Abstract
Hyperinsulinemia observed in obese subject is caused at least in part by low metabolic clearance rate of insulin (MCRI). However, the determinants of MCRI in non-obese subjects are not fully understood. To investigate the correlates of MCRI in healthy non-obese men (BMI <25 kg/m2), we studied 49 non-obese Japanese men free of cardiometabolic risk factors. Using a 2-step hyperinsulinemic euglycemic clamp, we evaluated MCRI and insulin sensitivity. We also calculated the rate of glucose disappearance (Rd) during the clamp and muscle insulin sensitivity was defined as Rd/steady state serum insulin (SSSI) at the second step. Based on the median value of MCRI, the subjects were divided into the low- and high-MCRI groups. Subjects of the low-MCRI group had significant impairment of muscle insulin sensitivity, although Rd levels were comparable between the two groups, probably due to elevated SSSI in the low-MCRI group. Subjects of the low-MCRI group had higher total body fat content and lower VO2peak and showed no deterioration of cardiometabolic risk factors. Our results suggest that low MCRI may be early change to maintain glucose uptake and metabolic status in the face of slight impairment of muscle insulin sensitivity caused by increased adiposity and lower fitness level.
Similar content being viewed by others
Introduction
Obesity and the associated metabolic abnormalities are worldwide health problems. Insulin resistance and coexistent hyperinsulinemia are important in the pathogenesis of type 2 diabetes mellitus (T2DM) and metabolic abnormalities in obese subjects1. Insulin resistance can be compensated by hyperinsulinemia; the latter is not only due to enhanced insulin secretion, but low insulin clearance. In fact, the latter was found to be associated with increased serum insulin level in overweight and obese subjects2, 3. In addition, low insulin clearance correlates with metabolic abnormalities4 and insulin resistance5, 6 in nondiabetic overweight and obese subjects. Thus, low insulin clearance seems to be a feature of obese subjects with insulin resistance and metabolic syndrome4.
Several studies demonstrated changes in insulin clearance even in non-obese subjects and that such change could be the initial event predating insulin resistance and obesity7, 8. In animal studies, healthy dogs fed high-fat diet for 12 weeks showed moderate accumulation of visceral fat without changes in body weight and mild insulin resistance accompanied by hyperinsulinemia7. In this setting, decreased hepatic insulin clearance rather than increased insulin secretion was the major determinant of the hyperinsulinemia7, 9. Similarly, a human study also demonstrated that intentional weight gain within the normal range (from BMI of 21.8 to 23.8 kg/m2) resulted in impaired insulin sensitivity and hyperinsulinemia and that the hyperinsulinemia was mainly due to reduced insulin clearance rather than enhanced insulin secretion8. Thus, insulin clearance can decrease during modest weight gain even when it is within the normal range and may be secondary to initial insulin resistance or can be the initial factor causing impaired insulin sensitivity and adiposity. However, impaired insulin clearance observed in the above human study in non-obese individuals8 occurred when body weight was acutely increased by overfeeding (6.2 kg over a 10-week period) and glucose disposal decreased. To our knowledge, there is little or no information on the factors that determine insulin clearance in apparently healthy non-obese subjects who do not undergo dietary intervention. In addition, insulin sensitivity could be impaired in muscle and liver in non-obese subjects, independently10; however, it has also not been determined which site of insulin resistance is more associated with changes in insulin clearance.
Based on the above background, the present study was designed to identify the clinical correlates of insulin clearance in apparently healthy non-obese subjects. For this purpose, we recruited healthy non-obese (<25 kg/m2) Japanese men without any cardiometabolic risk factors to measure insulin clearance and insulin resistance in muscle and liver by a 2-step euglycemic hyperinsulinemic clamp. The results were analyzed for the association of various metabolic parameters with insulin clearance.
Results
Anthropometric characteristics of low- and high- metabolic clearance rate of insulin (MCRI) groups
Table 1 summarizes the clinical characteristics of the study subjects. The mean values of the entire group for cardiometabolic risk factors, fasting serum insulin (FSI), homeostasis model assessment of insulin resistance (HOMA-IR) and other parameters were within the normal ranges. For further analysis, we divided the subjects into low-MCRI group (n = 25) and high-MCRI group (n = 24), based on the median value of MCRI [594.1 (561.3–663.2) ml/min/m2]. As shown in Table 1, subcutaneous fat area and total fat content were significantly higher, while visceral fat area tended to be higher, in the low-MCRI group than the high-MCRI group. In addition, VO2peak was significantly lower in the low-MCRI group than the high-MCRI group. Although fasting C-peptide was comparable between the two groups, the FSI was significantly higher in the low-MCRI group than the high-MCRI group probably due to the difference in insulin clearance between the two groups. While FPG levels were comparable between the two groups, HOMA-IR was higher in the low-MCRI group than the high-MCRI group. Similarly, during oral glucose tolerance test (OGTT), the insulin level was higher and MATSUDA index was lower in the low-MCRI group compared with the high-MCRI group. The above data suggest that subjects of the low-MCRI group were characterized by moderate fat accumulation, low fitness level, higher insulin level, and impaired insulin sensitivity, compared to those of the high-MCRI group. Although these features seem to be associated with deterioration in cardiometabolic risk factors, such as hypertension, dyslipidemia and hyperglycemia, even in non-obese subjects with BMI of 23–25 kg/m2 10, the cardiometabolic risk factors were comparable between the two groups (Table 1).
Insulin sensitivity in muscle and liver evaluated by glucose clamp
We evaluated insulin sensitivity in muscle and liver by the gold standard method; the 2-step hyperinsulinemic euglycemic clamp (Table 2). The steady-state serum insulin (SSSI) level during glucose clamp was higher in the low-MCRI group at the first and second steps compared with the high-MCRI group. Nevertheless, the rate of disappearance of glucose (Rd) levels at both the first and second steps were comparable between the two groups, suggesting impaired insulin sensitivity in peripheral tissue (mainly muscle) in subjects of the low-MCRI group. Since Rd is known to be enhanced in parallel with serum insulin concentration11, we divided Rd by SSSI at the second step and used the product as an index of muscle insulin sensitivity12. As shown in Table 2, muscle insulin sensitivity (Rd/SSSI at the second step) was significantly lower in the low-MCRI group than in the high-MCRI group. These data suggest impaired muscle insulin sensitivity in the low-MCRI group; although Rd during glucose clamp was maintained by increased SSSI due to decreased MCRI.
Similar to Rd, endogenous glucose production (EGP) is also known to be suppressed in accordance with insulin concentration at low insulin levels (~20 μU/ml)11. Accordingly, we divided the %reduction of EGP by SSSI at the first step and used it as an index of hepatic insulin sensitivity13. As shown in Table 2, hepatic insulin sensitivity was comparable between the two groups. In addition, the basal EGP level was significantly lower in the low-MCRI group than in the high-MCRI group, probably due to higher insulin level in the low-MCRI group.
Correlations between MCRI and other parameters
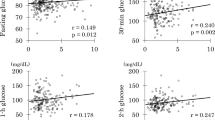
To further investigate the association of MCRI with various metabolic parameters, correlation analysis was performed between MCRI and several metabolic parameters (Table 3). In this analysis, parameters with p values less than 0.1 shown in Table 1 and glucose clamp data shown in Table 2 were selected. MCRI correlated significantly with body fat content and subcutaneous fat area (SFA), but not with the visceral fat area (VFA). In addition, MCRI correlated significantly with both FSI and AUC-insulin during OGTT, again suggesting the contribution of MCRI to hyperinsulinemia at fasting and during OGTT. The results also showed a significant correlation between MCRI and VO2peak.
MCRI correlated negatively with basal EGP and %reduction of EGP at the first step, but not with hepatic insulin sensitivity (%reduction of EGP/SSSI at the first step). In contrast, MCRI did not correlate with Rd at the first and second steps, but correlated significantly with muscle insulin sensitivity (Rd/SSSI at the second step).
Discussion
We investigated the clinical correlates of insulin clearance in non-obese Japanese men free of cardiometabolic risk factors. The results showed that subjects of the low-MCRI group exhibited moderate fat accumulation, low fitness level, high insulin level and impaired insulin sensitivity in muscle, but not in liver, compared with the high-MCRI group. In subjects of the low-MCRI group, the Rd level during the glucose clamp was maintained by elevated SSSI level and no deterioration of cardiometabolic risk factors was observed.
Previous studies suggested that low MCRI values were associated with obesity14, 15, insulin resistance5, 6 and cardiometabolic risk factors4, 16, 17. However, there is little or no information on the factors that determine insulin clearance in apparently healthy non-obese subjects. This is in part because sequential 2-step hyperinsulinemic euglycemic clamp study, the gold standard method to precisely evaluate hepatic and muscle insulin sensitivity, has not been applied previously for a large number of apparently healthy non-obese subjects. The present study demonstrated impaired muscle insulin sensitivity (Rd/SSSI at the second step) in the low-MCRI group compared with the high-MCRI group; however, hepatic insulin sensitivity and cardiometabolic risk factors were comparable between the two groups. In addition, previous studies demonstrated that decreased MCRI is observed in obese subjects with decreased glucose utilization during glucose clamp14, 15; however in the present study, despite impaired muscle insulin sensitivity in subjects of the low-MCRI group, the Rd at the second step during the glucose clamp test did not decrease probably due to elevated serum insulin concentration induced by low MCRI (Table 2). These data suggest that MCRI is well correlated to insulin resistance in muscle, but not in liver. In addition, the data also suggest that the insulin resistance in muscle seen in the low-MCRI group was mild, thus increased serum insulin concentrations induced by lower MCRI was sufficient to maintain the Rd level. Thus, one can hypothesize that in non-obese apparently healthy subjects, low MCRI is a compensatory mechanism to maintain muscle glucose uptake and metabolic status in the face of minuscule impairment of insulin sensitivity in muscle.
On the other hand, it has been shown that low MCRI could be a primary change for insulin resistance. Indeed, in several animal models impaired insulin clearance induced an insulin resistant state18, 19. For example, carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1) promotes receptor-mediated insulin uptake and degradation in hepatocytes18, 19. Liver-specific inactivation or global null-mutation of CEACAM1 impaired hepatic insulin clearance and induced chronic hyperinsulinemia, resulting in insulin resistance and adiposity18, 19. In addition, high-fat diet diminished CEACAM1 expression and induced impaired insulin clearance, hyperinsulinemia and insulin resistance in C57/BL6J mice, and these effects were counteracted by liver-specific inducible CEACAM1 expression19. These data suggest that impaired insulin clearance could be the primary culprit in insulin resistant states. Furthermore, low MCRI can also induce insulin resistance via hyperinsulinemia, such as that seen after insulin-stimulated fat accumulation, downregulation of insulin receptor20 and by post receptor mechanisms such as serine-threonine phosphorylation by insulin-stimulated Erk or S6K21. Thus, it is still unclear whether low MCRI is a consequence of slightly impaired insulin sensitivity or a cause of insulin resistance and adiposity. The existence of a vicious cycle between insulin resistance and low insulin clearance renders the search for the primary defect difficult.
Although the mechanisms of how low MCRI can cause insulin resistance have been reported as mentioned above, the mechanisms of how insulin resistance can cause a decrease in MCRI are less known. One possibility is that certain factors that influence both MCRI and muscle insulin sensitivity may mediate a link between MCRI and insulin sensitivity in muscle. For example, previous studies showed that free fatty acids (FFA) do not only induce muscle insulin resistance22, but also decrease MCRI23, 24. It has been also suggested that FFA is a major source of intrahepatic lipid (IHL) accumulation25 and fatty liver is associated with decreased MCRI26. Thus, FFA and IHL accumulation can be key factors linking MCRI to insulin resistance; however, both FFA and IHL accumulation were not associated with MCRI in the present study. Another possibility is that muscle activity by aerobic exercise may mediate the association between muscle insulin sensitivity and MCRI. For example, one-year aerobic exercise (jogging) enhanced muscle insulin sensitivity in healthy young men27; however, glucose infusion rate during glucose clamp was not increased as anticipated, because aerobic exercise greatly increased MCRI by 87% and reduced SSSI during glucose clamp. A recent animal study indicated that one bout of exercise increased insulin clearance in parallel with increased expression of insulin degrading enzyme in the liver and skeletal muscle28. Taken together, we hypothesize that aerobic exercise enhances both muscle insulin sensitivity and MCRI and mediates the link between these factors. Given that aerobic exercise enhances fitness level (VO2peak)29, our observation that MCRI correlated significantly with VO2peak is in agreement with this hypothesis.
The present study has several limitations. First, we did not perform a pancreatic clamp to completely suppress endogenous insulin secretion throughout the experiment (basal period and clamp). To take this fact into consideration, we adjusted the calculation of MCRI according to the suppression of C-peptide level using a previously published method30, 31. Second, we recruited only Japanese in this study, thus our results may not be applicable to other ethnic groups. In fact, previous reports suggested that insulin clearance is lower in South Asians than East Asians and Caucasians32. Third, we included men only in this study though metabolism and fat distribution are known to be different between men and women. In addition, we only included middle aged men (30–50 y.o.). Previous study reported decreased MCRI in elderly compared with young33, while age was not associated with MCRI in the present study. Finally and most importantly, the present study is cross-sectional and thus cannot address causality.
In conclusion, in non-obese apparently healthy Japanese men, low MCRI was associated with impaired muscle insulin sensitivity, lower fitness level and increased adiposity. However, the Rd level was maintained during glucose clamp test through elevated steady state serum insulin, and there was no increase in cardiometabolic risk factors. These data suggest that in non-obese apparently healthy men, low MCRI could be viewed as an early change observed with modest insulin resistance and could be a compensatory phenomenon to maintain glucose uptake and metabolic status in the face of slightly impaired insulin sensitivity in muscle. However, which of the two factors occurs first, insulin resistance or decreased insulin clearance, is a question that requires further longitudinal studies.
Research Design and Methods
Study subjects
The MCRI was assessed in participants of the Sportology Center Core Study, a prospective observational study to support hypothesis-driven, hypothesis-generating research on the underlying mechanisms of metabolic abnormalities in non-obese subjects10. In the study, we recruited non-diabetic Japanese men with BMI of 21 to 27.5 kg/m2 (≥21.0 to <27.5 kg/m2), aged between 30 and 50 years. The study participants were described in detail previously10. To assess the role of MCRI in apparently healthy non-obese men, we selected from the cohort those with BMI of 21 to 25 kg/m2 (≥21.0 to <25.0 kg/m2) who were free of cardiometabolic risk factors [based on the definition of metabolic syndrome in Japan34, we used three cardiometabolic risk factors, including hyperglycemia (fasting plasma glucose (FPG) ≥110 mg/dl), dyslipidemia (triglycerides (TG) ≥150 mg/dl and/or high-density lipoprotein-C (HDL-C) <40 mg/dl) and hypertension (systolic blood pressure (SBP) ≥130 mmHg and/or diastolic BP (DBP) ≥85 mmHg)] (n = 52). Among the selected group, the values of C-peptide were not available in 3 participants, who were, accordingly, excluded from the group. Thus, in the present study, we analyzed the baseline data of 49 men (Table 1). All participants gave written informed consent to the study, which was approved by the ethics committee of Juntendo University. This study was carried out in accordance with the principles outlined in the Declaration of Helsinki.
Study design
The design of the Sportology Center Core Study was described previously in detail10. Briefly, after the screening session, all participants visited our institute three times for baseline evaluation. At the first or second visit, each participant underwent OGTT or peak oxygen uptake test as described previously10, 35. The participants were instructed to quit regular exercise for 10 days before the third visit and the mean daily physical activity level was evaluated over 7 days with an accelerometer (Lifecorder; Suzuken, Nagoya, Japan). Then, each participant was asked to maintain daily physical activity at mean daily physical activity level ±10% during the last 3 days, which was monitored by an accelerometer.
On the day of the experiment, we measured intramyocellular lipid (IMCL) and IHL by 1H-magnetic resonance spectroscopy (MRS) after an overnight fast. Total body fat content and fat-free mass (FFM) were measured by the bioimpedance method (InBody; Biospace, Tokyo)36. Furthermore, VFA and SFA were also estimated by magnetic resonance imaging (MRI). Then, euglycemic hyperinsulinemic clamp was performed to measure insulin sensitivity in muscle and liver (see below). Surrogate markers of insulin resistance [i.e., HOMA-IR and Matsuda index] were calculated as described previously10, 37.
Euglycemic hyperinsulinemic glucose clamp
The participants were instructed to consume a weight-maintaining standard diet on the 3 days immediately preceding the clamp study. In addition, they were asked to refrain from alcohol the day before the clamp study. After an overnight fast, a two-step euglycemic hyperinsulinemic glucose clamp study was performed with an artificial endocrine pancreas (STG 22; Nikkiso, Shizuoka, Japan)10. Briefly, after securing an intravenous cannula in the forearm, a bolus dose [200 mg/m2 body surface area (BSA)] of [6,6-2H2]glucose (Cambridge Isotope Laboratories, Tewksbury, MA) was injected intravenously, followed by constant infusion of 2 mg/m2 BSA per min for 3-h (−180 to 0 min) to measure fasting EGP38. This was followed by primed insulin infusion (40 mU/m2 per min followed by 20 mU/m2 per min; each lasting 5 min) and continuous insulin infusion at 10 mU/m2 per min for 3 hours (first step) (0 to 180 min). In the second step of the clamp, after a priming insulin infusion (80 mU/m2 per min followed by 40 mU/m2 per min; each lasted 5 min), insulin was infused continuously at 20 mU/m2 per min for 3 hours (180 to 360 min). The infusion of [6,6-2H2]glucose was decreased by 75% of the initial infusion rate during the first step and 85% of the basal rate during the second step of the clamp to maintain constant plasma glucose enrichment39. We used a warming blanket for arterialization of hand vein. Plasma glucose level in arterialized blood was maintained at ~95 mg/dl by a variable 20% glucose infusion containing ~2.5% [6,6-2H2]glucose. Blood samples were withdrawn for biochemical analysis at 10 min intervals during the last 30 min before the clamp and during the steady state periods of the first and second steps of the clamp. Enrichment of [6,6-2H2]glucose in plasma was measured by high-performance liquid chromatography (LTQ-XL-Orbitrap mass spectrometer, Therm Scientific, CA) as described previously10. A steady state equation was used to calculate the rates of EGP and Rd at each step40. EGP and Rd were normalized by BSA and FFM, respectively10. We divided % reduction of EGP at the first step by SSSI and used it as an index of hepatic insulin sensitivity13. Similarly, Rd at the second step was divided by SSSI and used as an index of muscle insulin sensitivity12.
Calculation of metabolic clearance rate for serum insulin (MCRI)
The MCRI during glucose clamp at the second step was calculated by the following equation30: MCRI = {IIR/[SSSI − (BSI*SSSC/BSC)]}, where IIR = insulin infusion rate, SSSI = steady state serum insulin during glucose clamp, BSI = basal serum insulin, SSSC = steady-state serum C-peptide during glucose clamp, BSC = basal serum C-peptide. The C-peptide data were entered to take into account the suppression of endogenous insulin secretion by the exogenous insulin.
1H-MRS
The IMCL values of the right tibialis anterior (TA) and soleus (SOL) muscles and IHL of segment 6 in the liver were measured by 1H-MRS (VISART EX V4.40, Toshiba, Tokyo)41, 42. After the measurements, IMCL was quantified by methylene signal intensity (S-fat) using creatine signal (Cre) as the reference and calculated as the ratio S-fat/Cre. IHL was quantified by S-fat with H2O as the internal reference, and calculated as a percentage of H2O + S-fat [S-fat × 100/(H2O + S-fat)]41, 42.
Intra-abdominal and subcutaneous fat areas
Intra-abdominal and subcutaneous fat areas were measured with MRI as described previously42. Briefly, T1-weighted trans-axial scans were obtained and intra-abdominal and subcutaneous fat areas at the fourth and fifth lumbar interspaces were measured as described previously by using specific software (AZE Virtual Place, Tokyo, Japan)42.
Statistical analysis
Data are presented as mean ± SD or median (range: 25–75%). To approximate normal distribution, log-transformed values were used in the analysis, as appropriate. Data of two groups were compared by unpaired Student t-test. The relation between MCRI at the second step and various metabolic parameters were assessed by Pearson or Spearman correlation coefficient, as appropriate. All statistical tests were two-sided with 5% significance level.
References
Reaven, G. M. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 37, 1595–1607, doi:10.2337/diabetes.37.12.1595 (1988).
Meistas, M. T., Margolis, S. & Kowarski, A. A. Hyperinsulinemia of obesity is due to decreased clearance of insulin. Am J Physiol 245, E155–159 (1983).
Peiris, A. N., Mueller, R. A., Smith, G. A., Struve, M. F. & Kissebah, A. H. Splanchnic insulin metabolism in obesity. Influence of body fat distribution. J Clin Invest 78, 1648–1657, doi:10.1172/JCI112758 (1986).
Pivovarova, O. et al. Hepatic insulin clearance is closely related to metabolic syndrome components. Diabetes care 36, 3779–3785, doi:10.2337/dc12-1203 (2013).
Kim, M. K., Reaven, G. M., Chen, Y. D., Kim, E. & Kim, S. H. Hyperinsulinemia in individuals with obesity: Role of insulin clearance. Obesity 23, 2430–2434, doi:10.1002/oby.21256 (2015).
Goodarzi, M. O. et al. Fasting insulin reflects heterogeneous physiological processes: role of insulin clearance. American journal of physiology. Endocrinology and metabolism 301, E402–408, doi:10.1152/ajpendo.00013.2011 (2011).
Mittelman, S. D. et al. Longitudinal compensation for fat-induced insulin resistance includes reduced insulin clearance and enhanced beta-cell response. Diabetes 49, 2116–2125, doi:10.2337/diabetes.49.12.2116 (2000).
Erdmann, J. et al. Development of hyperinsulinemia and insulin resistance during the early stage of weight gain. American journal of physiology 294, E568–575, doi:10.1152/ajpendo.00560.2007 (2008).
Ader, M. et al. Hepatic insulin clearance is the primary determinant of insulin sensitivity in the normal dog. Obesity 22, 1238–1245, doi:10.1002/oby.20625 (2014).
Takeno, K. et al. Relation between insulin sensitivity and metabolic abnormalities in Japanese men with BMI of 23–25 kg/m2. J Clin Endocrinol Metab 101, 3676–3684, doi:10.1210/jc.2016-1650 (2016).
Groop, L. C. et al. Glucose and free fatty acid metabolism in non-insulin-dependent diabetes mellitus. Evidence for multiple sites of insulin resistance. J Clin Invest 84, 205–213, doi:10.1172/JCI114142 (1989).
Abdul-Ghani, M. & DeFronzo, R. A. Fasting hyperglycemia impairs glucose- but not insulin-mediated suppression of glucagon secretion. J Clin Endocrinol Metab 92, 1778–1784, doi:10.1210/jc.2006-1515 (2007).
Kotronen, A., Juurinen, L., Tiikkainen, M., Vehkavaara, S. & Yki-Jarvinen, H. Increased liver fat, impaired insulin clearance, and hepatic and adipose tissue insulin resistance in type 2 diabetes. Gastroenterology 135, 122–130, doi:10.1053/j.gastro.2008.03.021 (2008).
Zuniga-Guajardo, S., Jimenez, J., Angel, A. & Zinman, B. Effects of massive obesity on insulin sensitivity and insulin clearance and the metabolic response to insulin as assessed by the euglycemic clamp technique. Metabolism 35, 278–282, doi:10.1016/0026-0495(86)90214-3 (1986).
Marini, M. A. et al. Differences in insulin clearance between metabolically healthy and unhealthy obese subjects. Acta Diabetol 51, 257–261, doi:10.1007/s00592-013-0511-9 (2014).
Salvatore, T. et al. Decreased insulin clearance as a feature of essential hypertension. The Journal of clinical endocrinology and metabolism 74, 144–149, doi:10.1210/jcem.74.1.1727814 (1992).
Bonora, E., Zavaroni, I., Coscelli, C. & Butturini, U. Decreased hepatic insulin extraction in subjects with mild glucose intolerance. Metabolism 32, 438–446, doi:10.1016/0026-0495(83)90004-5 (1983).
Poy, M. N. et al. CEACAM1 regulates insulin clearance in liver. Nature genetics 30, 270–276, doi:10.1038/ng840 (2002).
Al-Share, Q. Y. et al. Forced Hepatic Overexpression of CEACAM1 Curtails Diet-Induced Insulin Resistance. Diabetes 64, 2780–2790, doi:10.2337/db14-1772 (2015).
Virkamaki, A., Ueki, K. & Kahn, C. R. Protein-protein interaction in insulin signaling and the molecular mechanisms of insulin resistance. The Journal of clinical investigation 103, 931–943, doi:10.1172/JCI6609 (1999).
Copps, K. D. & White, M. F. Regulation of insulin sensitivity by serine/threonine phosphorylation of insulin receptor substrate proteins IRS1 and IRS2. Diabetologia 55, 2565–2582, doi:10.1007/s00125-012-2644-8 (2012).
Bachmann, O. P. et al. Effects of intravenous and dietary lipid challenge on intramyocellular lipid content and the relation with insulin sensitivity in humans. Diabetes 50, 2579–2584, doi:10.2337/diabetes.50.11.2579 (2001).
Wiesenthal, S. R. et al. Free fatty acids impair hepatic insulin extraction in vivo. Diabetes 48, 766–774, doi:10.2337/diabetes.48.4.766 (1999).
Hennes, M. M., Dua, A. & Kissebah, A. H. Effects of free fatty acids and glucose on splanchnic insulin dynamics. Diabetes 46, 57–62, doi:10.2337/diab.46.1.57 (1997).
Donnelly, K. L. et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest 115, 1343–1351, doi:10.1172/JCI23621 (2005).
Kotronen, A., Vehkavaara, S., Seppala-Lindroos, A., Bergholm, R. & Yki-Jarvinen, H. Effect of liver fat on insulin clearance. American journal of physiology. Endocrinology and metabolism 293, E1709–1715, doi:10.1152/ajpendo.00444.2007 (2007).
Oshida, Y., Yamanouchi, K., Hayamizu, S. & Sato, Y. Long-term mild jogging increases insulin action despite no influence on body mass index or VO2 max. Journal of applied physiology 66, 2206–2210 (1989).
Kurauti, M. A. et al. Acute Exercise Improves Insulin Clearance and Increases the Expression of Insulin-Degrading Enzyme in the Liver and Skeletal Muscle of Swiss Mice. PloS one 11, e0160239, doi:10.1371/journal.pone.0160239 (2016).
Wenger, H. A. & Bell, G. J. The interactions of intensity, frequency and duration of exercise training in altering cardiorespiratory fitness. Sports medicine 3, 346–356, doi:10.2165/00007256-198603050-00004 (1986).
Elahi, D. et al. Feedback Inhibition of Insulin Secretion by Insulin : Relation to the Hyperinsulinemia of Obesity. The New England journal of medicine 306, 1196–1202, doi:10.1056/NEJM198205203062002 (1982).
Vasquez, B., Reaven, G. M. & Andrews, W. J. Elevated in vivo insulin clearance in pima indians with non-insulin-dependent diabetes mellitus. Diabetes 34, 671–676, doi:10.2337/diab.34.7.671 (1985).
Liew, C. F., Seah, E. S., Yeo, K. P., Lee, K. O. & Wise, S. D. Lean, nondiabetic Asian Indians have decreased insulin sensitivity and insulin clearance, and raised leptin compared to Caucasians and Chinese subjects. Int J Obes Relat Metab Disord 27, 784–789, doi:10.1038/sj.ijo.0802307 (2003).
Minaker, K. L., Rowe, J. W., Tonino, R. & Pallotta, J. A. Influence of age on clearance of insulin in man. Diabetes 31, 851–855, doi:10.2337/diab.31.10.851 (1982).
Matsuzawa, Y. Metabolic syndrome – definition and diagnostic criteria in Japan. J Jpn Soc Intern Med 94, 188–203, doi:10.5551/jat.12.301 (2005).
Nishitani, M. et al. Impact of diabetes on muscle mass, muscle strength, and exercise tolerance in patients after coronary artery bypass grafting. J Cardiol 58, 173–180, doi:10.1016/j.jjcc.2011.05.001 (2011).
Shafer, K. J., Siders, W. A., Johnson, L. K. & Lukaski, H. C. Validity of segmental multiple-frequency bioelectrical impedance analysis to estimate body composition of adults across a range of body mass indexes. Nutrition 25, 25–32, doi:10.1016/j.nut.2008.07.004 (2009).
Matsuda, M. & DeFronzo, R. A. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22, 1462–1470, doi:10.2337/diacare.22.9.1462 (1999).
Kelley, D. E., McKolanis, T. M., Hegazi, R. A., Kuller, L. H. & Kalhan, S. C. Fatty liver in type 2 diabetes mellitus: relation to regional adiposity, fatty acids, and insulin resistance. American journal of physiology 285, E906–916, doi:10.1152/ajpendo.00117.2003 (2003).
Lindegaard, B. et al. Inhibition of lipolysis stimulates peripheral glucose uptake but has no effect on endogenous glucose production in HIV lipodystrophy. Diabetes 56, 2070–2077, doi:10.2337/db07-0144 (2007).
Steele, R. Influences of glucose loading and of injected insulin on hepatic glucose output. Ann N Y Acad Sci 82, 420–430, doi:10.1111/j.1749-6632.1959.tb44923.x (1959).
Tamura, Y. et al. Effects of diet and exercise on muscle and liver intracellular lipid contents and insulin sensitivity in type 2 diabetic patients. J Clin Endocrinol Metab 90, 3191–3196, doi:10.1210/jc.2004-1959 (2005).
Sato, F. et al. Effects of diet-induced moderate weight reduction on intrahepatic and intramyocellular triglycerides and glucose metabolism in obese subjects. J Clin Endocrinol Metab 92, 3326–3329, doi:10.1210/jc.2006-2384 (2007).
Acknowledgements
We thank Mrs Mutsuko Yoshikawa, Miyuki Iwagami, Naoko Daimaru, Eriko Magoshi, and Emi Miyazawa for the excellent technical assistance. We also thank Hikari Taka and Tsutomu Fujimura (Juntendo University) for performing LC-MS analysis. High Technology Research Center Grant, Strategic Research Foundation at Private Universities and KAKENHI (Grants-in-Aid for Young Scientists (A), #23680069) from the Ministry of Education, Culture, Sports, Science and Technology of Japan, Japan Diabetes Foundation, Suzuken Memorial Foundation, Mitsukoshi Welfare Foundation, and Diabetes Masters Conference.
Author information
Authors and Affiliations
Contributions
H.K., K.T. and Y.T. researched the data and contributed to study design, data collection, interpretation of results, wrote and edited the manuscript. S.K., T.F., Y.F., M.N.-Y. and K.S. participated in data collection, data analysis and contributed to the discussion. H.D., S.A., A.G., A.K., and R.K. contributed to the discussion. H.W. contributed to study design, reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kaga, H., Tamura, Y., Takeno, K. et al. Correlates of insulin clearance in apparently healthy non-obese Japanese men. Sci Rep 7, 1462 (2017). https://doi.org/10.1038/s41598-017-01469-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-01469-x
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.