Abstract
Portfolio analysis is a fundamental practice of organizational leadership and is a necessary precursor of strategic planning. Successful application requires a highly detailed model of research options. We have constructed a model, the first of its kind, that accurately characterizes these options for the biomedical literature. The model comprises over 18 million PubMed documents from 1996–2019. Document relatedness was measured using a hybrid citation analysis + text similarity approach. The resulting 606.6 million document-to-document links were used to create 28,743 document clusters and an associated visual map. Clusters are characterized using metadata (e.g., phrases, MeSH) and over 20 indicators (e.g., funding, patent activity). The map and cluster-level data are embedded in Tableau to provide an interactive model enabling in-depth exploration of a research portfolio. Two example usage cases are provided, one to identify specific research opportunities related to coronavirus, and the second to identify research strengths of a large cohort of African American and Native American researchers at the University of Michigan Medical School.
Measurement(s) | Biomedical Research • document relatedness |
Technology Type(s) | digital curation • machine learning • Cluster Analysis • text similarity • citation analysis |
Machine-accessible metadata file describing the reported data: https://doi.org/10.6084/m9.figshare.13010537
Similar content being viewed by others
Background & Summary
Portfolio analysis is a common practice in the finance world where options (e.g., stocks, bonds) are well defined. Portfolio analysis is also being increasingly done in research institutions. It is a more difficult problem here, however, because research options – the topics of research – are not rigorously defined. Research administrators thus often have a somewhat cloudy view of their institution’s research activity which means that their visions and missions are difficult to translate into plans involving concrete choices. Due to ambiguity in the definition of research options, evaluation of potential responses to societal and economic pressures are likewise ambiguous.
Fuzzy descriptions of research options are now being replaced by highly detailed and accurate models of the scientific literature. For instance, tens of millions of documents in the Scopus database have been grouped into 91,000 document clusters using extended direct citation among documents1, a process that has been shown to create coherent clusters2,3,4. This same process was replicated and made available in Elsevier’s SciVal product where document clusters (called “Topics”) are now used by many institutions for portfolio analysis and research decision-making5. The most recent versions of the Leiden Ranking6, developed by the Centre for Science and Technology Studies (CWTS) at Leiden University, are based on a model of science that consists of 4,535 document clusters7 (referred to as micro-level fields) partitioned from the citation network. While these previous works made use of subscription-based citation databases (Scopus and the Web of Science), the goal of this work was to create a similarly accurate model based on the (openly available) PubMed literature for strategic decision-making in biomedical research.
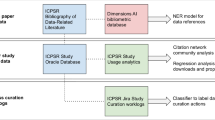
Figure 1 illustrates the process used to create the model along with the resulting framework. The first major step is the creation of a detailed model of science. Using over 18 million PubMed records from 1996–2019, each with a PubMed identifier (PMID), we retrieved similar article (SA) scores using an Entrez e-utility and citation links from the OCC8 and COCI9 databases. After merging, we used the resulting 606.6 million document-document links to cluster the documents into 28,889 clusters using the Leiden algorithm10. The second major step is to characterize each cluster using the document level metadata along with US patent reference data, US National Institutes of Health (NIH) and National Science Foundation (NSF) project data from Star Metrics, paper-to-project link tables from NIH ExPORTER, and additional metrics from the NIH iCite2.0 database11,12. We also created a visual map of the clusters using the OpenOrd layout routine13 and cluster-level relatedness. Some clusters were removed from the model at this point. Finally, we loaded the resulting cluster-level metadata, indicators and cluster positions into Excel and Tableau workbooks; the Excel workbook makes the data readily available for re-use while the Tableau interface enables visual exploration and filtering of the model for detailed analysis.
This model is the first highly granular characterization of the PubMed literature that can be used for portfolio analysis at the level of research topics. It is also the first large scale model of any literature that is based on both citation links and the textual relatedness of documents and is thus among the most accurate characterizations of the literature ever created. The open database and tool contain detailed information that can be used to search and explore topics related to biomedical science, and to analyze these topics within the context of funding, industrial application, clinical application, translational potential, and other features.
In addition, this model is complementary to the recently published PubMed Knowledge Graph (PKG)14 which contains document level information from PubMed and other sources such as extracted bioentities, disambiguated authors and institutions. The cluster-level analytics enabled by our model are an important addition to the type of data provided by PKG, enabling both macro- and micro-level analysis of the research landscape.
Methods
The methodology used to create and characterize our PubMed model uses and combines data from many sources as listed in Table 1.
For the model we chose to include PubMed documents from 1996–2019. Given that the model is intended to be used for planning and evaluation of recent trends, we felt no need to include historical documents much older than 20 years. 1996 was chosen as the starting year to enable comparison to our Scopus-based models1 if need arises. Documents were also limited to those that either had references in the NIH Open Citation Collection (OCC)8,15 or for which the National Library of Medicine (NLM) had already calculated similar article (SA) scores, resulting in a set of 18,765,313 documents.
Before calculating relatedness between pairs of papers, we further filtered the documents by removing those from a set of 42 journals, primarily from the physical sciences, that were found in a preliminary study to create clusters that were unconnected to the biomedical core of the document set. These clusters, which contained papers from disciplines such as high energy physics, physical chemistry and crystallography, were distracting to early users of our models since they had nothing to do with biomedicine. Although many other journals could have been removed, we chose those that were large and whose removal would most affect the overall model. Upon removal of the 550,659 documents in these journals, our set was left with 18,214,654 documents, which were then used to create the model.
Relatedness measure and clustering
Our model is constructed using a hybrid relatedness measure composed of direct citation (DC) and textual SA scores. We use a 50:50 DC + SA hybrid measure that our most recent study found to be more accurate than either a pure citation-based measure or pure text-based measure16.
The relatedness rij between papers i and j is calculated as
The parameter α is set such that α ∑ rijDC = (1 - α) ∑ rijSA to achieve a 50:50 weighting of citation and textual relatedness across the entire set of document pairs.
where cij = 1/nref if i cites j and is 0 if not, and nref is the number of references in document i within the OCC set, and
using SA scores (S). Since SA scores are symmetrical, in cases were document pairs ij and ji were both within the set, only the ij pair was included. Each type of relatedness value was normalized to its corresponding maximum. Thus, all values of rijDC and rijSA ranged between 0 and 1.
Citation links for the citation portion of the relatedness measure were obtained from two sources. First, we used the January 2020 version of the OCC linkage set which contained references for 13,013,385 (71.44%) of the documents in our set. Of these, 12,578,393 cited other PMID within the set, comprising 315,512,095 citation links. It is important to use complete data where possible to obtain optimum results. We note that complete reference data are not available from any source, paid or open. Reference data can be missing for several reasons including publishers not making references available to aggregators (such as Scopus or WOS) and lack of open data. For comparison, while the OCC is missing references for 13.1% of PubMed documents in 2017, Scopus is also missing references for 7.4% of the same set of documents. Overall, the OCC is a relatively complete source of reference data (over 80%) for recent years and is thus very suitable for use in science mapping studies.
Second, we used data from OpenCitations (COCI) that were downloaded on January 20, 2020 and converted those data to PMID using DOIs. After matching the OCC and COCI data, it was found that the COCI data contain 186,399,013 links between pairs of PMID within our set, of which only 1,488,388 are not in the OCC linkage set. The COCI set contains references for only 27,065 documents that are not covered in the OCC set. Thus, the OCC data are a relatively complete set on their own and the COCI data add little to the total. The combined OCC/COCI sets of citation links contained 317,000,482 links between PMID within our document set.
For the textual component of the hybrid relatedness measure, we used the similar article (SA) scores (https://www.ncbi.nlm.nih.gov/books/NBK3827/#pubmedhelp.Computation_of_Similar_Articl) calculated by NLM using words from titles, abstracts and MeSH terms17, and which can be retrieved using an Entrez e-utility (https://www.ncbi.nlm.nih.gov/books/NBK25499/, see cmd = neighbor_score). We chose to use the top 20 SA scores for each document, limited to those where the paired document was also within the set, resulting in a set of 363,404,050 links of which 314,316,142 were unique; i.e., Sji and Sij pairs were not both present. The choice to use the top 20 scores per paper was made because previous research showed that there was little difference in clustering accuracy whether 12 or 40 links per document were used18.
Combining the citation and SA links resulted in a set of 601,605,944 links of which 30,560,355 had a direct citation link and a top 20 SA score. This overlap (9.64% of the direct citation links also had a top 20 SA link) is quite low, suggesting that citation-based and text-based relatedness are complementary and that both are ultimately important to accurate clustering. There were 286 million pairs of documents that had a direct citation link but that did not have a top 20 SA score. This does not mean that there is no textual relatedness between these pairs of documents. In most cases there is topical overlap between pairs of documents linked through citation19. Rather than assuming no textual relatedness for these pairs, we used SA scores even though they were outside of the top 20. Actual SA scores were used for pairs for which they were available, and estimated scores (half the minimum SA score for either document in the pair) were added where a calculated score was not available. The summed normalized SA and DC scores were ∑ rijDC = 9359293 and ∑ rijSA = 26297313; solving α ∑ rijDC = (1 - α) ∑ rijSA gives α = 0.7375 to achieve a 50:50 weighting of citation and textual relatedness. Equation (1) was then used to calculate hybrid relatedness for each document pair. For example, PMID 18637048 cites 26 papers within the set, one of which is PMID 15000003. For this pair of documents rijDC = 0.038462 and rijSA = 0.136923 which leads to rijHYB = 0.064307.
The full list of document-document pairs and their relatedness values (which are used as edge weights) were used to cluster the documents. We desired a model with approximately 30,000 clusters at a minimum size of 75 documents each. This specification is based on previous work where we have found that models with an average cluster size of several hundred documents tend to contain clusters that are sufficiently large to be meaningful and well differentiated to experts20 but without being so large as to be about multiple topics. A three-level hierarchical clustering was created using the Leiden algorithm10 with input resolutions as indicated in Table 2. Different quality functions can be used with the Leiden algorithm; we used the original quality function introduced in an earlier version of Leiden University’s clustering methodology21. The most granular level is designated PM5 to denote that this is a PubMed (PM) model and that the cluster sizes are similar to those of our Scopus model with around 105 (5) clusters. Higher level clusters are also designated by their rough order of magnitude – e.g., PM4 contains on the order of 104 clusters.
Visual map
Once the clustering was completed, a visual map of the clusters was created. Relatedness (R) between clusters m and n was calculated as the summed relatedness values
where Nm and Nn are the number of documents in clusters m and n, respectively. The denominator serves to normalize for cluster size so that the relatedness values are not inherently biased toward large clusters.
Cluster-cluster relatedness values were then filtered to the top 15 per cluster, and OpenOrd13 was used to create a graph layout of the clusters with the cut parameter set to 0.7. OpenOrd returns [x, y] positions for each cluster on a 2D plane.
Each cluster is assigned to one of 12 major fields (e.g., Biology, Medicine, Brain Science, etc.) based on the journal distribution within the cluster1 and a journal-to-field mapping previously developed22 and the cluster is colored in the map based on its dominant field (see Fig. 2). Clusters that are highly related are close to each other in the map and those that have little or no relationship to each other have more distance between them. A two-dimensional map is simply a visual approximation of a multi-dimensional system. In addition to showing the position of each cluster, the map can be used as a basis for the overlay of other information or to show filtered results.
The high-level layout of the map is similar to that found in other maps of science. For instance, chemistry and biology are proximate, and infectious disease tends to fall between biology and medicine. Health sciences, which mostly comprise clinical areas, nursing, etc., are at the outer edge of the map but also have interfaces with medicine and brain sciences. Relatively few clusters in physics, computer science, engineering, earth sciences, social science and humanities are found in the map.
In our initial exploration of the map we noticed several groups of clusters that were either not connected to the main component of the map or that were dominated by physics. The group of physics clusters was reviewed, and 25 clusters were found to have no biomedical content and manually discarded from the map and model. Investigation of other groups of clusters showed several groups of clusters with no discernible topic focus. In many cases they were clusters of documents with errata/corrigendum/correction in their titles, or clusters of documents with no abstracts and few references. 121 of these clusters were also removed manually. The final PM5 map consists of 28,743 clusters containing 18,160,327 documents.
Model characterization
A variety of metadata and indicators were used to characterize the clusters in the model to enable practitioners and decision makers to recognize and analyze topic-level structures. Figure 3 shows an example of this characterization for a single cluster. Except for the chart showing the number of documents by year in the upper right corner, characterization was done using only those documents published from 2015–2019 to focus on recent content.
Lists of top ten phrases, idiosyncratic phrases, MeSH terms, sources (journals), and journal categories are provided to characterize the topic embodied by each cluster. Additional nuance is added by listing the ten most central papers and five most central review papers.
Central papers and reviews are chosen by calculating the sum of the relatedness scores rijHYB from Eq. (1) where papers i and j are in the same cluster and then sorting by summed scores. Those with the highest within-cluster relatedness scores are assumed to be the most central to the cluster.
Phrases are identified using the following method:
-
(1)
The NLTK library for Python is used to extract noun phrases (NP) from titles and abstracts using the grammar NP: {<JJ.*|VBG > * < NN.* > + <VBG> ?} which matches a sequence of zero or more adjectives (JJ.*) or gerunds (VBG), followed by one or more nouns (NN.*), followed by 0 or 1 gerund.
-
(2)
The process in step 1 is repeated 20 times for each cluster using a random one-third sample of its titles and abstracts. Phrase counts are then summed over the 20 bootstrapped samples. We have noticed that phrase scores obtained from bootstrapping are more well separated than those obtained from simply running over the cluster contents.
-
(3)
Counts were then transformed to scores as sc = count/(20*np/3) where np is the number of papers in the cluster (2015–2019). Thus, the score is an estimate of the number of times the phrase occurs per paper in the cluster.
For idiosyncratic phrases, we take the full list of scores from step 3 above, and then re-score them using idio = 10*log(1 + sc/nptot)*sc/sctot, where sc is the score from step 3, sctot is the sum of scores for that phrase over all clusters, and nptot is the total number of papers in all clusters in which the phrase appears. This formula effectively re-ranks phrases by how much they differentiate one cluster from another. If a phrase is common and occurs in many clusters, the score adjusts down in a relative sense and vice versa. Having access to the most common phrases in a cluster and to those that differentiate a cluster from other clusters provides greater perspective on the cluster contents than if only one or the other were reported.
This method for labeling clusters is similar to that of Waltman & van Eck21 in some respects, but differs in others. While the two methods use different toolkits, the functions are similar, both focusing on noun phrases with some allowance for adjectives. Also, our method for re-scoring phrases based on their relative prevalence in the cluster and corpus is similar in intent to Waltman’s calculation of term relevance scores.
Top categories uses the Scopus ASJC classification system and is based on the journal-to-ASJC file that is publicly available at the Scopus website23. ASJC categories were chosen because the list of categories is larger and seems more intuitive to us than those in either the ScienceMetrix or Australian Research Council journal classification systems.
Metrics are also calculated for each cluster. For many of these the actual metric value is given, and a percentile score is also given, where the percentile is related to the cluster ranking for that metric. For instance, for the cluster shown in Fig. 3, the average cites per paper is 9.20, and this value is in the 97th percentile among all clusters. The metrics reported by category for each cluster include:
-
Impact
-
Mean cites per paper from iCite2.08.
-
Mean relative citation ratio (RCR) from iCite2.012. RCR is a metric developed several years ago by NIH that normalizes citation counts of each paper based on its local co-citation network.
-
Mean SNIP journal impact24 using the Scopus journal file23. SNIP is a journal level impact factor that accounts for differences in citation practices by field.
-
-
Industry involvement
-
Fraction of papers with an industry address. Papers with an industry address were identified using a method similar to that of Tijssen25 by searching addresses for abbreviations associated with companies (e.g., Corp., Inc., Ltd., GmbH, BV) and prominent company names (e.g., Merck, Novartis).
-
Mean patent-to-paper citations per paper. We have over many years systematically mined non-patent references from US patents and matched those reference strings to indices built from Scopus and PubMed article data to find likely matches. The resulting matches are used to calculate patent-based metrics.
-
-
Clinical involvement
-
Fraction of papers with a clinical address. Papers with a clinical address were identified by search addresses for strings associated with clinical institutions such as hospitals and medical centers (e.g., spital, clinic, klinik, medical center, cancer center, NHS)25.
-
Mean research level on a scale of basic to applied using the method for calculating research level by paper. The machine learning approach used here was trained on titles and abstracts of papers from over 4,000 journals using the journal research levels26.
-
-
Funding
-
Mean number of funding types per paper (using PubMed “Research Support” tags).
-
Mean number of grants per paper.
-
Mean funding per paper (in $$M).
-
-
Document type
-
Fraction of papers classified as a method paper27. Method papers were identified using citing sentences and citing locations from PubMed Central full text. Machine learning was based on a training set of 1000 manually classified papers with the best classifier achieving 92% accuracy.
-
Fraction of papers classified as a discovery paper28. Discovery papers were identified using specific terms in citing sentences from PubMed Central full text. Machine learning was based on a manually curated set of 135 discovery papers (158 non-discovery papers were excluded) with a classifier accuracy of 94%.
-
Fraction of papers classified as reviews by PubMed.
-
Fraction of papers classified as clinical trials by PubMed.
-
-
Translation
-
Mean translational potential using Approximate Potential to Translate (APT) from iCite2.011. APT is a metric recently developed by NIH based on multiple features including citation patterns between different types of MeSH terms (e.g., papers with Human terms citing papers with Molecular, Cellular and Animal terms).
-
-
Authorship/community strength
-
Number of authors with at least 2 papers in the cluster.
-
Number of authors with at least 5 papers in the cluster. These metrics are based on the principle that communities (or topics) with many active authors are stronger than those with few.
-
-
Age and momentum
-
Transparency indicators extracted from PubMed Central open access (PMCOA) full text articles, 2015–201930. Researchers at Stanford manually identified the below listed types of statements in 500 articles. Machine learning developed methods to identify such statements in other full text papers and was applied to the full PMCOA corpus. Specificity and sensitivity were both above 90%.
-
Fraction of papers that are open access (OA) from PMCOA.
-
Fraction of OA papers with COI statements.
-
Fraction of OA papers with funding statements.
-
Fraction of OA papers with registration statements.
-
Fraction of OA papers with data sharing statements.
-
Fraction of OA papers with code sharing statements.
-
Cursory explanatory details have been given above for several of the metrics along with associated references that contain more information about the scope and accuracy of the metric. Detail has not been given for metrics that are more common or that are self-explanatory.
Regarding funding information, PubMed contains a set of Research Support tags that specify different types of funding. For example, NIH-intramural, NIH-extramural, US-govt-non-NIH, and non-US-govt are four of the types. These data are used to calculate the mean number of funding types per paper. However, these numbers are questionable as recent research suggests that NIH may be indexing less than half of the acknowledged funding content available in articles30. PubMed indexes acknowledged grant numbers from a limited list of funding sources with a heavy emphasis on NIH grants. These data are used to calculate mean number of grants per paper. For funding amounts, grant-to-article links from NIH RePORTER and the US National Science Foundation (NSF) API were used to calculate the numbers of papers by grant per cluster. The funding amount for each grant from the StarMetrics Federal RePORTER data (see Table 1) was fractionally assigned to the clusters containing those papers, summed, and then used to calculate mean funding (from NIH and NSF) per paper.
Data Records
As explained in the method section, metadata from PubMed records and multiple other sources were used to characterize the 28,743 clusters in our PubMed model. These characterizations comprise a derivative database that is freely available on Figshare under the CC BY 4.0 license in two formats – an Excel workbook and a Tableau workbook31. The Excel workbook makes the data readily available for re-use while the Tableau interface enables visual exploration and filtering of the model for detailed analysis.
Excel workbook
The Excel workbook is comprised of 15 different sheets as shown in Table 3. The majority of the sheets contain cluster-level metadata (e.g., top 10 phrases, top 10 MeSH headings) or metrics. PM5_SHEET allows the user to input a cluster number and then self-populates with data from other sheets to create the characterization shown in Fig. 3. One sheet contains the list of 42 journals that were excluded from our model and one sheet contains the list of PMID identified as method27 or discovery28 papers which is not available elsewhere. Field names are given in the first row of each sheet. Data across different sheets are linked through the cluster number (PM5).
Descriptions of the data fields for the CLUST and TRANSP tables are provided in Tables 4 and 5, respectively.
The COUNT sheet contains a separate field for each year, 1996–2019, with integer document counts by cluster and year. The PHRASE, IDIO, MESH, ASJC and JNL sheets all have a similar format, an example of which is given in Table 6. The AUTH table is similar to these, but also contains a cpp (cites per paper) field to reflect relative author impact within the cluster. Tables 7–9 contain descriptions of the QUERY, CORE/REVIEW and METHDISC sheets, respectively.
Updating of the PubMed model requires updating of all the databases that feed the model along with assignment of new papers to existing clusters, consideration of the formation of new clusters, recalculation of metrics, etc. We anticipate updating the model annually.
Tableau workbook
Due to the variety of metadata and indicators associated with each cluster in the PM5 model, exploration of topics could proceed in many ways. To accommodate this potential, data from the Excel file (with the exception of the JNL_EXCL and METHDISC sheets) were incorporated into a Tableau workbook. Only the free software, Tableau Reader (https://www.tableau.com/products/reader), is required to interact with the workbook. Three data views have been constructed in Tableau – a map view, a scatterplot view, and a cluster detail view.
Filters (sliders and dropdowns) can be used to limit the clusters in the map and scatterplot views to those that meet a set of desired parameters. Each slider is tied to an indicator; thus one can filter based on variables such as impact (e.g., cites per paper, RCR), the relative presence of different types of documents (e.g., discovery, method, review, clinical trial), funding (e.g., dollars per paper from NIH/NSF, number of funding types per paper), and clinical application (e.g., research level, translational potential). Dropdowns include journal categories and MeSH terms which can be selected from menus or typed in (with available choices automatically reducing as text is entered). The display will be limited to clusters matching those choices. There is also an institution dropdown which limits the clusters to those in which a given institution has published in the 2015–2019 time period (see Table 7). This allows users to focus on clusters in which an institution of interest already has a publishing presence. Clusters can be sized using different parameters (e.g., number of papers, number of authors with at least 5 papers from 2015–2019, fraction of discovery papers) and can be colored using many of the features as well.
Figure 4 shows map and scatterplot views of the clusters in which the University of Michigan has published in the 2015–2019 time period. Cluster coloring is based on research level, where dark red is the most basic and dark green is the most applied. An overview of cluster characteristics is shown when the mouse is used to hover over a cluster. Clicking on a cluster opens a cluster detail view that is equivalent to the Excel PM5_SHEET view (see Fig. 3).
The scatterplot view in Fig. 4b shows the relationship between translational potential (x-axis) and NIH/NSF funding (y-axis). The horizontal and vertical lines in the middle of the chart show medians, thus dividing the graph into four quadrants to enable quadrant-based analysis. In this view, basic research clusters tend to appear in the upper left quadrant while applied research clusters tend to appear at the lower right. The upper right quadrant is interesting in that it contains a mix of basic and applied content, although it is more heavily weighted toward applied.
PMID to cluster listing
In addition to the Excel and Tableau workbooks, we make available the listing of PMID to PM5 cluster assignments in a separate tab separated (TSV) file. This enables linking of other PubMed-based data sources such as PKG14 to our model through PMID.
Technical Validation
The validity of the model rests on the validity of the process used to create it. We start by acknowledging that there is no single clustering of a large-scale dataset that can be proven to be the most accurate. There are no ground truth data that can be used to determine the absolute accuracy of the clusters formed from a set of over 18 million documents. Nevertheless, there are ways to compare the results of different methodologies in a relative way that suggest that the clusters are coherent and useful.
Recently, a principled approach to comparing cluster solutions was introduced. It employs granularity-accuracy plots where cluster solutions are compared using their cluster size distributions (from which granularity is calculated) and pairwise relatedness data (from which relative accuracy is calculated)4. The most reliable results are obtained when the relatedness data used as the basis of comparison are independent of the relatedness measures used in clustering. It is also wise to use multiple bases of comparison where possible.
Prior to creating this PubMed model, we ran a large-scale experiment in which we compared seven relatedness measures, two citation-based, one text-based, and four hybrids using a set of nine million documents from PubMed16. The Leiden algorithm was used for each clustering run and each solution had roughly 20,000 clusters. Three different bases of comparison were used to determine the relative accuracies of the seven cluster solutions. One was based on the concentration of references of nearly 30,000 papers with large numbers of references within clusters. This measure is clearly biased toward citation-based relatedness measures. The second was based on the fraction of the top 20 SA scores in the entire set that were preserved within clusters. This measure is clearly biased toward text-based relatedness measures. The third was to calculate the fraction of papers with a common grant that were preserved within clusters. This measure is independent of both citation and text-based measures. Using a composite based on these three different relative accuracy measures, we found that the hybrid relatedness measure based on a 50:50 mix of direct citation and SA scores had the highest performance16. This hybrid solution preserved nearly 51% of the overall relatedness signal within clusters. These values suggest clusters of high quality given that there were nearly 20,000 clusters in the solution and also suggests that the PubMed model presented here is of similar quality in that the same process and relatedness measure were used.
To place this result in context, we note that other recent studies have similar findings regarding some specifics related to our model. Many studies at both small and large scale have found that hybrid relatedness measures produce better clusters than measures based solely on citations or text3,32,33,34,35,36,37. In addition, the principled approach for comparing cluster solutions has been used for multiple large-scale studies3,18,38,39 and is becoming a standard in the area of science mapping.
Usage Notes
The recent COVID-19 pandemic is not only affecting individuals in negative ways, but it is also affecting research institutions. Although it is creating some new opportunities for COVID-19-related research, it is threatening many current research programs and structures. Under conditions of lockdown and social distancing, many labs have had to close. Some will re-open without issue, some will face uncertain conditions upon re-opening, while others may not be able to open at all. Funding availability will likely decrease, not only from agencies but also from local and regional governments that rely on decreasing tax revenues associated with economic downturn. For medical schools with associated hospitals, clinical margin revenue (a portion of which is used to fund research) is also greatly reduced. This very real scenario is being faced by universities across the world. How can universities balance their fiduciary duties to the financial health of the organization while simultaneously supporting the research mission from within a contracting financial system? Our PubMed model provides a quantitative view of the research landscape that facilitates informed decision-making.
We provide here example workflows to show how the Tableau tool can be used. The examples show how one medical school (in this case the University of Michigan Medical School, UMMS) could navigate the current landscape, first by looking for opportunities related to historical literature associated with coronavirus, and then by looking at topics related to a cohort of African American and Native American researchers.
Since early 2020, the Semantic Scholar team at the Allen Institute for AI, along with other partners, has been compiling a set of literature related to coronavirus – the COVID-19 Open Research Dataset (CORD-19) – and has made it publicly available for use by researchers40. The May 31, 2020 version of this dataset contained 139,952 documents among which 98,228 unique PMID could be identified. Of these, 67,452 PMID were from 1996 through 2019 and were found in our PubMed model. CORD-19 contains literature associated not only with coronavirus (e.g., CoV, SARS, MERS) but since mid-May also contains literature associated with co-morbidity factors and related medical conditions (respiratory problems, etc.) It also contains over 30,000 documents from 2020, many of which have been recently added to PubMed from preprint servers such as medRxiv and bioRxiv despite not yet having completed the peer review process. These documents from 2020 are not in our model but will be added in the future.
Figure 5a shows the location of 85 clusters that contain at least 25 documents from CORD-19 and where the concentration of CORD-19 documents is at least 10%. Clusters are sized by the number of CORD-19 documents and colored using research level (red = basic, green = applied). These are the clusters that are most related to coronavirus as defined by CORD-19. A majority of the CORD-19 clusters are at the bottom of the map in an area focused around viruses known to affect the respiratory system (e.g., influenza, SARS, MERS, etc.) Clusters in other areas of the map focus on a variety of topics such as other viruses, proteins, and treatments or devices that have recently become associated with treatment of COVID-19 (e.g., cerebral oxygenation, mechanical thrombectomy).
This view of the coronavirus-related literature is quite different from others that have been published recently41,42 and separates the literature into distinct topics much more precisely. For example, Colavizza et al.41 recently characterized and mapped nearly 40k publications from the April 4, 2020 version of CORD-19. Using topic modeling they identified 15 topics, the largest of which is labeled ‘public health and epidemics’ and contains roughly 20% of the corpus. Citation-based clustering was also done, resulting in 23 clusters with at least 100 documents and a much larger number of very small clusters. The largest cluster contained 7,300 documents with a focus on coronaviruses and related molecular biology analyses. In contrast, our solution has two separate clusters for SARS (one related to the virus and the other to infection) along with separate clusters on MERS, human coronaviruses, equine coronaviruses, bat viruses, and many other different types of viruses. Both strategic and tactical decision making are thus better enabled using the type of granular classification of documents available in our model than in other related mapping exercises.
Figure 5b further limits to only those clusters in which UMMS has published from 2015–2019. Identification of the UMMS papers was not done using the Tableau application but were obtained from a query to PubMed. Once the list of UMMS documents was obtained, clusters were identified for each PMID, the number of documents was counted by PM5 cluster, and these numbers were then imported into the Tableau file and included in the filtering and labeling capabilities. The identification of UMMS-authored documents could have also been done by simply extracting a list of PMID from the institution’s research information management system (e.g., https://experts.umich.edu/), a task that any institution with such a system could accomplish with minimal effort.
In Fig. 5b, clusters are sized by the number of UMMS papers to show relevance to UMMS and are plotted as a function of funding percentile (y-axis) and patent reference percentile (x-axis). If UMMS were to choose to prioritize activities in which a) they have a strong publication presence, b) historical U.S. funding levels are high, and c) commercial potential exists, large clusters at the upper right of this graph might be prioritized. In this case, the top three candidates (largest three clusters in the upper right quadrant) are focused on murine norovirus infections, rhinovirus infections, and RNA viruses. Despite being represented in the CORD-19 dataset, none of these clusters has the type of overt relationship to COVID-19 that would suggest that immediate short-term funding could be obtained. However, one of the smaller clusters in that quadrant, #12391 on ribosomal frameshifting, could be a candidate for such funding in that frameshifting could play a role in mutation and was already investigated for SARS years ago. Also, the largest cluster in the upper left quadrant is clearly important in the current climate, #2673 on the effects of school closures and social distancing on epidemics.
As COVID-related research efforts continue to increase in both scale and scope, universities must not forget about other areas of research that, while not directly related to the current health crisis, are nonetheless vitally important to the health of a nation. For instance, it is important to support the research activities of groups that are currently under-represented, such as female and minority researchers.
UMMS has a substantial number of African-American/black (AA/B) and Native American (NA) researchers. Between 2010 and 2018, these individuals submitted 657 research proposals to external funding sources and published 4,489 papers from 1996–2019 that were indexed in PubMed. Of these, 3,995 appear in our model; most of the remainder are physics and chemistry papers in journals that we removed from the model. Note that this analysis requires that data from the university be linked to the model and shows how the model can be easily extended to include such data to facilitate advanced analysis.
Publication activities of AA/B and NA researchers at UMMS are consistent with the most recent in-depth study of racial bias at NIH. Hoppe et al. studied over 157,000 R01 applications to characterize how racial bias affects AA/B researchers during different stages of their career. They provided compelling evidence that the topic mix of R01 applications from AA/B researchers is very different from that of White researchers, and that AA/B applicants tend to propose research on topics with lower award rates43. They noted that AA/B applications were far more likely to involve human subjects than those from White applicants. AA/B applications tended to focus on health disparities such as AIDS and patient-focused interventions. In contrast, clusters associated with osteoarthritis, cartilage, prion, corneal, skin, iron, and neuron contained no applications from AA/B researchers.
The 3,995 papers published by AA/B and NA researchers at UMMS are shown in Fig. 6 as colored circles overlaid on a gray version of the map. The distribution of publications over the map is notably unbalanced with relatively few in the lower right quadrant (which is dominated by biology and infectious disease) and much higher in the lower left quadrant in areas that reflect racial disparities. For example, the topic in which AA/B and NA researchers at UMMS have the largest number of publications is #6953 which focuses on physical health, mental health and economic disparities in minority populations.
Figure 6 suggests that the topic choices of AA/B and NA researchers at UMMS are skewed in a way that is consistent with NIH findings on AA/B R01 applications. By extension, given that these topics are, on the whole, less well-funded by NIH than other topics, this suggests that minority researchers are disadvantaged in a fundamental way. It behooves universities to be aware of the topic choices of their researchers, how these topics and researchers might be disadvantaged, and then to use this knowledge to inform equity focused interventions.
We note that recently published PubMed Knowledge Graph14 contains complementary document level information such as extracted bioentities, disambiguated authors and institutions that could be added to the PubMed model by linking through PubMed IDs to facilitate additional types of analysis.
Code availability
The Leiden algorithm was used for clustering and is freely available at https://github.com/vtraag/leidenalg.
References
Klavans, R. & Boyack, K. W. Research portfolio analysis and topic prominence. Journal of Informetrics 11, 1158–1174, https://doi.org/10.1016/j.joi.2017.10.002 (2017).
Klavans, R. & Boyack, K. W. Which type of citation analysis generates the most accurate taxonomy of scientific and technical knowledge? Journal of the Association for Information Science and Technology 68, 984–998, https://doi.org/10.1002/asi.23734 (2017).
Ahlgren, P., Chen, Y., Colliander, C. & van Eck, N. J. Enhancing direct citations: A comparison of relatedness measures for community detection in a large set of PubMed publications. Quantitative Science Studies 1, 714–729, https://doi.org/10.1162/qss_a_00027 (2020).
Waltman, L., Boyack, K. W., Colavizza, G. & Van Eck, N. J. A principled methodology for comparing relatedness measures for clustering publications. Quantitative Science Studies 1, 691–713, https://doi.org/10.1162/qss_a_00035 (2020).
Baas, J., Schotten, M., Plume, A., Côté, G. & Karimi, R. Scopus as a curated, high-quality bibliometric data source for academic research in quantitative science studies. Quantitative Science Studies 1, 377–386 (2020).
Waltman, L. et al. The Leiden Ranking 2011/2012: Data collection, indicators, and interpretation. Journal of the American Society for Information Science and Technology 63, 2419–2432 (2012).
Ruiz-Castillo, J. & Waltman, L. Field-normalized citation impact indicators using algorithmically constructed classification systems of science. Journal of Informetrics 9, 102–117 (2015).
Hutchins, B. I. et al. The NIH Open Citation Collection: A public access, broad coverage resource. PLoS Biology 17, e03000385, https://doi.org/10.1371/journal.pbio.3000385 (2019).
Peroni, S. & Shotton, D. OpenCitations, an infrastructure organization for open scholarship. Quantitative Science Studies 1, 428–444 (2020).
Traag, V. A., Waltman, L. & Van Eck, N. J. From Louvain to Leiden: Guaranteeing well-connected communities. Scientific Reports 9, 5233, https://doi.org/10.1038/s41598-019-41695-z (2019).
Hutchins, B. I., Davis, M. T., Meseroll, R. A. & Santangelo, G. M. Predicting translational progress in biomedical research. PLoS Biology 17, e3000416 (2019).
Hutchins, B. I., Yuan, X., Anderson, J. M. & Santangelo, G. M. Relative Citation Ratio (RCR): A new metric that uses citation rates to measure influence at the article level. PLoS Biology 14, e1002541, https://doi.org/10.1371/journal.pbio.1002541 (2016).
Martin, S., Brown, W. M., Klavans, R. & Boyack, K. W. OpenOrd: An open-source toolbox for large graph layout. Proceedings of SPIE - The International Society for Optical Engineering 7868, 786806 (2011).
Xu, J. et al. Building a PubMed knowledge graph. Scientific Data 7, 205 (2020).
iCite, Hutchins, B. I. & Santangelo, G. M. iCite Database Snapshots (NIH Open Citation Collection). National Institutes of Health. https://doi.org/10.35092/yhjc.c.4586573.v8 (2019).
Boyack, K. W. & Klavans, R. A comparison of large-scale science models based on textual, direct citation and hybrid relatedness. Quantitative Science Studies 1, qss_a_00085, https://doi.org/10.1162/qss_a_00085 (2020).
Lin, J. & Wilbur, W. J. PubMed related articles: A probabilistic topic-based model for content similarity. BMC Bioinformatics 8, 423, https://doi.org/10.1186/1471-2105-8-423 (2007).
Boyack, K. W. & Klavans, R. Accurately identifying topics using text: Mapping PubMed. In 23rd International Conference on Science and Technology Indicators (STI 2018) (Leiden, The Netherlands 2018).
Ding, Y. et al. Content-based citation analysis: The next generation of citation analysis. Journal of the Association for Information Science and Technology 65, 1820–1833 (2014).
Boyack, K. W., Klavans, R., Small, H. & Ungar, L. Characterizing the emergence of two nanotechnology topics using a contemporaneous global micro-model of science. Journal of Engineering and Technology Management 32, 147–159, https://doi.org/10.1016/j.jengtecman.2013.07.001 (2014).
Waltman, L. & van Eck, N. J. A new methodology for constructing a publication-level classification system of science. Journal of the American Society for Information Science and Technology 63, 2378–2392, https://doi.org/10.1002/asi.22748 (2012).
Börner, K. et al. Design and update of a classification system: The UCSD map of science. PLoS ONE 7, e39464 (2012).
Elsevier. Scopus Source List. (2020).
Waltman, L., van Eck, N. J., van Leeuwen, T. & Visser, M. S. Some modifications to the SNIP journal impact indicator. Journal of Informetrics 7, 272–285, https://doi.org/10.1016/j.joi.2012.11.011 (2013).
Tijssen, R. J. W. Discarding the ‘basic science/applied science’ dichotomy: A knowledge utilization triangle classification system of research journals. Journal of the American Society for Information Science and Technology 61, 1842–1852 (2010).
Boyack, K. W., Patek, M., Ungar, L. H., Yoon, P. & Klavans, R. Classification of individual articles from all of science by research level. Journal of Informetrics 18, 1–12 (2014).
Small, H. Characterizing highly cited method and non-method papers using citation contexts: The role of uncertainty. Journal of Informetrics 12, 461–480 (2018).
Small, H., Tseng, H. & Patek, M. Discovering discoveries: Identifying biomedical discoveries using citation contexts. Journal of Informetrics 11, 46–62 (2017).
Klavans, R., Boyack, K. W. & Murdick, D. A. A novel approach to predicting exceptional growth in research. PLoS One 15, e0239177, https://doi.org/10.1371/journal.pone.0239177 (2020).
Serghiou, S. et al. Assessment of transparency across the biomedical literature: How open is open? Preprint at https://doi.org/10.1101/2020.10.30.361618v1 (2020).
Boyack, K. W., Smith, C. & Klavans, R. STS model of the PubMed literature. figshare https://doi.org/10.6084/m9.figshare.12743639 (2020).
Ahlgren, P. & Colliander, C. Document-document similarity approaches and science mapping: Experimental comparison of five approaches. Journal of Informetrics 3, 49–63, https://doi.org/10.1016/j.joi.2008.11.003 (2009).
Zhang, L., Glänzel, W. & Ye, F. Y. The dynamic evolution of core documents: An experimental study based on h-related literature (2005–2013). Scientometrics 106, 369–381, https://doi.org/10.1007/s11192-015-1705-4 (2016).
Thijs, B., Schiebel, E. & Glänzel, W. Do second-order similarities provide added-value in a hybrid approach? Scientometrics 96, 667–677, https://doi.org/10.1007/s11192-012-0896-1 (2013).
Boyack, K. W. & Klavans, R. Co-citation analysis, bibliographic coupling, and direct citation: Which citation approach represents the research front most accurately? Journal of the American Society for Information Science and Technology 61, 2389–2404, https://doi.org/10.1002/asi.21419 (2010).
Boyack, K. W. et al. Clustering more than two million biomedical publications: Comparing the accuracies of nine text-based similarity approaches. PLoS One 6, e18029, https://doi.org/10.1371/journal.pone.0018029 (2011).
Glänzel, W. & Thijs, B. Using hybrid methods and ‘core documents’ for the representation of clusters and topics: The astronomy dataset. Scientometrics 111, 1071–1087, https://doi.org/10.1007/s11192-017-2301-6 (2017).
Sjögårde, P. & Ahlgren, P. Granularity of algorithmically constructed publication-level classifications of research publications: Identification of topics. Journal of Informetrics 12, 133–152, https://doi.org/10.1016/j.joi.2017.12.006 (2018).
Sjögårde, P. & Ahlgren, P. Granularity of algorithmically constructed publication-level classifications of research publications: Identification of specialties. Quantitative Science. Studies 1, 207–238, https://doi.org/10.1162/qss_a_00004 (2020).
Wang, L. L. et al. CORD-19: The COVID-19 Open Research Dataset. arXiv: 2004.10706 (2020).
Colavizza, G. et al. A scientometric overview of CORD-19. biorXiv 2020.04.20.046144 (2020).
Jia, Q. et al. Analysis of knowledge bases and research hotspots of coronavirus from the perspective of mapping knowledge domain. Medicine 99, e20378 (2020).
Hoppe, T. A. et al. Topic choice contributes to the lower rate of NIH awards to African-American/black scientists. Science Advances 5, eaaw7238 (2019).
Acknowledgements
This work was supported by NIH award HHSN271201800033C.
Author information
Authors and Affiliations
Contributions
K.W.B. proposed and managed the project, K.W.B., C.S. and R.K. wrote this manuscript, K.W.B. created the model, characterization and Excel workbook, C.S. created the Tableau workbook, K.W.B., C.S. and R.K. conducted analysis leading to the usage notes.
Corresponding author
Ethics declarations
Competing interests
Two of the authors (K.W.B. and R.K.) are employed by a small company that received the award mentioned above under which this work was funded. The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
The Creative Commons Public Domain Dedication waiver http://creativecommons.org/publicdomain/zero/1.0/ applies to the metadata files associated with this article.
About this article
Cite this article
Boyack, K.W., Smith, C. & Klavans, R. A detailed open access model of the PubMed literature. Sci Data 7, 408 (2020). https://doi.org/10.1038/s41597-020-00749-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-020-00749-y
This article is cited by
-
Normalization of direct citations for clustering in publication-level networks: evaluation of six approaches
Scientometrics (2024)
-
Academic information retrieval using citation clusters: in-depth evaluation based on systematic reviews
Scientometrics (2023)
-
Making Biomedical Sciences publications more accessible for machines
Medicine, Health Care and Philosophy (2022)
-
Reply to issues about entitymetrics and paper-entity citation network
Scientometrics (2022)
-
Finding citations for PubMed: a large-scale comparison between five freely available bibliographic data sources
Scientometrics (2021)