Abstract
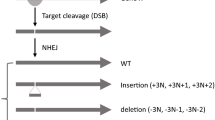
Gene knock outs (KOs) are efficiently engineered through CRISPR–Cas9-induced frameshift mutations. While the efficiency of DNA editing is readily verified by DNA sequencing, a systematic understanding of the efficiency of protein elimination has been lacking. Here we devised an experimental strategy combining RNA sequencing and triple-stage mass spectrometry to characterize 193 genetically verified deletions targeting 136 distinct genes generated by CRISPR-induced frameshifts in HAP1 cells. We observed residual protein expression for about one third of the quantified targets, at variable levels from low to original, and identified two causal mechanisms, translation reinitiation leading to N-terminally truncated target proteins or skipping of the edited exon leading to protein isoforms with internal sequence deletions. Detailed analysis of three truncated targets, BRD4, DNMT1 and NGLY1, revealed partial preservation of protein function. Our results imply that systematic characterization of residual protein expression or function in CRISPR–Cas9-generated KO lines is necessary for phenotype interpretation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The transcriptomics and proteomics data of the 19 KO lines shown in Fig. 1b and the NGLY1 KO lines were deposited to publicly available repositories. The mass spectrometry proteomics data were deposited to the ProteomeXchange Consortium via the PRIDE57 partner repository with the dataset identifier PXD010335. RNA-seq data were deposited in the ArrayExpress database at EMBL-EBI (www.ebi.ac.uk/arrayexpress) under accession number E-MTAB-7061. The data for the remaining 174 KO lines shown in Fig. 1a are available from the corresponding author upon request.
References
Doudna, J. A. & Charpentier, E. The new frontier of genome engineering with CRISPR–Cas9. Science 346, 1258096 (2014).
Hsu, P. D., Lander, E. S. & Zhang, F. Development and applications of CRISPR–Cas9 for genome engineering. Cell 157, 1262–1278 (2014).
Sander, J. D. & Joung, J. K. CRISPR–Cas systems for genome editing, regulation and targeting. Nat. Biotechnol. 32, 347–355 (2014).
Jinek, M. et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337, 816–821 (2012).
Jinek, M. et al. RNA-programmed genome editing in human cells. eLife 2, e00471 (2013).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Lieber, M. R. The mechanism of double-strand DNA break repair by the nonhomologous DNA end-joining pathway. Annu. Rev. Biochem. 79, 181–211 (2010).
Lykke-Andersen, J. & Bennett, E. J. Protecting the proteome: eukaryotic cotranslational quality control pathways. J. Cell Biol. 204, 467–476 (2014).
Fu, Y. et al. High-frequency off-target mutagenesis induced by CRISPR–Cas nucleases in human cells. Nat. Biotechnol. 31, 822–826 (2013).
Hsu, P. D. et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nat. Biotechnol. 31, 827–832 (2013).
Pattanayak, V. et al. High-throughput profiling of off-target DNA cleavage reveals RNA-programmed Cas9 nuclease specificity. Nat. Biotechnol. 31, 839–843 (2013).
Cho, S. W. et al. Analysis of off-target effects of CRISPR/Cas-derived RNA-guided endonucleases and nickases. Genome Res. 24, 132–141 (2014).
Tsai, S. Q. et al. GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR–Cas nucleases. Nat. Biotechnol. 33, 187–198 (2015).
Ran, F. A. et al. Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 154, 1380–1389 (2013).
Shen, B. et al. Efficient genome modification by CRISPR–Cas9 nickase with minimal off-target effects. Nat. Methods 11, 399–402 (2014).
Doench, J. G. et al. Rational design of highly active sgRNAs for CRISPR–Cas9-mediated gene inactivation. Nat. Biotechnol. 32, 1262–1267 (2014).
Doench, J. G. et al. Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR–Cas9. Nat. Biotechnol. 34, 184–191 (2016).
Kleinstiver, B. P. et al. High-fidelity CRISPR–Cas9 nucleases with no detectable genome-wide off-target effects. Nature 529, 490–495 (2016).
Slaymaker, I. M. et al. Rationally engineered Cas9 nucleases with improved specificity. Science 351, 84–88 (2016).
Kotecki, M., Reddy, P. S. & Cochran, B. H. Isolation and characterization of a near-haploid human cell line. Exp. Cell Res. 252, 273–280 (1999).
Carette, J. E. et al. Haploid genetic screens in human cells identify host factors used by pathogens. Science 326, 1231–1235 (2009).
Lackner, D. H et al. A generic strategy for CRISPR–Cas9-mediated gene tagging. Nat. Commun. 6, 10237 (2015).
Moll, P., Ante, M., Seitz, A. & Reida, T. QuantSeq 3′ mRNA sequencing for RNA quantification. Nat. Methods 11, 972 (2014).
Lindeboom, R. G. H., Supek, F. & Lehner, B. The rules and impact of nonsense-mediated mRNA decay in human cancers. Nat. Genet. 48, 1112–1118 (2016).
Ting, L., Rad, R., Gygi, S. P. & Haas, W. MS3 eliminates ratio distortion in isobaric multiplexed quantitative proteomics. Nat. Methods 8, 937–940 (2011).
Grossmann, J. et al. Implementation and evaluation of relative and absolute quantification in shotgun proteomics with label-free methods. J. Proteom. 73, 1740–1746 (2010).
Zhang, J. & Maquat, L. E. Evidence that translation reinitiation abrogates nonsense-mediated mRNA decay in mammalian cells. EMBO J. 16, 826–833 (1997).
Makino, S., Fukumura, R. & Gondo, Y. Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles created by CRISPR–Cas9. Sci. Rep. 6, 39608 (2016).
Filippakopoulos, P. et al. Histone recognition and large-scale structural analysis of the human bromodomain family. Cell 149, 214–231 (2012).
Bechtel, S et al. The full-ORF clone resource of the German cDNA Consortium. BMC Genomics 8, 399 (2007).
Filippakopoulos, P. et al. Selective inhibition of BET bromodomains. Nature 468, 1067–1073 (2010).
Dawson, M. A. et al. Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia. Nature 478, 529–533 (2011).
Zerbino, D. R. et al. Ensembl 2018. Nucleic Acids Res. 46, D754–D761 (2017).
Kar, S. et al. An insight into the various regulatory mechanisms modulating human DNA methyltransferase 1 stability and function. Epigenetics 7, 994–1007 (2012).
Tomlin, F. M. et al. Inhibition of NGLY1 inactivates the ranscription factor Nrf1 and potentiates proteasome inhibitor cytotoxicity. ACS Cent. Science 3, 1143–1155 (2017).
Misaghi, S., Pacold, M. E., Blom, D., Ploegh, H. L. & Korbel, G. A. Using a small molecule inhibitor of peptide: N-glycanase to probe its role in glycoprotein turnover. Chem. Biol. 11, 1677–1687 (2004).
Enns, G. M. et al. Mutations in NGLY1 cause an inherited disorder of the endoplasmic reticulum-associated degradation pathway. Genet. Med. 16, 751 (2014).
Kapahnke, M., Banning, A. & Tikkanen, R. Random splicing of several exons caused by a single base hange in the target exon of CRISPR/Cas9 mediated gene knockout. Cells 5, 45 (2016).
Mou, H et al. CRISPR/Cas9-mediated genome editing induces exon skipping by alternative splicing or exon deletion. Genome Biol. 18, 108 (2017).
Mitchell, A. L. et al. InterPro in 2019: improving coverage, classification and access to protein sequence annotations. Nucleic Acids Res. 47, D351–D360 (2019).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Robinson, J. T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24 (2011).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Hahne, F. & Ivanek, R. in Statistical Genomics: Methods and Protocols (Eds. Mathé, E & Davis, S) 335–351 (Humana Press, 2016).
Durinck, S., Spellman, P. T., Birney, E. & Huber, W. Mapping identifiers for the integration of genomic datasets with the R/ Bioconductor package biomaRt. Nat. Protoc. 4, 1184–1191 (2009).
Moggridge, S., Sorensen, P. H., Morin, G. B. & Hughes, C. S. Extending the compatibility of the SP3 paramagnetic bead processing approach for proteomics. J. Proteome Res. 17, 1730–1740 (2018).
Werner, T. et al. High-resolution enabled TMT 8-plexing. Anal. Chem. 84, 7188–7194 (2012).
Werner, T. et al. Ion coalescence of neutron encoded TMT 10-plex reporter ions. Anal. Chem. 86, 3594–3601 (2014).
Savitski, M. M. et al. Multiplexed proteome dynamics profiling reveals mechanisms controlling protein homeostasis. Cell 173, 260–274 (2018).
Savitski, M. M. et al. Targeted data acquisition for improved reproducibility and robustness of proteomic mass spectrometry assays. J. Am. Soc. Mass Spectrom. 21, 1668–1679 (2010).
Savitski, M. M. et al. Measuring and managing ratio compression for accurate iTRAQ/TMT quantification. J. Proteome Res. 12, 3586–3598 (2013).
Franken, H. et al. Thermal proteome profiling for unbiased identification of direct and indirect drug targets using multiplexed quantitative mass spectrometry. Nat. Protoc. 10, 1567–1593 (2015).
Huber, W. et al. Orchestrating high-throughput genomic analysis with Bioconductor. Nat. Methods 12, 115–121 (2015).
Wickham, H. Elegant Graphics for Data Analysis (Springer-Verlag, New York, 2009).
Fortin, J.-P., Triche, T. J. Jr & Hansen, K. D. Preprocessing, normalization and integration of the Illumina HumanMethylationEPIC array with minfi. Bioinformatics 33, 558–560 (2016).
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014).
Vizcaino, J. A. et al. 2016 update of the PRIDE database and its related tools. Nucleic Acids Res. 44, D447–D456 (2015).
Acknowledgements
We would like to thank D. Pavlinic, F. Jung and V. Benes for RNA-seq; J. Stuhlfauth and team for cell banking; M. Boesche and team for mass spectrometry analyses; S. Shimamura for cell culture and sample preparation; and the microarray unit of the DKFZ Genomics and Proteomics Core Facility for providing the Illumina Human Methylation arrays and related services. A.H.S. was supported by a fellowship from the EMBL Interdisciplinary Postdoc (EIPOD) Programme under a grant from the Marie Sklodowska-Curie Actions COFUND (664726). The NGLY1 work was supported by the Grace Science Foundation.
Author information
Authors and Affiliations
Contributions
A.H.S. and F.Z. analyzed the data. A.H.S. and D.E. performed transcriptomics experiments. N.Z. performed mass spectrometry measurements. G.J. resequenced cell lines and analyzed BRD4 and DNMT1 truncation data. W.F.M., K.T. and H.S. performed all NGLY1 KO experiments and initial analyses. P.J. and S.C.-J. created and validated the NGLY1 KO cell lines and established the functional reporter in K562 to test NGLY1 functionality. A.-M.M. performed the BRD4 functional experiments. P.G., T.B., M.F.S., M.B., L.M.S., W.H. and G.D. supervised the work. A.H.S., F.Z., W.H. and G.D. wrote the manuscript with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
G.J., N.Z., D.E., M.F.S., P.G., M.B., G.D. are employees and/or shareholders of Cellzome and GlaxoSmithKline. T.B. was an employee of Horizon Genomics GmbH.
Additional information
Peer review information Nicole Rusk was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6 and Supplementary Tables 1 and 2
Data of Figure 1
Residual RNA and protein levels for 193 KO cell lines.
Data of Figure 2
Residual peptide levels aggregated per exon of four BRD4 KO replicates.
Data of Figure 3
Residual peptide levels aggregated per exon for two DNMT1 KO lines.
Data of Figure 4
Residual mRNA and protein levels, and deglycosylation quantification of two NGLY1-KO clones.
Rights and permissions
About this article
Cite this article
Smits, A.H., Ziebell, F., Joberty, G. et al. Biological plasticity rescues target activity in CRISPR knock outs. Nat Methods 16, 1087–1093 (2019). https://doi.org/10.1038/s41592-019-0614-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-019-0614-5
This article is cited by
-
Escaping from CRISPR–Cas-mediated knockout: the facts, mechanisms, and applications
Cellular & Molecular Biology Letters (2024)
-
HiHo-AID2: boosting homozygous knock-in efficiency enables robust generation of human auxin-inducible degron cells
Genome Biology (2024)
-
Functional analysis of Cdc20 reveals a critical role of CRY box in mitotic checkpoint signaling
Communications Biology (2024)
-
Comprehensive behavioral analyses of mice with a glycine receptor alpha 4 deficiency
Molecular Brain (2023)
-
DNA replication initiation factor RECQ4 possesses a role in antagonizing DNA replication initiation
Nature Communications (2023)