Abstract
Coral reefs are under increasingly severe threat from climate change and other anthropogenic stressors. Anomalously high seawater temperatures in particular are known to cause coral bleaching (loss of algal symbionts in the family Symbiodiniaceae), which frequently leads to coral mortality. Remote sensing of sea surface temperature (SST) has served as an invaluable tool for monitoring physical conditions that can lead to bleaching events over relatively large scales (e.g. few kms to 100 s of kms). But, it is also well known that seawater temperatures within a site can vary significantly across depths due to the combined influence of solar heating of surface waters, water column thermal stratification, and cooling from internal waves and upwelling. We deployed small autonomous benthic temperature sensors at depths ranging from 0–40 m in fore reef, back reef, and lagoonal reef habitats on the Belize Mesoamerican Barrier Reef System from 2000–2019. These data can be used to calculate depth-specific climatologies across reef depths and sites, and emphasize the dynamic and spatially-variable nature of coral reef physical environments.
Measurement(s) | temperature of water |
Technology Type(s) | Temperature Sensor Device |
Factor Type(s) | temporal interval • geographic location • depth |
Sample Characteristic - Environment | sea • marine reef • coral reef • marine coral reef back reef • marine coral reef fore reef |
Sample Characteristic - Location | Belize Barrier Reef |
Machine-accessible metadata file describing the reported data: https://doi.org/10.6084/m9.figshare.13095506
Similar content being viewed by others
Background & Summary
Reef-building corals and their algal symbionts (family Symbiodiniaceae) are highly sensitive to changes in temperature1,2 and in particular to deviations from local climatic norms3,4. Increases in seawater temperature of only one or a few degrees above a bleaching threshold can lead to the breakdown of the relationship between coral hosts and their photosynthetic algal symbionts in a process known as coral bleaching or dysbiosis, which often cascades into widespread coral mortality5 and subsequent reef ecosystem collapse6. The application of remotely sensed sea surface temperature (SST) data has served as a valuable predictor of coral bleaching, especially the metric of Degree Heating Weeks that incorporates both the magnitude and duration of excursions in temperature above regional climatological norms3,4.
However, susceptibility to bleaching is both patchy in space and time and is affected strongly by local factors7,8 such as reduced water flow that can lead both to increased localized heating9,10,11 and decreased gas exchange12. Further, temperatures often vary sharply with depth on coral reefs13 as a result of shallow-water solar heating14, and from the delivery of deeper, colder water from upwelling5 and internal waves15,16,17. What is less well understood is how temperature patterns change with depth over coral reefs18,19. Specifically, it is not clear if and when SST is diagnostic of temperatures at deeper locations on the same reef20. Furthermore, it is not well understood whether deviations from surface-recorded climatic norms necessarily reflect deviations from the depth-specific climatic norm13,14,19.
There is an urgent need to calibrate our understanding of the relationship between surface temperatures and benthic temperatures at specific reef locations. First, bleaching is correlated not only with the temperature, per se but rather with the magnitude and duration of deviations in temperature from the local climatic norm. The “bleaching threshold,” an analytical construct but one that has proved useful in bleaching predictions, is generally defined as 1 °C above the climatological mean SST of the warmest month21. Degree Heating Weeks is used as a cumulative measure combining both the extent and duration of heating above this bleaching threshold over the prior 12 weeks22,23. Defined in units of °C-time, the magnitude of the deviation from the bleaching threshold is multiplied by the duration that temperature remains above the threshold over a predetermined time window. For example, the cumulative number of degree days over any 12-week period is often chosen to indicate the likelihood of bleaching. Recent refinements of this approach have been proposed, including the inclusion of taxon-specific vulnerability to temperature anomalies24 and the suggestion to use in situ data collected at depth to calculate depth-specific climatologies and anomalies17,19.
Second, in the face of increasing coral mortality from thermal stress and other climate-related stressors, increasing attention has been paid to the concept of “rescue sites”25. By providing thermal refugia during heat waves26, these rescue sites may serve as larval sources that permit the repopulation of nearby assemblages that have experienced localized mass mortality25,27 (but see28). In addition to adjacent or nearby rescue sites, it has been proposed that deeper reefs (down to the mesophotic zone) may serve as reservoirs capable of rescuing their shallow counterparts with larval re-supply29. Whether or not deeper reefs could actually serve as rescue sites, however, depends heavily on whether the magnitude of temperature anomalies are lower at depth than they are at the surface19, as well as whether corals adapted to cooler temperatures at depth could effectively repopulate shallower and warmer locations. Our ability to detect potential rescue sites or reservoirs would be much better informed with an accurate understanding of whether deviations at different depths occur in synchrony with thermal stresses at or near the sea surface.
Here, we present 15 years of seawater temperature data collected in situ using small autonomous temperature loggers over a range of depths (0–40 m) and in fore reef, back reef, and lagoonal reef environments on the Belize Mesoamerican Barrier Reef System. This long-term dataset provides a novel mechanism for exploring the climatology on a site- and depth-specific basis.
Methods
Logger deployment sites
Temperature data loggers were deployed across multiple depths at 2 different locations (Carrie Bow Caye and Curlew Caye) near the Smithsonian Institution’s Carrie Bow Caye field station, (16° 48′ 08″ N, 88° 04′ 49″ W); at 3 different locations (Cat, Channel and Manatee Cayes) near the Pelican Cayes, (16° 38′ 59″ N, 88° 11′ 54″ W); and at one location each near the Sapodilla Cayes (16° 07′ 06″ N, 88° 15′ 26″ W), and Snakes Cayes (16° 11′ 30″ N, 88° 34′ 20″ W). These four different groupings of Cayes (Islands) extend from the central to southern portions of the Belize Mesoamerican Barrier Reef System (MBRS) (Fig. 1). The benthic community of the fore reef near Carrie Bow Caye represents quintessential Belizean Barrier Reef structure. It hosts four geographically distinct reef zones in close proximity: a low-relief spur-and-groove zone, the inner reef slope, the outer ridge, and the forereef slope30. Visiting researchers to the Smithsonian research station have documented 41 species of Scleractinian corals and 51 sponge species31. There are different abundances and distributions of sponge and coral species across each zone, which are thought to be driven by environmental factors30,31.
These sites represent a diversity of environmental conditions including exposed fore reefs with predicted upwelling (barrier reef), buffered and sheltered sites (lagoonal environments), and areas of comparatively high flow30. Near Carrie Bow Caye, loggers were deployed on a double shelf on the fore reef (Fig. 2). On the inner shelf (more proximal to the reef crest) loggers were installed at five depths ranging from 2 to 28 m (~6 to 90 ft), and within the outer shelf, (towards the Cayman Trench) loggers were installed at three additional depths ranging from 18 to 37 m (~60 to 120 ft; Fig. 2). Additional loggers were deployed at Curlew Caye, approximately 1.6 km south of the Carrie Bow Caye field station, at four depths ranging from 5 to 20 m (~15–60 ft). At the Pelican Cayes, loggers were installed on the inside and the immediate outside portions of mangrove lagoons at Cat, Channel, and Manatee Cayes (Online-only Table 1). In the Sapodilla Cayes, loggers were deployed on the fore reef at White Reef at 3 m (~10 ft depth). At the Snake Cayes, loggers were deployed at 3 m (~10 ft depth) near Lagoon Snake Caye. Note that for simplification, while loggers were deployed at fixed intervals using non-metric depths we hereafter refer to depths using the approximate metric values (e.g. 10 m instead of 30 ft). All depths are referenced to the depth of the seafloor
In situ seawater temperature measurements
A series of small, autonomous data loggers (StowAway, Tidbit, and HOBO Water Temp Pro loggers) manufactured by the Onset Computer Corporation (Pocasset, MA) with an accuracy of ± 0.2 °C and resolution 0.02 °C and 5 min response time were used to record temperature at all the study sites. Each instrument made seawater temperature measurements every 0.5 seconds and recorded 10 min averages (except for Hobo Water Temperature Pro loggers which acquired instantaneous measurements at 10 or 15 min intervals) from 2000–2019. At all sites deeper than 2 m loggers were affixed to a bottom-mounted float that suspended the logger 1 m above the seafloor, but the depths recorded for these loggers are referenced to the seafloor. At shallower sites (0 and 2 m) loggers were affixed to coral rubble or mangrove roots using cable ties, with the exception of the 2 m and surface loggers in the protected backreef of Carrie bow Caye, which we affixed to floats. Delayed start times and deployment of multiple loggers per site permitted deployment times of up to a year, at which time loggers were replaced. Overlapping records by multiple loggers at each location also permitted cross-calibration. All loggers were certified for accuracy by the manufacturer (Onset Computer Corporation) and were tested for accuracy following each retrieval (See Technical Validation below).
Data Records
Data from all loggers are archived in two databases. The first is an interactive database maintained by Northeastern University: http://www.northeastern.edu/helmuthlab/belize/database.html. This database provides a map-based, user-friendly interface for unrestricted access to data. Data can be filtered, viewed, and downloaded by selecting a series of map layers: depth, site location, and reef type on the ESRI web-based map. Given the range of selections, the database provides the range of years over which data meeting those criteria are available for download. Metadata for each dataset are included as a downloadable spreadsheet, which includes, for each site: Country (BZ), District (a subnational jurisdiction in Belize), Site Name, and GPS coordinates (see the associated Metadata Record). The metadata file also includes information specific to each microsite, including reef location, depth of each logger, and deployment years. This database also provides an http option for SQL access. Data from each query can be downloaded as raw .csv data.
Raw data in text file format are archived in a second database hosted by Figshare Digital repository32. The database is organized through a series of subfolders that follows the same query order as the interactive database hosted by Northeastern University. An associated metadata spreadsheet is also included as a downloadable zip file. Each datum is named with information that is specific to the logger information: a 6-10 letter/number naming convention as follows: 2 letter country code, two-letter regional code, 2 letter site code, and numerical digits to identify microsite location (Table 1). While all data are from Belize (BZ), the inclusion of country code is to ensure interoperability with a related database33.
Technical Validation
All loggers were factory calibrated before their initial deployment. For each deployment, multiple loggers at each depth/site recorded for overlapping intervals of up to a week. Differences of more than 0.2 °C between the two loggers were flagged as potential drift and the logger was tested further in the laboratory. In rare instances where drift was detected (greater than 0.5 °C difference between adjacent loggers), logger data were discarded. On average, + /−0.05 °C differences were found across loggers deployed simultaneously at the same location. This is within the accuracy range (0.2 °C) of the manufacturer.
Data retrieved from the field were cropped to remove any data recorded by the loggers during deployment and retrieval. This was done by matching the recorded deployment and retrieval dates and times with the timestamps of the loggers. In addition, quality control for each time series was accomplished by flagging unrealistic temperature drops or rises in the dataset, which we defined as a change of greater than 1 °C in a single (10–15 min) time step. These errors can occur when loggers undergo physical trauma. Flagged data were removed from the dataset.
Usage Notes
The majority of the Belizean Barrier Reef is experiencing “a phase of transition” toward a community dominated by macroalgae and coral rubble31, similar to reefs throughout the Caribbean. Total coral cover across all reef zones has decreased by 90% since 1995 with reef-building corals now comprising less than 8% of benthic cover31. Sponges remain present, covering about 3% of the benthos, a slight increase since the 1990s. The dramatic decline of coral species near Carrie Bow Caye likely has multiple drivers. McField34 first established bleaching-induced tissue mortality as a driver across the wider barrier reef. Villamizar et al.31 proposed hurricane activity as a driver decreasing physical structure and increasing rubble abundance in the benthic community near Carrie Bow Caye, noting eight hurricanes that have struck Belize with force since 1993. However, the relative importance of in situ temperature change at multiple depths as a driver of this transition has not been fully established.
This high temporal resolution dataset is valuable for examining sub-daily variability (daily ranges recorded by some loggers was as high as 3–4 °C) at different depths that traditional SST datasets cannot provide. The resolution of 10–15 mins is also valuable as a means of observing discrete events, including internal waves that are rarely detectable in hourly datasets and are only detectable from the surface under limited conditions35.
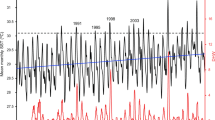
Furthermore, deeper (e.g. depth >15 m) temperature series on coral reefs are relatively rare, generally of short duration, and/or often interrupted by data gaps associated with funding constraints, shifting research priorities, and instrument losses and failures. While our data are also subject to some of these issues, they nevertheless provide relevant analysis opportunities due to their longevity and large distribution of sites and depths. One example of an application of this data set is to produce depth-specific temperature climatologies that can be used to predict depth-specific bleaching threshold temperatures (Fig. 3) and can be combined with time series of biological observations to predict and test taxon-specific bleaching patterns in relation to temperature stresses. Long-term data sets also provide a means of assessing the spatiotemporal frequencies over which measurements conducted at one location and depth are correlated with or decoupled from patterns at other locations. For example, analyses of the extent of frequency-specific correlation, or coherence, of temperature variation among depths can be conducted. Combining these records with remotely sensed SST observations can also provide insight into the ability to predict or approximate thermal extremes at depth from remotely sensed observations.
Climatology of daily maximum water temperature recorded at (a) 5 m depth inner reef slope and (b) 40 m depth outer reef slope at Carrie Bow Caye from 2001–2006, calculated as the average of each day of the year. Shaded areas represent 95% confidence intervals of interannual temperature variation of each day of the year.
References
Doney, S. C. et al. Climate change impacts on marine ecosystems. Annual Review of Marine Science 4, 11–37, https://doi.org/10.1146/annurev-marine-041911-111611 (2012).
Fabina, N. S. et al. The differential effects of increasing frequency and magnitude of extreme events on coral populations. Ecological Applications 25, 1534–1545 (2015).
Gleeson, M. W. & Strong, A. E. Applying MCSST to coral reef bleaching. Advances in Space Research 16, 151–154 (1995).
van Hooidonk, R. & Huber, M. Quantifying the quality of coral bleaching predictions. Coral Reefs 28, 579–587 (2009).
Glynn, P. W. & D’Croz, L. Experimental evidence for high temperature stress as the cause of El Nino-coincident coral mortality. Coral Reefs 8, 181–191 (1990).
Aronson, R. B. et al. Causes of coral reef degradation. Science 302, 1502–1502 (2003).
Carilli, J. E. et al. Century-scale records of coral growth rates indicate that local stressors reduce coral thermal tolerance threshold. Global Change Biology 16, 1247–1257 (2010).
Palumbi, S. R. et al. Mechanisms of reef coral resistance to future climate change. Science 344, 895–898 (2014).
Castillo, K. D. & Helmuth, B. S. T. Influence of thermal history on the response of Montastraea annularis to short-term temperature exposure. Marine Biology 148, 261–270 (2005).
Jimenez, I. M. et al. Heat budget and thermal microenvironment of shallow-water corals: Do massive corals get warmer than branching corals? Limnology and Oceanography 53, 1548–1561 (2008).
McCabe, R. et al. Temperature variability in a shallow, tidally-isolated coral reef lagoon. Journal of Geophysical Research 115, C12011, https://doi.org/10.1029/2009JC006023 (2010).
Finelli, C. M. et al. Water flow controls oxygen transport and photosynthesis in corals: potential links to coral bleaching. Coral Reefs 25, 47–57 (2006).
Leichter, J. J., Helmuth, B. & Fischer, A. M. Variation beneath the surface: Quantifying complex thermal environments on coral reefs in the Caribbean, Bahamas and Florida. Journal of Marine Research 64, 563–588 (2006).
Castillo, K. D. & Lima, F. P. Comparison of in situ and satellite-derived (MODIS-Aqua/Terra) methods for assessing temperatures on coral reefs. Limnology and Oceanography: Methods 8, 107–117 (2010).
Leichter, J. J. et al. Breaking internal waves on a Florida (USA) coral reef: a plankton pump at work? Marine Ecology Progress Series 166, 83–97 (1998).
Leichter, J. J. et al. Pulsed delivery of subthermocline water to Conch Reef (Florida Keys) by internal tidal bores. Limnology and Oceanography 41, 1490–1501 (1996).
Wyatt, A. S. J. et al. Heat accumulation on coral reefs mitigated by internal waves. Nature Geoscience 13, 28–34 (2020).
Kahng, S. E. et al. Community ecology of mesophotic coral reef ecosystems. Coral Reefs 29, 255–275 (2010).
Neal, B. P. et al. When depth is no refuge: cumulative thermal stress increases with depth in Bocas del Toro, Panama. Coral Reefs 33, 193–205, https://doi.org/10.1007/s00338-013-1081-6 (2014).
Brewin, R. J. W. et al. Evaluating operational AVHRR sea surface temperature data at the coastline using benthic temperature loggers. Remote Sensing 10, https://doi.org/10.3390/rs10060925 (2018).
Liu, G. et al. NOAA Coral Reef Watch 50 km Satellite Sea Surface temperature‐based decision support system for coral bleaching management. (Silver Spring, MD, 2013).
Liu, G., Strong, A. E. & Skirving, W. Remote sensing of sea surface temperatures during 2002 Barrier Reef coral bleaching. Eos 84, 137–141 (2003).
Strong, A. E. et al. In Coral Reefs and Climate Change: Science Management, (Coastal and Estuarine Studies) Vol. 61 (eds Jonathan T. Phinney et al.) 163-180 (American Geophysical Union, 2006).
Swain, T. et al. Coral bleaching response index: a new tool to standardize and compare susceptibility to thermal bleaching. Global Change Biology 22, 2475–2488 (2016).
Hannah, L. et al. Fine-grain modeling of species’ response to climate change: holdouts, stepping-stones, and microrefugia. Trends in Ecology & Evolution 29, 390–397 (2014).
Bridge, T. C. L. et al. Depth-dependent mortality of reef corals following a severe bleaching event: implications for thermal refuges and population recovery. F1000 Research 2, https://doi.org/10.12688/f1000research.2-187.v3 (2014).
Hock, K. et al. Connectivity and systemic resilience of the Great Barrier Reef. PLoS Biol 15, e2003355 (2017).
Bongaerts, P. et al. Deep reefs are not universal refuges: Reseeding potential varies among coral species. Science Advances 3, e1602373 (2017).
Holstein, D. M. et al. Modeling vertical coral connectivity and mesophotic refugia. Coral Reefs 35, 25–37 (2016).
Rützler, K. & Macintyre, I. G. The Atlantic Barrier Reef Ecosystem at Carrie Bow Cay, Belize. I. Structure and Communities. Vol. 12 (Smithsonian Institution Press, 1982).
Villamizar, E. et al. Biodiversity, ecological structure, and change in the sponge community of different geomorphological zones of the barrier fore reef at Carrie Bow Cay, Belize. Marine Ecology 35, 425–435 (2014).
Helmuth, B. et al. High resolution spatiotemporal patterns of seawater temperatures across the Belize Mesoamerican Barrier Reef. figshare https://doi.org/10.6084/m9.figshare.c.5003012 (2020).
Helmuth, B. et al. Long-term, high frequency in situ measurements of intertidal mussel bed temperatures using biomimetic sensors. Scientific Data 3, 160087 (2016).
McField, M. D. Coral response during and after mass bleaching in Belize. Bulletin of Marine Science 64, 155–172 (1999).
Varela, R. et al. Coastal warming and wind-driven upwelling: A global analysis. Science of the Total Environment 639, 1501–1511 (2018).
Acknowledgements
The authors wish to thank Rich Aronson, Emily Carrington, Mike Carpenter, Ian McIntyre, Bill Precht, Klaus Ruetzler and Ken Sebens for their support with data collection and logistics and Lauren Szathmary, Sarah Gilman, and Allison Matzelle for assistance in data management. The authors also wish to thank the Smithsonian Caribbean Coral Reef Ecosystem Program for their support. This is contribution number 1044 of the Smithsonian’s Caribbean Coral Reef Ecosystems Program and contribution 412 of the Marine Science Center, Northeastern University.
Author information
Authors and Affiliations
Contributions
B.H., J.L., R.R., K.C., C.F. and S.J. oversaw instrument deployment, retrieval, and data collection. F.C. oversaw data management and database design and implementation. F.C. and B.H. led the writing of the manuscript and all authors contributed to the writing and editing of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Online-only Table
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
The Creative Commons Public Domain Dedication waiver http://creativecommons.org/publicdomain/zero/1.0/ applies to the metadata files associated with this article.
About this article
Cite this article
Helmuth, B., Leichter, J.J., Rotjan, R.D. et al. High resolution spatiotemporal patterns of seawater temperatures across the Belize Mesoamerican Barrier Reef. Sci Data 7, 396 (2020). https://doi.org/10.1038/s41597-020-00733-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-020-00733-6