Abstract
The marine iodine cycle has significant impacts on air quality and atmospheric chemistry. Specifically, the reaction of iodide with ozone in the top few micrometres of the surface ocean is an important sink for tropospheric ozone (a pollutant gas) and the dominant source of reactive iodine to the atmosphere. Sea surface iodide parameterisations are now being implemented in air quality models, but these are currently a major source of uncertainty. Relatively little observational data is available to estimate the global surface iodide concentrations, and this data has not hitherto been openly available in a collated, digital form. Here we present all available sea surface (<20 m depth) iodide observations. The dataset includes values digitised from published manuscripts, published and unpublished data supplied directly by the originators, and data obtained from repositories. It contains 1342 data points, and spans latitudes from 70°S to 68°N, representing all major basins. The data may be used to model sea surface iodide concentrations or as a reference for future observations.
Measurement(s) | iodide |
Technology Type(s) | digital curation |
Factor Type(s) | sampling time and place |
Sample Characteristic - Environment | sea water • ocean |
Sample Characteristic - Location | Earth (planet) |
Machine-accessible metadata file describing the reported data: https://doi.org/10.6084/m9.figshare.10130129
Similar content being viewed by others
Background & Summary
There has recently been a resurgence of interest in the marine iodine cycle, reflecting its involvement in a diverse range of processes, from influencing air quality (e.g.1) to recording ocean deoxygenation in sediments (e.g.2). Iodine is a redox active element that is present in seawater in two main forms, iodide (I−) and iodate (IO3−). Sea-to-air transfer is the dominant source of iodine to the atmosphere, where it is subject to atmospheric processing prior to deposition back to the sea or onto land. It is an essential nutrient for many organisms including humans, and deficiency in humans leads to goitre, cretinism and is the leading cause of preventable mental retardation globally3. Iodine radionuclides are also released to the oceans by anthropogenic activities, where they will be subject to the same processes of biogeochemical cycling and volatilisation as the naturally occurring stable isotope4. Despite the wide ranging impacts of iodine biogeochemistry, the distribution of iodine species in the oceans remains relatively poorly understood. Here we present an updated compilation of all currently available sea surface iodide concentrations. The data set is specifically intended to inform studies of the sea-air exchange of iodine species, but may also be of use in improving understanding of the marine iodine cycle more generally.
The reaction of iodide with ozone at the surface of the ocean has been established as an important sink for ozone, thought to be responsible for around one third of the total ozone loss by dry deposition5. The reaction liberates reactive iodine compounds to the atmosphere, which in turn contribute to further ozone removal processes. Gas phase reactions involving iodine are estimated to account for up to 15% of tropospheric ozone losses6. To incorporate this chemistry, global and regional air quality and atmospheric chemistry models have begun to include predicted sea-surface iodide fields derived from parameterisations (e.g.5,7,8,9). However, current sea surface iodide parameterisations are known to have biases10, are subject to substantial uncertainty6, and do not take advantage of recent and substantial increases in the number of available observations (e.g.11).
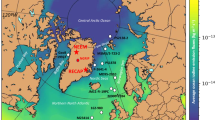
The observational data underpinning iodide parameterisations is sparse, and has hitherto not been publicly available in a collated form. In many cases, iodide observations are not readily accessible in a digital form (i.e. are only presented in graphical format). To facilitate the development and validation of improved sea surface iodide parameterisations, we have compiled all available sea surface iodide observations. The dataset is an extended version of that used in our earlier publication12, in which we described the large scale sea surface iodide distribution and presented correlations between iodide and other oceanographic variables, but did not publish the observations themselves. The dataset we now present incorporates more than 400 new observations (see Fig. 1), including new, basin scale transects from the Indian Ocean (currently unpublished) and the tropical eastern Pacific11, both of which were previously undersampled12. This new extended dataset is freely available via the British Oceanographic Data Centre (BODC; http://doi.org/czhx)13.
We anticipate that the primary use of the dataset will be modelling of ozone deposition to the sea surface and/or associated trace gas emissions to the atmosphere. It has been used to generate new monthly parameterised sea-surface iodide fields (12 × 12 km resolution) using a machine learning approach, described in our accompanying partner publication10. The dataset may also be of interest in other areas of iodine research. In particular, improved understanding of the marine iodine cycle is needed to refine the use of iodine speciation as a paleo-oceanographic tracer of past ocean oxygenation (e.g.2), and to better predict the impacts of iodine radionuclides released to the environment by anthropogenic activities (e.g.4).
Methods
Data compilation
The data set includes iodide measurements made by a number of different research groups (Online-only Table 1). These were collated from the following sources:
- A.
Published manuscripts. Data was digitised from tables and graphics, either by hand or using the free online tool WebPlotDigitizer (https://automeris.io/WebPlotDigitizer).
- B.
Data originators. Data (both published and unpublished) was provided directly by the owners.
- C.
Data repositories. Data was obtained by request or on-demand download from hosting repositories (e.g. BODC, PANGAEA, the US JGOFS Data System).
Following the approach adopted previously12, ‘surface’ concentrations are considered to be those from depths of less than 20 m. As discussed in Chance et al.12, the ocean is usually considered well mixed to this depth, and to restrict ‘surface data’ to shallower depths would substantially reduce the number of observations included. We examined a sub-set of data (n = 93) where observations were available from multiple depths within the upper 20 m of the water column. While significant differences were occasionally found between individual pairs of samples collected from depths of ~1-2 m and ~10 m at a given station, concentrations were within 10 nM in almost 50% of pairs (49.5%), and 80% were within 26 nM. Statistical analysis (using a paired students t-test) found no significant difference between samples from different depths within the upper 20 m. The exact depth of near surface samples can itself have high relative uncertainty, as factors such as sea swell can lead to metre scale fluctuations to the exact depth of e.g. a ship seawater inlet. Furthermore, the exact depths of such inlets, or the ‘surface’ sample bottle, was not always stated in the original data sources. Therefore, we have not included depth as a parameter in our compiled data set and no distinction has been made between samples obtained using a CTD rosette fitted with Niskin bottles (or similar), a pumped underway seawater supply or a manual method (such as bucket sampling).
Each data set was entered onto an individual Excel spreadsheet in a standard format. Rarely, source values were below the limit of detection (LoD) for the method used. Where this was the case, we have used a substitute value of 0.75 x the estimated LoD and the data point was flagged (column ‘ErrorMethod’). No further processing has been applied to any of the data. It has not been normalised e.g. to salinity. Required fields from individual ‘input’ files were then collated into a single comma-separated value (.csv) file using open-source Python code, including the Pandas package14.
A total of 1342 observations, from 57 individual data sets has been collated (Online-only Table 1). This is an increase of 417 observations (45%) on that included in our earlier compilation12. Locations of individual data points are shown in Fig. 1, which highlights how the expanded dataset increases spatial coverage. The earliest observations were made in 1967 and the most recent in 2018. For some data points (n = 32) the date of sampling is not specified as this was not given in the original publication. Ten of the input data sets are currently unpublished.
Additional fields
Each iodide observation is associated with the record fields listed in Table 1. In addition to spatial and temporal co-ordinates, the estimated uncertainty and analytical method used to generate the observations are provided.
Method
Analytical methods are summarised in Table 2. In the majority of cases (~53%), iodide was measured by cathodic stripping square wave voltammetry (CSSWV) according to the method of Campos15. However a range of other measurements techniques were also used. Iodide was sometimes measured as the difference between the total inorganic iodine (TII) concentration and the iodate concentration.
Uncertainty
Measurements of iodide in seawater are subject to non-trivial analytical uncertainties, which should be considered when using the data set. An estimate of the uncertainty associated with each observation has been included, using either information provided by the data source where available, or comparison to other measurements using the same analytical method. The uncertainty estimates provided are typically derived from replicate analyses of the same sample, and so represent the precision of the measurements. As insufficient information was available to quantify the precision in the same way for all observations, the approach used to estimate the precision is also included (see Table 1). Relative uncertainty estimates for each analytical method, for typical ambient concentrations in a seawater matrix, are also provided in Table 2. The precision given for each data set is often 5% (Table 2), which reflects the stated repeatability of the CSSWV method15 and a number of other measurements used. However, we note that repeat analyses of samples using this method can sometimes give lower precision (e.g. ~10%)16. Considering all data points in our dataset, we find ~75% have a precision of 10% or less, and ~51% have an precision of 5% or less. Such uncertainties are modest in comparison to the global scale variation in sea surface iodide concentrations (from less than 10 to more than 200 nM; Fig. 2).
Estimated probability density function (PDF; kernel density estimate) for sea surface iodide observations. Plot shows all data (blue) combined, and open ocean (green) and coastal (red) data treated separately. Expanded inset shows values <400 nM only. Figure produced with Python Matplotlib79 and Seaborn80 packages.
As the uncertainty estimates provided are typically derived from replicate analyses of the same sample they only estimate the short (days) to medium term (approx. monthly) repeatability. A fuller consideration of the uncertainty should also include the longer term (months to years) reproducibility, and an estimate of any uncertainties arising from bias, and thus may result in a larger uncertainty value. These sources of uncertainty are as yet poorly documented for the determination of iodide in seawater. At least in the case of the most commonly used method (CSSWV), we believe the contribution of long term reproducibility and bias to be small relative to the short-term precision. This is because the key of sources of uncertainty (e.g. that associated with making standard additions and sample dilutions by pipette, or variation in mercury electrode drop size) operate over a short time scale. Within our own laboratory, we have been monitoring long term reproducibility of the CSSWV method using aliquots of a near shore seawater sample, and estimate it at ~12% RSD over a period of 11 months (analysis by three operators using two different instruments; individual aliquots stored at −20 °C and defrosted within a few days of analysis), compared to ~monthly repeatability of 7–12% and repeatability over a few days of 5 to 18%. Changes taking place during storage will also contribute to the overall uncertainty of reported observations; for samples stored frozen (−16 °C), average iodide recovery after one year was 95–96%, compared to an average standard deviation of 5–8%15. In the majority of data sets we include, samples were stored frozen for less than one year prior to analysis, others were either analysed immediately following collection or stored for a shorter period refrigerated. Therefore we assume that storage artefacts were minimal. This view is supported by the oceanographic consistency found between stored and freshly analysed samples.
Assessment of bias in iodide in seawater determinations is hindered by the lack of a suitable reference material – many similar reference materials e.g. for trace metals, are acidified, which is unsuitable for the preservation of iodine speciation. Inaccuracy in standard preparation will contribute to bias in the short-term (all samples analysed using same standard), but are likely to become a random error in the longer term (several standards used over time). In either case, this should be a small contribution, as the uncertainty associated with preparing a typical analytical standard (e.g. 10 μM standard) should be less than 1% in a competent lab with well-maintained and calibrated equipment (e.g. balance, pipette). Other contributions to bias, such as matrix effects, are minimised by the use of standard additions rather than external calibration in the CSSWV protocol. In the absence of an iodide reference material, Campos15 tested the accuracy of the CSSWV method using solutions of known iodate concentration and a reduction step, and found it to be 99 ± 5.7% for 34 analyses. Given the current interest in marine iodide concentrations2,10,11, we believe that an inter-laboratory calibration exercise leading to development of a saline iodide reference material with a consensus value would be very timely. Such an exercise could follow the model of the recent GEOTRACES inter-calibration scheme (http://www.geotraces.org/Intercalibration).
Geographical categorisation
Data points are categorised as either ‘coastal’ or ‘non-coastal’. Following the approach used in Chance et al.12, this is determined by the designation of their static Longhurst biogeochemical province17. In most cases, the Longhurst province was assigned automatically, according to the nearest whole number degree of latitude and longitude. For a small number of samples collected very close to the coast, province (and hence coastal/non-coastal) was assigned manually - these samples are flagged (see Table 1). As in Chance et al.12, a small number of samples collected near Bermuda were also categorised as ‘coastal’ despite being located in an open ocean province (North Atlantic Subtropical Gyre Province (West)), as they were collected from an inshore area18. These samples are identified as such in the ‘Locator Method’ column.
Ancillary data
Note that original ancillary data such as temperature and salinity is not included, as this was not reliably available for all data sets. Instead we recommend the use of climatological data (e.g. the World Ocean Database and World Ocean Atlas Series) selected according to user needs.
Data Records
The compiled dataset is hosted by BODC (https://doi.org/10.5285/7e77d6b9-83fb-41e0-e053-6c86abc069d0)13, and is available as a single.csv file (plus a separate metadata file). It includes the fields listed in Table 1. It is anticipated that updated versions will be made available periodically, as new sea surface iodide observations become available. The current iteration is termed Version 1.0, future iterations will be named sequentially (i.e. version 2.0 etc). The lead authors would be very pleased to be contacted regarding new or omitted iodide observations for inclusion in future iterations of the dataset.
Technical Validation
Of the records included in our database, the majority (47/57) are described in peer-reviewed literature, and a further two are from PhD theses, and so their quality has already been subject to scientific scrutiny. Unpublished data sets made use of well-established analytical techniques, including the use of calibration standards and replicate analyses. In addition, the majority of data points were described in our earlier peer reviewed manuscript12, and were shown to have to a cohesive global distribution. The distribution of observations in the extended dataset continues to conform to this distribution (not shown), with concentrations remaining in the expected range (Fig. 2).
A very small number of unusually high concentration points (19 with iodide levels higher than 400 nM) are present in the data set. These are not representative of the overall iodide distribution, all being above the 98th percentile and also defined as outliers under the Tukey definition19. Where present, these extreme outlier values have been subject to rigorous scrutiny and are believed to be real.
We have not evaluated the data set to look for systematic differences between measurement techniques, as method used and location (and hence iodide concentration) are not independent variables. In most cases, only a small number of geographically limited points are available for a given method (Table 2). As noted, more than half the observations have been made using the same CSSWV technique. The remainder have been analysed using a wide range of other approaches, including, for some of the earliest datasets, labour intensive ‘wet chemical’ procedures which have since been superseded. In particular, a large proportion of the Pacific measurements were made in between 1968 and 197020,21 using a revised version of the Sugawara precipitation method22. The scarcity of more modern data from the Pacific limits comparisons, but we note that the range of this early Pacific data (3–168 nM) falls within that of the global data set, with a well-defined latitudinal distribution consistent with that observed overall. Regional concentrations (e.g. high latitudes, north Pacific23) are in agreement with those measured subsequently using different methods. Furthermore, the original data sources report vertical iodide profiles consistent in shape and magnitude with more recent measurements. Data obtained using the original, unmodified Sugawara method24 (1955) is not included, as this method is known to have poor performance22.
As described earlier, iodide observations are subject to non-negligible analytical uncertainty; we have reviewed the uncertainty estimation for each data set, and present this alongside the observations. As noted above, precision has usually been taken to represent method uncertainty. A variety of different methods have been used to estimate this, and so uncertainty magnitudes may not be directly comparable across all datasets.
Usage Notes
For computational convenience, iodide concentrations and associated uncertainties are provided to one decimal place (units are nM for both). However, note that this does not usually reflect the precision of the data points correctly, as this is typically a few percent.
For the purposes of investigating large-scale trends and creating regional iodide parameterisations, it may be appropriate to exclude the very high outlier values noted in the preceding section. Similarly, a number of points are from relatively low salinity estuarine areas (e.g. the Skaggerak), and so may not be representative of true marine trends in iodine speciation.
Missing fields are shown as not a number (“NaN”) in the output data file.
Code availability
The Python code used to prepare the archived data, and to enable incorporation of any subsequent observational data files, has also been made permanently available (https://doi.org/10.5281/zenodo.3271678)25.
References
Sherwen, T. et al. Effects of halogens on European air-quality. Faraday Discuss. 200, 75–100 (2017).
Lu, W. et al. Late inception of a resiliently oxygenated upper ocean. Science, https://doi.org/10.1126/science.aar5372, 361(6398), 174–177, (2018).
Andersson, M., Karumbunathan, V. & Zimmermann, M. B. Global Iodine Status in 2011 and Trends over the Past Decade. J. Nutr 142, 744–750 (2012).
Kadowaki, M. et al. Impacts of anthropogenic source from the nuclear fuel reprocessing plants on global atmospheric iodine-129 cycle: A model analysis. Atmos. Environ. 184, 278–291 (2018).
Ganzeveld, L., Helmig, D., Fairall, C. W., Hare, J. & Pozzer, A. Atmosphere‐ocean ozone exchange: A global modeling study of biogeochemical, atmospheric, and waterside turbulence dependencies. Global Biogeochem. Cycles. 23, GB4021, https://doi.org/10.1029/2008GB003301 (2009).
Sherwen, T. et al. Iodine’s impact on tropospheric oxidants: a global model study in GEOS-Chem. Atmos. Chem. Phys. 16, 1161–1186 (2016).
Oh, I.-B., Byun, D. W., Kim, H.-C., Kim, S. & Cameron, B. Modeling the effect of iodide distribution on ozone deposition to seawater surface. Atmos. Environ. 42, 4453–4466 (2008).
Sarwar, G. et al. Technical note: Examining ozone deposition over seawater. Atmos. Environ. 141, 255–262 (2016).
Sherwen, T., Evans, M. J., Carpenter, L. J., Schmidt, J. A. & Mickley, L. J. Halogen chemistry reduces tropospheric O3 radiative forcing. Atmos. Chem. Phys. 17, 1557–1569 (2017).
Sherwen, T. et al. A machine learning based global sea-surface iodide distribution. Earth Syst. Sci. Data Discuss 2019, 1–40 (2019).
Cutter, G. A., Moffett, J. G., Nielsdottir, M. C. & Sanial, V. Multiple oxidation state trace elements in suboxic waters off Peru: In situ redox processes and advective/diffusive horizontal transport. Mar. Chem. 201, 77–89 (2018).
Chance, R., Baker, A. R., Carpenter, L. & Jickells, T. D. The distribution of iodide at the sea surface. Environ. Sci.-Proc. Imp 16, 1841–1859 (2014).
Chance, R. et al. Global sea-surface iodide observations, 1967–2018. British Oceanographic Data Centre, Natural Environment Research Council, UK, https://doi.org/10.5285/7e77d6b9-83fb-41e0-e053-6c86abc069d0 (2019).
McKinney, W. Data Structures for Statistical Computing in Python, In: Proceedings of the 9th Python in Science Conference, (eds. van der Walt, S. & Millman, J.) 51–56 (2010).
Campos, M. L. A. M. New approach to evaluating dissolved iodine speciation in natural waters using cathodic stripping voltammetry and a storage study for preserving iodine species. Mar. Chem. 57, 107–117 (1997).
Chance, R. et al. Seasonal and interannual variation of dissolved iodine speciation at a coastal Antarctic site. Mar. Chem. 118, 171–181 (2010).
Longhurst, A. R. Ecological geography of the sea. (Academic, 1998).
Jickells, T. D., Boyd, S. S. & Knap, A. H. Iodine cycling in the Sargasso Sea and the Bermuda Inshore waters. Mar. Chem. 24, 61–82 (1988).
Frigge, M., Hoaglin, D. C. & Iglewicz, B. Some implementations of the Boxplot. The American Statistician 43, 50–54 (1989).
Tsunogai, S. Iodine in the deep water of the ocean. Deep-Sea Res 18, 913–919 (1971).
Tsunogai, S. & Henmi, T. Iodine in the surface water of the Pacific Ocean. J. Oceanogr. Soc. Japan 27, 67–72 (1971).
Tsunogai, S. Determination of iodine in seawater by an improved Sugawara method. Anal. Chim. Acta 55, 444–447 (1971).
Nakayama, E., Kimoto, T., Isshiki, K., Sohrin, Y. & Okazaki, S. Determination and distribution of iodide-iodine and total-iodine in the north Pacific ocean by using a new automated electrochemical method. Mar. Chem. 27, 105–116 (1989).
Sugawara, K., Koyama, T. & Terada, K. A new method of spectrophotometric determination of iodine in natural waters. Bull. Chem. Soc. Jpn. 28, 494–497 (1955).
Sherwen, T. tsherwen/sparse2spatial: sparse2spatial v0.1.0, Zenodo, https://doi.org/10.5281/zenodo.3349646 (2019).
Campos, M. L. A. M., Farrenkopf, A. M., Jickells, T. D. & Luther, G. W. A comparison of dissolved iodine cycling at the Bermuda Atlantic Time-Series station and Hawaii Ocean Time-Series Station. Deep-Sea Res. Pt. II 43, 455–466 (1996).
Wong, G. T. F. & Brewer, P. G. Determination and distribution of iodate in South Atlantic waters. J. Mar. Res. 32, 25–36 (1974).
Truesdale, V. W. Automatic determination of Iodate-iodine and Total-iodine in seawater. Mar. Chem. 6, 253–273 (1978).
Truesdale, V. W. & Smith, P. J. Automatic determination of iodide or iodate in solution by catalytic spectrophotometry, with particular reference to river water. Analyst 100, 111–123 (1975).
Elderfield, H. & Truesdale, V. W. On the biophilic nature of iodine in seawater. Earth Planet. Sci. Lett. 50, 105–114 (1980).
Truesdale, V. W., Bale, A. J. & Woodward, E. M. S. The meridional distribution of dissolved iodine in near-surface waters of the Atlantic Ocean. Prog.Oceanogr. 45, 387–400 (2000).
Herring, J. R. & Liss, P. S. New method for determination of iodine species in seawater. Deep-Sea Res 21, 777–783 (1974).
Takayanagi, K. & Wong, G. T. F. The oxidation of iodide to iodate for the polarographic determination of total iodine in natural waters. Talanta 33, 451–454 (1986).
Wong, G. T. F. & Zhang, L. Changes in iodine speciation across coastal hydrographic fronts in south-eastern United States continental shelf waters. Cont. Shelf Res. 12, 717–733 (1992).
Liss, P. S., Herring, J. R. & Goldberg, E. D. The Iodide/Iodate system in seawater as a possible measure of redox potential. Nat. Phys. Sci 242, 108–109 (1973).
Luther, G. W., Swartz, C. B. & Ullman, W. J. Direct determination of iodide in seawater by cathodic stripping square-wave voltammetry. Anal. Chem. 60, 1721–1724 (1988).
Farrenkopf, A. M. & Luther, G. W. Iodine chemistry reflects productivity and denitrification in the Arabian Sea: evidence for flux of dissolved species from sediments of western India into the OMZ. Deep-Sea Res. Pt II 49, 2303–2318 (2002).
Ullman, W. J., Luther, G. W., Delange, G. J. & Woittiez, J. R. W. Iodine chemistry in deep anoxic basins and overlying waters of the Mediterranean Sea. Mar. Chem. 31, 153–170 (1990).
Wong, G. T. F. & Zhang, L. S. Chemical removal of oxygen with sulfite for the polarographic or voltammetric determination of iodate or iodid in seawater. Mar. Chem. 38, 109–116 (1992).
Rue, E. L., Smith, G. J., Cutter, G. A. & Bruland, K. W. The response of trace element redox couples to suboxic conditions in the water column. Deep-Sea Res. Pt I 44, 113–134 (1997).
Nakayama, E., Kimoto, T. & Okazaki, S. Automatic determination of iodine species in determination of iodine species in natural waters by a new flow-through electrode system. Anal. Chem. 57, 1157–1160 (1985).
Wong, G. T. F. & Brewer, P. G. Determination of iodide in seawater by instrumental neutron activation analysis. Anal. Chim. Acta 81, 81–90 (1976).
Hou, X. et al. Speciation of I-129 and I-127 in seawater and implications for sources and transport pathways in the North Sea. Environ. Sci. Technol. 41, 5993–5999 (2007).
Hou, X. L., Dahlgaard, H. & Nielsen, S. P. Chemical speciation analysis of I-129 in seawater and a preliminary investigation to use it as a tracer for geochemical cycle study of stable iodine. Mar. Chem. 74, 145–155 (2001).
He, P., Aldahan, A., Hou, X. L. & Possnert, G. Tracing variability in the iodine isotopes and species along surface water transect from the North Sea to the Canary Islands. J. Radioanal. Nucl. Ch 307, 1407–1412 (2016).
Schwehr, K. A. & Santschi, P. H. Sensitive determination of iodine species, including organo-iodine, for freshwater and seawater samples using high performance liquid chromatography and spectrophotometric detection. Anal. Chim. Acta 482, 59–71 (2003).
McTaggart, A. R., Butler, E. C. V., Haddad, P. R. & Middleton, J. H. Iodide and iodate concentrations in eastern Australian subtropical waters, with iodide by ion chromatography. Mar. Chem. 47, 159–172 (1994).
Huang, Z., Ito, K., Timerbaev, A. R. & Hirokawa, T. Speciation studies by capillary electrophoresis - simultaneous determination of iodide and iodate in seawater. Anal. Bioanal. Chem. 378, 1836–1841 (2004).
Huang, Z. et al. Sensitive monitoring of iodine species in sea water using capillary electrophoresis: vertical profiles of dissolved iodine in the Pacific Ocean. J. Environ. Monit. 7, 804–808 (2005).
Woittiez, J. R. W., Vandersloot, H. A., Wals, G. D., Nieuwendijk, B. J. T. & Zonderhuis, J. The determination of iodide, iodate, total inorganic iodine and charcoal adsorbable iodine in seawater. Mar. Chem. 34, 247–259 (1991).
Chance, R. The influence of marine algae on the reduction of iodate to iodide in the sea Ph.D. thesis, University of East Anglia (2007).
Truesdale, V. W., Danielssen, D. S. & Waite, T. J. Summer and winter distributions of dissolved iodine in the Skagerrak. Estuar. Coast. Shelf Sci. 57, 701–713 (2003).
Truesdale, V. W., Nausch, G. & Baker, A. The distribution of iodine in the Baltic Sea during summer. Mar. Chem. 74, 87–98 (2001).
Truesdale, V. W., Kennedy, H., Agusti, S. & Waite, T. J. On the relative constancy of iodate and total-iodine concentrations accompanying phytoplankton blooms initiated in mesocosm experiments in Antarctica. Limnol. Oceanogr. 48, 1569–1574 (2003).
Waite, T. J., Truesdale, V. W. & Olafsson, J. The distribution of dissolved inorganic iodine in the seas around Iceland. Mar. Chem. 101, 54–67 (2006).
Wong, G. T. F. & Brewer, P. G. Marine chemistry of iodine in anoxic basins. Geochim. Cosmochim. Acta 41, 151–159 (1977).
Truesdale, V. W. & Bailey, G. W. Iodine distribution in the Southern Benguela system during an upwelling episode. Cont. Shelf Res. 22, 39–49 (2002).
Tian, R. C. et al. Iodine speciation: A potential indicator to evaluate new production versus regenerated production. Deep-Sea Res. Pt I 43, 723–738 (1996).
Tian, R. C. & Nicolas, E. Iodine speciation in the north-westen Mediterranean Sea - Method and vertical profile. Mar. Chem. 48, 151–156 (1995).
Tian, R. C. & Nicolas, E. Iodine speciation at time series station DYFAMED from date 1994-06-04. PANGAEA, https://doi.org/10.1594/PANGAEA.174586 (1995).
Truesdale, V. W. Iodine in inshore and off-shore marine waters. Mar. Chem. 6, 1–13 (1978).
Truesdale, V. W. & Upstill-Goddard, R. Dissolved iodate and total iodine along the British east coast. Estuar. Coast. Shelf Sci. 56, 261–270 (2003).
Wong, G. T. F. & Zhang, L. S. Geochemical dynamics of iodine in marginal seas: the southern East China Sea. Deep-Sea Res. Pt II 50, 1147–1162 (2003).
Campos, M. L. A. M., Sanders, R. & Jickells, T. The dissolved iodate and iodide distribution in the South Atlantic from the Weddell Sea to Brazil. Mar. Chem. 65, 167–175 (1999).
Luther, G.W. & Lewis, B. Iodine speciation. United States JGOFS Data Server. Woods Hole Oceanographic Institution, USA: U.S. JGOFS Data Management Office. Accessed:2018, http://usjgofs.whoi.edu/jg/dir/jgofs/arabian/ttn-045/ (1997).
Wong, G. T. F., Takayanagi, K. & Todd, J. F. Dissolved iodine in waters overlying and in the Orca Basin, Gulf of Mexico. Mar. Chem. 17, 177–183 (1985).
Wong, G. T. F. & Cheng, X. H. Dissolved organic iodine in marine waters: Determination, occurrence and analytical implications. Mar. Chem. 59, 271–281 (1998).
Bluhm, K., Croot, P. L., Huhn, O., Rohardt, G. & Lochte, K. Distribution of iodide and iodate in the Atlantic sector of the southern ocean during austral summer. Deep-Sea Res. Pt II 58, 2733–2748 (2011).
Wong, G. T. F. & Cheng, X.-H. Dissolved inorganic and organic iodine in the Chesapeake Bay and adjacent Atlantic waters: Speciation changes through an estuarine system. Mar. Chem. 111, 221–232 (2008).
Truesdale, V. W. & Jones, K. Steady-state mixing of iodine in shelf seas off the British Isles. Cont. Shelf Res. 20, 1889–1905 (2000).
Ducklow, H. W. et al. Spring-summer net community production, new production, particle export and related water column biogeochemical processes in the marginal sea ice zone of the Western Antarctic Peninsula 2012–2014. Philos. Tr. Roy. Soc. A 376, 15 (2018).
Hou, X. et al. Iodine-129 in Seawater Offshore Fukushima: Distribution, Inorganic Speciation, Sources, and Budget. Environ. Sci. Technol. 47, 3091–3098 (2013).
Luther, G. W. & Campbell, T. Iodine speciation in the water column of the Black Sea. Deep-Sea Res 38, S875–S882 (1991).
Wong, G. T. F. Dissolved and Particulate Iodine in the Oceans Ph.D. Thesis thesis, Massachusetts Institute of Technology and Woods Hole Oceanographic Institution (1976).
Wong, G. T. F. Distribution of iodine in upper layers of the equatorial. Atlantic. Deep-Sea Res. 24, 115–125 (1977).
He, P., Hou, X. L., Aldahan, A., Possnert, G. & Yi, P. Iodine isotopes species fingerprinting environmental conditions in surface water along the northeastern Atlantic Ocean. Sci. Rep 3, 8 (2013).
He, P., Aldahan, A., Possnert, G. & Hou, X. L. Temporal Variation of Iodine Isotopes in the North Sea. Environ. Sci. Technol. 48, 1419–1425 (2014).
Zhou, P. et al. Intrusion pattern of the Kuroshio Subsurface Water onto the East China Sea continental shelf traced by dissolved inorganic iodine species during the spring and autumn of 2014. Mar. Chem. 196, 24–34 (2017).
Hunter, J. D. Matplotlib: A 2D graphics environment. Computing In Science & Engineering 9, 90–95 (2007).
Waskom, M. et al. mwaskom/seaborn: seaborn v0.8.1, Zenodo, https://doi.org/10.5281/zenodo.883859 (2017).
Acknowledgements
We would like to thank the research teams, support staff and ships’ crews on all of our field campaigns, for invaluable assistance with the collection, transport and analysis of iodide samples. For funding details of published paper sets, please refer to the original publications. For unpublished and directly provided data sets, we particularly thank the following funding bodies who have made our research possible: Natural Environment Research Council (NERC) in the UK (grants NE/N00109983/1, NE/D006538/1, NE/D006511/1, NE/C001737/1, and studentship NER/S/A/2003/11224); The British Antarctic Survey, and Antarctic Funding Initiative (project NER/G/S/2003/00024); The Ministry of Earth Sciences, India; The Strategic Priority Research Program of the Chinese Academy of Sciences (grant XDA11020601); US National Science Foundation Office of Polar Programs (award PLR-1440435); German Research Foundation (DFG), via Collaborative Research Centre 754 (SFB 754); The Federal Ministry of Education and Research (BMBF), Germany. European Research Council (Advanced Grant agreement No. 669947); The World Bank Millennium Science Initiative (grant IC 120019); National Natural Science Foundation of China (no. 41603122). We acknowledge BODC, PANGAEA, the US JGOFS Data System for the provision of archived data sets, and BODC for hosting the compiled dataset. Assembly and dissemination of this data set has taken place as part of our project ‘Iodide in the ocean: distribution and impact on iodine flux and ozone loss.’ (NERC NE/N009983/1). Earlier work on the compilation also took place as part of Dr Chance’s PhD thesis (studentship NER/S/A/2003/11224), and as unfunded work during the period 2007–2016. Dr Sherwen’s contribution was also partially supported by NE/L01291X/1.
Author information
Authors and Affiliations
Contributions
All data originators who have provided data directly are authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Online-only Table
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
The Creative Commons Public Domain Dedication waiver http://creativecommons.org/publicdomain/zero/1.0/ applies to the metadata files associated with this article.
About this article
Cite this article
Chance, R.J., Tinel, L., Sherwen, T. et al. Global sea-surface iodide observations, 1967–2018. Sci Data 6, 286 (2019). https://doi.org/10.1038/s41597-019-0288-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-019-0288-y