Abstract
The behavioral dynamics within a social group not only could depend on individual traits and social-experience of each member, but more importantly, emerges from inter-individual interactions over time. Herein, we first present a dataset, as well as the corresponding original video recordings, of the results of 4 behavioral tests associated with fear and aggressive response performed on 106 Japanese quail. In a second stage, birds were housed with conspecifics that performed similarly in the behavioral tests in groups of 2 females and 1 male. By continuously monitoring each bird in these small social groups, we obtained time series of social and reproductive behavior, and high-resolution locomotor time series. This approach provides the opportunity to perform precise quantification of the temporal dynamics of behavior at an individual level within different social scenarios including when an individual showing continued aggressive behaviors is present. These unique datasets and videos are publicly available in Figshare and can be used in further analysis, or for comparison with existing or future data sets or mathematical models across different taxa.
Measurement(s) | locomotor behavior trait • social interaction measurement |
Technology Type(s) | video camera |
Factor Type(s) | sex • day of testing |
Sample Characteristic - Organism | Coturnix japonica |
Sample Characteristic - Environment | laboratory environment |
Machine-accessible metadata file describing the reported data: https://doi.org/10.6084/m9.figshare.10289750
Similar content being viewed by others
Background & Summary
The behavioral dynamics within a social group depends on many factors (i.e. individual traits, prior social-experience of its members, environmental context) and emerges from the interactions between its members over time. For example, individuals considered to be more aggressive toward conspecifics, may show more dominant behavior, and be more explorative, bold and active1,2. In farm animals it is well known that animals can be selected based on certain behavioral traits and this impacts, at least on a population level, on the overall behavioral performance in a wide variety of contexts. For example, quail selected by their high andrenocortical response to restraint, are more fearful in a wide variety of tests3 but also more aggressive in social groups, in comparison with those with low responsiveness4. Also, quail selected as chicks as highly sociable, are less fearful and less aggressive in social groups as juveniles than less sociable birds. Thus, the use of selection tests can favor a higher proportion of individuals with a desired characteristic5.
Although individual traits such as fearfulness and aggressiveness can affect the outcome of social interactions and the establishment of dominance hierarchies6,7,8, other factors such as previous fighting experience9, group size10,11,12 and housing conditions (i.e. size of box or cage10,13, presence or not of enrichment14,15) as well as the dynamical interaction between them16 can determine the social dynamics of a group. For instance, in large groups it has been proposed that tolerant social dynamics, that does not require individual recognition per se, emerges as the predominate social strategy10,11, while hierarchy formation is predominant in small groups12. Housing that is relatively small in relation to group size, can lead to high stocking density hence an increase of frequency of agonistic acts10,13.
In poultry, like other birds, within small groups hierarchies are established through a peck-order, according to which the animal that rank highest pecks at conspecifics and it is not pecked in return, and the opposite happens to the animals at the extreme bottom of the rank17,18. Hence, when two or more unacquainted adult birds are brought together, fights and pecks usually occur until each bird has established a dominance-subordination relationship with each other12. Thus, dominance is an emergent property that springs from the interaction of at least two individuals19, where a more aggressive bird in a specific environmental/social context becomes dominant. In this context, the study of hierarchical social groups in farm animals, and in poultry in particular12,20,21, has been widely addressed due to welfare implication. These welfare concerns arise from the observation in farms that, especially in small social groups, aggressive interactions target subordinates leading to high levels of social stress and in the worst cases to death.
Although important, the in-depth study on the behavioral patterns of individuals in their social environment is difficult from a methodological standpoint. In particular, tracking animals automatically within social groups present a unique difficulty, provided that animals in groups touch each other, move in paths that cross, and interact in complex ways, leading to an undesired switch of identities of unmarked individuals22. This is especially a problem when tracking poultry in groups, given that not only do animals frequently lay close to each other, but also during reproductive behavior male’s mount females (thus are literally on top of females) rendering automatic individualization during mounts impossible. Recent development of software has assessed this problem22,23,24. In particular, idTracker22 uses a multitracking algorithm that extracts a characteristic fingerprint from each animal in a video recording of a group. It then uses these fingerprints to identify every individual throughout the video. Tracking by identification minimizes propagation of errors, and thus correct identities can be maintained.
Once high-resolution individual tracking is achieved using specialized software, we are able to asses’ temporal patterns of locomotion of all individuals in the social group. Locomotor temporal patterns are particularly interesting given that they reflect both motivations to move (i.e. to feed, drink, or escape) and to remain immobile (i.e. when resting, fearful, threatened or are hiding). Moreover it is well known that locomotion does not occur randomly over time but rather presents long-term correlations (i.e. present behavior depends on past behavior25) and fractal dynamics (i.e. fluctuations occur on a broad range of time scales). These properties can be evaluated using the appropriate mathematical tools, thus providing insight on the temporal fractal complexity26,27,28. The degree of fractal complexity of behavior has been associated with health status29,30,31,32, stress33,34,35, welfare26,36, and environmental complexity28,32. Specifically, social stressors have been shown to induce changes in behavioral complexity26,32,36 highlighting the usefulness of this strategy in the study of organization of behavior within social groups.
Herein, in a first stage of the experimental setup we evaluate 106 Japanese quail (53 males and 53 females) in 4 experimental situations that can be associated with level of fearfulness or aggressiveness. In this context, longer latency to ambulate in a novel environment, longer tonic immobility reactions and more pronounced silence and inactivity during mechanical restraint have all been equated with increased fearfulness in several genetic lines of chickens and Japanese quail6,37,38,39,40,41,42,43. Aggressive behavior displayed during social interactions with an unknown conspecific or with a cagemate have been associated with levels of male aggressiveness44,45,46. In a second stage, social groups were arranged based on performance in preselection tests. The social groups were triads of 2 females and 1 male. This proportion 2:1 (female:male) allowed assessment of female—female as well as female-male interactions, while avoiding well documented violent male-male aggressions. Moreover, triads were used given that hierarchy can easily be visualized and it is well documented that in triads, predominately, linear hierarchy (i.e. if bird A dominates B and B dominates C, then A also dominates C) are established16,47,48. By continuously monitoring each bird in these social groups, we obtained time series of social and reproductive behavior and high-resolution locomotor time series. This approach provides the opportunity to perform precise quantification of the temporal dynamics of behavior at an individual level within their social environment including when one of the group members is showing continued aggressive behaviors. Elsewhere49 we show that subordinate animals (i.e., none or low levels of aggressive interactions; neutral groups) that are continuously pecked at during a 1 h period show quantitatively distinct dynamics of locomotion (i.e. lower level of fluctuation between immobility and mobility events, thus longer durations of events) in comparison to those that receive few or no aggression from conspecifics, deemed dominants. Moreover we show that subordinates also showed a high level of synchronization in locomotor pattern with the dominant member, likely reflecting a lack of “freedom” to perform locomotor behavior49. The data sets of all behavioral tests, the behavioral time series obtained in social groups of divergent characteristics described herein, as well as the corresponding original video recordings are publicly available on Figshare50,51,52,53. This data can be used to for comparison with existing or future data sets, and mathematical models developed in other species.
Methods
In this section we describe in more detail the methods described in Alcala et al.49. All the procedures were in compliance with the Guide for the Care and Use of Laboratory Animals issued by the National Institute of Health (NIH Publications, Eighth Edition). Experimental protocol was approved by the Institutional Council for the Care of Laboratory Animals (CICUAL, Comité Institutional de Cuidado de Animales de Laboratorio) of the Instituto de Investigaciones Biologicas y Technologicas (IIByT, CONICET - Universidad Nacional de Córdoba).
Animals and husbandry
The study was performed with Japanese quail (Coturnix japonica) a species widely used for studies covering neuroendocrine and social behaviors studies54,55. Also, they are considered an excellent laboratory model for the extrapolation of data to other poultry species with higher commercial relevance because of its high physiological similarity39,54. The animals were bred according to standard laboratory protocols56,57. Mixed-sex Japanese quail hatchlings were randomly housed in groups of 50–60 in white wooden brood boxes measuring 90 × 80 × 60 cm (length × width × height respectively) with a feeder along one wall, and 16 automatic nipple drinkers. A wire-mesh floor (1 cm grid) was raised 5 cm to allow the passage of excreta to the collection tray to facilitate cleaning and comfort of the animals, and a lid prevented the birds from escaping. Brooding temperature was 37.0 °C during the first week of life, with a weekly decline of 3.0 °C until room temperature (24 to 27 °C) was achieved. Food and water were provided ad libitum. The first week of life all animals were raised under the same standard conditions. Quail were subjected to a daily cycle of 14 h light (300 to 320 lx): 10 h dark (long photoperiod; photostimulated) throughout the study, with the exception of Photocastrated stimulus birds (for the Social interaction test, see below) that were submitted to a short photoperiod light cycle (06 h light: 18 h dark) beginning at 4 weeks of age until testing ended58.
At 28 days of age, test animals were sexed by plumage coloration, marked with a numbered wing band and randomly housed in pairs of 1 male and 1 female in cages of 20 × 40 × 20 cm (width × length × height respectively).
If an animal showed any indication of illness or escaped from their cage during rearing, they and their companion cagemate were completed excluded from the experiment.
Preselection of quail
One-hundred six quail were first evaluated in 4 preselection tests, separated between each other roughly by 30 days in order to favor independence between tests. These tests were used as a preselection criterion for social group testing. All data registered during the preselection tests are available in file “PreselectionTestsQuail.xls” and stored in the public repository Figshare52. Original video recordings are also available53. A schematically representation of the experimental design, tests and variables registered is shown in Fig. 1. In each test the order of testing of cages was randomized avoiding evaluation of adjacent cages consecutively. Both birds of the cage were always evaluated simultaneously. Moreover, the experimenters were always blinded regarding the prior history of the animals allocated in each group. With the exception of the tonic immobility test, all tests were recorded onto a computer and video recordings were analyzed the following days after testing by one previously trained experimenter.
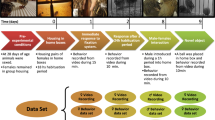
Schematically representation on experimental design. On the left the 4 preselection tests assed, namely, Partial mechanical restraint, Tonic Immobility, Social Interaction and Welfare assessment, and performance assessment. On the right a picture of the social group tests, where the individuals (1 male and 2 females) are observed as well as the feeder in the bottom left corner and automatic nipple in bottom right corner of the apparatus. The variables registered in each test is written in the proximity of the test name.
Partial mechanical restraint
This test has been proposed as a method to measure fear in quail6,59,60. Moreover, in juveniles, subsedative57 anxiolytic doses of Propofol have shown to reduce struggling (see below) to durations bellow 60 s (See pilot study results in Fig. 2). This test was performed at 40 days of age. This test consists in restricting the movement of the animal between two walls of a melamine box of 20 × 10 cm (height per width, respectively) with the characteristic that the front wall was made of glass (it allows the visualization of the animal and video recording of its behavior), and the back wall was adjustable to induce immobility in such a way that the animal cannot open the wings, but can move the head and legs9. The experimenters retreated out of the birds’ sight, and the test was during 5 minutes recorded with a video camera place in front of the box. All the birds were tested in 31 batches of 4 animals each, where the birds had no visual or physical contact between each other. The video was analyzed manually, and the following variables were recorded: the latency to struggle considered as time between the initiation of restraint until the first struggling episode (defined as the birds making fast movement with their legs when aiming to escape from the test apparatus) and the number of struggles events during the observation period. The struggle events were considered different if they were separated by 5 s or more. The immobility of the animal during the test has been widely considered in the literature as an indicator of fear6,59,60. Struggling during such restraint is known to be more pronounced in genetic lines of quail showing low rather than high levels of underlying fearfulness6,40. Those whose latency of struggle was >60 s were considered as fearful (Fig. 2)61.
Pilot study showing 60 s threshold for latency to stuggle during Partial Mechanical Restraint in Japanese quail treated with a subsedative anxiolytic dose of propofol. Latency to struggle during partial mechanical restraint in juvenile (31–32 days old) Japanese quail treated with either vehicle (i.e. distilled water and 20% Tween-80 (Sigma Chem. Co.)) or a propofol 10 mg/kg57. Birds were placed into the restraint apparatus ten min after intraperitoneal administration. Note that birds treated with Vehicle showed struggling latencies from 0–300 s while none of the birds treated with propofol showed latencies above 60 s. Dotted line indicates this 60 s threshold.
Social interaction (SI) test
This test is described in detail in Caliva et al. (2017) and measures levels of aggressiveness towards a non-aggressive photocastrated male opponent in a novel environment58. The SI test was performed over a five day period, between 70 and 74 days of age. Briefly, the SI test consists in a 5 min encounter between an unfamiliar test adult bird and a photocastrated stimulus adult male, in the presence of the test bird’s cagemate (audience). A video-camera was positioned 1 m above the apparatus and connected to a computer that allowed constant monitoring and recording during the test while out of the sight of the birds. Using commercially available behavioral tracking software (ANY-maze™, 2015) the number of events (i.e. continuous time performing the behavior separated from the following event by at least 5 s), duration of behavior (i.e. seconds performing behavior) and latency to initiate behavior (i.e. time from the start of the test until bird shows the first event) of the following aggressive behaviors were recorded:
Pecks: when one bird raises its head and vigorously pecks the other bird’s body (usually on the head).
Grabs: when a male catches (“grabs”) with their beak the neck or head region of the female.
Mounts: while performing a grab, the male approaches a female from behind, and places both feet on the dorsal surface of its torso, stepping over the females’ tail (adapted from62).
Cloacal contacts: during mounting, the male lifts his tail and tilts his pelvis underneath the other bird and briefly presses its cloaca against the female (adapted from62).
Threats: one bird stands with its neck and head raised in front of the other bird that usually has its’ head at a lower level than the first (adapted from62).
Chase: a bird runs after another that is escaping (adapted from63).
Herein, when grabs, mounts or cloacal contacts were performed by one male towards another male, they were considered as aggressive behaviors64. Birds that performed more than 5 aggressiveness behaviors were considered aggressive, and birds that did not perform any aggressive behavior towards the photocastrated opponent were considered non-aggressive58. If during the interaction a quail received more than 5 consecutive aggressive pecks, showed a clear and continued escaping (retrieval) behavior, and/or showed any sign of physical damage, the interaction was immediately interrupted65. Caliva et al. (2017) showed that in the SI test only 8% of the photostimulated females showed clear signs of aggression towards photocastrated opponent. The authors proposed that this is most likely due to the short duration of the test (up to 5 min), and that longer test durations are needed in females to observe significant aggressiveness (i.e. 3 h tests are performed in hens to establish dominance16,48,66). Thus, expression of aggressive behavior in females was very low.
Tonic immobility (TI)
The TI test was performed at 100 days of age. According to Jones67 this test induces an unlearned antipredator response that is triggered by a brief period of physical restraint. In the test the individual was place in the left lateral decubitus and hold for 15 seconds (the necessary time required to unleash the muscular immobilized tonic behavior), holding him with both hands against a support base (one hand on the head and another in the body). We recorded the number of inductions to achieve an immobility of at least 10 seconds and the duration of TI once induced. Maximum duration of TI was fixed at 5 minutes. Duration of TI implies both a behavioral and physiological response modulated by frightening situations and is considered as a measure of the level of fearfulness68,69. Thus, a long duration of TI and a smaller number of necessary inductions is indicative of a high level of fear as opposed to a short response67. If IT was not attained after 5 successive attempts, the bird was considered not to be susceptible and scores of 0 were given for TI duration. Thus “non-fearful” birds were selected based on those that needed 4 or more inductions, while the birds considered “fearful” required a single induction in the test.
Welfare assessment
At 96 and 108 days of age female skin lesions and plumage status were evaluated following a procedure proposed by Pellegrini et al.46 that is an adapted version of the protocol proposed by Welfare Quality® consortium70. Pellegrini et al. showed in Japanese quail that male aggressions toward a female cagemate can predict aggressiveness toward unknown conspecifics46. Head, neck and back skin lesions were determined using a score scale from 0 to 2 where “0” represents no lesions (punctiform damage <0.25 cm diameter) or scratches, “1” represents less than 3 lesion or scratches, and “2” reflects 3 or more lesion or scratches. Head, neck and back plumage damage was also determined using a score scale from 0 to 2 as follows: “0” represents individuals with no plumage damage or slight wear (only single feathers lacking), “1” represent individuals with one or more body parts that have moderate wear (i.e. damaged feathers worn or deformed) or one or more featherless areas <1.5 cm in diameter at the larger extent and “2” corresponded to individuals that have at least one featherless area >1.5 cm in diameter at the largest extent. None of the males showed plumage damage or lesions, thus, only male aggression towards females were considered in this analysis. Plumage damage induced by males (score > 0) to their female cagemates were considered as indicative of male aggressiveness46. Non-aggressive males were those in which no plumage damage was seen in female cagemates46. It should be noted that if at any point in the study a bird showed severe lesions they were separated from their cagemates and thus both cagemates were eliminated from the study in order to protect the welfare of the animals. Due to this systematic standard laboratory procedure, very aggressive birds were excluded from the study even at a young age.
Performance assessment
Birds were weighed at 28 days of age. The weight of birds transferred to cages ranged between 100–150 g. Thereafter, weight and male cloacal gland width and length, and male foam production were recorded weekly until 9 weeks of age, when all males showed completed gonadal development (Cloacal gonadal volume CGV > 1000 mm3). Cloacal gland volume was estimated as (4/3 × 3.5414 × a × b2), where a = 0.5 × length, and b = 0.5 × width71. Foam production was quantified by subjective scaling of the amount of foam ejected upon manual expression (squeezing) of the foam gland, using a scale of 1 (no foam expressed) to 5 (maximum amount of foam expressed). Female quail egg production was monitored throughout the study and all females reached peak egg production. All birds were also weighed after the last behavioral test at 92 days of age, and male cloacal gland size and foam production also assessed. This data is available in file “PreselectionTestsQuail.xls” and stored in the public repository Figshare52, and Frequency distribution of variables are shown in Fig. 3.
Frequency distribution of preselection test variables. Frequency distribution of (a) latency to struggle during Partial Mechanical Restraint, (b) number of inductions in Tonic Immobility test and (c) the duration of the tonic immobility, (d) latency to ambulate during the first stage and (e) total duration of aggressions in the Social Interaction test, and (f) aggressive in home cage valued by welfare assessment of cagemate. Frequency distribution of variables “a”-“d” are shown for data pooled from females and males, while variables “e”-“f” are only from males. A total of 106 animals were studied, half female and half males.
Principal component analysis of preselection tests
Principal Component Analysis biplots of the preselection tests are shown in Fig. 4, and illustrates the relationship between variables, and selection criteria. Statistical independence (R2 = 0.04 and ~90° angles in the PCA biplot (lines in Fig. 4)) was observed between variables of two different tests, namely latency to struggle during partial mechanical restraint and number of inductions for tonic immobility. However, 74% of females that were fearful in the tonic immobility test (i.e. only needed one induction) also showed low level of struggling (i.e. ≤3 struggling bouts), in comparison to 34% (P < 0.05, 2-tailed proportion test) that showed low struggling and were found to be less fearful (i.e. tonic immobility was not induced or only after 5 inductions). This was not evident in males (65% and 68% fearful and non-fearful during tonic immobility, respectively). In all, these results show that, at least in females, highly fearful birds during tonic immobility on average were also more fearful during restraint.
Principal components analysis biplot of the preselection tests data. Each point represents (a) a female or (b) a male quail. Variables are ploted as vectors from the origen ending in black triangles. Birds used for social groups type A are shown in black circles while those used for type B are shown in gray circles (see main tet for definition). The percent of the eigenvalues of each PC are shown in brackets next to each component. Only variables with low levels (R2 < 0.40) of correlations between them were used in the analysis: latency to struggle during Partial Mechanical Restraint (Lat. struggle (PMR)), number of inductions (Numb. Inductions (TI)) and the duration of the tonic immobility (Durat. t. immobility (TI)) during the Tonic Immobility test, latency to ambulate during the first stage (Lat. ambulation (SI)) and total duration of aggressions (Durat. aggression (SI)) in the Social Interaction test, and aggressive in home cage valued by welfare assessment of cagemate (Aggressive HC (WA)). A total of 106 animals were studied, half female and half males.
Considering results obtained in preselection test birds were classified in 2 types: 1) Type A (Fig. 4 black circles) had birds that were fearful in both the Tonic immobility and the partial mechanical restraint test. These males also were aggressive in the Social interaction test or their female cagemate showed higher scores during Welfare assessment. 2) Type B (Fig. 4 gray circles) had females and males that were not fearful in the Tonic immobility test, and males that tested non-aggressive in both the Social interaction test and in their home cage. No differences between body weight, cloacal gland volume or foam production was observed between types. It should be noted that there are males that were not used for the social group tests (Fig. 4 open circles) because they were used in a separate neurobiology experiment.
Social group testing
Novel social groups (2 females: 1 male) of animals (156–171 days old) that behaved coherently in the 4 preselection tests described in the previous section, thus half of the 12 social groups evaluated had type A birds and the other half type B birds (see previous subsection). Social groups were housed in a white wooden apparatus measuring 80 × 40 × 40 cm (width × length × height, respectively) with wood-shavings on the floor. A feeder and an automatic nipple drinker were positioned in opposite corners of the apparatus (Fig. 1, left and right bottom corner of box in the photograph, respectively). Nylon monofilament line was extended over the top of the boxes with a 1 cm separation in order to prevent the birds from escaping without interfering with their visualization. A video camera was suspended 1.5 m above the box. Since only 4 social groups could be tested simultaneously, the setup was repeated 3 consecutive times. For convenience, boxes in which each social group were placed were numbered from 1 to 12. Boxes 1–4 were tested simultaneously first, 5–8 second and 9–12 last. Video recordings are publicly available on Figshare53.
We used IdTracker22 in MATLAB R2017a to register x, y coordinates of each animal within the social group during a 1 h period immediately after being placed in the test apparatus between 9 and 10 am, and 48 hours after testing began. Locomotion was than estimated at 0.5 s intervals (7200 time intervals) using customized code Locomotion.m that calculates the distance moved by the animal, converts distance expressed in pixel to centimeters, and if the distance moved is above a threshold of 1 cm that the animal is considered mobile72. Thus, the recorded behavioral data is expressed in the form of a time series of mutually exclusive states. At any given time, if the bird was moving a number one was recorded or a zero if immobile. These locomotor time series are publicly available and stored in the public repository Figshare51.
Time series of non-locomotor behaviors were obtained through visual observation of video recordings using as an interface ANY-MAZE@ to register behavior. For each bird, when the specific behavior was performed the corresponding key was pressed until the bird finished performing the behavior, thus a binary time series, xi, sampled at up to 2 data points per second was constructed for each behavior.
Only one observer recorded all data in order to avoid inter-individual variability. Prior to video analysis observer performed training sessions than consisted in analyzing the same three behavioral video at least two times. Then, reliability was estimated to be >95%. (formula: number of agreements/number of agreements + number of disagreements).
The following behaviors were recorded: Pecks, Grabs, Mounts, Cloacal contacts, Threats, Chase, as described previously for the SI test, and additionally, Foraging: pecking at the ground or actively moving litter with beak, Feeding: peaking at food in the feeding trough, and Dust bathing: vertical wing shakes in a lying position73. From the behavioral time series both frequency and durations of behaviors can be easily estimated. These behavioral time series are also publicly available and stored in the public repository Figshare50.
Data Records
Original video recordings53 of Partial Mechanical Restraint, Social Interaction test and Social groups are provided in avi or mod format. File names include the abbreviation, PMR, SI or Box, respectively. For video file of the Partial Mechanical Restraint and Social Interaction tests file names also include the ID of the animals tested. In the case of social groups box number (1–12) and the day of testing (day1 or day3) is provided in the file name.
The results of the preselection tests are presented in the excel file “PreselectionTestsQuail.xls” stored in the public repository Figshare52 with the headers of the columns representing the variable analyzed in each test for each animal (rows). Table 1 presents all the column headers as well as a brief definition of the variable.
All time series from this study are stored in Figshare as text files (.txt). For practical purposes, locomotor data obtained from IdTracker are in separate files51 from the behavioral data time series obtained from AnyMaze52. Locomotor data consists of a single column of data sampled at 0.5 s intervals (as explained previously). For the behavioral data the first column refers to the time while the following columns refer to the behavioral data. Considering that the 36 animals were evaluated in 12 mixed-sex groups of 3 birds in individual experimental boxes on the first hour (Day1) and 48 hours (Day3) after test initiation, each subject quail was identified by their experimental group number (Box), ID number of wing band, and sex (femaleA, femaleB or male). In the case of females, an indication A or B is used to discriminate between the two. In the file name, an indication of the corresponding bird is also provided as “BoxN°_IDN°_sex_DayN°” (Table 2) for recorded obtained.
Technical Validation
All data analysis and technical validation was performed by one observer both in Any-Maze as well as in IdTracker. In both cases the observer was blinded regarding the prior history of the animals allocated in each group. One of the advantages of IdTracker is that the researcher can perform visual observation of the tracking performed on each frame analyzed using the complementary software IdPlayer. A number (see Fig. 1) in the center of the of the animal a number indicates the identity of the bird. In the case of identification errors, they were corrected manually using this software. In order to validate the correct tracking and identification of the animal, visual observations of tracking were performed for all birds. The high contrast between the white, well illuminated, box and the dark brown quail feathers facilitated an accurate tracking of the animal. Also animals had small white markings of their backs that allowed identification from video recordings.
Behavioral data sets were collected using the commercially available ANY-maze™ Video Tracking System software that can be downloaded at www.anymaze.com. Since in this software keystrokes allow observer to register manually behaviors from video recording, a validation period to guarantee reproducibility was first performed. Observer performed validation sessions than consisted in analyzing the same three behavioral video at least two times. Then, reliability was estimated to be >95%. (formula: number of agreements/number of agreements + number of disagreements).
Code availability
IdTracker22 is a videotracking software that keeps the correct identity of each individual during video behavioral analysis and is publicly available at http://www.idtracker.es/. ANY-MAZE@ is a licensed video tracking program, that can be downloaded from http://www.anymaze.co.uk/. The customized Matlab code customized code Locomotion.m in publicly available on Figshare72.
References
Finkemeier, M. A., Langbein, J. & Puppe, B. Personality Research in Mammalian Farm Animals: Concepts, Measures, and Relationship to Welfare. Front Vet Sci 5, 131, https://doi.org/10.3389/fvets.2018.00131 (2018).
Koolhaas, J. M. et al. Coping styles in animals: current status in behavior and stress-physiology. Neurosci Biobehav Rev 23, 925–935 (1999).
Koolhaas, J. M. & Van Reenen, C. G. ANIMAL BEHAVIOR AND WELL-BEING SYMPOSIUM: Interaction between coping style/personality, stress, and welfare: Relevance for domestic farm animals. J Anim Sci 94, 2284–2296, https://doi.org/10.2527/jas.2015-0125 (2016).
Favati, A., Leimar, O., Radesater, T. & Lovlie, H. Social status and personality: stability in social state can promote consistency of behavioural responses. Proc Biol Sci 281, 20132531, https://doi.org/10.1098/rspb.2013.2531 (2014).
Portugal, S. J. et al. Boldness traits, not dominance, predict exploratory flight range and homing behaviour in homing pigeons. Philos Trans R Soc Lond B Biol Sci 372, https://doi.org/10.1098/rstb.2016.0234 (2017).
Jones, R. B. & Satterlee, D. G. Threat-induced behavioural inhibition in Japanese quail genetically selected for contrasting adrenocortical response to mechanical restraint. Brit Poultry Sci 37, 465–470 (1996).
Koski, S. E. Broader horizons for animal personality research. Frot Ecol Evol 28, https://doi.org/10.3389/fevo.2014.00070 (2014).
van der Meer, E. & van Oers, K. Gender and Personality Differences in Response to Social Stressors in Great Tits (Parus major). Plos One 10, e0127984, https://doi.org/10.1371/journal.pone.0127984 (2015).
Jones, R. B., Satterlee, D. G., Waddington, D. & Cadd, G. G. Effects of repeated restraint in Japanese quail genetically selected for contrasting adrenocortical responses. Physiol Behav 69, 317–324 (2000).
Estevez, I., Anderson, I. L. & Nævdal, E. Group size, density and social dynamics in farm animals. Appl Anim Behav Sci 103, 185–204 (2007).
Estevez, I., Keeling, L. J. & Newberry, R. C. Decreasing aggression with increasing group size in young domestic fowl. Appl Anim Behav Sci 84, 213–218 (2003).
Guhl, A. M. Social Behavior of the Domestic Fowl. Trans Kans Acad Sci 71, 379–384 (1968).
Al-Rawi, B. & Craig, J. V. Agonistic behavior of caged chickens related to group size and area per bird. Appl Animal. Ethol 2, 68–80 (1975).
Haemisch, A., Voss, T. & Gartner, K. Effects of environmental enrichment on aggressive behavior, dominance hierarchies, and endocrine states in male DBA/2J mice. Physiol Behav 56, 1041–1048 (1994).
Moroki, Y. & Tanaka, T. A pecking device as an environmental enrichment for caged laying hens. Anim Sci J 87, 1055–1062, https://doi.org/10.1111/asj.12525 (2016).
Chase, I. D. Dynamics of hierarchy formation: the sequential development of dominance relationships. Behav 80, 218–239 (1982).
Masure, R. H. & Allee, W. C. The social order in flocks of the common chicken and the pigeon. Auk 51, 306–327 (1934).
Schjelderup-Ebbe, T. Beitr/ige zur Sozialpsychologie des Haushuhns. Z. Psychol 88, 225–252 (1922).
Barrette, C. The ‘inheritance of dominance’, or of an aptitude to dominate? Anim Behav 46, 591–593 (1993).
Marin, R. H., Liste, M. G., Campderrich, I. & Estevez, I. The impact of phenotypic appearance on body weight and egg production in laying hens: a group-size- and experience-dependent phenomenon. Poult Sci 93, 1623–1635, https://doi.org/10.3382/ps.2013-03705 (2014).
McBride, G., Parer, I. P. & Foenander, J. M. The social organization and behaviour of the feral domestic fowl. Anim Behav Monogr 2, 125–181 (1969).
Perez-Escudero, A., Vicente-Page, J., Hinz, R. C., Arganda, S. & de Polavieja, G. G. idTracker: tracking individuals in a group by automatic identification of unmarked animals. Nat Methods 11, 743–748, https://doi.org/10.1038/nmeth.2994 (2014).
Branson, K., Robie, A. A., Bender, J., Perona, P. & Dickinson, M. H. High-throughput ethomics in large groups of Drosophila. Nat Methods 6, 451–457, https://doi.org/10.1038/nmeth.1328 (2009).
Swierczek, N. A., Giles, A. C., Rankin, C. H. & Kerr, R. A. High-throughput behavioral analysis in C. elegans. Nat Methods 8, 592–598, https://doi.org/10.1038/nmeth.1625 (2011).
Kantelhard, J. W., Koscielny-Bunde, E., Rego, H. H. A., Havlin, S. & Bunde, A. Detecting long-range correlations with detrended fluctuation analysis. Phys A 295, 441–454 (2001).
Rutherford, K. M. D., Haskell, M. J., Glasbey, C., Jones, R. B. & Lawrence, A. B. Fractal analysis of animal behaviour as an indicator of animal welfare. Anim Welfare 13, 99–103 (2004).
Paulus, M. P. & Geyer, M. A. Three independent factors characterize spontaneous rat motor activity. Behav Brain Res 53, 11–20 (1993).
Kembro, J. M., Perillo, M. A., Pury, P. A., Satterlee, D. G. & Marín, R. H. Fractal analysis of the ambulation pattern of Japanese quail. Brit Poultry Sci 87, 2186–2195 (2009).
Alados, C. L. & Huffman, M. A. Fractal long-range correlations in behavioural sequences of wild chimpanzees: a non-invasive analytical tool for the evaluation of health. Ethology 106, 105–116 (2000).
Burgunder, J., Petrželková, K. J., Modrý, D., Kato, A. & MacIntosh, A. J. J. Fractal measures in activity patterns: Do gastrointestinal parasites affect the complexity of sheep behaviour? Appl Anim Behav Sci 205, 44–53 (2018).
Goldberger, A. L. et al. Fractal dynamics in physiology: Alterations with disease and aging. Proc Natl Acad Sci USA 99, 2466–2472 (2002).
Macintosh, A. J., Alados, C. L. & Huffman, M. A. Fractal analysis of behaviour in a wild primate: behavioural complexity in health and disease. J R Soc Interface 8, 1497–1509, https://doi.org/10.1098/rsif.2011.0049 (2011).
Kembro, J. M., Satterlee, D. G., Schmidt, J. B., Perillo, M. A. & Marín, R. H. Open-Field Temporal Pattern of Ambulation in Japanese Quail Genetically Selected for Contrasting Adrenocortical Responsiveness to Brief Manual Restraint. Poult Sci 87, 2186–2195 (2008).
Rutherford, K. M., Haskell, M., Glasbey, C., Jones, R. B. & Lawrence, A. Detrended fluctuation analysis of behavioural responses to mild acute stressors in domestic hens. Appl Anim Behav Sci 83, 125–139 (2003).
Rutherford, K. M., Haskell, M. J., Glasbey, C. & Lawrence, A. B. The responses of growing pigs to a chronic-intermittent stress treatment. Physiol Behav 89, 670–680, https://doi.org/10.1016/j.physbeh.2006.08.006 (2006).
María, G. A., Escós, J. & Alados, C. L. Complexity of behavioural sequences and their relation to stress conditions in chickens: a non-invasive technique to evaluate animal welfare. Appl Anim Behav Sci 86, 93–104 (2004).
Craig, J. V., Craig, T. P. & Dayton, A. D. Fearful behavior by hens of two genetic stocks. Appl Anim. Ethol 10, 263–273 (1983).
Gallup, G. G. Jr. Tonic immobility as a measure of fear in domestic fowl. Anim Behav 20, 166–169 (1979).
Jones, R. B. Fear and adaptability in poultry: insights. World’s Poultry Sc J 52, 131–170 (1996).
Jones, R. B., Mills, A. D., Faure, J. M. & Williams, J. B. Restraint, fear and distress in Japanese quail genetically selected for long or short tonic immobility reactions. Physiol Behav 56, 529–534 (1994).
Jones, R. B., Satterlee, D. G., Hughes, H. L. M. & Black, A. J. The effect of environmental factors on activity, selected behaviour patterns and “fear” of fowls in cages and pens. Br Poult Sci 15, 375–380 (1974).
Mauldin, J. M. & Siegel, P. B. “Fear”, head shaking and production in five populations of caged chickens. Br Poult Sci 20, 39–44 (1979).
Sefton, A. E. The interactions of cage size, cage level, social density, fearfulness and production of single comb White Leghorns. Poult Sci 55, 1922–1926 (1976).
Hirschenhauser, K., Gahr, M. & Goymann, W. Winning and losing in public: audiences direct future success in Japanese quail. Horm Behav 63, 625–633, https://doi.org/10.1016/j.yhbeh.2013.02.010 (2013).
Hirschenhauser, K., Wittek, M., Johnston, P. & Mostl, E. Social context rather than behavioral output or winning modulates post-conflict testosterone responses in Japanese quail (Coturnix japonica). Physiol Behav 95, 457–463, https://doi.org/10.1016/j.physbeh.2008.07.013 (2008).
Pellegrini, S., Condat, L., Marin, R. H. & Guzman, D. A. Can Japanese quail male aggressions toward a female cagemate predict aggressiveness toward unknown conspecifics? Livest Sci 22, 65–70 (2019).
Chase, I. D., Tovey, C., Spangler-Martin, D. & Manfredonia, M. Individual differences versus social dynamics in the formation of animal dominance hierarchies. Proc Natl Acad Sci U S A 99, 5744–5749, https://doi.org/10.1073/pnas.082104199 (2002).
Cloutier, S., Beaugrand, J. P. & Lague, P. C. The role of individual differences and patterns of resolution in the formation of dominance orders in domestic hen triads. Behav Processes 38, 227–239 (1996).
Alcala, R., Caliva, J. M., Flesia, A. G., Marin, R. H. & Kembro, J. M. Aggressive dominance can decrease behavioral complexity on subordinates through synchronization of locomotor activities. Comunications Biology https://doi.org/10.1038/s42003-019-0710-1 (2019).
Alcala, R., Caliva, J. M., Marin, R. H. & Kembro, J. M. One-hour social and reproductive behavioral time series of Japanese quail in diverse social environments. figshare, https://doi.org/10.6084/m9.figshare.7117679.v1 (2019).
Alcala, R., Caliva, J. M., Marin, R. H. & Kembro, J. M. High-resolution, 1-hour, locomotor time series of Japanese quail in diverse social environments. figshare, https://doi.org/10.6084/m9.figshare.7117631.v1 (2019.
Caliva, J. M., Alcala, R., Guzman, D. A., Marin, R. & Kembro, J. M. Four behavioral tests associated with fear and aggressiveness in Japanese quail. figshare, https://doi.org/10.6084/m9.figshare.7122926.v1 (2019).
Kembro, J. M., Guzmán, D., Caliva, J. M., Alcala, R. & Marin, R. H. High-resolution behavioral time series of Japanese quail within their social environment. figshare, https://doi.org/10.6084/m9.figshare.c.4424327 (2019).
Ball, G. F. & Balthazart, J. Japanese quail as a model system for studying the neuroendocrine control of reproductive and social behaviors. ILAR J 51, 310–325 (2010).
Balthazart, J., Baillien, M., Charlier, T. D., Cornil, C. A. & Ball, G. F. The neuroendocrinology of reproductive behavior in Japanese quail. Domestic Animal Endocrinology 25, 69–82 (2003).
Shanaway, M. M. Quail productiion systems. (FAO, 1994).
Kembro, J. M., Guzman, D. A., Perillo, M. A. & Marin, R. H. Temporal pattern of locomotor activity recuperation after administration of propofol in Japanese quail (Coturnix coturnix japonica). Res Vet Sci 93, 156–162, https://doi.org/10.1016/j.rvsc.2011.06.011 (2012).
Caliva, J. M., Kembro, J. M., Pellegrini, S., Guzman, D. A. & Marin, R. H. Unexpected results when assessing underlying aggressiveness in Japanese quail using photocastrated stimulus birds. Poult Sci 96, 4140–4150, https://doi.org/10.3382/ps/pex258 (2017).
Moriarty, A. G. Anxiogenic effects of a β-Carboline on a tonic immobility and open field behaviour in chickens (Gallus gallus). Pharm Bioch Behav 51, 795–798 (1995).
Faure, J. M., Jones, R. B. & Bessei, W. Fear and social motivation in open-field behaviour of the domestic chick. A theoretical consideration. Biol Behav 8, 103–116 (1983).
Kembro, J. M. Estudios sobre la dinámica temporal de locomoción y su modulación por eventos estresantes y sustancias neuroactivas Ph.D. thesis, Universidad Nacional de Cordoba (2010).
McGary, S., Estevez, I. & Russek-Cohen, E. Reproductive and aggressive behavior in male broiler breeders with varying fertility. Appl Anim Behav Sci 82, 29–44 (2003).
Ramenofsky, M. Agonistic behaviour and endogenous plasma hormones in male Japanese quail. Anim Behav 32, 698–708 (1984).
Adkins-Regan, E. Male-male sexual behavior in Japanese quail: Being “on top” reduces mating and fertilization with females. Beh Proc 108, 71–79 (2015).
Dominchin, M. F., Busso, J. M., Kembro, J. M., Marin, R. H. & Guzman, D. A. Divergent cloacal gland photo-responsiveness in male Japanese quail exposed to short days and associated differences in social interactions and reproduction. Poult Sci 96, 5–13, https://doi.org/10.3382/ps/pew287 (2017).
Cloutier, S., Beaugrand, J. P. & Lague, P. C. The effect of prior victory or defeat in the same site as that of subsequent encounter on the determination of dyadic dominance in the domestic hen. Behav Processes 34, 293–298 (1995).
Jones, R. B. The tonic immobility reaction of the domestic fowl: a review. World’s Poultry Sc J 42, 82–96 (1986).
Forkman, B., Boissy, A., Meunier-Salaün, M.-C., Canali, E. & Jones, R. B. A critical review of fear tests used on cattle, pigs, sheep, poultry and horses. Physiol Behav 92, 340–374 (2007).
Jones, R. B., Mills, A. D. & Faure, J. M. Genetic and experiential manipulation of fear-related behavior in Japanese quail chicks (Coturnix coturnix japonica). J Comp Psychol 105, 15–24 (1991).
WelfareQuality®. Welfare Quality® Assessment protocol for poultry (broilers, laying hens). (Welfare Quality® Consortium, 2009).
Marín, R. H. & Satterlee, D. G. Cloacal Gland and testes Development in Male Japanese Quail Selected for Divergent Adrenocortical Responsiveness. Poultry Science 83, 1028–1034 (2004).
Kembro, J. M. Source code for: Locomotor time series from x,y IdTracker coordinates in Matlab. figshare, https://doi.org/10.6084/m9.figshare.7716284 (2019).
Louton, H., Bergmann, S., Reese, S., Erhard, M. H. & Rauch, E. Dust-bathing behavior of laying hens in enriched colony housing systems and an aviary system. Poult Sci 95, 1482–1491, https://doi.org/10.3382/ps/pew109 (2016).
Acknowledgements
This research was financially supported by Fondo para la Investigación Científica y Tecnológica (FONCyT) grant N° PICT-2016-0282, Consejo Nacional para Investigaciones Científicas y Técnicas (CONICET), and Secretaría de Ciencia y Técnica (SeCyT), Universidad Nacional de Córdoba, Argentina. A.G.F., D.A.G., J.M.K. and R.H.M. are career members of CONICET. J.M.C. has a PhD scholarship from the later institution.
Author information
Authors and Affiliations
Contributions
J.M.K., J.M.C., R.S.A., D.A.G. and R.H.M. conceived and designed research strategy and edited and revised manuscript; R.A., J.M.C. and J.M.K. performed experiments; J.M.K. drafted the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
The Creative Commons Public Domain Dedication waiver http://creativecommons.org/publicdomain/zero/1.0/ applies to the metadata files associated with this article.
About this article
Cite this article
Caliva, J.M., Alcala, R.S., Guzmán, D.A. et al. High-resolution behavioral time series of Japanese quail within their social environment. Sci Data 6, 300 (2019). https://doi.org/10.1038/s41597-019-0299-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-019-0299-8
This article is cited by
-
The relevance of a right scale for sampling when studying high-resolution behavioral dynamics
Scientific Reports (2023)
-
Aggressive dominance can decrease behavioral complexity on subordinates through synchronization of locomotor activities
Communications Biology (2019)