Abstract
Photochemical conversion in oxygenic photosynthesis takes place in two large protein–pigment complexes named photosystem II and photosystem I (PSII and PSI, respectively). Photosystems associate with antennae in vivo to increase the size of photosynthetic units to hundreds or thousands of pigments. Regulation of the interactions between antennae and photosystems allows photosynthetic organisms to adapt to their environment. In low-iron environments, cyanobacteria express IsiA, a PSI antenna, critical to their survival. Here we describe the structure of the PSI–IsiA complex isolated from the mesophilic cyanobacterium Synechocystis sp. PCC 6803. This 2-MDa photosystem–antenna supercomplex structure reveals more than 700 pigments coordinated by 51 subunits, as well as the mechanisms facilitating the self-assembly and association of IsiA with multiple PSI assemblies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The final model (PDB 6NWA) and map (EMD 0524) were deposited in the Protein Databank and Electro-Magnetic Database, respectively. All other data are available from the authors upon reasonable request.
Code availability
Dipole orientation values were calculated using an R script available from the author upon reasonable request.
References
Nelson, N. & Yocum, C. F. Structure and function of photosystems I and II. Annu. Rev. Plant Biol. 57, 521–565 (2006).
Croce, R. & van Amerongen, H. Natural strategies for photosynthetic light harvesting. Nat. Chem. Biol. 10, 492–501 (2014).
Buchel, C. Evolution and function of light harvesting proteins. J. Plant Physiol. 172, 62–75 (2015).
Saer, R. G. & Blankenship, R. E. Light harvesting in phototrophic bacteria: structure and function. Biochem. J. 474, 2107–2131 (2017).
La Roche, J. et al. Independent evolution of the prochlorophyte and green plant chlorophyll a/b light-harvesting proteins. Proc. Natl Acad. Sci. USA 93, 15244–15248 (1996).
Guikema, J. A. & Sherman, L. A. Organization and function of chlorophyll in membranes of cyanobacteria during iron starvation. Plant Physiol. 73, 250–256 (1983).
Laudenbach, D. E. & Straus, N. A. Characterization of a cyanobacterial iron stress-induced gene similar to psbC. J. Bacteriol. 170, 5018–5026 (1988).
Burnap, R. L., Troyan, T. & Sherman, L. A. The highly abundant chlorophyll-protein complex of iron-deficient Synechococcus sp. PCC7942 (CP43′) is encoded by the isiA gene. Plant Physiol. 103, 893–902 (1993).
Havaux, M. et al. The chlorophyll-binding protein IsiA is inducible by high light and protects the cyanobacterium Synechocystis PCC6803 from photooxidative stress. FEBS Lett. 579, 2289–2293 (2005).
Singh, A. K., Li, H. & Sherman, L. A. Microarray analysis and redox control of gene expression in the cyanobacterium Synechocystis sp. PCC 6803. Physiol. Plant. 120, 27–35 (2004).
Bibby, T. S., Nield, J. & Barber, J. Three-dimensional model and characterization of the iron stress-induced CP43′-photosystem I supercomplex isolated from the cyanobacterium Synechocystis PCC 6803. J. Biol. Chem. 276, 43246–43252 (2001).
Boekema, E. J. et al. A giant chlorophyll-protein complex induced by iron deficiency in cyanobacteria. Nature 412, 745–748 (2001).
Bibby, T. S., Nield, J. & Barber, J. Iron deficiency induces the formation of an antenna ring around trimeric photosystem I in cyanobacteria. Nature 412, 743–745 (2001).
Kouril, R. et al. Structure and functional role of supercomplexes of IsiA and Photosystem I in cyanobacterial photosynthesis. FEBS Lett. 579, 3253–3257 (2005).
Chauhan, D. et al. A novel photosynthetic strategy for adaptation to low-iron aquatic environments. Biochemistry 50, 686–692 (2011).
Yeremenko, N. et al. Supramolecular organization and dual function of the IsiA chlorophyll-binding protein in cyanobacteria. Biochemistry 43, 10308–10313 (2004).
Ihalainen, J. A. et al. Aggregates of the chlorophyll-binding protein IsiA (CP43′) dissipate energy incyanobacteria. Biochemistry 44, 10846–10853 (2005).
Melkozernov, A. N., Bibby, T. S., Lin, S., Barber, J. & Blankenship, R. E. Time-resolved absorption and emission show that the CP43′ antenna ring of iron-stressed Synechocystis sp. PCC6803 is efficiently coupled to the Photosystem I reaction center core. Biochemistry 42, 3893–3903 (2003).
Andrizhiyevskaya, E. G., Frolov, D., Van Grondelle, R. & Dekker, J. P. Energy transfer and trapping in the Photosystem I complex of Synechococcus PCC 7942 and in its supercomplex with IsiA. Biochim. Biophys. Acta 1656, 104–113 (2004).
Garczarek, L., Hess, W. R., Holtzendorff, J., van der Staay, G. W. M. & Partensky, F. Multiplication of antenna genes as a major adaptation to low light in a marine prokaryote. Proc. Natl Acad. Sci. USA 97, 4098 LP–4094101 (2000).
Bibby, T. S., Mary, I., Nield, J., Partensky, F. & Barber, J. Low-light-adapted Prochlorococcus species possess specific antennae for each photosystem. Nature 424, 1051 (2003).
Scheres, S. H. W. & Chen, S. Prevention of overfitting in cryo-EM structure determination. Nat. Methods 9, 853–854 (2012).
Andrizhiyevskaya, E. G. et al. Spectroscopic properties of PSI-IsiA supercomplexes from the cyanobacterium Synechococcus PCC 7942. Biochim. Biophys. Acta 1556, 265–272 (2002).
Qin, X., Suga, M., Kuang, T. & Shen, J.-R. Photosynthesis. Structural basis for energy transfer pathways in the plant PSI-LHCI supercomplex. Science 348, 989–995 (2015).
Su, X. et al. Structure and assembly mechanism of plant C2S2M2-type PSII-LHCII supercomplex. Science 357, 815–820 (2017).
Mazor, Y., Borovikova, A., Caspy, I. & Nelson, N. Structure of the plant photosystem I supercomplex at 2.6 Å resolution. Nat. Plants 3, 17014 (2017).
Pi, X. et al. Unique organization of photosystem I-light-harvesting supercomplex revealed by cryo-EM from a red alga. Proc. Natl Acad. Sci. USA 115, 4423–4428 (2018).
Antoshvili, M., Caspy, I., Hippler, M. & Nelson, N. Structure and function of photosystem I in Cyanidioschyzon merolae. Photosynth. Res. 139, 499–508 (2018).
Mazor, Y., Borovikova, A. & Nelson, N. The structure of plant photosystem I super-complex at 2.8 Å resolution. eLife 4, e07433 (2015).
Chen, H.-Y. S., Liberton, M., Pakrasi, H. B. & Niedzwiedzki, D. M. Reevaluating the mechanism of excitation energy regulation in iron-starved cyanobacteria. Biochim. Biophys. Acta 1858, 249–258 (2017).
Berera, R., van Stokkum, I. H. M., Kennis, J. T. M., van Grondelle, R. & Dekker, J. P. The light-harvesting function of carotenoids in the cyanobacterial stress-inducible IsiA complex. Chem. Phys. 373, 65–70 (2010).
Kouril, R. et al. Supercomplexes of IsiA and photosystem I in a mutant lacking subunit PsaL. Biochim. Biophys. Acta 1706, 262–266 (2005).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Scheres, S. H. W. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 180, 519–530 (2012).
Kucukelbir, A., Sigworth, F. J. & Tagare, H. D. Quantifying the local resolution of cryo-EM density maps. Nat. Methods 11, 63 (2013).
Malavath, T., Caspy, I., Netzer-El, S. Y., Klaiman, D. & Nelson, N. Structure and function of wild-type and subunit-depleted photosystem I in Synechocystis. Biochim. Biophys. Acta 1859, 645–654 (2018).
Pettersen, E. F. et al. UCSF Chimera–a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Umena, Y., Kawakami, K., Shen, J.-R. & Kamiya, N. Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9A. Nature 473, 55–60 (2011).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, (213–221 (2010).
Afonine, P. V., Headd, J. J., Terwilliger, T. C. & Adams, P. D. New tool: phenix.real_space_refine. Comput. Crystallogr. Newsl. 4, 43–44 (2013).
The PyMOL Molecular Graphics System v.1.8 (Schrödinger, 2015).
Förster, T. Ein beitrag zur theorie der photosynthese. Z. Naturforsch. 2b, 174–182 (1947).
Acknowledgements
We would like to thank N. Nelson and P. Fromme for critical reading of the manuscript, and O. Rog for many discussions. We would like to acknowledge the use of the Titan Krios at the Erying Materials Center at Arizona State University, and the funding of this instrument by the National Science Foundation (No. MRI 1531991). This study is funded by a startup grant from Arizona State University.
Author information
Authors and Affiliations
Contributions
Y.M. and H.T. performed experiments, analyzed data and wrote the manuscript. J.L. performed experiments. D.W. performed experiments. P.-L.C. analyzed data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Cryo-EM structure of PSI – IsiA.
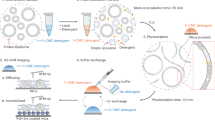
A. Sucrose gradients (10% – 60% sucrose) of IsiA-containing complexes showing that at our growth conditions there is one major fraction of PSI – IsiA. B. An SDS-PAGE gel showing the subunit composition of the main chlorophyll containing fraction from the first (I) and second (II) sucrose gradient showing the presence of IsiA. The same subunit composition is shown for the sample used for the cryo-EM experiments (Grid). C. Flow chart describing the workflow of image processing. D. A representative micrograph together with the power spectrum and CTF fit. In vitreous ice, PSI – IsiA particles are visible in different orientations as projections (a subset is surrounded by a red ring). E. Representative 2D class averages generated from unsupervised 2D classification. F. Top and side views, respectively, of the Euler angle distributions of particles obtained in the final refinement with C3 symmetry.
Supplementary Figure 2 Model resolution and map examples.
A, B, C and D: The final 3D map colored according to the local resolution estimates obtained from ResMap, seen from the Lumen ‘A’, Stroma ‘B’, membrane ‘C’ and membrane section with surface capping along the dashed line ‘D’ orientations. E. Plots of Fourier shell correlation (FSC) against resolution. F. Representative map sections showing transmembrane helices, carotenoids, chlorophylls and lipids from PSI – IsiA. The identity of each one is indicated above.
Supplementary Figure 3 IsiA orientation and alignment with CP43.
A. The orientation of IsiA monomers in PSI-IsiA. Only transmembrane segments are shown. Subunits are colored as in Fig. 1 in the main text. The six IsiA transmembrane helices are marked in position ‘a’. B. A detailed view of the E loop and two IsiA unique chlorophylls together with their coordinating residues, Gln 316 for chlorophyll 8 and the backbone carbonyl of Ile 282 for chlorophyll 17. C. The orientation of CP43 and D1 compared with that of the IsiA dimer. The figure shows the transmembrane helices of the respective subunits (labeled according to sequence position). D. Comparing the chlorophylls and carotenoids of IsiA and CP43 shows the position of two new chlorophylls (numbered 8 and 17) as well as a new carotenoid (B1) in addition to the three similar positions in CP43.
Supplementary information
Supplementary Information
Supplementary Figures 1–3, Supplementary Tables 1–3
Rights and permissions
About this article
Cite this article
Toporik, H., Li, J., Williams, D. et al. The structure of the stress-induced photosystem I–IsiA antenna supercomplex. Nat Struct Mol Biol 26, 443–449 (2019). https://doi.org/10.1038/s41594-019-0228-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-019-0228-8
This article is cited by
-
Unveiling large charge transfer character of PSII in an iron-deficient cyanobacterial membrane: A Stark fluorescence spectroscopy study
Photosynthesis Research (2024)
-
Uphill energy transfer mechanism for photosynthesis in an Antarctic alga
Nature Communications (2023)
-
Energetic robustness to large scale structural fluctuations in a photosynthetic supercomplex
Nature Communications (2023)
-
Structural insights into a unique PSI–LHCI–LHCII–Lhcb9 supercomplex from moss Physcomitrium patens
Nature Plants (2023)
-
Structural insights into the assembly and energy transfer of the Lhcb9-dependent photosystem I from moss Physcomitrium patens
Nature Plants (2023)