Abstract
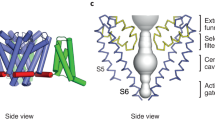
Voltage-gated ion channels (VGICs) contain positively charged residues within the S4 helix of the voltage-sensing domain (VSD) that are displaced in response to changes in transmembrane voltage, promoting conformational changes that open the pore. Pacemaker hyperpolarization-activated cyclic nucleotide-gated (HCN) channels are unique among VGICs because their open probability is increased by membrane hyperpolarization rather than depolarization. Here we measured the precise movement of the S4 helix of a sea urchin HCN channel using transition metal ion fluorescence resonance energy transfer (tmFRET). We show that the S4 undergoes a substantial (~10 Å) downward movement in response to membrane hyperpolarization. Furthermore, by applying distance constraints determined from tmFRET experiments to Rosetta modeling, we reveal that the carboxy-terminal part of the S4 helix exhibits an unexpected tilting motion during hyperpolarization activation. These data provide a long-sought glimpse of the hyperpolarized state of a functioning VSD and also a framework for understanding the dynamics of reverse gating in HCN channels.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The coordinate files of the State 1 and State 2 models were deposited in PDB-dev (https://pdb-dev.wwpdb.org/) with accession code PDBDEV_00000032. Source data for Figs. 3 and 4 and Supplementary Fig. 6 are available online. Other data are available from the corresponding author upon reasonable request.
Code availability
Rosetta scripts used for the modeling are in Supplementary Dataset 1.
References
Hille, B. Ion Channels of Excitable Membranes 3rd edn (Sinauer Associates, 2001).
Armstrong, C. M. & Hille, B. Voltage-gated ion channels and electrical excitability. Neuron 20, 371–380 (1998).
Long, S. B., Campbell, E. B. & Mackinnon, R. Voltage sensor of Kv1.2: structural basis of electromechanical coupling. Science 309, 903–908 (2005).
Stühmer, W. et al. Structural parts involved in activation and inactivation of the sodium channel. Nature 339, 597–603 (1989).
Papazian, D. M., Timpe, L. C., Jan, Y. N. & Jan, L. Y. Alteration of voltage-dependence of Shaker potassium channel by mutations in the S4 sequence. Nature 349, 305–310 (1991).
Yang, N. & Horn, R. Evidence for voltage-dependent S4 movement in sodium channels. Neuron 15, 213–218 (1995).
Larsson, H. P., Baker, O. S., Dhillon, D. S. & Isacoff, E. Y. Transmembrane movement of the Shaker K+ channel S4. Neuron 16, 387–397 (1996).
Mannikko, R., Elinder, F. & Larsson, H. P. Voltage-sensing mechanism is conserved among ion channels gated by opposite voltages. Nature 419, 837–841 (2002).
Catterall, W. A., Wisedchaisri, G. & Zheng, N. The chemical basis for electrical signaling. Nat. Chem. Biol. 13, 455–463 (2017).
Tao, X., Lee, A., Limapichat, W., Dougherty, D. A. & MacKinnon, R. A gating charge transfer center in voltage sensors. Science 328, 67–73 (2010).
Long, S. B., Tao, X., Campbell, E. B. & MacKinnon, R. Atomic structure of a voltage-dependent K+ channel in a lipid membrane-like environment. Nature 450, 376–382 (2007).
Clairfeuille, T. et al. Structural basis of alpha-scorpion toxin action on Nav channels. Science 363, eaav8573 (2019).
Xu, H. et al. Structural basis of Nav1.7 inhibition by a gating-modifier spider toxin. Cell 176, 1238–1239 (2019).
Guo, J. et al. Structure of the voltage-gated two-pore channel TPC1 from Arabidopsis thaliana. Nature 531, 196–201 (2016).
Biel, M., Wahl-Schott, C., Michalakis, S. & Zong, X. Hyperpolarization-activated cation channels: from genes to function. Physiol. Rev. 89, 847–885 (2009).
DiFrancesco, D. Characterization of single pacemaker channels in cardiac sino-atrial node cells. Nature 324, 470–473 (1986).
Santoro, B. et al. Identification of a gene encoding a hyperpolarization-activated pacemaker channel of brain. Cell 93, 717–729 (1998).
Ludwig, A., Zong, X., Jeglitsch, M., Hofmann, F. & Biel, M. A family of hyperpolarization-activated mammalian cation channels. Nature 393, 587–591 (1998).
Lee, C. H. & MacKinnon, R. Structures of the human HCN1 hyperpolarization-activated channel. Cell 168, 111–120.e11 (2017).
James, Z. M. & Zagotta, W. N. Structural insights into the mechanisms of CNBD channel function. J. Gen. Physiol. 150, 225–244 (2018).
Vemana, S., Pandey, S. & Larsson, H. P. S4 movement in a mammalian HCN channel. J. Gen. Physiol. 123, 21–32 (2004).
Cowgill, J. et al. Bipolar switching by HCN voltage sensor underlies hyperpolarization activation. Proc. Natl Acad. Sci. USA 116, 670–678 (2018).
Flynn, G. E. & Zagotta, W. N. Insights into the molecular mechanism for hyperpolarization-dependent activation of HCN channels. Proc. Natl Acad. Sci. USA 115, E8086–E8095 (2018).
Gauss, R., Seifert, R. & Kaupp, U. B. Molecular identification of a hyperpolarization-activated channel in sea urchin sperm. Nature 393, 583–587 (1998).
Flynn, G. E., Black, K. D., Islas, L. D., Sankaran, B. & Zagotta, W. N. Structure and rearrangements in the carboxy-terminal region of SpIH channels. Structure 15, 671–682 (2007).
Shin, K. S., Maertens, C., Proenza, C., Rothberg, B. S. & Yellen, G. Inactivation in HCN channels results from reclosure of the activation gate: desensitization to voltage. Neuron 41, 737–744 (2004).
Taraska, J. W., Puljung, M. C., Olivier, N. B., Flynn, G. E. & Zagotta, W. N. Mapping the structure and conformational movements of proteins with transition metal ion FRET. Nat. Methods 6, 532–537 (2009).
Gordon, S. E., Senning, E. N., Aman, T. K. & Zagotta, W. N. Transition metal ion FRET to measure short-range distances at the intracellular surface of the plasma membrane. J. Gen. Physiol. 147, 189–200 (2016).
Zagotta, W. N., Gordon, M. T., Senning, E. N., Munari, M. A. & Gordon, S. E. Measuring distances between TRPV1 and the plasma membrane using a noncanonical amino acid and transition metal ion FRET. J. Gen. Physiol. 147, 201–216 (2016).
Aman, T. K., Gordon, S. E. & Zagotta, W. N. Regulation of CNGA1 channel gating by interactions with the membrane. J. Biol. Chem. 291, 9939–9947 (2016).
Dai, G., James, Z. M. & Zagotta, W. N. Dynamic rearrangement of the intrinsic ligand regulates KCNH potassium channels. J. Gen. Physiol. 150, 625–635 (2018).
Dai, G. & Zagotta, W. N. Molecular mechanism of voltage-dependent potentiation of KCNH potassium channels. eLife 6, e26355 (2017).
Chatterjee, A., Guo, J., Lee, H. S. & Schultz, P. G. A genetically encoded fluorescent probe in mammalian cells. J. Am. Chem. Soc. 135, 12540–12543 (2013).
Kalstrup, T. & Blunck, R. Dynamics of internal pore opening in Kv channels probed by a fluorescent unnatural amino acid. Proc. Natl Acad. Sci. USA 110, 8272–8277 (2013).
Zheng, J. & Zagotta, W. N. Patch-clamp fluorometry recording of conformational rearrangements of ion channels. Sci. STKE 2003, PL7 (2003).
Ryu, S. & Yellen, G. Charge movement in gating-locked HCN channels reveals weak coupling of voltage sensors and gate. J. Gen. Physiol. 140, 469–479 (2012).
Stryer, L. Fluorescence energy transfer as a spectroscopic ruler. Annu. Rev. Biochem 47, 819–846 (1978).
Taraska, J. W., Puljung, M. C. & Zagotta, W. N. Short-distance probes for protein backbone structure based on energy transfer between bimane and transition metal ions. Proc. Natl Acad. Sci. USA 106, 16227–16232 (2009).
Bruening-Wright, A., Elinder, F. & Larsson, H. P. Kinetic relationship between the voltage sensor and the activation gate in spHCN channels. J. Gen. Physiol. 130, 71–81 (2007).
Gordon, S. E., Munari, M. & Zagotta, W. N. Visualizing conformational dynamics of proteins in solution and at the cell membrane. eLife 7, e37248 (2018).
Kodama, M. & Kimura, E. Equilibria and kinetics of complex formation between zinc(II), lead(II), and cadmium(II), and 12-, 13-, 14-, and 15-membered macrocyclic tetra-amines. Dalton Trans. 1977, 2269–2276 (1977).
Polyhach, Y. & Jeschke, G. Prediction of favourable sites for spin labelling of proteins. Spectroscopy 24, 651–659 (2010).
Jeschke, G. DEER distance measurements on proteins. Annu. Rev. Phys. Chem. 63, 419–446 (2012).
Vieira-Pires, R. S. & Morais-Cabral, J. H. 310 helices in channels and other membrane proteins. J. Gen. Physiol. 136, 585–592 (2010).
Lorinczi, E. et al. Voltage-dependent gating of KCNH potassium channels lacking a covalent link between voltage-sensing and pore domains. Nat. Commun. 6, 6672 (2015).
Tomczak, A. P. et al. A new mechanism of voltage-dependent gating exposed by KV10.1 channels interrupted between voltage sensor and pore. J. Gen. Physiol. 149, 577–593 (2017).
Cha, A., Snyder, G. E., Selvin, P. R. & Bezanilla, F. Atomic scale movement of the voltage-sensing region in a potassium channel measured via spectroscopy. Nature 402, 809–813 (1999).
Glauner, K. S., Mannuzzu, L. M., Gandhi, C. S. & Isacoff, E. Y. Spectroscopic mapping of voltage sensor movement in the Shaker potassium channel. Nature 402, 813–817 (1999).
Bell, D. C., Yao, H., Saenger, R. C., Riley, J. H. & Siegelbaum, S. A. Changes in local S4 environment provide a voltage-sensing mechanism for mammalian hyperpolarization-activated HCN channels. J. Gen. Physiol. 123, 5–19 (2004).
Posson, D. J., Ge, P., Miller, C., Bezanilla, F. & Selvin, P. R. Small vertical movement of a K+ channel voltage sensor measured with luminescence energy transfer. Nature 436, 848–851 (2005).
Chanda, B., Asamoah, O. K., Blunck, R., Roux, B. & Bezanilla, F. Gating charge displacement in voltage-gated ion channels involves limited transmembrane movement. Nature 436, 852–856 (2005).
Kintzer, A. F. et al. Structural basis for activation of voltage sensor domains in an ion channel TPC1. Proc. Natl Acad. Sci. USA 115, E9095–E9104 (2018).
She, J. et al. Structural insights into the voltage and phospholipid activation of the mammalian TPC1 channel. Nature 556, 130–134 (2018).
Varnum, M. D., Black, K. D. & Zagotta, W. N. Molecular mechanism for ligand discrimination of cyclic nucleotide-gated channels. Neuron 15, 619–625 (1995).
Pian, P., Bucchi, A., Decostanzo, A., Robinson, R. B. & Siegelbaum, S. A. Modulation of cyclic nucleotide-regulated HCN channels by PIP2 and receptors coupled to phospholipase C. Pflugers Arch. 455, 125–145 (2007).
Rohl, C. A., Strauss, C. E., Misura, K. M. & Baker, D. Protein structure prediction using Rosetta. Methods Enzymol. 383, 66–93 (2004).
Song, Y. et al. High-resolution comparative modeling with RosettaCM. Structure 21, 1735–1742 (2013).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Selvin, P. R. Principles and biophysical applications of lanthanide-based probes. Annu. Rev. Biophys. Biomol. Struct. 31, 275–302 (2002).
Acknowledgements
We thank X. Optiz-Araya and S. Haraguchi for animal care and surgical support; S.E. Gordon, L. Delemotte and all members of the Zagotta laboratory for their helpful advice and support; D. Farrell for help with the PDB-dev submission; and L. Anson for comments on the manuscript. This work was funded by NIH Grants (nos. R01EY010329, R01MH102378 and R01GM125351 to W.N.Z. and no. F32NS077622 to T.K.A.) and an American Heart Association Award (no. 14CSA20380095 to W.N.Z.).
Author information
Authors and Affiliations
Contributions
G.D., T.K.A. and W.N.Z. conceived and designed experiments and G.D. performed experiments. T.K.A. performed pilot experiments. F.D. designed and performed Rosetta-based computational modeling. G.D., T.K.A., F.D. and W.N.Z. analyzed data. W.N.Z. and G.D. wrote the manuscript. G.D., T.K.A., F.D. and W.N.Z. edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Inês Chen was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Electrophysiological properties of spHCN channels with incorporated Anap.
a, Left: representative current traces of wild-type spHCN channels in the absence or the presence of 1 mM cAMP. In the absence of cyclic nucleotide, the spHCN channel rapidly inactivates with hyperpolarization. Right: representative current traces from wild-type spHCN channels elicited by a series of hyperpolarizing voltage pulses from 0 mV to -110 mV with 10 mV steps, in the presence of 1 mM cAMP. b, Representative current traces from spHCN channels with L-Anap incorporated at different sites elicited by a series of hyperpolarizing voltage pulses from 0 mV to -120 mV, in the presence of 1 mM cAMP.
Supplementary Figure 2 Correlation of the Anap fluorescence and the YFP fluorescence and estimation of the quantum yields of L-Anap.
a-e, Plots of Anap versus YFP fluorescence intensities from multiple patches for the various L-Anap sites in spHCN. The slopes of the linear fit were used to calculate the L-Anap brightness relative to YFP. f, From the brightness of L-Anap at 0 mV and -100 mV, the quantum yields were estimated assuming the extinction coefficient of L-Anap was unchanged at different sites and different voltages.
Supplementary Figure 3 Emission spectra for L-Anap at each position in spHCN.
a-e, Emission spectra of L-Anap measured with a spectrograph for the indicated positions in spHCN at 0 mV and -100 mV. These spectra were fit with emission spectra for free L-Anap in solution, measured with a fluorometer shifted so that the peak positions are the same (black traces). The small peak at about 530 nm most likely corresponds to direct excitation of YFP. Also shown is the absorption spectra of Cu2+-TETAC (blue). f, Summary of the peak wavelength of the Anap emission for the various Anap sites in spHCN (n = 3 - 4, *p < 0.05). Error bars are s.e.m.
Supplementary Figure 4 ACCuRET between an Anap site in the S1 helix and the transition metal ion site in the HCN domain.
a, Cartoon showing the W218 Anap site and L186C modified by Cu2+-TETAC. b, Simultaneous current and Anap fluorescence recordings of spHCN-W218Anap channels. The Anap fluorescence did not change appreciably with -100 mV voltage pulses in the presence of 1 mM cAMP. c, Summary of the fractional Cu2+-TETAC quenching of Anap fluorescence at the W218 site, without and with the introduced cysteine L186C, at 0 mV and -100 mV (n = 4, error bars are s.e.m.). d, FRET efficiency calculated using the quenching data in panel c and Equation 1 (see Methods). e, Summary showing the distances of the W218/L186 FRET pair at 0 mV and -100 mV, calculated from the FRET efficiency in panel d and the FCG equation.
Supplementary Figure 5 Cu2+-TETAC binding to L186C site does not significantly change the electrophysiological properties of spHCN channels.
a-d, Left: representative current traces of spHCN-L186C channels with L-Anap incorporated at S346, L348, S353, and W355 sites of the S4 helix before and after applying 10 µM Cu2+-TETAC. Currents were recorded in the inside-out patch configuration and elicited by a series of hyperpolarizing voltage pulses from 0 mV to -110 mV (-100 mV for S353) with 10 mV steps, in the presence of 1 mM cAMP. Right: Normalized G-V relationships for the same recordings on the left before and after applying 10 µM Cu2+-TETAC. e, Time course of the Anap fluorescence for spHCN-W355Anap, L186C channels before and during application of 10 µM Cu2+-TETAC, illustrating that Anap fluorescence was not further decreased after 2 mins of applying Cu2+-TETAC compared to 1 min. This suggests the labeling of L186C was complete within 1 minute of Cu2+-TETAC application. n = 3, error bars are s.e.m.
Supplementary Figure 6 Different methods for correcting background quenching produce similar measurements of FRET efficiency.
Supplementary Figure 7 Alternative Rosetta models at -100 mV using different weights for experimentally-determined distance constraints.
Structure of 11 models varying the weights of experimentally-determined distance constraints from 50% to 200% of that used for the model in Fig. 5 (weight of 15, also see Methods). The green-colored S4 helix is the model shown in Fig. 5. The S4 helices in the other 10 models are shown in different colors.
Supplementary Figure 8 Fluorescence properties of L-Anap.
a, Emission spectra of free L-Anap in different solvents. Insert: overlap of spectra in the different solvents illustrates that the shape of the spectra is not appreciably affected by these different solvents. b, Emission spectra of L-Anap using the spectrograph attached to the patch-clamp microscope, using different slit widths. Slit 1 width: 5.9 µm; slit 2 width: 10.7 µm; slit 3 width: 24.8 µm. The shape of the spectra is not appreciably affected by different slit widths up to 25 µm, much larger than the diameter of our patches. c, Absorption spectra of free L-Anap in different solvents. The spectra were normalized to the extinction coefficient (17,500 M-1 cm-1) of L-Anap in EtOH at 360 nm as previously reported by Chatterjee et al. 2013. In our estimates of the quantum yield of Anap incorporated into the channel, we assume the environmental change in fluorescence intensity was entirely due to a change in quantum yield and not extinction coefficient.
Supplementary information
Supplementary Information
Supplementary Figures 1–8, Supplementary Table 1
Supplementary Dataset 1
Rosetta.sym file, Rosetta.xml file, Rosetta.sh file, Rosetta State 1 cen.cst file, Rosetta State 1 fa.cst file, Rosetta State 2 cen.cst file, Rosetta State 2 fa.cst file.
Supplementary Video 1
Patch-clamp fluorometry image of spHCN-S346Anap channels in response to a repetitive –100 mV hyperpolarizing voltage in the presence of 1 mM cAMP in the bath. The upper left bar indicates three one-second pulses to –100 mV interspersed with one-second intervals at 0 mV. The holding potential is 0 mV. Scale bar on the lower left is 15 μm.
Supplementary Video 2
Patch-clamp fluorometry image of spHCN-W355Anap channels in response to a –100 mV hyperpolarizing voltage in the presence of 1 mM cAMP in the bath. The upper left bar indicates a two-second pulse to –100 mV (the same duration as in Fig. 2b,d). Scale bar on the lower left is 15 μm.
Supplementary Video 3
A morph between the 0 mV (State 1) and –100 mV (State 2) state Rosetta models. The morph highlights the red/blue charge-smoothed surface of the HCN domain and the S1–S3 helices which the charged arginines and lysines of the S4 helix are interacting with. This qualitative vacuum electrostatic surface is automatically generated using the PyMOL software (https://pymol.org) with red color representing the negatively charged surface and blue color representing positively charged surface.
Supplementary Video 4
Rosetta model of the hyperpolarization-induced S4 voltage sensor movement in the spHCN channel. The movie shows a morph between the Rosetta model at 0 mV (State 1) and the Rosetta model at –100 mV (State 2), highlighting the distance changes for the specific Anap sites between these two states.
Supplementary Video 5
Comparison of three different examples of S4 movement in VGIC family. Model of S4 movement in spHCN channels (left), S4 movement in TPCs (middle) and S4 movement in VSD2 of Nav channels (right). The model in the middle is a morph between the ‘up’ state of VSD2 S4 in mouse TPC1 (PDB: 6C96) and the ‘down’ state of the VSD2 S4 in Arabidopsis TPC1 (PDB: 5E1J). The model on the right is a morph between the ‘up’ state of S4 in Nav1.7-VSD2 (PDB: 6N4Q) and the ‘down’ state of S4 in Nav1.7- VSD2 (PDB: 6N4R) in complex with the gating modifier toxin ProTx2 (not shown).
Rights and permissions
About this article
Cite this article
Dai, G., Aman, T.K., DiMaio, F. et al. The HCN channel voltage sensor undergoes a large downward motion during hyperpolarization. Nat Struct Mol Biol 26, 686–694 (2019). https://doi.org/10.1038/s41594-019-0259-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-019-0259-1
This article is cited by
-
Structures of a sperm-specific solute carrier gated by voltage and cAMP
Nature (2023)
-
Direct regulation of the voltage sensor of HCN channels by membrane lipid compartmentalization
Nature Communications (2023)
-
Gating intermediates reveal inhibitory role of the voltage sensor in a cyclic nucleotide-modulated ion channel
Nature Communications (2022)
-
Structural basis for the activity regulation of a potassium channel AKT1 from Arabidopsis
Nature Communications (2022)
-
Electromechanical coupling mechanism for activation and inactivation of an HCN channel
Nature Communications (2021)