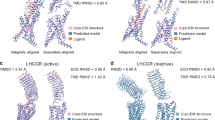
Abstract
G-protein-coupled receptors (GPCRs) transduce physiological and sensory stimuli into appropriate cellular responses and mediate the actions of one-third of drugs. GPCR structural studies have revealed the general bases of receptor activation, signaling, drug action and allosteric modulation, but so far cover only 13% of nonolfactory receptors. We broadly surveyed the receptor modifications/engineering and methods used to produce all available GPCR crystal and cryo-electron microscopy (cryo-EM) structures, and present an interactive resource integrated in GPCRdb (http://www.gpcrdb.org) to assist users in designing constructs and browsing appropriate experimental conditions for structure studies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hauser, A. S., Attwood, M. M., Rask-Andersen, M., Schiöth, H. B. & Gloriam, D. E. Trends in GPCR drug discovery: new agents, targets and indications. Nat. Rev. Drug Discov. 16, 829–842 (2017). Landmark reference for GPCR drugs, targets and indications, describing recent drug discovery successes and new strategies in clinical trials.
Anonymous. Structure statistics. GPCRdb http://gpcrdb.org/structure/statistics (2018).
Rasmussen, S. G. et al. Crystal structure of the β2 adrenergic receptor–Gs protein complex. Nature 477, 549–555 (2011).
Liang, Y. L. et al. Phase-plate cryo-EM structure of a class B GPCR–G-protein complex. Nature 546, 118–123 (2017).
Koehl, A. et al. Structure of the µ-opioid receptor–Gi protein complex. Nature 558, 547–552 (2018).
Kang, Y. et al. Cryo-EM structure of human rhodopsin bound to an inhibitory G protein. Nature 558, 553–558 (2018).
Kang, Y. et al. Crystal structure of rhodopsin bound to arrestin by femtosecond X-ray laser. Nature 523, 561–567 (2015).
Tautermann, C. S. & Gloriam, D. E. Editorial overview: New technologies: GPCR drug design and function—exploiting the current (of) structures. Curr. Opin. Pharmacol. 30, vii–x (2016).
Manglik, A. et al. Structural insights into the dynamic process of β2-adrenergic receptor signaling. Cell 161, 1101–1111 (2015).
Van Eps, N. et al. Conformational equilibria of light-activated rhodopsin in nanodiscs. Proc. Natl Acad. Sci. USA 114, E3268–E3275 (2017).
Staus, D. P. et al. Allosteric nanobodies reveal the dynamic range and diverse mechanisms of G-protein-coupled receptor activation. Nature 535, 448–452 (2016).
Ye, L., Van Eps, N., Zimmer, M., Ernst, O. P. & Prosser, R. S. Activation of the A2A adenosine G-protein-coupled receptor by conformational selection. Nature 533, 265–268 (2016).
Van Eps, N. et al. Gi- and Gs-coupled GPCRs show different modes of G-protein binding. Proc. Natl Acad. Sci. USA 115, 2383–2388 (2018).
Violin, J. D., Crombie, A. L., Soergel, D. G. & Lark, M. W. Biased ligands at G-protein-coupled receptors: promise and progress. Trends Pharmacol. Sci. 35, 308–316 (2014).
Congreve, M., Oswald, C. & Marshall, F. H. Applying structure-based drug design approaches to allosteric modulators of GPCRs. Trends Pharmacol. Sci. 38, 837–847 (2017).
Munk, C., Harpsøe, K., Hauser, A. S., Isberg, V. & Gloriam, D. E. Integrating structural and mutagenesis data to elucidate GPCR ligand binding. Curr. Opin. Pharmacol. 30, 51–58 (2016).
Isberg, V. et al. GPCRdb: an information system for G protein-coupled receptors. Nucleic Acids Res. 44, D356–D364 (2016).
Isberg, V. et al. GPCRDB: an information system for G protein-coupled receptors. Nucleic Acids Res. 42, D422–D425 (2014).
Flock, T. et al. Selectivity determinants of GPCR–G-protein binding. Nature 545, 317–322 (2017).
Pándy-Szekeres, G. et al. GPCRdb in 2018: adding GPCR structure models and ligands. Nucleic Acids Res. 46, D440–D446 (2018).
Munk, C. et al. GPCRdb: the G protein-coupled receptor database—an introduction. Br. J. Pharmacol. 173, 2195–2207 (2016).
Rose, P. W. et al. The RCSB Protein Data Bank: integrative view of protein, gene and 3D structural information. Nucleic Acids Res. 45, D271–D281 (2017).
Velankar, S. et al. SIFTS: Structure Integration with Function, Taxonomy and Sequences resource. Nucleic Acids Res. 41, D483–D489 (2013). Resource for residue-level mapping of UniProt (protein) and PDB (structure) entries that also integrates annotations from many more major databases.
Anonymous. Construct alignments. GPCRdb http://gpcrdb.org/construct/ (2018).
Hutchings, C. J., Koglin, M., Olson, W. C. & Marshall, F. H. Opportunities for therapeutic antibodies directed at G-protein-coupled receptors. Nat. Rev. Drug Discov. 16, 787–810 (2017).
Anonymous. Structure. GPCRdb http://gpcrdb.org/structure (2018).
Renaud, J. P. et al. Cryo-EM in drug discovery: achievements, limitations and prospects. Nat. Rev. Drug Discov. 17, 471–492 (2018). Reviews the recent advances in cryo-EM and provides an outlook of what to expect in the near future.
García-Nafría, J., Lee, Y., Bai, X., Carpenter, B. & Tate, C. G. Cryo-EM structure of the adenosine A2A receptor coupled to an engineered heterotrimeric G protein. eLife 7, e35946 (2018).
Su, X. et al. Structure and assembly mechanism of plant C2S2M2-type PSII-LHCII supercomplex. Science 357, 815–820 (2017).
Liang, Y. L. et al. Phase-plate cryo-EM structure of a biased agonist-bound human GLP-1 receptor–Gs complex. Nature 555, 121–125 (2018).
Khoshouei, M., Radjainia, M., Baumeister, W. & Danev, R. Cryo-EM structure of haemoglobin at 3.2 Å determined with the Volta phase plate. Nat. Commun. 8, 16099 (2017).
Zhang, Y. et al. Cryo-EM structure of the activated GLP-1 receptor in complex with a G protein. Nature 546, 248–253 (2017).
Draper-Joyce, C. J. et al. Structure of the adenosine-bound human adenosine A1 receptor–Gi complex. Nature 558, 559–563 (2018).
Tsai, C. J. et al. Crystal structure of rhodopsin in complex with a mini-Go sheds light on the principles of G protein selectivity. Sci. Adv. 4, eaat7052 (2018).
García-Nafría, J., Nehmé, R., Edwards, P. C. & Tate, C. G. Cryo-EM structure of the serotonin 5-HT1B receptor coupled to heterotrimeric Go. Nature 558, 620–623 (2018).
Chun, E. et al. Fusion partner toolchest for the stabilization and crystallization of G protein-coupled receptors. Structure 20, 967–976 (2012).
Anonymous. Fusion construct analysis. GPCRdb http://gpcrdb.org/construct/analysis#fusions (2018).
Isberg, V. et al. Generic GPCR residue numbers—aligning topology maps while minding the gaps. Trends Pharmacol. Sci. 36, 22–31 (2015). Generic numbering of receptor residues crucial for all GPCR structure–function studies.
Anonymous. Mutation construct analysis. GPCRdb http://gpcrdb.org/construct/analysis#mutations (2018).
Pace, C. N. & Scholtz, J. M. A helix propensity scale based on experimental studies of peptides and proteins. Biophys. J. 75, 422–427 (1998).
Jazayeri, A. et al. Crystal structure of the GLP-1 receptor bound to a peptide agonist. Nature 546, 254–258 (2017).
Venkatakrishnan, A. J. et al. Molecular signatures of G-protein-coupled receptors. Nature 494, 185–194 (2013). Pioneering GPCR structure analysis uncovering common contact networks stabilizing the receptor fold, and characteristic features of ligand binding and receptor activation.
Anonymous. Stabilising Mutation Analyser. GPCRdb http://gpcrdb.org/construct/stabilisation (2018).
Roth, C. B., Hanson, M. A. & Stevens, R. C. Stabilization of the human β2-adrenergic receptor TM4-TM3-TM5 helix interface by mutagenesis of Glu122(3.41), a critical residue in GPCR structure. J. Mol. Biol. 376, 1305–1319 (2008).
White, K. L. et al. Structural connection between activation microswitch and allosteric sodium site in GPCR signaling. Structure 26, 259–269 (2018).
Nygaard, R., Frimurer, T. M., Holst, B., Rosenkilde, M. M. & Schwartz, T. W. Ligand binding and micro-switches in 7TM receptor structures. Trends Pharmacol. Sci. 30, 249–259 (2009).
Katritch, V., Cherezov, V. & Stevens, R. C. Structure-function of the G protein-coupled receptor superfamily. Annu. Rev. Pharmacol. Toxicol. 53, 531–556 (2013).
Carpenter, B. & Tate, C. G. Active state structures of G protein-coupled receptors highlight the similarities and differences in the G protein and arrestin coupling interfaces. Curr. Opin. Struct. Biol. 45, 124–132 (2017).
Anonymous. Truncation analysis. GPCRdb http://gpcrdb.org/construct/analysis#truncations (2018).
Cilia, E., Pancsa, R., Tompa, P., Lenaerts, T. & Vranken, W. F. The DynaMine webserver: predicting protein dynamics from sequence. Nucleic Acids Res. 42, W264–W270 (2014).
Petersen, T. N., Brunak, S., von Heijne, G. & Nielsen, H. SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat. Methods 8, 785–786 (2011).
Anonymous. Mutation rules. GPCRdb http://files.gpcrdb.org/mutation_rules.html (2018).
Anonymous. Local installation. GPCRdb http://docs.gpcrdb.org/local_installation.html (2018).
Peng, Y. et al. 5-HT2C receptor structures reveal the structural basis of GPCR polypharmacology. Cell 172, 719–730 (2018).
Magnani, F., Shibata, Y., Serrano-Vega, M. J. & Tate, C. G. Co-evolving stability and conformational homogeneity of the human adenosine A2a receptor. Proc. Natl Acad. Sci. USA 105, 10744–10749 (2008).
Anonymous. Experiment Browser. GPCRdb http://gpcrdb.org/construct/experiments (2018).
Zhang, X., Stevens, R. C. & Xu, F. The importance of ligands for G protein-coupled receptor stability. Trends Biochem. Sci. 40, 79–87 (2015).
Milić, D. & Veprintsev, D. B. Large-scale production and protein engineering of G protein-coupled receptors for structural studies. Front. Pharmacol. 6, 66 (2015).
Lv, X. et al. In vitro expression and analysis of the 826 human G protein-coupled receptors. Protein Cell 7, 325–337 (2016).
Luo, P. & Baldwin, R. L. Origin of the different strengths of the (i,i+4) and (i,i+3) leucine pair interactions in helices. Biophys. Chem. 96, 103–108 (2002).
Anonymous. Mutations. GPCRdb http://gpcrdb.org/construct/mutations (2018).
Acknowledgements
We acknowledge A. Tsolakou, D. Milic and K.S. Harpsøe for help with data annotation; I. Carson for development of the preliminary version of the Stabilising Mutation Analyser; and C.-J. Tsai for input on the description of cryo-EM construct engineering and experiments. This work was supported in part by the ERC (Starting Grant 639125 to D.E.G.), the Lundbeck Foundation (grants R163-2013-16327 and R218-2016-1266 to D.E.G.), the Swiss National Science Foundation (grant CRSII2_160805 to X.D.), the European Commisions Seventh Framework Program (FP7/2007-2013; grant 290605 (COFUND: PSI-FELLOW) to E.M.) and the COST Action CM1207 (‘GLISTEN’).
Author information
Authors and Affiliations
Contributions
C.M., D.E.G., J.M.B. and T.F. made the construct analyses and figures; C.M. and V.I. developed the online resources; E.M. and C.M. annotated and analyzed published experimental data; L.F.N. conducted the mutagenesis experiments; M.A.H. and R.C.S. provided critical input on the project, manuscript writing and data analysis; X.D. and D.E.G. drafted the paper; all authors commented on the drafted manuscript; D.E.G. and X.D. designed the project; and D.E.G. managed the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Text and Figures
Supplementary Note 1 and Supplementary Tables 1–5
Rights and permissions
About this article
Cite this article
Munk, C., Mutt, E., Isberg, V. et al. An online resource for GPCR structure determination and analysis. Nat Methods 16, 151–162 (2019). https://doi.org/10.1038/s41592-018-0302-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-018-0302-x
This article is cited by
-
SpaceGrow: efficient shape-based virtual screening of billion-sized combinatorial fragment spaces
Journal of Computer-Aided Molecular Design (2024)
-
Modeling the expansion of virtual screening libraries
Nature Chemical Biology (2023)
-
AlphaFold2 versus experimental structures: evaluation on G protein-coupled receptors
Acta Pharmacologica Sinica (2023)
-
Structure-based drug discovery of a corticotropin-releasing hormone receptor 1 antagonist using an X-ray free-electron laser
Experimental & Molecular Medicine (2023)
-
The dual roles of autophagy and the GPCRs-mediating autophagy signaling pathway after cerebral ischemic stroke
Molecular Brain (2022)