Abstract
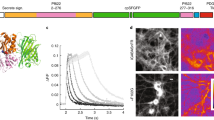
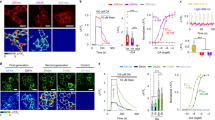
Fluorescent sensors are an essential part of the experimental toolbox of the life sciences, where they are used ubiquitously to visualize intra- and extracellular signaling. In the brain, optical neurotransmitter sensors can shed light on temporal and spatial aspects of signal transmission by directly observing, for instance, neurotransmitter release and spread. Here we report the development and application of the first optical sensor for the amino acid glycine, which is both an inhibitory neurotransmitter and a co-agonist of the N-methyl-d-aspartate receptors (NMDARs) involved in synaptic plasticity. Computational design of a glycine-specific binding protein allowed us to produce the optical glycine FRET sensor (GlyFS), which can be used with single and two-photon excitation fluorescence microscopy. We took advantage of this newly developed sensor to test predictions about the uneven spatial distribution of glycine in extracellular space and to demonstrate that extracellular glycine levels are controlled by plasticity-inducing stimuli.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Okumoto, S. et al. Detection of glutamate release from neurons by genetically encoded surface-displayed FRET nanosensors. Proc. Natl. Acad. Sci. USA 102, 8740–8745 (2005).
Namiki, S., Sakamoto, H., Iinuma, S., Iino, M. & Hirose, K. Optical glutamate sensor for spatiotemporal analysis of synaptic transmission. Eur. J. Neurosci. 25, 2249–2259 (2007).
Marvin, J. S. et al. An optimized fluorescent probe for visualizing glutamate neurotransmission. Nat. Methods 10, 162–170 (2013).
Masharina, A., Reymond, L., Maurel, D., Umezawa, K. & Johnsson, K. A fluorescent sensor for GABA and synthetic GABA(B) receptor ligands. J. Am. Chem. Soc. 134, 19026–19034 (2012).
Betz, H. Glycine receptors: heterogeneous and widespread in the mammalian brain. Trends Neurosci. 14, 458–461 (1991).
Xu, T.-L. & Gong, N. Glycine and glycine receptor signaling in hippocampal neurons: diversity, function and regulation. Prog. Neurobiol. 91, 349–361 (2010).
Johnson, J. W. & Ascher, P. Glycine potentiates the NMDA response in cultured mouse brain neurons. Nature 325, 529–531 (1987).
Schell, M. J. The N-methyl d-aspartate receptor glycine site and d-serine metabolism: an evolutionary perspective. Philos. Trans. R. Soc. Lond. B 359, 943–964 (2004).
Citri, A. & Malenka, R. C. Synaptic plasticity: multiple forms, functions, and mechanisms. Neuropsychopharmacology 33, 18–41 (2008).
Nabavi, S. et al. Engineering a memory with LTD and LTP. Nature 511, 348–352 (2014).
Nong, Y. et al. Glycine binding primes NMDA receptor internalization. Nature 422, 302–307 (2003).
Ferreira, J. S. et al. Co-agonists differentially tune GluN2B-NMDA receptor trafficking at hippocampal synapses. Elife 6, e25492 (2017).
Henneberger, C., Papouin, T., Oliet, S. H. R. & Rusakov, D. A. Long-term potentiation depends on release of d-serine from astrocytes. Nature 463, 232–236 (2010).
Papouin, T. et al. Synaptic and extrasynaptic NMDA receptors are gated by different endogenous coagonists. Cell 150, 633–646 (2012).
Le Bail, M. et al. Identity of the NMDA receptor coagonist is synapse specific and developmentally regulated in the hippocampus. Proc. Natl. Acad. Sci. USA 112, E204–E213 (2015).
Chen, R.-Q. et al. Role of glycine receptors in glycine-induced LTD in hippocampal CA1 pyramidal neurons. Neuropsychopharmacology 36, 1948–1958 (2011).
Winkelmann, A. et al. Changes in neural network homeostasis trigger neuropsychiatric symptoms. J. Clin. Invest. 124, 696–711 (2014).
Hashimoto, A., Oka, T. & Nishikawa, T. Extracellular concentration of endogenous free d-serine in the rat brain as revealed by in vivo microdialysis. Neuroscience 66, 635–643 (1995).
Berntsson, R. P.-A., Smits, S. H. J., Schmitt, L., Slotboom, D.-J. & Poolman, B. A structural classification of substrate-binding proteins. FEBS Lett. 584, 2606–2617 (2010).
Planamente, S. et al. A conserved mechanism of GABA binding and antagonism is revealed by structure-function analysis of the periplasmic binding protein Atu2422 in Agrobacterium tumefaciens. J. Biol. Chem. 285, 30294–30303 (2010).
Van Durme, J. et al. A graphical interface for the FoldX forcefield. Bioinformatics 27, 1711–1712 (2011).
Morris, G. M. et al. AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J. Comput. Chem. 30, 2785–2791 (2009).
Bajar, B. T., Wang, E. S., Zhang, S., Lin, M. Z. & Chu, J. A guide to fluorescent protein FRET pairs. Sensors (Basel) 16, 1488 (2016).
Whitfield, J. H. et al. Construction of a robust and sensitive arginine biosensor through ancestral protein reconstruction. Protein Sci. 24, 1412–1422 (2015).
Deuschle, K. et al. Construction and optimization of a family of genetically encoded metabolite sensors by semirational protein engineering. Protein Sci. 14, 2304–2314 (2005).
Fritz, R. D. et al. A versatile toolkit to produce sensitive FRET biosensors to visualize signaling in time and space. Sci. Signal. 6, rs12 (2013).
Chen, X., Zaro, J. L. & Shen, W.-C. Fusion protein linkers: property, design and functionality. Adv. Drug Deliv. Rev. 65, 1357–1369 (2013).
Piston, D. W. & Kremers, G.-J. Fluorescent protein FRET: the good, the bad and the ugly. Trends Biochem. Sci. 32, 407–414 (2007).
Hamberger, A. & Nyström, B. Extra- and intracellular amino acids in the hippocampus during development of hepatic encephalopathy. Neurochem. Res. 9, 1181–1192 (1984).
Clifton, B. E. & Jackson, C. J. Ancestral protein reconstruction yields insights into adaptive evolution of binding specificity in solute-binding proteins. Cell Chem. Biol. 23, 236–245 (2016).
Okubo, Y. et al. Imaging extrasynaptic glutamate dynamics in the brain. Proc. Natl. Acad. Sci. USA 107, 6526–6531 (2010).
Ermolyuk, Y. S. et al. Independent regulation of basal neurotransmitter release efficacy by variable Ca2+influx and bouton size at small central synapses. PLoS Biol. 10, e1001396 (2012).
Maravall, M., Mainen, Z. F., Sabatini, B. L. & Svoboda, K. Estimating intracellular calcium concentrations and buffering without wavelength ratioing. Biophys. J. 78, 2655–2667 (2000).
Roux, M. J. & Supplisson, S. Neuronal and glial glycine transporters have different stoichiometries. Neuron 25, 373–383 (2000).
Tinberg, C. E. & Khare, S. D. Computational Protein Design. 363–373 (Humana Press, New York, NY, 2017).
Tannous, B. A. et al. Metabolic biotinylation of cell surface receptors for in vivo imaging. Nat. Methods 3, 391–396 (2006).
Panatier, A. et al. Glia-derived d-serine controls NMDA receptor activity and synaptic memory. Cell 125, 775–784 (2006).
Horio, M. et al. Levels of d-serine in the brain and peripheral organs of serine racemase (Srr) knock-out mice. Neurochem. Int. 59, 853–859 (2011).
Matsui, T. et al. Functional comparison of d-serine and glycine in rodents: the effect on cloned NMDA receptors and the extracellular concentration. J. Neurochem. 65, 454–458 (1995).
Tønnesen, J., Inavalli, V. V. G. K. & Nägerl, U. V. Super-resolution imaging of the extracellular space in living brain tissue. Cell 172, 1108–1121.e15 (2018).
Rusakov, D. A. & Kullmann, D. M. Extrasynaptic glutamate diffusion in the hippocampus: ultrastructural constraints, uptake, and receptor activation. J. Neurosci. 18, 3158–3170 (1998).
Bethge, P., Chéreau, R., Avignone, E., Marsicano, G. & Nägerl, U. V. Two-photon excitation STED microscopy in two colors in acute brain slices. Biophys. J. 104, 778–785 (2013).
Cubelos, B., Giménez, C. & Zafra, F. Localization of the GLYT1 glycine transporter at glutamatergic synapses in the rat brain. Cereb. Cortex 15, 448–459 (2005).
Bergeron, R., Meyer, T. M., Coyle, J. T. & Greene, R. W. Modulation of N-methyl-d-aspartate receptor function by glycine transport. Proc. Natl Acad. Sci. USA 95, 15730–15734 (1998).
Martina, M. et al. Glycine transporter type 1 blockade changes NMDA receptor-mediated responses and LTP in hippocampal CA1 pyramidal cells by altering extracellular glycine levels. J. Physiol. (Lond.) 557, 489–500 (2004).
Danglot, L., Rostaing, P., Triller, A. & Bessis, A. Morphologically identified glycinergic synapses in the hippocampus. Mol. Cell. Neurosci. 27, 394–403 (2004).
Langer, J. & Rose, C. R. Synaptically induced sodium signals in hippocampal astrocytes in situ. J. Physiol. (Lond.) 587, 5859–5877 (2009).
Chen, P. E. et al. Modulation of glycine potency in rat recombinant NMDA receptors containing chimeric NR2A/2D subunits expressed in Xenopus laevis oocytes. J. Physiol. (Lond.) 586, 227–245 (2008).
Le Meur, K., Galante, M., Angulo, M. C. & Audinat, E. Tonic activation of NMDA receptors by ambient glutamate of non-synaptic origin in the rat hippocampus. J. Physiol. (Lond.) 580, 373–383 (2007).
Zhang, L.-H., Gong, N., Fei, D., Xu, L. & Xu, T.-L. Glycine uptake regulates hippocampal network activity via glycine receptor-mediated tonic inhibition. Neuropsychopharmacology 33, 701–711 (2008).
Woitecki, A. M. H. et al. Identification of synaptotagmin 10 as effector of NPAS4-mediated protection from excitotoxic neurodegeneration. J. Neurosci. 36, 2561–2570 (2016).
Anders, S. et al. Spatial properties of astrocyte gap junction coupling in the rat hippocampus. Philos. Trans. R. Soc. Lond. B 369, 20130600 (2014).
Ortinski, P. I. et al. Selective induction of astrocytic gliosis generates deficits in neuronal inhibition. Nat. Neurosci. 13, 584–591 (2010).
Minge, D. et al. Heparan sulfates support pyramidal cell excitability, synaptic plasticity, and context discrimination. Cereb. Cortex 27, 903–918 (2017).
Acknowledgements
We thank Dr. O'Mara (Australian National University) for helpful discussions. Research was funded by the Human Frontiers Science Program Young Investigator Award (HFSP to H.J., C.H., and C.J.J.; grant number: RGY0084/2012), German Academic Exchange Service (DAAD-Go8) Travel Fellowship (to C.H. and C.J.J.), NRW-Rückkehrerprogramm (to C.H.), the European Union (ITN EU-GliaPhD) and German Research Foundation (DFG, SFB1089 B03, SPP1757 HE6949/1 and HE6949/3, to C.H.).
Author information
Authors and Affiliations
Contributions
W.H.Z., J.A.M., V.V., J.H.W. and C.J.J. designed, produced and analyzed the sensor. M.K.H., J.H.W., A.B.W., W.H.Z., D.M., B.B. and C.H. performed and analyzed all experiments using two-photon excitation and electrophysiology in acute brain slices. M.K.H., J.H.W., I.S.-R., P.E.G., H.J., S.S. and A.B.W. performed studies on GlyFS expressed by cells. C.H., C.J.J. and H.J. designed the study. C.H., C.J.J. and W.H.Z. wrote the initial manuscript, to which all authors subsequently contributed.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–3, Supplementary Figures 1–15
Rights and permissions
About this article
Cite this article
Zhang, W.H., Herde, M.K., Mitchell, J.A. et al. Monitoring hippocampal glycine with the computationally designed optical sensor GlyFS. Nat Chem Biol 14, 861–869 (2018). https://doi.org/10.1038/s41589-018-0108-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41589-018-0108-2
This article is cited by
-
Pushing the frontiers: tools for monitoring neurotransmitters and neuromodulators
Nature Reviews Neuroscience (2022)
-
Genetically encoded tools for measuring and manipulating metabolism
Nature Chemical Biology (2022)
-
An astrocytic signaling loop for frequency-dependent control of dendritic integration and spatial learning
Nature Communications (2022)
-
Semi-Implantable Bioelectronics
Nano-Micro Letters (2022)
-
An l-2-hydroxyglutarate biosensor based on specific transcriptional regulator LhgR
Nature Communications (2021)