000 dilution: Sigma-Aldrich). After exposure to the primary antibody, the membranes were incubated with peroxidase goat anti-rabbit, goat anti-mouse or rabbit anti-sheep IgG (Thermo Scientific) as the secondary antibody. The immunoreactive bands were made visible with ImmunoStar LD (Wako).
Statistical analysis
The data are presented as the means ± standard error of the means (SEMs). The significance of the differences were determined by two-tailed Student’s t-test [STAT VIEW version 5.0 (SAS Institute, Cary, NC, USA)]. A P < 0.05 was considered statistically significant.
References
Freeman, M. R. Specification and morphogenesis of astrocytes. Science 330, 774–8, doi:10.1126/science.1190928 (2010).
Tao, C. & Zhang, X. Development of astrocytes in the vertebrate eye. Dev. Dyn. 243, 1501–1510, doi:10.1002/dvdy.v243.12 (2014).
Yonehara, K. et al. Expression of SPIG1 reveals development of a retinal ganglion cell subtype projecting to the medial terminal nucleus in the mouse. PLoS One 3 (2008).
Vecino, E., Rodriguez, F. D., Ruzafa, N., Pereiro, X. & Sharma, S. C. Progress in Retinal and Eye Research Glia e neuron interactions in the mammalian retina. Prog. Retin. Eye Res. 51, 1–40, doi:10.1016/j.preteyeres.2015.06.003 (2016).
Howell, G. R. et al. Axons of retinal ganglion cells are insulted in the optic nerve early in DBA/2J glaucoma. J. Cell Biol. 179, 1523–37, doi:10.1083/jcb.200706181 (2007).
Bialas, A. R. & Stevens, B. TGF-β signaling regulates neuronal C1q expression and developmental synaptic refinement. Nat. Neurosci. 16, 1773–82, doi:10.1038/nn.3560 (2013).
Clarke, L. E. & Barres, B. A. Emerging roles of astrocytes in neural circuit development. Nat. Rev. Neurosci. 14, 311–21, doi:10.1038/nrn3484 (2013).
Fruttiger, M. et al. PDGF mediates a neuron-astrocyte interaction in the developing retina. Neuron 17, 1117–31, doi:10.1016/S0896-6273(00)80244-5 (1996).
Wallace, V. a. & Raff, M. C. A role for Sonic hedgehog in axon-to-astrocyte signalling in the rodent optic nerve. Development 126, 2901–2909 (1999).
Olivera-Bravo, S. et al. Neonatal astrocyte damage is sufficient to trigger progressive striatal degeneration in a rat model of glutaric acidemia-I. PLoS One 6, e20831, doi:10.1371/journal.pone.0020831 (2011).
Song, H., Stevens, C. F. & Gage, F. H. Astroglia induce neurogenesis from adult neural stem cells. Nature 417, 39–44, doi:10.1038/417039a (2002).
Van Damme, P. et al. Progranulin functions as a neurotrophic factor to regulate neurite outgrowth and enhance neuronal survival. J. Cell Biol. 181, 37–41, doi:10.1083/jcb.200712039 (2008).
Tang, W. et al. The growth factor progranulin binds to TNF receptors and is therapeutic against inflammatory arthritis in mice. Science 332, 478–84, doi:10.1126/science.1199214 (2011).
Egashira, Y. et al. The growth factor progranulin attenuates neuronal injury induced by cerebral ischemia-reperfusion through the suppression of neutrophil recruitment. J. Neuroinflammation 10, 105, doi:10.1186/1742-2094-10-105 (2013).
Tsuruma, K. et al. Progranulin, a major secreted protein of mouse adipose-derived stem cells, inhibits light-induced retinal degeneration. Stem Cells Transl. Med. 3, 42–53, doi:10.5966/sctm.2013-0020 (2014).
Kanazawa, M. et al. Multiple therapeutic effects of progranulin on experimental acute ischaemic stroke. Brain 138, 1932–1948, doi:10.1093/brain/awv079 (2015).
Kuse, Y. et al. Progranulin promotes the retinal precursor cell proliferation and the photoreceptor differentiation in the mouse retina. Sci. Rep. 6, 23811, doi:10.1038/srep23811 (2016).
Krencika, R. & Zhang, S.-C. Directed Differentiation of Functional Astroglial Subtypes from Human Pluripotent Stem Cells. Nat Protoc. 6, 1710–1717, doi:10.1038/nprot.2011.405 (2011).
Molofsky, A. V. et al. Astrocytes and disease: a neurodevelopmental perspective. Genes Dev. 26, 891–907, doi:10.1101/gad.188326.112 (2012).
Lambert, W. S., Ruiz, L., Crish, S. D., Wheeler, L. A. & Calkins, D. J. Brimonidine prevents axonal and somatic degeneration of retinal ganglion cell neurons. Mol. Neurodegener. 6, 4, doi:10.1186/1750-1326-6-4 (2011).
Buckingham, B. P. et al. Progressive ganglion cell degeneration precedes neuronal loss in a mouse model of glaucoma. J Neurosci 28, 2735–2744, doi:10.1523/JNEUROSCI.4443-07.2008 (2008).
Harada, T., Harada, P. & Parada, L. F. Molecular regulation of visual system development: more than meets the eye. Genes Dev. 21, 367–378, doi:10.1101/gad.1504307 (2007).
Li, Y.-H. et al. Progranulin A-mediated MET signaling is essential for liver morphogenesis in zebrafish. J. Biol. Chem. 285, 41001–9, doi:10.1074/jbc.M110.138743 (2010).
Tönges, L. et al. Hepatocyte growth factor protects retinal ganglion cells by increasing neuronal survival and axonal regeneration in vitro and in vivo. J. Neurochem. 117, 892–903, doi:10.1111/jnc.2011.117.issue-5 (2011).
Wong, W.-K., Cheung, A. W.-S., Yu, S.-W., Sha, O. & Cho, E. Y. P. Hepatocyte growth factor promotes long-term survival and axonal regeneration of retinal ganglion cells after optic nerve injury: comparison with CNTF and BDNF. CNS Neurosci. Ther. 20, 916–29, doi:10.1111/cns.2014.20.issue-10 (2014).
Menzel, L. et al. Progranulin protects against exaggerated axonal injury and astrogliosis following traumatic brain injury. Glia 65, 278–292, doi:10.1002/glia.v65.2 (2017).
Petkau, T. L. et al. Progranulin expression in the developing and adult murine brain. J. Comp. Neurol. 518, 3931–3947, doi:10.1002/cne.22430 (2010).
Lü, L. et al. Progranulin expression in neural stem cells and their differentiated cell lineages: an immunocytochemical study. Mol. Med. Rep. 8, 1359–64, doi:10.3892/mmr.2013.1664 (2013).
Daniel, R., Daniels, E., He, Z. & Bateman, A. Progranulin (acrogranin/PC cell-derived growth factor/granulin-epithelin precursor) is expressed in the placenta, epidermis, microvasculature, and brain during murine development. Dev. Dyn. 227, 593–9, doi:10.1002/dvdy.10341 (2003).
Daniel, R., He, Z., Carmichael, K. P., Halper, J. & Bateman, A. Cellular localization of gene expression for progranulin. J. Histochem. Cytochem. 48, 999–1009, doi:10.1177/002215540004800713 (2000).
Petkau, T. L. & Leavitt, B. R. Progranulin in neurodegenerative disease. Trends in Neurosciences 37, 388–398, doi:10.1016/j.tins.2014.04.003 (2014).
Kayasuga, Y. et al. Alteration of behavioural phenotype in mice by targeted disruption of the progranulin gene. Behav. Brain Res. 185, 110–118, doi:10.1016/j.bbr.2007.07.020 (2007).
Author information
Authors and Affiliations
Contributions
Y.K., K.T., T.M., M.S. and H.H. conceived and designed the experiments. Y.K. and T.M. performed the analysis and the experiments. Y.K., K.T. and H.H. wrote the paper. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kuse, Y., Tsuruma, K., Mizoguchi, T. et al. Progranulin deficiency causes the retinal ganglion cell loss during development. Sci Rep 7, 1679 (2017). https://doi.org/10.1038/s41598-017-01933-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-01933-8
This article is cited by
-
Temporal single-cell atlas of non-neuronal retinal cells reveals dynamic, coordinated multicellular responses to central nervous system injury
Nature Immunology (2023)
-
Progranulin deficiency in Iba-1+ myeloid cells exacerbates choroidal neovascularization by perturbation of lysosomal function and abnormal inflammation
Journal of Neuroinflammation (2021)
-
VGF nerve growth factor inducible is involved in retinal ganglion cells death induced by optic nerve crush
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.