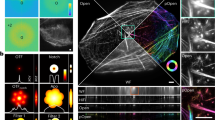
Abstract
vLUME is a virtual reality software package designed to render large three-dimensional single-molecule localization microscopy datasets. vLUME features include visualization, segmentation, bespoke analysis of complex local geometries and exporting features. vLUME can perform complex analysis on real three-dimensional biological samples that would otherwise be impossible by using regular flat-screen visualization programs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
All the experimental data used in the present communication can be found in the Supplementary Software.
Code availability
vLUME is available as Supplementary Software. Updated versions of the software can also be found at https://github.com/lumevr/vLume/releases, vLUME software for Windows (with manual, license, samples and scripts). Open-source plugins for vLUME and a forum for collaborative creation and improvement can be found at https://github.com/lumevr/vLume_OpenSourcePlugins.
References
Von Diezmann, A., Shechtman, Y. & Moerner, W. E. Three-dimensional localization of single molecules for super-resolution imaging and single-particle tracking. Chem. Rev. 117, 7244–7275 (2017).
Lee, M. K., Rai, P., Williams, J., Twieg, R. J. & Moerner, W. E. Small-molecule labeling of live cell surfaces for three-dimensional super-resolution microscopy. J. Am. Chem. Soc. 136, 14003–14006 (2014).
Sage, D. et al. Super-resolution Fight Club: assessment of 2D and 3D single-molecule localization microscopy software. Nat. Methods 16, 387–395 (2019).
Theart, R. P., Loos, B. & Niesler, T. R. Virtual reality assisted microscopy data visualization and colocalization analysis. BMC Bioinf. 18, 64 (2017).
Caroline, S. & Adam, L. H. ConfocalVR: immersive visualization for confocal microscopy. J. Mol. Biol. 430, 4028–4035 (2018).
El Beheiry, M. & Dahan, M. ViSP: representing single-particle localizations in three dimensions. Nat. Methods 10, 689–690 (2013).
Henriques, R. et al. QuickPALM: 3D real-time photoactivation nanoscopy image processing in ImageJ. Nat. Methods 7, 339–340 (2010).
Ovesný, M., Křížek, P., Borkovec, J., Švindrych, Z. & Hagen, G. M. ThunderSTORM: a comprehensive imageJ Plug-In for PALM and STORM data analysis and super-resolution imaging. Bioinformatics 30, 2389–2390 (2014).
Takashina, T., Ito, M. & Kokumai, Y. Evaluation of navigation operations in immersive microscopic visualization. VRST ‘19 Proc. 68, 68 (2019).
Culley, S. et al. Quantitative mapping and minimization of super-resolution optical imaging artifacts. Nat. Methods 15, 263–266 (2018).
Lee, S. F., Thompson, M. A., Schwartz, M. A., Shapiro, L. & Moerner, W. E. Super-resolution Imaging of the nucleoid-associated protein HU in Caulobacter crescentus. Biophys. J. 100, L31–L33 (2011).
Griffié, J. et al. 3D Bayesian cluster analysis of super-resolution data reveals LAT recruitment to the T cell synapse. Sci. Rep. 7, 4077 (2017).
Lillemeier, B. F. et al. TCR and Lat are expressed on separate protein islands on T cell membranes and concatenate during activation. Nat. Immunol. 11, 90–96 (2010).
Broadhead, M. J. et al. PSD95 nanoclusters are postsynaptic building blocks in hippocampus circuits. Sci. Rep. 6, 24626 (2016).
Leterrier, C., Dubey, P. & Roy, S. The nano-architecture of the axonal cytoskeleton. Nat. Rev. Neurosci. 18, 713–726 (2017).
Legant, W. R. et al. High-density three-dimensional localization microscopy across large volumes. Nat. Methods 13, 359–365 (2016).
Carr, A. R. Development of Three-Dimensional Super-Resolution Imaging Using a Double-Helix Point Spread Function. Doctoral thesis, Univ. Cambridge (2019); https://doi.org/10.17863/CAM.26413
Li, Y. et al. Real-time 3D single-molecule localization using experimental point spread functions. Nat. Methods 15, 367–369 (2018).
Jimenez, A., Friedl, K. & Leterrier, C. About samples, giving examples: optimized procedures for single molecule localization microscopy. Methods 174, 100–114 (2020).
Vassilopoulos, S. et al. Ultrastructure of the axonal periodic scaffold reveals a braid-like organization of actin rings. Nat. Commun. 10, 5803 (2019).
Acknowledgements
We thank M. Lee, J. Yoon and M. Lee from the laboratory of W.E. Moerner (Stanford) for kindly providing the C. crescentus datasets (Fig. 1d,e left). We thank the laboratory of J. Ries (EMBL Heidelberg) for the publicly available NPC data shown in Fig. 1c and for the microtubule datasets shown in Fig. 1d,e, left. We thank F. Boroni-Rueda and K. Friedl for preparation of the neuron samples. D.E.-F. thanks the European Union’s Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement no, 712949 (TECNIOspring Plus) and the Agency for Business Competitiveness of the Government of Catalonia for the research funding leading to these results. A.H. thanks the Engineering and Physical Sciences Research Council (EP/N509620/1). We thank the Royal Society for S.F.L.’s University Research Fellowship (no. UF120277). R.H. was funded by grants from the UK Biotechnology and Biological Sciences Research Council (no. BB/S507532/1), Wellcome Trust (no. 203276/Z/16/Z) and core funding by the MRC Laboratory for Molecular Cell Biology, University College London (grant no. MC_UU12018/7). We thank The Imagination Group, Imagination Europe and Imagination Labs for their support.
Author information
Authors and Affiliations
Contributions
S.F.L., A.S. and A.K. conceived the initial idea of visualizing 3D SMLM data in VR. A.S. and A.K. wrote, developed and designed the software. A.S, A.H. and D.E.-F. cowrote the open-source C# scripts. A.H. and D.E.-F. beta-tested the software for SMLM applications. A.H., D.E.-F., L.-M.N., R.H. and S.F.L. cowrote the manuscript. A.R.C. and A.P. performed and analyzed the T cell experiments. A.M.S., S.J.D. and J.M. provided the cell samples and labeling methodology. C.L. provided the spectrin data. All authors edited and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
A.S. and A.K. are cofounders of Lume VR Ltd, a company dedicated to creating image analysis software for life sciences and working with groups to tackle visualization and analysis problems. Lume VR Ltd. is providing a free for academic use licence of vLUME governed by Terms of Use that can be found in the Supplementary Software folder. All intellectual property rights in the vLUME software (not the user data), throughout the world, belong to Lume VR Ltd. The remaining authors declare no competing interests.
Additional information
Peer review information Rita Strack was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Microtubules and Clatherin in COS cells.
Two-channel data of Microtubules (purple) and Clathrin (green) in COS cells from dataset 5. Scale bar is approximately 1 um. a, A ‘birds- eye’ projection of the two channels in vLUME. b, The same data set from a different point of view closer to the ground to show the 3D nature of the data. To achieve this superposition the first channel has to be opened in vLUME and the color changed. Then the second channel also needs to be opened and changed in color. Subsequently with simple data translation of one of the datasets the two axes need to be overlapped (this task is very simple in VR, manual). The 3D nature of the oblique figure makes it difficult to render a scale bar.
Extended Data Fig. 2 Nearest Neighbours plot on Caulobacter stalk.
Nearest Neighbor plot using the C# script (CalcNearestNeighbour.cs) after selecting Caulobacter crescentus’ stalk from dataset 4 of the Supplementary Information (Fig. 1d, e, left). The red to blue gradient of the image shows an increasing density of nearest neighbors within a radius of 50 nm (user defined). The color-gradient scale bar goes from 0 to 34 neighbors. Scale bar is approximately 200 nm.
Supplementary information
Supplementary Information
Supplementary Table 1, videos and references.
Supplementary Video 1
Overview of vLUME. Overview of the main GUI and functionality in vLUME. The video shows a variety of datasets and the ease of going from the micron scale to nanoscale regions.
Supplementary Video 2
Selecting and annotating data. Demonstration of a user isolating a single microtubule from a complex tangle in vLUME to be saved as isolated data.
Supplementary Video 3
Loading and filtering data. Demonstration of a user isolating a stalk from a predivision stage of a C. crescentus in vLUME to be saved as isolated data.
Supplementary Video 4
Manipulation of data. Data manipulation features of vLUME on a single C. crescentus, showing the user actions simultaneously.
Supplementary Video 5
Selecting data and running scripts. Maximum and minimum distances, and nearest neighbor script application to a NPC dataset.
Supplementary Video 6
Outputting a video. Setting waypoints in the 3D space using vLUME and saving these points as a video.
Supplementary Video 7
Case study. Exploring annotating and analyzing spectrin rings in plated neurons.
Supplementary Software
vLume software for Windows.
Supplementary Sample Datasets
Sample datasets for resubmission.
Rights and permissions
About this article
Cite this article
Spark, A., Kitching, A., Esteban-Ferrer, D. et al. vLUME: 3D virtual reality for single-molecule localization microscopy. Nat Methods 17, 1097–1099 (2020). https://doi.org/10.1038/s41592-020-0962-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-020-0962-1
This article is cited by
-
Extended reality for biomedicine
Nature Reviews Methods Primers (2023)