Abstract
The adipokine Leptin activates its receptor LEP-R in the hypothalamus to regulate body weight and exerts additional pleiotropic functions in immunity, fertility and cancer. However, the structure and mechanism of Leptin-mediated LEP-R assemblies has remained unclear. Intriguingly, the signaling-competent isoform of LEP-R is only lowly abundant amid several inactive short LEP-R isoforms contributing to a mechanistic conundrum. Here we show by X-ray crystallography and cryo-EM that, in contrast to long-standing paradigms, Leptin induces type I cytokine receptor assemblies featuring 3:3 stoichiometry and demonstrate such Leptin-induced trimerization of LEP-R on living cells via single-molecule microscopy. In mediating these assemblies, Leptin undergoes drastic restructuring that activates its site III for binding to the Ig domain of an adjacent LEP-R. These interactions are abolished by mutations linked to obesity. Collectively, our study provides the structural and mechanistic framework for how evolutionarily conserved Leptin:LEP-R assemblies with 3:3 stoichiometry can engage distinct LEP-R isoforms to achieve signaling.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Crystallographic coordinates and structure factors have been deposited in the PDB, and cryo-EM maps and models have been deposited in the Electron Microscopy Data Bank (EMDB) and PDB data banks with accession codes listed in Tables 1 and 2. Briefly, the crystal structure of mLeptin:mLEP-RCRH2 is deposited under the accession code PDB 7z3p, hLeptin:hLEP-RCRH2 under PDB 7z3q, mLeptin:mLEP-RIgCRH2 under PDB 7z3r, and mLEP-RFNIII:VHH-4.80 under PDB 8av2. The cryo-EM map of the open 1:2 mLeptin:mLEP-RECD complex has been deposited under the accession codes EMD-15677 and PDB 8avb. The cryo-EM map of the closed 3:3 mLeptin:mLEP-RECD-tGCN4 complex has been deposited under the accession codes EMD-15678 and PDB 8avc, after local refinement under accession codes EMD-15679 and PDB 8avd, and after local refinement and symmetry expansion under accession codes EMD-15899 and PDB 8b7q. The cryo-EM map of the open 2:2 hLeptin:hLEP-RECD-tGCN4 complex has been deposited under the accession codes EMD-15680 and PDB 8ave, the closed 3:3 hLeptin:hLEP-RECD-tGCN4 complex under EMD-15681 and PDB 8avf, and the open 3:3 hLeptin:hLEP-RECD-tGCN4 complex under EMD-15683 and PDB 8avo. Search models for molecular replacement were prepared using PDB 1ax8 (hLeptin), PDB 3v6o (hLEP-RCRH2 fragment), PDB 1OHQ (VHH model) and AF-P48356-F1-model_v1 (for mLEP-RFnIII). Coordinates for the IL6 (PDB 1p9m) and GCSF (PDB 2d9q) receptor complexes were used for comparative analysis. Structural predictions via Alphafold were used to generate starting models for real-space cryo-EM refinement: mLEP-R (AF-P48356-F1-model_v1), hLeptin (AF-P41159-F1-model_v2) and hLEP-R (AF-P48357-F1-model_v2). TIRF microscopy data from localization and co-tracking analysis have been uploaded into the publicly available repository Zenodo together with representative raw imaging data and are available at https://doi.org/10.5281/zenodo.7419603. AUC data are publicly available at https://doi.org/10.5281/zenodo.7566572. All remaining data generated or analyzed during this study are included in this published article (and its supplementary information files). Source data are provided with this paper.
References
Zhang, F. et al. Crystal structure of the obese protein leptin-E100. Nature 387, 206–209 (1997).
Wauman, J., Zabeau, L. & Tavernier, J. The leptin receptor complex: heavier than expected? Front. Endocrinol. 8, 30 (2017).
de Candia, P. et al. The pleiotropic roles of leptin in metabolism, immunity, and cancer. J. Exp. Med. 218, e20191593 (2021).
Friedman, J. M. Leptin and the endocrine control of energy balance. Nat. Metab. 1, 754–764 (2019).
Salum, K. C. R. et al. When leptin is not there: a review of what nonsyndromic monogenic obesity cases tell us and the benefits of exogenous leptin. Front. Endocrinol. 12, 722441 (2021).
Farooqi, I. S. et al. Clinical and molecular genetic spectrum of congenital deficiency of the leptin receptor. N. Engl. J. Med. 356, 237–247 (2007).
Gorska, E. et al. Leptin receptors. Eur. J. Med. Res. 15, 50–54 (2010).
Fei, H. et al. Anatomic localization of alternatively spliced leptin receptors (Ob-R) in mouse brain and other tissues. Proc. Natl Acad. Sci. USA 94, 7001–7005 (1997).
Haniu, M. et al. Human leptin receptor. Determination of disulfide structure and N-glycosylation sites of the extracellular domain. J. Biol. Chem. 273, 28691–28699 (1998).
Zabeau, L. et al. Functional analysis of leptin receptor activation using a Janus kinase/signal transducer and activator of transcription complementation assay. Mol. Endocrinol. 18, 150–161 (2004).
Moharana, K. et al. Structural and mechanistic paradigm of leptin receptor activation revealed by complexes with wild-type and antagonist leptins. Structure 22, 866–877 (2014).
Peelman, F. et al. Mapping of binding site III in the leptin receptor and modeling of a hexameric leptin.leptin receptor complex. J. Biol. Chem. 281, 15496–15504 (2006).
Zabeau, L. et al. Leptin receptor activation depends on critical cysteine residues in its fibronectin type III subdomains. J. Biol. Chem. 280, 22632–22640 (2005).
Tu, H., Hsuchou, H., Kastin, A. J., Wu, X. & Pan, W. Unique leptin trafficking by a tailless receptor. FASEB J. 24, 2281–2291 (2010).
Ge, H., Huang, L., Pourbahrami, T. & Li, C. Generation of soluble leptin receptor by ectodomain shedding of membrane-spanning receptors in vitro and in vivo. J. Biol. Chem. 277, 45898–45903 (2002).
Ferrao, R. D., Wallweber, H. J. & Lupardus, P. J. Receptor-mediated dimerization of JAK2 FERM domains is required for JAK2 activation. eLife 7, e38089 (2018).
Glassman, C. R. et al. Structure of a Janus kinase cytokine receptor complex reveals the basis for dimeric activation. Science 376, 163–169 (2022).
Mancour, L. V. et al. Ligand-induced architecture of the leptin receptor signaling complex. Mol. Cell 48, 655–661 (2012).
White, D. W., Kuropatwinski, K. K., Devos, R., Baumann, H. & Tartaglia, L. A. Leptin receptor (OB-R) signaling. Cytoplasmic domain mutational analysis and evidence for receptor homo-oligomerization. J. Biol. Chem. 272, 4065–4071 (1997).
Bacart, J. et al. Evidence for leptin receptor isoforms heteromerization at the cell surface. FEBS Lett. 584, 2213–2217 (2010).
Iserentant, H. et al. Mapping of the interface between leptin and the leptin receptor CRH2 domain. J. Cell Sci. 118, 2519–2527 (2005).
Peelman, F. et al. Mapping of the leptin binding sites and design of a leptin antagonist. J. Biol. Chem. 279, 41038–41046 (2004).
Danielsson, J. et al. The pierced lasso topology leptin has a bolt on dynamic domain composed by the disordered loops I and III. J. Mol. Biol. 432, 3050–3063 (2020).
Niv-Spector, L. et al. Mapping leptin-interacting sites in recombinant leptin-binding domain (LBD) subcloned from chicken leptin receptor. Biochem. J. 390, 475–484 (2005).
Verploegen, S. A., Plaetinck, G., Devos, R., Van der Heyden, J. & Guisez, Y. A human leptin mutant induces weight gain in normal mice. FEBS Lett. 405, 237–240 (1997).
Carpenter, B. et al. Structure of the human obesity receptor leptin-binding domain reveals the mechanism of leptin antagonism by a monoclonal antibody. Structure 20, 487–497 (2012).
Spangler, J. B., Moraga, I., Mendoza, J. L. & Garcia, K. C. Insights into cytokine–receptor interactions from cytokine engineering. Annu. Rev. Immunol. 33, 139–167 (2015).
Varadi, M. et al. AlphaFold Protein Structure Database: massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res. 50, D439–D444 (2022).
Niv-Spector, L. et al. Identification of the hydrophobic strand in the A–B loop of leptin as major binding site III: implications for large-scale preparation of potent recombinant human and ovine leptin antagonists. Biochem. J. 391, 221–230 (2005).
Gromada, J. S. P., Altarejos, J., & Murphy, A. J. A leptin receptor agonist antibody for use in treating a metabolic dysfunction or hypoleptinemia. Patent No. WO2019195796A1 (2019).
Harbury, P. B., Kim, P. S. & Alber, T. Crystal structure of an isoleucine-zipper trimer. Nature 371, 80–83 (1994).
Walls, A. C. et al. Cryo-electron microscopy structure of a coronavirus spike glycoprotein trimer. Nature 531, 114–117 (2016).
Xu, Y. et al. Crystal structure of the entire ectodomain of gp130: insights into the molecular assembly of the tall cytokine receptor complexes. J. Biol. Chem. 285, 21214–21218 (2010).
Mirdita, M. et al. ColabFold: making protein folding accessible to all. Nat. Methods 19, 679–682 (2022).
Evans, R. et al. Protein complex prediction with AlphaFold-Multimer. Preprint at bioRxiv https://doi.org/10.1101/2021.10.04.463034 (2022).
Liongue, C., Sertori, R. & Ward, A. C. Evolution of cytokine receptor signaling. J. Immunol. 197, 11–18 (2016).
Sotolongo Bellon, J. et al. Four-color single-molecule imaging with engineered tags resolves the molecular architecture of signaling complexes in the plasma membrane. Cell Rep. Methods 2, 100165 (2022).
Morris, R., Kershaw, N. J. & Babon, J. J. The molecular details of cytokine signaling via the JAK/STAT pathway. Protein Sci. 27, 1984–2009 (2018).
Richter, D. et al. Ligand-induced type II interleukin-4 receptor dimers are sustained by rapid re-association within plasma membrane microcompartments. Nat. Commun. 8, 15976 (2017).
Wilmes, S. et al. Mechanism of homodimeric cytokine receptor activation and dysregulation by oncogenic mutations. Science 367, 643–652 (2020).
Martinez-Fabregas, J. et al. Kinetics of cytokine receptor trafficking determine signaling and functional selectivity. eLife 8, e49314 (2019).
Wilmes, S. et al. Receptor dimerization dynamics as a regulatory valve for plasticity of type I interferon signaling. J. Cell Biol. 209, 579–593 (2015).
Gorby, C. et al. Engineered IL-10 variants elicit potent immunomodulatory effects at low ligand doses. Sci. Signal. 13, eabc0653 (2020).
Mazen, I., El-Gammal, M., Abdel-Hamid, M. & Amr, K. A novel homozygous missense mutation of the leptin gene (N103K) in an obese Egyptian patient. Mol. Genet. Metab. 97, 305–308 (2009).
Wabitsch, M. et al. Biologically inactive leptin and early-onset extreme obesity. N. Engl. J. Med. 372, 48–54 (2015).
Wabitsch, M. et al. Measurement of immunofunctional leptin to detect and monitor patients with functional leptin deficiency. Eur. J. Endocrinol. 176, 315–322 (2017).
Zastrow, O. et al. The soluble leptin receptor is crucial for leptin action: evidence from clinical and experimental data. Int. J. Obes. Relat. Metab. Disord. 27, 1472–1478 (2003).
Djogo, T. et al. Adult NG2-glia are required for median eminence-mediated leptin sensing and body weight control. Cell Metab. 23, 797–810 (2016).
Butiaeva, L. I. et al. Leptin receptor-expressing pericytes mediate access of hypothalamic feeding centers to circulating leptin. Cell Metab. 33, 1433–1448.e5 (2021).
Langendonk, J. G. et al. Circadian rhythm of plasma leptin levels in upper and lower body obese women: influence of body fat distribution and weight loss. J. Clin. Endocrinol. Metab. 83, 1706–1712 (1998).
Li, Z., Ceccarini, G., Eisenstein, M., Tan, K. & Friedman, J. M. Phenotypic effects of an induced mutation of the ObRa isoform of the leptin receptor. Mol. Metab. 2, 364–375 (2013).
Bjorbaek, C. et al. Expression of leptin receptor isoforms in rat brain microvessels. Endocrinology 139, 3485–3491 (1998).
Bjorbaek, C., Uotani, S., da Silva, B. & Flier, J. S. Divergent signaling capacities of the long and short isoforms of the leptin receptor. J. Biol. Chem. 272, 32686–32695 (1997).
Bahrenberg, G. et al. Identification of the critical sequence elements in the cytoplasmic domain of leptin receptor isoforms required for Janus kinase/signal transducer and activator of transcription activation by receptor heterodimers. Mol. Endocrinol. 16, 859–872 (2002).
Vernooy, J. H. et al. Leptin modulates innate and adaptive immune cell recruitment after cigarette smoke exposure in mice. J. Immunol. 184, 7169–7177 (2010).
Aricescu, A. R., Lu, W. & Jones, E. Y. A time- and cost-efficient system for high-level protein production in mammalian cells. Acta Crystallogr. D 62, 1243–1250 (2006).
Zabeau, L. et al. A novel leptin receptor antagonist uncouples leptin’s metabolic and immune functions. Cell. Mol. Life Sci. 76, 1201–1214 (2019).
Reeves, P. J., Callewaert, N., Contreras, R. & Khorana, H. G. Structure and function in rhodopsin: high-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc. Natl Acad. Sci. USA 99, 13419–13424 (2002).
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010).
Evans, P. R. & Murshudov, G. N. How good are my data and what is the resolution? Acta Crystallogr. D 69, 1204–1214 (2013).
Liebschner, D. et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr. D 75, 861–877 (2019).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Bricogne, G. B. E. et al. BUSTER version 2.10.4. (Global Phasing Ltd., 2017); https://www.globalphasing.com/buster/
Eisenberg, D., Schwarz, E., Komaromy, M. & Wall, R. Analysis of membrane and surface protein sequences with the hydrophobic moment plot. J. Mol. Biol. 179, 125–142 (1984).
Schuck, P. Size-distribution analysis of macromolecules by sedimentation velocity ultracentrifugation and lamm equation modeling. Biophys. J. 78, 1606–1619 (2000).
Schuck, P. On the analysis of protein self-association by sedimentation velocity analytical ultracentrifugation. Anal. Biochem. 320, 104–124 (2003).
Brautigam, C. A. Calculations and publication-quality illustrations for analytical ultracentrifugation data. Methods Enzymol. 562, 109–133 (2015).
DuBridge, R. B. et al. Analysis of mutation in human cells by using an Epstein–Barr virus shuttle system. Mol. Cell. Biol. 7, 379–387 (1987).
Eyckerman, S. et al. Analysis of Tyr to Phe and fa/fa leptin receptor mutations in the PC12 cell line. Eur. Cytokine Netw. 10, 549–556 (1999).
Götzke, H. et al. The ALFA-tag is a highly versatile tool for nanobody-based bioscience applications. Nat. Commun. 10, 4403 (2019).
You, C., Richter, C. P., Löchte, S., Wilmes, S. & Piehler, J. Dynamic submicroscopic signaling zones revealed by pair correlation tracking and localization microscopy. Anal. Chem. 86, 8593–8602 (2014).
Serge, A., Bertaux, N., Rigneault, H. & Marguet, D. Dynamic multiple-target tracing to probe spatiotemporal cartography of cell membranes. Nat. Methods 5, 687–694 (2008).
Niewidok, B. et al. Single-molecule imaging reveals dynamic biphasic partition of RNA-binding proteins in stress granules. J. Cell Biol. 217, 1303–1318 (2018).
Jaqaman, K. et al. Robust single-particle tracking in live-cell time-lapse sequences. Nat. Methods 5, 695–702 (2008).
Blanchet, C. E. et al. Versatile sample environments and automation for biological solution X-ray scattering experiments at the P12 beamline (PETRA III, DESY). J. Appl. Crystallogr. 48, 431–443 (2015).
Manalastas-Cantos, K. et al. ATSAS 3.0: expanded functionality and new tools for small-angle scattering data analysis. J. Appl. Crystallogr. 54, 343–355 (2021).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Bepler, T. et al. Positive-unlabeled convolutional neural networks for particle picking in cryo-electron micrographs. Nat. Methods 16, 1153–1160 (2019).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Cianfrocco MA, W. M., Youn, C., Wagner, R. & Leschziner, A. E. COSMIC2: a science gateway for cryo-electron microscopy structure determination. In Proc. Practice and Experience in Advanced Research Computing 2017 on Sustainability, Success and Impact (eds. Hart, D.) 22 (ACM, 2017).
Sanchez-Garcia, R. et al. DeepEMhancer: a deep learning solution for cryo-EM volume post-processing. Commun. Biol. 4, 874 (2021).
Rosenthal, P. B. & Henderson, R. Optimal determination of particle orientation, absolute hand, and contrast loss in single-particle electron cryomicroscopy. J. Mol. Biol. 333, 721–745 (2003).
Chen, S. et al. High-resolution noise substitution to measure overfitting and validate resolution in 3D structure determination by single particle electron cryomicroscopy. Ultramicroscopy 135, 24–35 (2013).
Cardone, G., Heymann, J. B. & Steven, A. C. One number does not fit all: mapping local variations in resolution in cryo-EM reconstructions. J. Struct. Biol. 184, 226–236 (2013).
Pettersen, E. F. et al. UCSF ChimeraX: structure visualization for researchers, educators, and developers. Protein Sci. 30, 70–82 (2021).
The PyMOL Molecular Graphics System, Version 2.3.3 (Schrödinger, 2019).
Peelman, F., Zabeau, L., Moharana, K., Savvides, S. N. & Tavernier, J. 20 years of leptin: insights into signaling assemblies of the leptin receptor. J. Endocrinol. 223, T9–T23 (2014).
Boulanger, M. J., Chow, D. C., Brevnova, E. E. & Garcia, K. C. Hexameric structure and assembly of the interleukin-6/IL-6 alpha-receptor/gp130 complex. Science 300, 2101–2104 (2003).
Tamada, T. et al. Homodimeric cross-over structure of the human granulocyte colony-stimulating factor (GCSF) receptor signaling complex. Proc. Natl Acad. Sci. USA 103, 3135–3140 (2006).
Amemiya, C. T. et al. The African coelacanth genome provides insights into tetrapod evolution. Nature 496, 311–316 (2013).
Londraville, R. L., Prokop, J. W., Duff, R. J., Liu, Q. & Tuttle, M. On the molecular evolution of leptin, leptin receptor, and endospanin. Front. Endocrinol. 8, 58 (2017).
Acknowledgements
We thank the staff of beamlines P12, P13 and P14 (Petra III, Deutsches Elektronen-Synchrotron), and Proxima 2A (SOLEIL) for technical support and beamtime allocation. We are grateful to the staff of the VIB-VUB facility for Bio Electron Cryogenic Microscopy (BECM, Brussels, Belgium), the staff of the Laboratory of Cell Biology & Histology (CBH) and the Antwerp Centre for Advanced Microscopy (ACAM) (UAntwerpen, Antwerp, Belgium), and the electron Bio-Imaging Centre (eBIC) at the Diamond Light Source (Didcot, UK) for technical support and infrastructural access, and H. Kenneweg for production of labeled NBs. The pGL3-rPAPluc plasmid containing the luciferase gene was a kind gift of F. Peelman (VIB-UGent, Ghent, Belgium). This work was supported by grants from the Research Foundation-Flanders (grant G0G0619N to K.V.) and the VIB (to S.N.S.). This work benefited from access to the Integrated Structural Biology platform of the Strasbourg Instruct-ERIC center IGBMC-CBI. Financial support was provided by FRISBI (ANR-10-INBS-0005 to C.B.), Instruct-ERIC (PID 15107 to K.V.), iNext-Discovery (project number 17947 to K.V. and funded by the Horizon 2020 program of the European Commission) and the DFG (SFB 944, projects P8 and Z, to J.P.).
Author information
Authors and Affiliations
Contributions
A.T. prepared constructs, and performed protein expression and purification with contributions from A.D. and K.V. A.T., K.H.G.V. and K.V. determined and analyzed crystallographic structures with contributions from S.N.S. K.V. collected and analyzed cryo-EM data, with contributions from J.F., W.V.P. and S.N.S. A.T. performed biolayer interferometry, and SEC–MALLS experiments. SAXS data were analyzed by K.V. and A.T. I.M. performed cellular assays. M.H. and C.P. performed smTIRFM experiments and data analysis. J.F.B. carried out evolutionary structural analyses, and J.P. supervised the smTIRFM experiments. C.B. performed SV–AUC experiments and analyzed data. D.C. and J.T. contributed critical reagents. A.T., K.V. and S.N.S. wrote the paper with contributions and approval from all authors. K.V. and S.N.S. conceived and supervised the project and procured funding.
Corresponding authors
Ethics declarations
Competing interests
W.V.P. is the founder and chief executive officer of PUXANO. All other authors declare no competing interests.
Peer review
Peer review information
Nature Structural & Molecular Biology thanks Eunhee Choi, Ellinor Haglund and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Florian Ullrich and Katarzyna Ciazynska, in collaboration with the Nature Structural & Molecular Biology team. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Structural and biophysical characterization of the Leptin:LEP-R assembly.
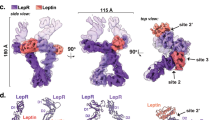
a-c, Characterization of the minimal Leptin:LEP-R recognition complex. a, Structural superposition of the glycan-trimmed hLeptin:hLEP-RCRH2 complex crystal structure (LEP-RCRH2 fragment 428-635 N516Q/C604S in blue; Leptin in yellow) with the mouse complex crystal structure (colored in gray). The structure of the individual components and the interaction interface thereof is highly similar between the two homologues, consistent with their high sequence identity (83.2% for Leptin, 86.9% for LEP-RCRH2, Supplementary Fig. 1). (r.m.s.d.: root mean square deviation of alignment, 161 Cα atoms) b, Molar mass determination of the glycan-trimmed mLeptin:mLEP-RCRH2 complex using Size Exclusion Chromatography coupled with Multi-Angle Laser Light Scattering (SEC-MALLS). The complex (50 μM) was resolved in a Superdex 200 increase column pre-equilibrated in HBS buffer (20 mM HEPES pH 7.4, 150 mM NaCl). The measured mass (39.3 kDa) approximates the theoretical of 40.3 kDa for 1:1 stoichiometry. (n = 1) c, Biolayer interferometry (BLI) sensograms of the mouse Leptin:LEP-R interaction. Left: mLeptin was titrated, as indicated on the side, against the in vitro biotinylated mLEP-RCRH2 fragment immobilized on Streptavidin sensors (n = 4). Right: mLEP-RECD was titrated against in vitro biotinylated mLeptin (n = 3). Comparison of the kinetic rates and derived affinities suggest that the high-affinity Leptin:LEP-R interaction is attributed to the Leptin:LEP-RCRH2 interface, in agreement with previous studies89. d–g, Characterization of the mLeptin:LEP-RIgCRH2 oligomeric assembly. d, The asymmetric unit of the glycan-trimmed mLeptin:mLEP-RIgCRH2 crystal structure resolved at 2.9 Å (coloured as in Fig. 1a) superimposed to the mLeptin:mLEP-RCRH2 structure (grey) (r.m.s.d. of alignment =0.4 Å, 257 Cα atoms). Interacting residues at the Ig-CRH2 interface are highlighted in the insert. A glycan chain (green sticks) was resolved at position Asn393 of the LEP-RIg domain. e, The oligomeric assembly of mLeptin:mLEP-RIgCRH2 (left) derived from the 3- and 2-fold crystallographic symmetry operations as detailed on the right (space group H 3 2). The assembly comprised of a 3:3 mLeptin:mLEP-RIgCRH2 stoichiometric complex through site II and site III contacts, and an artifactual head-to-head dimerization thereof, resulting in a 6:6 complex. The head-to-head dimer associates purely by mirroring Ig-Ig’ contacts at an exposed hydrophobic patch shown in the insert. The LEP-R N-terminal domain is expected to disrupt this interface (N-termini indicated). f, In solution molar mass determination of LEP-RIgCRH2 and complexes thereof with Leptin and the Leptin antagonist a1 (140ST141/AA)22,29 using SEC-MALLS (Superdex 200 increase column). Protein molar masses are plotted after protein conjugate analysis of the glycosylated complexes. Experimentally determined and theoretical masses are given on the right. h: human; m: mouse complexes. n = 1. g, SDS-PAGE (17%) of isolated 6:6 and 1:1 Leptin:LEP-RIgCRH2 complexes under different sample preparation conditions. Multimeric artifacts are observed only with the combination of reduction and boiling, raising awareness for sample preparation for determining stoichiometry from SDS-PAGE. Leptin migrates slower under reducing conditions. (β-merc: β-mercaptoethanol).
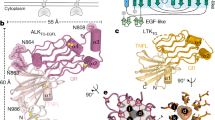
Extended Data Fig. 2 Evolutionary and co-evolutionary features of LEP-RIgCRH2 and its interaction interfaces with Leptin.
a, Structural superposition (top) and sequence alignment (bottom) of the LEP-RIgCRH2 domains with those of human gp130 (IL6Rβ)90 and GCSFR91 in their cytokine-bound state (pdb: 1p9m, 2d9q respectively). After alignment at the first subdomain of the CRH2 module (r.m.s.d. indicated; 75 and 60 Cα atoms respectively), the shift distance was determined between the highly conserved cysteine residues of the Ig motif, as indicated. Structure-based sequence alignment was performed using UCSF Chimera and shows a shorter linker between the Ig and CRH2 domains for LEP-R relatively to its structural homologues. b, Structural superposition of the site III interface in evolutionary relatives. For visualization purposes only the α-helix D is shown for mLeptin, hIL6 and hGCSF, featuring the aromatic blueprint of site III in sticks. IL6Rβ:IL6 and GCSFR:GCSF were aligned to the Ig domain of mLeptin:LEP-RIgCRH2 (r.m.s.d. indicated; 41 and 59 Cα atoms respectively). c-f, Structural superposition of the crystallographically-distilled mLeptin:mLEP-RIgCRH2 assembly with AlphaFold models, as indicated, accompanied with predicted Aligned Error plots (PAE) of each prediction. AlphaFold2-ptm was used for the prediction of mLEP-RIgCRH2 (panels c-d) and AlphaFold-Multimer (version 2.2) for the prediction of the mLeptin:mLEP-RIgCRH2 interaction interfaces (panels e-f). g, Evolutionary conservation of the Leptin:LEP-RIgCRH2 hexameric assembly across the animal phylogenetic spectrum, tested by AlphaFold-Multimer (version 2.2, with associated ipTM scores)34,35 with orthologous 3:3 pairings of Leptin and LEP-R sequences harvested from UniProt. Drawing the branching of jawed vertebrates from the coelacanth phylogenetic tree92, birds are represented by the chicken G. gallus Leptin:LEP-R complex (respectively UniProt codes O42164 and Q9I8V6), reptiles by the alligator A. sinensis (A0A1U8DFZ0 and A0A3Q0H8E2), mammals by human H. sapiens (P41159 and P48357), amphibians by X. tropicalis (A0A803KEF7 and F6RVW6), lobe-finned fishes by the living fossil Coelacanth L. chalumnae (H3AP27 and H3AG22), cartilaginous fishes by the Australian ghostshark C. milii (A0A4W3H871 and V9KNM1), and ray-finned fishes by the eel A. anguilla (A0A0C7AV37 and A0A0C7AV33) and tilapia O. niloticus (I3KCE8 and A0A067Z8Z1). Sequence evolutionary relationships were based on LEP_R orthologues and were adapted from Londraville et al., 201793. Complexes were visualized and superposed to the X-ray structure of mLeptin:mLEP-RIgCRH2 by PyMOL 2.3.4 (www.pymol.org) with listed r.m.s.d fit to the 3:3 complex.
Extended Data Fig. 3 Biophysical and functional characterization of LEP-R complexes.
a-e, Biophysical analysis of mouse Leptin:LEP-RECD in solution. a, Sedimentation coefficient distributions c(s) for different concentrations of glycan-trimmed mLeptin:mLEP-RECD. b, SEC-MALLS analysis (Superdex 200 increase column) of glycan-trimmed wild type mLEP-RECD and mLEP-RECD-A407 mutant and their complexes with mLeptin. Similar results were obtained from at least 3 independent repetitions. c and e, SEC-MALLS analysis (Superdex 200 increase column) of glycan-trimmed (panel c) or glycosylated (panel e) mLEP-RECD-ΔFNIII and their complexes with mLeptin. Similar results were obtained from n = 2 at selected similar concentrations for panel c, n = 3 for panel e. d, SEC-MALLS analysis (Superose 6 increase column) of glycosylated mLEP-RECD and its complexes with mLeptin, as a function of complex concentration. Similar results were obtained from at least n = 2 independent experiments at a range of concentrations. f, j, k, Characterization of human LEP-R complexes. f, SEC-MALLS analysis (Superdex 200 increase column) of glycosylated hLEP-RECD and its complexes with hLeptin. Similar results were obtained from n = 2 independent experiments at selected concentrations. j, SEC-MALLS analysis (Superdex 200 increase column) of glycosylated hLEP-RECD and its complexes with the agonistic antibody mibavademab30. n = 1. k, Activation of hLEP-Rb by Leptin and variants, as well as by the agonistic antibody mibavademab, probed by a STAT3-responsive luciferase reporter in HEK293T cells. n = 3 independent experiments with 3 technical replicates each. Here, mean values and standard deviations from a representative independent experiment are shown. g, h, i, Interspecies cross-reactivity probed with SEC-MALLS (Superdex 200 increase column). n = 1. g, Complexes of hLEP-RECD with mouse and human Leptin. h, Complexes of glycan-trimmed mLEP-RECD with mouse and human Leptin. i, Complexes of glycan-trimmed mLEP-RECD with ‘murinized’ human Leptin. Specifically, residues close to the Site III interface that differ between mouse and human Leptin (Supplementary Fig. 1a) were mutated on hLeptin to the corresponding of the mouse homologue (that is CD-loop residues 118-129; G139L; I85V/M89L/G132D/G139L).
Extended Data Fig. 4 Cryo-EM analysis of mLep:mLEP-RECD complexes.
a. The COSMIC2 webserver was used to align projections of the trimeric mLeptin:mLEP-RIgCRH2 crystal structure, the 1:2 mLeptin:LEP-RIgCRH2FnIII cryo-EM model, and projections of a 1:1 mLeptin:mLEP-RECD model (based on the Alphafold2-model for mLEP-RECD and the mLeptin:mLEP-RCRH2 crystal structure) with high-resolution 2D-classes obtained for the mLeptin:mLEP-RECD complex sample. In the gallery 2D cryo-EM classes matching projections are indicated with a colored dot. b, Cryo-EM workflow towards the reconstruction of the 1:2 mLeptin:LEP-RECD complex. We note that 3D reconstruction of the remaining two observed states was hindered due to preferred orientation. NU, non-uniform refinement; GSFSC: gold-standard Fourier shell correlation.
Extended Data Fig. 5 Cryo-EM data analysis workflow for the mLeptin:mLEP-RtGCN4 complex.
a, SEC-MALLS analysis of the glycosylated mLeptin:mLEP-RECD-tGCN4 complex. The SEC-elution profile is plotted as the ultraviolet absorbance at 280 nm (left Y-axis) in function of elution volume. The total, protein and glycan molecular mass (right Y-axis) as determined by MALLS are reported as the average molecular mass (and s.d.) across the elution peak. Number of samples for mLeptin:mLEP-RECD-tGCN4 analyzed: n = 1. b, SAXS scattering curves of EndoH-treated samples for mLEP-RECD and mLeptin:mLEP-RECD complexes plotted as the scattered intensity in function of scattering vector s = 4πsinθ/λ. (i) mLEP-RECD (grey), (ii) non-stabilized mLeptin:mLEP-RECD complex (black), (iii) stabilized mLeptin:mLEP-RECD-tGCN4 complex (yellow), (iv) scaled and overlayed scattering profiles for mLeptin:mLEP-RECD and mLeptin:mLEP-RECD-tGCN4. Similar results were obtained from n = 2 independent experiments for mLeptin:mLEP-RECD. For the rest n = 1. c. Cryo-EM data processing workflow in cryoSPARC for the glycosylated mLeptin:mLEP-RECD-ECD-tGCN4 complex. GSFSC: gold-standard Fourier shell correlation. d. Fourier shell correlation plots, local resolution map and sharpened map following symmetry expansion of the particle data set and local refinement around one LEP-RCRH2:Leptin:LEP-RIg' subcomplex.
Extended Data Fig. 6 Comparative analysis between mouse and human Leptin:LEP-R complex structures as derived from Cryo-EM and X-ray crystallographic analysis.
a, Real-space refined atomic model for the trimeric mLeptin:mLEP-RIgCRH2 core region overlayed with the sharpened cryo-EM map in C1 symmetry for the mLeptin:mLEP-RECD-tGCN4 complex following local map refinement. The cryo-EM map is contoured at 0.313 V. In the cryo-EM map strong additional density is observed at all three LEP-RCRH2:Leptin interface regions (site II) that likely corresponds to a trapped Ni-ion coordinated by histidine residues in the LEP-RCRH2 module and the N-terminal His-tag present on mLeptin. b, Structural superposition of the crystallographically-distilled 3:3 mLeptin:mLEP-RIgCRH2 assembly (black) with the real-space refined mLeptin:mLEP-RIgCRH2 assembly via cryo-EM. c–d, Cryo-EM maps and fitted atomic models for the mouse and human Leptin:LEP-RECD-tGCN4 complexes. The sharpened cryo-EM maps are colored per zone with Leptin in yellow, the CRH1 module in grey, the Ig domain in magenta, the CRH2 module in blue and the FnIII module in green. The fitted atomic model models are shown as a cartoon overlayed with the cryo-EM maps as a transparent volume. The mouse and human cryo-EM maps are contoured at 0.178 V and 0.550 V, respectively. e, Structural superposition of the real-space refined atomic models for the mouse and human Leptin:LEP-RECD-tGCN4 complexes. f, Structural superposition of the real-space refined model for the hLeptin:hLEP-RIgCRH2 core with the structural prediction for hLEP-RECD via AlphaFold2 overlayed with the sharpened cryo-EM map for the hLeptin:hLEP-RECD-tGCN4 complex.
Extended Data Fig. 7 Structural insights into the mLEP-RFNIII conformation and map of naturally occurring mutations likely related to obesity on the Leptin:receptor assembly.
a, Cartoon representation of the determined crystal structure of mLEP-RFnIII:VHH-4.80 complex. Evolutionary conserved Trp across the interdomain interface and crystallographically observed N-linked glycosylation sites are shown as sticks. b, Mass spectrometric confirmation of the disulfide bridge between residues Cys602-Cys672 of the CRH2 and FNIII domains respectively in mLEP-RECD. The MS (top) and MS/MS spectra (bottom) are shown for the disulfide-linked peptides that are indicated on the left. c–f, Map of naturally occurring mutations likely related to obesity on the Leptin:receptor assembly. c, Domain distribution of missense variants identified in the human LEP-R gene of obese individuals (Supplementary Table 1), that have no currently known effect on protein secretion and stability. Functionally validated pathogenic mutations are highlighted in bold. d-e, Localization of mutations in the LEP and LEP-R genes (Supplementary Table 1) in the assembly. Mutated residues are shown as spheres. Functionally validated pathogenic mutations are highlighted in bold. f, Structural superposition of the crystallographically-resolved hLEP-RCRH2 domain (gray) with the AlphaFold model of hLEP-R (r.m.s.d.=0.669 Å, 171 Cα atoms). Residues found in variants likely related to obesity are annotated. A disulfide bridge is predicted to be formed between the CRH2 and FNIII domains (Cys604-Cys674), as shown in the insert. Both residues are functionally important1113. The disulfide bridge was confirmed by peptide mapping for mLEP-RECD, as shown in panel b. Cysteine 602 (604 in human) was mutated to serine in FNIII-deletion permutations of LEP-R in this study to prevent artificial disulfide-linked clusters.
Extended Data Fig. 8 Cryo-EM data analysis workflow for the hLeptin:hLEP-RtGCN4 complex.
a, SEC-MALLS analysis of the glycosylated hLeptin:hLEP-RECD-tGCN4 complex. The SEC-elution profile is plotted as the ultraviolet absorbance at 280 nm (left Y-axis) in function of elution volume. The total, protein and glycan molecular mass (right Y-axis) as determined by MALLS are reported as the average molecular mass (and s.d.) across the elution peak. Number of samples for hLeptin:hLEP-RECD-tGCN4 analyzed: n = 1. b, Cryo-EM data processing workflow in cryoSPARC for the glycosylated hLeptin:hLEP-RECD-tGCN4 complex. NU, non-uniform refinement; GSFSC: gold-standard Fourier shell correlation.
Extended Data Fig. 9 Receptor co-localization at the cell surface probed by smTIRFM.
a, Schematic overview of single molecule tracking and data evaluation. b, Dual-color (2 C) co-tracking. c, Triple-color (3 C) co-tracking. d, Diffusion properties; arrows indicate percentage of diffusional decrease e, fraction of immobilized emitters. For b-e, the number of cells (N) examined over n = 2 independent experiments for all LepR experiments, and n = 1 control experiment for TpoR, is indicated in the top row. Statistical differences were calculated with two-tailed two-sample Kolmogorov-Smirnov tests (**P ≤ 0.01, ***P ≤ 0.001; ns: not significant). Exact P-values for each comparison are given in the source data file of Fig. 6. Box and whisker plots show the five number summaries of the data: minimum, first quartile, median, third quartile, and maximum values. Outliers are indicated with an asterisk.
Supplementary information
Supplementary Information
Supplementary Figs. 1–5, Tables 1 and 2, and References.
mLep:mLEP-Rb assembly in living cells. Co-localization of mLEP-Rb in the absence (left) and presence of ligand (middle: two-color co-localization; right: three-color co-localization, 10 nM mLeptin, 10 min). Green, red and blue signals correspond to receptors labeled with Cy3BNB, Atto643NB and Dy752NB, respectively. Out-of-focus immobile signals observed in the presence of mLeptin can be explained by membrane-proximal endosomes. Acquisition frame rate: 33 Hz. Playback: real time.
mLep:mLEP-RΔ866-tFoldon assembly in living cells. Co-localization of mLEP-RΔ866-tFoldon in presence of ligand (left: two-color co-localization; right: three-color co-localization, 10 nM mLeptin, 10 min). Green, red and blue signals correspond to receptors labeled with Cy3BNB, Atto643NB and Dy752NB, respectively. Acquisition frame rate: 33 Hz. Playback: real time.
Source data
Source Data Fig. 3
SEC–MALLS and signaling source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 1
SEC–MALLS source data, biolayer interferometry source data and statistical analysis.
Source Data Extended Data Fig. 1
Uncropped SDS–PAGE gel.
Source Data Extended Data Fig. 3
SEC–MALLS and signaling source data.
Source Data Extended Data Fig. 5
SEC–MALLS and SAXS source data.
Source Data Extended Data Fig. 8
SEC–MALLS source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tsirigotaki, A., Dansercoer, A., Verschueren, K.H.G. et al. Mechanism of receptor assembly via the pleiotropic adipokine Leptin. Nat Struct Mol Biol 30, 551–563 (2023). https://doi.org/10.1038/s41594-023-00941-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-023-00941-9
This article is cited by
-
Structures of complete extracellular receptor assemblies mediated by IL-12 and IL-23
Nature Structural & Molecular Biology (2024)
-
Structural insights into the mechanism of leptin receptor activation
Nature Communications (2023)
-
How to control hunger
Nature Structural & Molecular Biology (2023)
-
Chemical shift assignments of wildtype human leptin
Biomolecular NMR Assignments (2023)