Abstract
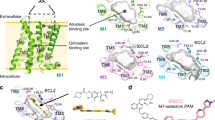
Human muscarinic receptor M2 is one of the five subtypes of muscarinic receptors belonging to the family of G-protein-coupled receptors. Muscarinic receptors are targets for multiple neurodegenerative diseases. The challenge has been designing subtype-selective ligands against one of the five muscarinic receptors. We report high-resolution structures of a thermostabilized mutant M2 receptor bound to a subtype-selective antagonist AF-DX 384 and a nonselective antagonist NMS. The thermostabilizing mutation S110R in M2 was predicted using a theoretical strategy previously developed in our group. Comparison of the crystal structures and pharmacological properties of the M2 receptor shows that the Arg in the S110R mutant mimics the stabilizing role of the sodium cation, which is known to allosterically stabilize inactive state(s) of class A GPCRs. Molecular dynamics simulations reveal that tightening of the ligand–residue contacts in M2 receptors compared to M3 receptors leads to subtype selectivity of AF-DX 384.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The atomic coordinates and structure factors for the reported crystal structures have been deposited in the Protein Data Bank under accession codes 5ZK8 (M2-BRIL–NMS), 5ZKC (S110R-BRIL–NMS), 5ZKB (S110R-BRIL–AF-DX 384), 5ZK3 (S110R-BRIL–QNB), 5YC8 (S110R-BRIL–NMS:Hg). Raw diffraction images have been also deposited in Zenodo data repository (https://doi.org/10.5281/zenodo.1172266 for S110R-BRIL–NMS:Hg, https://doi.org/10.5281/zenodo.1094808 for others).
References
Fredriksson, R., Lagerström, M. C., Lundin, L.-G. & Schiöth, H. B. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol. Pharmacol. 63, 1256–1272 (2003).
Wess, J., Eglen, R. M. & Gautam, D. Muscarinic acetylcholine receptors: mutant mice provide new insights for drug development. Nat. Rev. Drug Discov. 6, 721–733 (2007).
Kruse, A. C., Hu, J., Kobilka, B. K. & Wess, J. Muscarinic acetylcholine receptor X-ray structures: potential implications for drug development. Curr. Opin. Pharmacol. 16, 24–30 (2014).
Caulfield, M. P. & Birdsall, N. J. M. International Union of Pharmacology. XVII. Classification of muscarinic acetylcholine receptors. Pharmacol. Rev. 50, 279–290 (1998).
Haga, K. et al. Structure of the human M2 muscarinic acetylcholine receptor bound to an antagonist. Nature 482, 547–551 (2012).
Kruse, A. C. et al. Structure and dynamics of the M3 muscarinic acetylcholine receptor. Nature 482, 552–556 (2012).
Kruse, A. C. et al. Activation and allosteric modulation of a muscarinic acetylcholine receptor. Nature 504, 101–106 (2013).
Thal, D. M. et al. Crystal structures of the M1 and M4 muscarinic acetylcholine receptors. Nature 531, 335–340 (2016).
Thorsen, T. S. S., Matt, R., Weis, W. I. I. & Kobilka, B. K. K. Modified T4 lysozyme fusion proteins facilitate G protein-coupled receptor Crystallogenesis. Structure 22, 1657–1664 (2014).
Yasuda, S. et al. Hot-spot residues to be mutated common in G protein-coupled receptors of class A: identification of thermostabilizing mutations followed by determination of three-dimensional structures for two example receptors. J. Phys. Chem. B 121, 6341–6350 (2017).
Juan, A. & Ballesteros, H. W. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods Neurosci. 25, 366–428 (1995).
Katritch, V. et al. Allosteric sodium in class A GPCR signaling. Trends Biochem. Sci. 39, 233–244 (2014).
White, K. L. et al. Structural connection between activation microswitch and allosteric sodium site in GPCR signaling. Structure 26, 259–269.e5 (2018).
de Ligt, R. A., Kourounakis, A. P. & IJzerman, A. P. Inverse agonism at G protein-coupled receptors: (patho)physiological relevance and implications for drug discovery. Br. J. Pharmacol. 130, 1–12 (2000).
Miller, J. H., Gibson, V. A. & McKinney, M. Binding of [3H]AF-DX 384 to cloned and native muscarinic receptors. J. Pharmacol. Exp. Ther. 259, 601–607 (1991).
Tränkle, C., Andresen, I., Lambrecht, G. & Mohr, K. M2 receptor binding of the selective antagonist AF-DX 384: possible involvement of the common allosteric site. Mol. Pharmacol. 53, 304–312 (1998).
Zavitsanou, K., Katsifis, A., Yu, Y. & Huang, X. F. M2/M4 muscarinic receptor binding in the anterior cingulate cortex in schizophrenia and mood disorders. Brain Res. Bull. 65, 397–403 (2005).
Teaktong, T. et al. Muscarinic M2 and M4 receptors in anterior cingulate cortex: relation to neuropsychiatric symptoms in dementia with Lewy bodies. Behav. Brain Res. 161, 299–305 (2005).
Kitaichi, K., Day, J. C. & Quirion, R. A novel muscarinic M4 receptor antagonist provides further evidence of an autoreceptor role for the muscarinic M2 receptor sub-type. Eur. J. Pharmacol. 383, 53–56 (1999).
Piggott, M. et al. Comparative distribution of binding of the muscarinic receptor ligands pirenzepine, AF-DX 384, (R,R)-I-QNB and (R,S)-I-QNB to human brain. J. Chem. Neuroanat. 24, 211–223 (2002).
Gibbons, A. S., Scarr, E., McLean, C., Sundram, S. & Dean, B. Decreased muscarinic receptor binding in the frontal cortex of bipolar disorder and major depressive disorder subjects. J. Affect. Disord. 116, 184–191 (2009).
Hattori, M., Hibbs, R. E. & Gouaux, E. A fluorescence-detection size-exclusion chromatography-based thermostability assay for membrane protein precrystallization screening. Structure 20, 1293–1299 (2012).
Gregory, K. J., Sexton, P. M. & Christopoulos, A. Allosteric modulation of muscarinic acetylcholine receptors. Curr. Neuropharmacol. 5, 157–167 (2007).
Vickery, O. N. et al. Intracellular transfer of Na+ in an active-state G-protein-coupled receptor. Structure 26, 171–180.e2 (2018).
Heitz, F. et al. Site-directed mutagenesis of the putative human muscarinic M2 receptor binding site. Eur. J. Pharmacol. 380, 183–195 (1999).
Jakubı, J. & El-fakahany, E. E. Differences in kinetics of xanomeline binding and selectivity of activation of G proteins at M1 and M2 muscarinic acetylcholine receptors. Mol Pharmacol. 70, 656–666 (2006).
Bhattacharya, S., Lee, S., Grisshammer, R., Tate, C. G. & Vaidehi, N. Rapid computational prediction of thermostabilizing mutations for G protein-coupled receptors. J. Chem. Theory Comput. 10, 5149–5160 (2014).
Vaidehi, N., Grisshammer, R. & Tate, C. G. How can mutations thermostabilize G-protein-coupled receptors? Trends Pharmacol. Sci. 37, 37–46 (2016).
Hess, B., Bekker, H., Berendsen, H. J. C. & Fraaije, J. G. E. M. LINCS: A linear constraint solver for molecular simulations. J. Comput. Chem. 18, 1463–1472 (1997).
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: An N∙log(N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089 (1993).
Liu, W. et al. Structural basis for allosteric regulation of GPCRs by sodium ions. Science 337, 232–236 (2012).
Eddy, M. T. et al. Allosteric coupling of drug binding and intracellular signaling in the A2A adenosine receptor. Cell 172, 68–80.e12 (2018).
Dror, R. O. et al. Pathway and mechanism of drug binding to G-protein-coupled receptors. Proc. Natl. Acad. Sci. USA 108, 13118–13123 (2011).
Suno, R. et al. Crystal structures of human orexin 2 receptor bound to the subtype-selective antagonist EMPA. Structure 26, 7–19.e5 (2018).
Caffrey, M. & Porter, C. Crystallizing membrane proteins for structure determination using lipidic mesophases. Nat. Protoc. 4, 706–731 (2009).
Ueno, G. et al. Remote access and automation of SPring-8 MX beamlines. AIP Conf. Proc. 1741, 050021 (2016).
Hirata, K., Foadi, J., Evans G. & Hasegawa, K. Z. O. Structural biology with microfocus beamlines. Adv. Methods Struct. Biol. Springer Protoc. 241–273 (2016).
Yamashita, K., Hirata, K. & Yamamoto, M. KAMO: towards automated data processing for microcrystals. Acta Crystallogr. D Struct. Biol. 74, 441–449 (2018).
Kabsch, W. Xds. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Foadi, J. et al. Clustering procedures for the optimal selection of data sets from multiple crystals in macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 69, 1617–1632 (2013).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
OS. S. Grade, version 1.102. http://www.globalphasing.com (2011).
Schüttelkopf, A. W. & van Aalten, D. M. F. PRODRG: a tool for high-throughput crystallography of protein-ligand complexes. Acta Crystallogr. D Biol. Crystallogr. 60, 1355–1363 (2004).
Sheldrick, G. M. Experimental phasing with SHELXC/D/E: combining chain tracing with density modification. Acta Crystallogr. D Biol. Crystallogr. 66, 479–485 (2010).
Hess, B., Kutzner, C., van der Spoel, D. & Lindahl, E. GROMACS 4: algorithms for highly efficient, load balanced, and scalable molecular simulation. J. Chem. Theory Comput. 4, 435–447 (2008).
Oostenbrink, C., Villa, A., Mark, A. E. & van Gunsteren, W. F. A biomolecular force field based on the free enthalpy of hydration and solvation: the GROMOS force-field parameter sets 53A5 and 53A6. J. Comput. Chem. 25, 1656–1676 (2004).
Schrödinger, L. Maestro, version 9.0 (New York, 2009).
Berendsen, H. J. C., Postma, J. P. M., Van Gunsteren, W. F. & Hermans, J. Interaction models for water in relation to protein hydration. in Intermolecular Forces: The Jerusalem Symposia on Quantum Chemistry and Biochemistry, Vol. 14 (ed. Pullman, B.) 331–342 (Springer, Dordrecht, the Netherlands, 1981).
Miyamoto, S. & Kollman, P. A. Settle: an analytical version of the SHAKE and RATTLE algorithm for rigid water models. J. Comput. Chem. 13, 952–962 (1992).
Bennett, C. H. Efficient estimation of free energy differences from Monte Carlo data. J. Comput. Phys. 22, 245–268 (1976).
Shirts, M. R. & Pande, V. S. Comparison of efficiency and bias of free energies computed by exponential averaging, the Bennett acceptance ratio, and thermodynamic integration. J. Chem. Phys. 122, 144107 (2005).
Acknowledgements
We acknowledge support from the Research Acceleration Program of the JST (S.I.); the Platform Project for Supporting Drug Discovery and Life Science Research (Platform for Drug Discovery, Informatics, and Structural Life Science) from the Japanese Ministry of Education, Culture, Sports, Science and Technology (MEXT); the JSPS-NSF International Collaboration in Chemistry (ICC) (T.K. and B.K.); the Takeda Science Foundation (T.K. (Kyoto University) and R.S.); the Japan Agency for Medical Research and Development (AMED) (T.K. and T.M.); JSPS KAKENHI (Grant No. 15K08268 to R.S.; 15H06862 to K.Y.); and the ImPACT Program of the Council for Science, Technology and Innovation (Cabinet Office, Government of Japan) (T.M.). The MD simulations performed by S.L. and N.V. were supported by NIH R01-GM097261 (N.V.). We thank the beamline staff at SPring-8 for data collection and processing, and T. Sumiyoshi for providing the information about the ligands. The X-ray crystallography data collection was performed at SPring-8 (Proposal No. 2013A1379, 2013B1092, 2013B1184, 2014A1301, 2014B1273, 2014B1355, 2015A1044, 2015A1080, 2015B2044, and 2015B2080). DNA sequencing analysis was performed at the Medical Research Support Center, Graduate School of Medicine, Kyoto University. T. Nakagita made a diagram of the structure formula of ligands.
Author information
Authors and Affiliations
Contributions
R.S. and T.K. designed the project. S.Y., T.M., and M.K. discovered the thermostabilizing mutant using the theoretical strategy. R.S., M.S.T., and H.T. carried out expression and purification of the receptor. H.T and S.M. carried out the binding assay. R.S. and M.S.T. crystallized the receptor. R.S., K.Y., and K.H. collected and processed the diffraction data. M.Y. supervised the data collection and data processing. R.S. and S.H solved and refined the structures. S.L. carried out MD simulations. S.L. and N.V. performed analysis and N.V. wrote the manuscript associated with the MD simulations. T.K, B.K.K., and S.I. supervised the overall project. R.S., S.M., N.V. and T.K. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–9, Supplementary Tables 1–5
Rights and permissions
About this article
Cite this article
Suno, R., Lee, S., Maeda, S. et al. Structural insights into the subtype-selective antagonist binding to the M2 muscarinic receptor. Nat Chem Biol 14, 1150–1158 (2018). https://doi.org/10.1038/s41589-018-0152-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-018-0152-y
This article is cited by
-
Xanomeline displays concomitant orthosteric and allosteric binding modes at the M4 mAChR
Nature Communications (2023)
-
Molecular basis for selective activation of DREADD-based chemogenetics
Nature (2022)
-
Activation and allosteric regulation of the orphan GPR88-Gi1 signaling complex
Nature Communications (2022)
-
Characteristic structural difference between inactive and active states of orexin 2 receptor determined using molecular dynamics simulations
Biophysical Reviews (2022)
-
Vibrational spectroscopy analysis of ligand efficacy in human M2 muscarinic acetylcholine receptor (M2R)
Communications Biology (2021)