Abstract
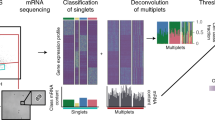
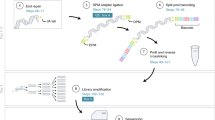
A cell’s function is influenced by the environment, or niche, in which it resides. Studies of niches usually require assumptions about the cell types present, which impedes the discovery of new cell types or interactions. Here we describe ProximID, an approach for building a cellular network based on physical cell interaction and single-cell mRNA sequencing, and show that it can be used to discover new preferential cellular interactions without prior knowledge of component cell types. ProximID found specific interactions between megakaryocytes and mature neutrophils and between plasma cells and myeloblasts and/or promyelocytes (precursors of neutrophils) in mouse bone marrow, and it identified a Tac1+ enteroendocrine cell–Lgr5+ stem cell interaction in small intestine crypts. This strategy can be used to discover new niches or preferential interactions in a variety of organs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Strogatz, S. H. Exploring complex networks. Nature 410, 268–276 (2001).
Albert, R., & Barabasi, A. L. Statistical mechanics of complex networks. Rev. Mod. Phys. 74, 47–97 (2002).
Newman, M. E. J. Networks: An Introduction (Oxford Univ. Press, New York, NY, 2010).
Raaijmakers, M. H. et al. Bone progenitor dysfunction induces myelodysplasia and secondary leukaemia. Nature 464, 852–857 (2010).
Scadden, D. T. Nice neighborhood: emerging concepts of the stem cell niche. Cell 157, 41–50 (2014).
Chu, V. T. et al. Eosinophils are required for the maintenance of plasma cells in the bone marrow. Nat. Immunol. 12, 151–159 (2011).
Chow, A. CD169+ macrophages provide a niche promoting erythropoiesis under homeostasis and stress. Nat. Med. 19, 429–436 (2013).
Mendelson, A., & Frenette, P. S. Hematopoietic stem cell niche maintenance during homeostasis and regeneration. Nat. Med. 20, 833–846 (2014).
Sato, T. et al. Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 469, 415–418 (2011).
Becker, A. et al. Extracellular vesicles in cancer: cell-to-cell mediators of metastasis. Cancer Cell 30, 836–848 (2016).
Chen, K. H., Boettiger, A. N., Moffitt, J. R., Wang, S., & Zhuang, X. Spatially resolved, highly multiplexed RNA profiling in single cells. Science 348, aaa6090 (2015).
Ke, R. et al. In situ sequencing for RNA analysis in preserved tissue and cells. Nat. Methods 10, 857–860 (2013).
Lee, J. H. et al. Highly multiplexed subcellular RNA sequencing in situ. Science 343, 1360–1363 (2014).
Lovatt, D. et al. Transcriptome in vivo analysis (TIVA) of spatially defined single cells in live tissue. Nat. Methods 11, 190–196 (2014).
Shah, S., Lubeck, E., Zhou, W.., & Cai, L. In situ transcription profiling of single cells reveals spatial organization of cells in the mouse hippocampus. Neuron 92, 342–357 (2016).
Junker, J. P. et al. Genome-wide RNA tomography in the zebrafish embryo. Cell 159, 662–675 (2014).
Achim, K. et al. High-throughput spatial mapping of single-cell RNA-seq data to tissue of origin. Nat. Biotechnol. 33, 503–509 (2015).
Satija, R., Farrell, J. A., Gennert, D., Schier, A. F., & Regev, A. Spatial reconstruction of single-cell gene expression data. Nat. Biotechnol. 33, 495–502 (2015).
Ståhl, P. L. et al. Visualization and analysis of gene expression in tissue sections by spatial transcriptomics. Science 353, 78–82 (2016).
Grün, D. et al. Single-cell messenger RNA sequencing reveals rare intestinal cell types. Nature 525, 251–255 (2015).
Jaitin, D. A. et al. Massively parallel single-cell RNA-seq for marker-free decomposition of tissues into cell types. Science 343, 776–779 (2014).
Patel, A. P. et al. Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science 344, 1396–1401 (2014).
Treutlein, B. et al. Reconstructing lineage hierarchies of the distal lung epithelium using single-cell RNA-seq. Nature 509, 371–375 (2014).
Zeisel, A. et al. Cell types in the mouse cortex and hippocampus revealed by single-cell RNA-seq. Science 347, 1138–1142 (2015).
Grün, D. et al. De novo prediction of stem cell identity using single-cell transcriptome data. Cell Stem Cell 19, 266–277 (2016).
Borregaard, N. & Cowland, J. B. Granules of the human neutrophilic polymorphonuclear leukocyte. Blood 89, 3503–3521 (1997).
Fouret, P. et al. Expression of the neutrophil elastase gene during human bone marrow cell differentiation. J. Exp. Med. 169, 833–845 (1989).
Pham, C. T. et al. Neutrophil serine proteases: specific regulators of inflammation. Nat. Rev. Immunol. 6, 541–550 (2006).
Chasis, J. A. & Mohandas, N. Erythroblastic islands: niches for erythropoiesis. Blood 112, 470–478 (2008).
Raj, A., van den Bogaard, P., Rifkin, S. A., van Oudenaarden, A. & Tyagi, S. Imaging individual mRNA molecules using multiple singly labeled probes. Nat. Methods 5, 877–879 (2008).
Janoff, A. Elastase in tissue injury. Annu. Rev. Med. 36, 207–216 (1985).
Klimenkova, O. et al. A lack of secretory leukocyte protease inhibitor (SLPI) causes defects in granulocytic differentiation. Blood 123, 1239–1249 (2014).
Herman, J. S., & Sagar & Grün, D. FateID infers cell fate bias in multipotent progenitors from single-cell RNA-seq data. Nat. Methods 15, 379–386 (2018).
Bruns, I. et al. Megakaryocytes regulate hematopoietic stem cell quiescence through CXCL4 secretion. Nat. Med. 20, 1315–1320 (2014).
Centurione, L. et al. Increased and pathologic emperipolesis of neutrophils within megakaryocytes associated with marrow fibrosis in GATA-1(low) mice. Blood 104, 3573–3580 (2004).
Aiken, K. D., & Roth, K. A. Temporal differentiation and migration of substance P, serotonin, and secretin immunoreactive enteroendocrine cells in the mouse proximal small intestine. Dev. Dyn. 194, 303–310 (1992).
Tan, C. W., Hirokawa, Y., Gardiner, B. S., Smith, D. W., & Burgess, A. W. Colon cryptogenesis: asymmetric budding. PLoS One 8, e78519 (2013).
Muraro, M. J. et al. A single-cell transcriptome atlas of the human pancreas. Cell Syst 3, 385–394 (2016).
Lyubimova, A. et al. Single-molecule mRNA detection and counting in mammalian tissue. Nat. Protoc. 8, 1743–1758 (2013).
Preibisch, S., Saalfeld, S., & Tomancak, P. Globally optimal stitching of tiled 3D microscopic image acquisitions. Bioinformatics 25, 1463–1465 (2009).
Acknowledgements
We thank J. Korving for help with the microdissection microscope; the animal facility; A. de Graaf and the microscope facility; the sequencing facility; K. Wiebrands for suggesting a name for the method and assistance in intestinal crypt dissociation; N. Battich and B. De Barbanson for help with machine learning; L. Kester for help with the Monte Carlo simulations; and R. van der Linden for help with the sorting experiments. This work was supported by the European Research Council (advanced grant ERC-AdG 742225-IntScOmics to A.v.O.) and a Nederlandse Organisatie voor Wetenschappelijk Onderzoek (NWO) OPEN award (NWO-ALWOP189 to A.v.O.). This work is part of the Oncode Institute, which is partly financed by the Dutch Cancer Society. In addition, we thank the Hubrecht Sorting Facility and the Utrecht Sequencing Facility, subsidized by the University Medical Center Utrecht, Hubrecht Institute, and Utrecht University.
Author information
Authors and Affiliations
Contributions
J.-C.B. and A.v.O. conceived the project. J.-C.B. performed experiments. J.V. performed single-cell RNA preparation for sorted BM and FL cells. M.J.M. performed single-cell sequencing of intestinal crypt single cells. A.L. prepared intestinal crypt cells. J.-C.B. and D.G. analyzed the data. J.-C.B. wrote the manuscript. A.v.O. guided experiments and data analysis and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Reproducibility and mixing within sub-dissected units.
(a,b) t-SNE map of transcriptome similarities with experimental replicates (a), and sub-dissected units (b) represented as color code. (c) Cumulative percentage of the fraction of different clusters present in the different units.
Supplementary Figure 2 Identification of the main cell types present in the BM.
(a-i) t-SNE map of transcriptome similarities with color-coded representation of transcript count for the sum of ribosomal genes (a), sum of S100a8 and S100a9 (b), Beta-s (c), sum of Ear1 and Ear2 (d), Ppbp (e), Igj (f), Hmox1 (g), sum of C1qa and C1qb (h), and Mcpt8 (i).
Supplementary Figure 3 Description of the neutrophil differentiation path at the single-cell level.
(a-f) t-SNE map of transcriptome similarities with color-coded representation of transcript count for Elane (a), Ltf (b), Ngp (c), Fpr1 (d), Slc11a1 (e), and Retnlg (f).
Supplementary Figure 4 Comparison of the frequency of picked versus sorted BM cells.
(a-c) t-SNE map of transcriptome similarities. (a) Colors represent the different RaceID2 clusters. The putative cell types for the different clusters are indicated. (b) Color representation of the origins of the cells. (c) Color indication of a significantly (p < 0.00001) higher frequency of picked cells (red) or sorted cells (blue). Non-significant differences are represented in grey.
Supplementary Figure 5 Filtering threshold value influences the network output.
(a) t-SNE map of transcriptome similarities with color-coded representation of raw transcript counts (UMIs) per cells. (b) Bar plot representing the average total number of transcripts per cell according to their RaceID2 clusters. The blue line represents the mean transcript count across all filtered cells, and the red line represents the filter threshold value. (c) Line chart showing the frequency of detected interactions: macrophages – erythroblasts (green), megakaryocytes – neutrophils (cyan), plasma cells – myeloblasts (magenta), and all 3 previously mentioned interactions in a single analysis (red), for 100 different downsampling, in function of the filtering threshold based on total transcript count per cells. (d) Line chart showing the frequency of detection of the megakaryocytes – neutrophils, plasma cells – promyelocytes/myeloblasts and macrophages – erythroblasts interaction as function of the number of sampled structures. The line and the shaded area are the running average and standard deviation respectively of 10 consecutive data points. Each data point is the frequency based on 20 different samplings.
Supplementary Figure 6 Network of physical interaction in the fetal mouse liver.
(a-f) t-SNE map of transcriptome similarities with color-coded representation of RaceID2 clusters (a), embryonic day (b), and expression of Beta-s (c) and Hmox1 (d). (e) Representation of enriched (red) and depleted (blue) interactions per clusters. Nodes represent cluster centers. Edges are interactions inter-clusters, and filled circles are interactions intra-clusters. (f) t-SNE map of transcriptome similarities with magenta edges representing all cell-cell interactions stemming from cluster 6 (macrophages, in cyan).
Supplementary Figure 7 Frequency of interstrain versus intrastrain interactions.
(a,b) t-SNE map of transcriptome similarities, with node colors representing the RaceID2 clusters (a) or strain of origin (b) (n=2 independent experiments). The edges connecting cells represent the physical interaction in between cells (a), with color-coding of inter- versus intra-strain interaction (b). 62% of the interactions are intra-strain. The putative cell types for the different clusters are indicated in (a). (c) Probability distribution of the frequency of inter-strain interactions after label permutation simulations (grey area, and blue bar representing the mean of the distribution, 10,000 simulations), compared to the actual number (red bar).
Supplementary Figure 8 Examination of unprocessed single-molecule FISH images.
(a,b) Raw single-plane images of BM cryosections corresponding to Figure 3a and 4a, stained with smFISH probes against Elane (green) and Igj (red) mRNAs, conjugated with Cy5 and TAMRA respectively (a) (n=1), or biotin anti-CD41 antibodies and alexa 488-streptavidin conjugates (grey), and smFISH probes against Retnlg (green) and Ngp (red) mRNAs, conjugated with Cy5 and TAMRA respectively (b) (n=1). Nucleuses were stained with DAPI (blue). The asterisk (*) denotes autofluorescent cells not used in the analysis, and the pound sign (#) denotes red blood cells. Scale bar corresponds to 10 μm.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8
Supplementary Software
Code and demo data for ProximID, ProximID with different cell-type-recognition methods, and image analysis of interacting cells
Supplementary Video 1
Doublet microdissection. The video shows a representative example of the separation of two interacting cells from the bone marrow using two glass needles, and their subsequent mouth-pipetting
Rights and permissions
About this article
Cite this article
Boisset, JC., Vivié, J., Grün, D. et al. Mapping the physical network of cellular interactions. Nat Methods 15, 547–553 (2018). https://doi.org/10.1038/s41592-018-0009-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-018-0009-z
This article is cited by
-
The diversification of methods for studying cell–cell interactions and communication
Nature Reviews Genetics (2024)
-
Advances in spatial transcriptomics and related data analysis strategies
Journal of Translational Medicine (2023)
-
Spatial transcriptomics: recent developments and insights in respiratory research
Military Medical Research (2023)
-
Sctensor detects many-to-many cell–cell interactions from single cell RNA-sequencing data
BMC Bioinformatics (2023)
-
Single-cell RNA sequencing to track novel perspectives in HSC heterogeneity
Stem Cell Research & Therapy (2022)