Abstract
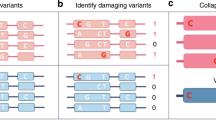
Exome association studies to date have generally been underpowered to systematically evaluate the phenotypic impact of very rare coding variants. We leveraged extensive haplotype sharing between 49,960 exome-sequenced UK Biobank participants and the remainder of the cohort (total n ≈ 500,000) to impute exome-wide variants with accuracy R2 > 0.5 down to minor allele frequency (MAF) ~0.00005. Association and fine-mapping analyses of 54 quantitative traits identified 1,189 significant associations (P < 5 × 10−8) involving 675 distinct rare protein-altering variants (MAF < 0.01) that passed stringent filters for likely causality. Across all traits, 49% of associations (578/1,189) occurred in genes with two or more hits; follow-up analyses of these genes identified allelic series containing up to 45 distinct ‘likely-causal’ variants. Our results demonstrate the utility of within-cohort imputation in population-scale genome-wide association studies, provide a catalog of likely-causal, large-effect coding variant associations and foreshadow the insights that will be revealed as genetic biobank studies continue to grow.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Access to the UKB Resource is available by application (http://www.ukbiobank.ac.uk/). Exome-wide summary association statistics for the 54 quantitative traits we analyzed are available at https://data.broadinstitute.org/lohlab/UKB_exomeWAS/ and data files containing allelic series for all gene–trait associations with multiple likely-causal variants are also available at this website.
Code availability
The following publicly available software packages were used to perform analyses: Eagle2 (v.2.3.5), https://data.broadinstitute.org/alkesgroup/Eagle/; Minimac4 (v.1.0.1), https://genome.sph.umich.edu/wiki/Minimac4; BOLT–LMM (v.2.3.4), https://data.broadinstitute.org/alkesgroup/BOLT-LMM/; FINEMAP (v.1.3.1), http://www.christianbenner.com/; plink (v.1.9 and v.2.0), https://www.cog-genomics.org/plink2/ and tsinfer (v.0.1.4), https://tsinfer.readthedocs.io/en/latest/. Information from the following databases were also used: VEP (v.95 on GRCh37 with GENCODE 19), https://www.ensembl.org/vep; CADD (v.1.5), https://cadd.gs.washington.edu/download; SpliceAI (v.1.2.1) https://github.com/Illumina/SpliceAI; NHGRI–EBI GWAS Catalog (v.1.0), https://www.ebi.ac.uk/gwas/home; TOPMed (v.r2, 97,256 TOPMed samples), https://imputation.biodatacatalyst.nhlbi.nih.gov/#!pages/about; Protein Data Bank, https://www.rcsb.org/; SWISS-MODEL, https://swissmodel.expasy.org/ and PANTHER (v.15.0), http://www.pantherdb.org/. Scripts used to perform the downstream analyses described above are available at https://data.broadinstitute.org/lohlab/UKB_exomeWAS/ (https://doi.org/10.5281/zenodo.4771214).
References
International Multiple Sclerosis Genetics Consortium. Low-frequency and rare-coding variation contributes to multiple sclerosis risk. Cell 175, 1679–1687.e7 (2018).
Marouli, E. et al. Rare and low-frequency coding variants alter human adult height. Nature 542, 186–190 (2017).
Liu, D. J. et al. Exome-wide association study of plasma lipids in >300,000 individuals. Nat. Genet. 49, 1758–1766 (2017).
Liu, C. et al. Meta-analysis identifies common and rare variants influencing blood pressure and overlapping with metabolic trait loci. Nat. Genet. 48, 1162–1170 (2016).
Fu, W. et al. Analysis of 6,515 exomes reveals a recent origin of most human protein-coding variants. Nature 493, 216–220 (2013).
Tennessen, J. A. et al. Evolution and functional impact of rare coding variation from deep sequencing of human exomes. Science 337, 64–69 (2012).
Dewey, F. E. et al. Distribution and clinical impact of functional variants in 50,726 whole-exome sequences from the DiscovEHR study. Science 354, aaf6814 (2016).
Van Hout, C. V. et al. Exome sequencing and characterization of 49,960 individuals in the UK Biobank. Nature 586, 749–756 (2020).
Cirulli, E. T. et al. Genome-wide rare variant analysis for thousands of phenotypes in over 70,000 exomes from two cohorts. Nat. Commun. 11, 542 (2020).
Flannick, J. et al. Exome sequencing of 20,791 cases of type 2 diabetes and 24,440 controls. Nature 570, 71–76 (2019).
Das, S. et al. Next-generation genotype imputation service and methods. Nat. Genet. 48, 1284–1287 (2016).
Bycroft, C. et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 562, 203–209 (2018).
DeBoever, C. et al. Medical relevance of protein-truncating variants across 337,205 individuals in the UK Biobank study. Nat. Commun. 9, 1612 (2018).
Emdin, C. A. et al. Analysis of predicted loss-of-function variants in UK Biobank identifies variants protective for disease. Nat. Commun. 9, 1–8 (2018).
Loh, P.-R., Palamara, P. F. & Price, A. L. Fast and accurate long-range phasing in a UK Biobank cohort. Nat. Genet. 48, 811–816 (2016).
Loh, P.-R. et al. Reference-based phasing using the haplotype reference consortium panel. Nat. Genet. 48, 1443–1448 (2016).
Nait Saada, J. et al. Identity-by-descent detection across 487,409 British samples reveals fine scale population structure and ultra-rare variant associations. Nat. Commun. 11, 6130 (2020).
Loh, P.-R., Genovese, G. & McCarroll, S. A. Monogenic and polygenic inheritance become instruments for clonal selection. Nature 584, 136–141 (2020).
Browning, B. L. & Browning, S. R. Genotype imputation with millions of reference samples. Am. J. Hum. Genet. 98, 116–126 (2016).
McCarthy, S. et al. A reference panel of 64,976 haplotypes for genotype imputation. Nat. Genet. 48, 1279–1283 (2016).
Huang, J. et al. Improved imputation of low-frequency and rare variants using the UK10K haplotype reference panel. Nat. Commun. 6, 8111 (2015).
Taliun, D. et al. Sequencing of 53,831 diverse genomes from the NHLBI TOPMed Program. Nature 590, 290–299 (2021).
Loh, P.-R. et al. Efficient Bayesian mixed-model analysis increases association power in large cohorts. Nat. Genet. 47, 284–290 (2015).
Loh, P.-R., Kichaev, G., Gazal, S., Schoech, A. P. & Price, A. L. Mixed-model association for biobank-scale datasets. Nat. Genet. 50, 906–908 (2018).
Rentzsch, P., Witten, D., Cooper, G. M., Shendure, J. & Kircher, M. CADD: predicting the deleteriousness of variants throughout the human genome. Nucleic Acids Res. 47, D886–D894 (2019).
McLaren, W. et al. The ensembl variant effect predictor. Genome Biol. 17, 122 (2016).
Jaganathan, K. et al. Predicting splicing from primary sequence with deep learning. Cell 176, 535–548.e24 (2019).
Benner, C. et al. FINEMAP: efficient variable selection using summary data from genome-wide association studies. Bioinforma. Oxf. Engl. 32, 1493–1501 (2016).
Schaid, D. J., Chen, W. & Larson, N. B. From genome-wide associations to candidate causal variants by statistical fine-mapping. Nat. Rev. Genet. 19, 491–504 (2018).
Szustakowski, J. D. et al. Advancing human genetics research and drug discovery through exome sequencing of the UK Biobank. Preprint at medRxiv https://doi.org/10.1101/2020.11.02.20222232 (2020).
Wang, Q. et al. Surveying the contribution of rare variants to the genetic architecture of human disease through exome sequencing of 177,882 UK Biobank participants. Preprint at bioRxiv https://doi.org/10.1101/2020.12.13.422582 (2020).
Vuckovic, D. et al. The polygenic and monogenic basis of blood traits and diseases. Cell 182, 1214–1231.e11 (2020).
Haworth, S. et al. Apparent latent structure within the UK Biobank sample has implications for epidemiological analysis. Nat. Commun. 10, 333 (2019).
Mathieson, I. & McVean, G. Differential confounding of rare and common variants in spatially structured populations. Nat. Genet. 44, 243–246 (2012).
Yasoda, A. et al. Natriuretic peptide regulation of endochondral ossification: Evidence for possible roles of the C-type natriuretic peptide/guanylyl cyclase-B pathway. J. Biol. Chem. 273, 11695–11700 (1998).
Gandotra, S. et al. Perilipin deficiency and autosomal dominant partial lipodystrophy. N. Engl. J. Med. 364, 740–748 (2011).
Aslan, J. E. & McCarty, O. J. T. Rho GTPases in platelet function. J. Thromb. Haemost. 11, 35–46 (2013).
Zhao, A. Z., Huan, J.-N., Gupta, S., Pal, R. & Sahu, A. A phosphatidylinositol 3-kinase–phosphodiesterase 3B–cyclic AMP pathway in hypothalamic action of leptin on feeding. Nat. Neurosci. 5, 727–728 (2002).
Boyle, E. A., Li, Y. I. & Pritchard, J. K. An expanded view of complex traits: from polygenic to omnigenic. Cell 169, 1177–1186 (2017).
Landrum, M. J. et al. ClinVar: improving access to variant interpretations and supporting evidence. Nucleic Acids Res. 46, D1062–D1067 (2018).
Ahituv, N. et al. Medical sequencing at the extremes of human body mass. Am. J. Hum. Genet. 80, 779–791 (2007).
The Gene Ontology Consortium. The Gene Ontology resource: 20 years and still GOing strong. Nucleic Acids Res. 47, D330–D338 (2019).
Mi, H., Muruganujan, A., Ebert, D., Huang, X. & Thomas, P. D. PANTHER version 14: more genomes, a new PANTHER GO-slim and improvements in enrichment analysis tools. Nucleic Acids Res. 47, D419–D426 (2019).
Sinnott-Armstrong, N. et al. Genetics of 38 blood and urine biomarkers in the UK Biobank. Nat. Genet. 53, 185–194 (2021).
Povysil, G. et al. Rare-variant collapsing analyses for complex traits: guidelines and applications. Nat. Rev. Genet. 20, 747–759 (2019).
Wu, M. C. et al. Rare-variant association testing for sequencing data with the sequence kernel association test. Am. J. Hum. Genet. 89, 82–93 (2011).
Zhou, W. et al. Efficiently controlling for case-control imbalance and sample relatedness in large-scale genetic association studies. Nat. Genet. 50, 1335–1341 (2018).
Chang, C. C. et al. Second-generation PLINK: rising to the challenge of larger and richer datasets. GigaScience 4, 7 (2015).
Yang, J. et al. Conditional and joint multiple-SNP analysis of GWAS summary statistics identifies additional variants influencing complex traits. Nat. Genet. 44, 369–S3 (2012).
Buniello, A. et al. The NHGRI-EBI GWAS Catalog of published genome-wide association studies, targeted arrays and summary statistics 2019. Nucleic Acids Res. 47, D1005–D1012 (2019).
Locke, A. E. et al. Exome sequencing of Finnish isolates enhances rare-variant association power. Nature 572, 323–328 (2019).
Cunningham, D. et al. Structural and biophysical studies of PCSK9 and its mutants linked to familial hypercholesterolemia. Nat. Struct. Mol. Biol. 14, 413–419 (2007).
Biterova, E., Esmaeeli, M., Alanen, H. I., Saaranen, M. & Ruddock, L. W. Structures of Angptl3 and Angptl4, modulators of triglyceride levels and coronary artery disease. Sci. Rep. 8, 6752 (2018).
LeCour, L. et al. The structural basis for Cdc42-induced dimerization of IQGAPs. Structure 24, 1499–1508 (2016).
Berman, H. M. et al. The protein data bank. Nucleic Acids Res. 28, 235–242 (2000).
Bienert, S. et al. The SWISS-MODEL repository—new features and functionality. Nucleic Acids Res. 45, D313–D319 (2017).
Acknowledgements
We thank A. Gusev, M. Hujoel, P. Palamara, A. Price and S. Sunyaev for helpful discussions. This research was conducted using the UKB Resource under application no. 10438. A.R.B. was supported by US NIH grant T32 HG229516 and fellowship F31 HL154537. M.A.S. was supported by the MIT John W. Jarve (1978) Seed Fund for Science Innovation and US NIH Fellowship F31 MH124393. R.E.M. was supported by US NIH grant K25 HL150334 and NSF grant DMS-1939015. P.-R.L. was supported by US NIH grant DP2 ES030554, a Burroughs Wellcome Fund Career Award at the Scientific Interfaces, the Next Generation Fund at the Broad Institute of MIT and Harvard, and a Sloan Research Fellowship. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. Computational analyses were performed on the O2 High Performance Compute Cluster, supported by the Research Computing Group, at Harvard Medical School (http://rc.hms.harvard.edu).
Author information
Authors and Affiliations
Contributions
A.R.B. and P.-R.L. performed statistical analyses and wrote the manuscript. M.A.S. and R.E.M. provided substantial input on all analyses and on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Genetics thanks S. Petrovski and S. Carmi for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes 1–5 and Figs. 1–11
Supplementary Tables
Supplementary Tables 1–15
Rights and permissions
About this article
Cite this article
Barton, A.R., Sherman, M.A., Mukamel, R.E. et al. Whole-exome imputation within UK Biobank powers rare coding variant association and fine-mapping analyses. Nat Genet 53, 1260–1269 (2021). https://doi.org/10.1038/s41588-021-00892-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-021-00892-1
This article is cited by
-
Exploring the mediating role of calcium homeostasis in the association between diabetes mellitus, glycemic traits, and vascular and valvular calcifications: a comprehensive Mendelian randomization analysis
Diabetology & Metabolic Syndrome (2024)
-
Human genetic associations of the airway microbiome in chronic obstructive pulmonary disease
Respiratory Research (2024)
-
Lipid levels and multiple myeloma risk: insights from Meta-analysis and mendelian randomization
Lipids in Health and Disease (2024)
-
Genetically predicted hypothyroidism, thyroid hormone treatment, and the risk of cardiovascular diseases: a mendelian randomization study
BMC Cardiovascular Disorders (2024)
-
Mendelian randomization analysis reveals the combined effects of epigenetics and telomere biology in hematologic cancers
Clinical Epigenetics (2024)