Abstract
Gene regulatory divergence is thought to play a central role in determining human-specific traits. However, our ability to link divergent regulation to divergent phenotypes is limited. Here, we utilized human–chimpanzee hybrid induced pluripotent stem cells to study gene expression separating these species. The tetraploid hybrid cells allowed us to separate cis- from trans-regulatory effects, and to control for nongenetic confounding factors. We differentiated these cells into cranial neural crest cells, the primary cell type giving rise to the face. We discovered evidence of lineage-specific selection on the hedgehog signaling pathway, including a human-specific sixfold down-regulation of EVC2 (LIMBIN), a key hedgehog gene. Inducing a similar down-regulation of EVC2 substantially reduced hedgehog signaling output. Mice and humans lacking functional EVC2 show striking phenotypic parallels to human–chimpanzee craniofacial differences, suggesting that the regulatory divergence of hedgehog signaling may have contributed to the unique craniofacial morphology of humans.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data were deposited in GEO under accession numbers GSE144825 and GSE146481. Source data are provided with this paper.
Code availability
Code used in this study is available at https://github.com/TheFraserLab/ASEr, https://github.com/TheFraserLab/Agoglia_HumanChimpanzee2020 and https://github.com/TheFraserLab/Hornet/tree/master.
Change history
24 March 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41588-021-00849-4
References
Aiello, L. & Dean, C. An Introduction to Human Evolutionary Anatomy (Elsevier, 2002).
King, M. C. & Wilson, A. C. Evolution at two levels in humans and chimpanzees. Science 188, 107–116 (1975).
Enard, D., Messer, P. W. & Petrov, D. A. Genome-wide signals of positive selection in human evolution. Genome Res. https://doi.org/10.1101/gr.164822.113 (2014).
Fraser, H. B. Gene expression drives local adaptation in humans. Genome Res. 23, 1089–1096 (2013).
Wittkopp, P. J. & Kalay, G. Cis-regulatory elements: molecular mechanisms and evolutionary processes underlying divergence. Nat. Rev. Genet. https://doi.org/10.1038/nrg3095 (2012).
Tirosh, I., Reikhav, S., Levy, A. A. & Barkai, N. A yeast hybrid provides insight into the evolution of gene expression regulation. Science https://doi.org/10.1126/science.1169766 (2009).
Wittkopp, P. J., Haerum, B. K. & Clark, A. G. Evolutionary changes in cis and trans gene regulation. Nature https://doi.org/10.1038/nature02698 (2004).
Pastinen, T. Genome-wide allele-specific analysis: insights into regulatory variation. Nat. Rev. Genet. https://doi.org/10.1038/nrg2815 (2010).
Fraser, H. B. Genome-wide approaches to the study of adaptive gene expression evolution. BioEssays https://doi.org/10.1002/bies.201000094 (2011).
Combs, P. A. et al. Tissue-specific cis-regulatory divergence implicates eloF in inhibiting interspecies mating in Drosophila. Curr. Biol. https://doi.org/10.1016/j.cub.2018.10.036 (2018).
Wang, X., Soloway, P. D. & Clark, A. G. Paternally biased X inactivation in mouse neonatal brain. Genome Biol. https://doi.org/10.1186/gb-2010-11-7-r79 (2010).
Agoglia, A. et al. Generation of human–chimpanzee hybrid stem cell-derived organoids to investigate cis-regulatory evolution of the cerebral cortex. Nature (in the press).
Shakhova, O. & Sommer, L.. Neural crest-derived stem cells. StemBook https://doi.org/10.3824/stembook.1.51.1 (2010).
Øvrebø, J. I. & Edgar, B. A. Polyploidy in tissue homeostasis and regeneration. Development https://doi.org/10.1242/dev.156034 (2018).
Shin, D.-H. et al. Characterization of tetraploid somatic cell nuclear transfer-derived human embryonic stem cells. Dev. Reprod. 21, 425–434 (2017).
Cowan, C. A., Atienza, J., Melton, D. A. & Eggan, K. Nuclear reprogramming of somatic cells after fusion with human embryonic stem cells. Science 309, 1369–1373 (2005).
Broughton, K. M. et al. Cardiac interstitial tetraploid cells can escape replicative senescence in rodents but not large mammals. Commun. Biol. https://doi.org/10.1038/s42003-019-0453-z (2019).
International Stem Cell Initiative et al. Screening ethnically diverse human embryonic stem cells identifies a chromosome 20 minimal amplicon conferring growth advantage. Nat. Biotechnol. 29, 1132–1144 (2011).
Van De Geijn, B., Mcvicker, G., Gilad, Y. & Pritchard, J. K. WASP: allele-specific software for robust molecular quantitative trait locus discovery. Nat. Methods https://doi.org/10.1038/nmeth.3582 (2015).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. https://doi.org/10.1186/s13059-014-0550-8 (2014).
Liu, X., Li, Y. I. & Pritchard, J. K. Trans effects on gene expression can drive omnigenic inheritance. Cell https://doi.org/10.1016/j.cell.2019.04.014 (2019).
Wittkopp, P. J., Haerum, B. K. & Clark, A. G. Regulatory changes underlying expression differences within and between Drosophila species. Nat. Genet. https://doi.org/10.1038/ng.77 (2008).
Peyrégne, S., Boyle, M. J., Dannemann, M. & Prüfer, K. Detecting ancient positive selection in humans using extended lineage sorting. Genome Res. 27, 1563–1572 (2017).
Racimo, F., Kuhlwilm, M. & Slatkin, M. A test for ancient selective sweeps and an application to candidate sites in modern humans. Mol. Biol. Evol. https://doi.org/10.1093/molbev/msu255 (2014).
Kronenberg, Z. N. et al. High-resolution comparative analysis of great ape genomes. Science https://doi.org/10.1126/science.aar6343 (2018).
Prüfer, K. et al. A high-coverage Neandertal genome from Vindija Cave in Croatia. Science 358, 655–658 (2017).
Prabhakar, S., Noonan, J. P., Pääbo, S. & Rubin, E. M. Accelerated evolution of conserved noncoding sequences in humans. Science https://doi.org/10.1126/science.1130738 (2006).
Lindblad-Toh, K. et al. A high-resolution map of human evolutionary constraint using 29 mammals. Nature 478, 476–482 (2011).
Kostka, D., Holloway, A. K. & Pollard, K. S. Developmental loci harbor clusters of accelerated regions that evolved independently in ape lineages. Mol. Biol. Evol. https://doi.org/10.1093/molbev/msy109 (2018).
McLean, C. Y. et al. Human-specific loss of regulatory DNA and the evolution of human-specific traits. Nature https://doi.org/10.1038/nature09774 (2011).
Gittelman, R. M. et al. Comprehensive identification and analysis of human accelerated regulatory DNA. Genome Res. https://doi.org/10.1101/gr.192591.115 (2015).
Marnetto, D., Molineris, I., Grassi, E. & Provero, P. Genome-wide identification and characterization of fixed human-specific regulatory regions. Am. J. Hum. Genet. https://doi.org/10.1016/j.ajhg.2014.05.011 (2014).
Lek, M. et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature https://doi.org/10.1038/nature19057 (2016).
Gayà-Vidal, M. & Albà, M. M. Uncovering adaptive evolution in the human lineage. BMC Genomics https://doi.org/10.1186/1471-2164-15-599 (2014).
Glinsky, G. V. Transposable elements and DNA methylation create in embryonic stem cells human-specific regulatory sequences associated with distal enhancers and noncoding RNAs. Genome Biol. Evol. https://doi.org/10.1093/gbe/evv081 (2015).
Gokhman, D. et al. Differential DNA methylation of vocal and facial anatomy genes in modern humans. Nat. Commun. 11, 1189 (2020).
Shibata, Y. et al. Extensive evolutionary changes in regulatory element activity during human origins are associated with altered gene expression and positive selection. PLoS Genet. https://doi.org/10.1371/journal.pgen.1002789 (2012).
Swain-Lenz, D. et al. Comparative analyses of chromatin landscape in white adipose tissue suggest humans may have less beigeing potential than other primates. Genome Biol. Evol. https://doi.org/10.1093/gbe/evz134 (2019).
Edsall, L. E. et al. Evaluating chromatin accessibility differences across multiple primate species using a joint modelling approach. Genome Biol. Evol. https://doi.org/10.1093/gbe/evz218 (2019).
Romero, I. G., Gopalakrishnan, S. & Gilad, Y. Widespread conservation of chromatin accessibility patterns and transcription factor binding in human and chimpanzee induced pluripotent stem cells. Preprint at bioRxiv https://doi.org/10.1101/466631 (2018).
Glinsky, G. V. Mechanistically distinct pathways of divergent regulatory DNA creation contribute to evolution of human-specific genomic regulatory networks driving phenotypic divergence of homo sapiens. Genome Biol. Evol. https://doi.org/10.1093/gbe/evw185 (2016).
Eres, I. E., Luo, K., Hsiao, C. J., Blake, L. E. & Gilad, Y. Reorganization of 3D genome structure may contribute to gene regulatory evolution in primates. PLoS Genet. https://doi.org/10.1371/journal.pgen.1008278 (2019).
Prescott, S. L. et al. Enhancer divergence and cis-regulatory evolution in the human and chimp neural crest. Cell 163, 68–84 (2015).
Reilly, S. K. & Noonan, J. P. Evolution of gene regulation in humans. Annu. Rev. Genomics Hum. Genet. https://doi.org/10.1146/annurev-genom-090314-045935 (2016).
Cotney, J. et al. The evolution of lineage-specific regulatory activities in the human embryonic limb. Cell 154, 185–196 (2013).
Gokhman, D. et al. Gene ORGANizer: linking genes to the organs they affect. Nucleic Acids Res. 45, W138–W145 (2017).
Köhler, S. et al. The human phenotype ontology project: linking molecular biology and disease through phenotype data. Nucleic Acids Res. 42, D966–D974 (2014).
Gokhman, D. et al. Reconstructing Denisovan anatomy using DNA methylation maps. Cell 179, 180–192.e10 (2019).
Orr, H. A. Testing natural selection vs. genetic drift in phenotypic evolution using quantitative trait locus data. Genetics 149, 2099–2104 (1998).
Kanehisa, M., Sato, Y., Kawashima, M., Furumichi, M. & Tanabe, M. KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res. 44, D457–D462 (2016).
Xavier, G. M. et al. Hedgehog receptor function during craniofacial development. Dev. Biol. https://doi.org/10.1016/j.ydbio.2016.02.009 (2016).
Ramsbottom, S. A. & Pownall, M. E. Regulation of hedgehog signalling inside and outside the cell. J. Dev. Biol. https://doi.org/10.3390/jdb4030023 (2016).
Wang, S. H., Hsiao, C. J., Khan, Z. & Pritchard, J. K. Post-translational buffering leads to convergent protein expression levels between primates. Genome Biol. https://doi.org/10.1186/s13059-018-1451-z (2018).
Dorn, K. V., Hughes, C. E. & Rohatgi, R. A Smoothened-Evc2 complex transduces the hedgehog signal at primary cilia. Dev. Cell https://doi.org/10.1016/j.devcel.2012.07.004 (2012).
Zhang, H. et al. Elevated fibroblast growth factor signaling is critical for the pathogenesis of the dwarfism in Evc2/Limbin mutant mice. PLoS Genet. https://doi.org/10.1371/journal.pgen.1006510 (2016).
Pipes, L. et al. The non-human primate reference transcriptome resource (NHPRTR) for comparative functional genomics. Nucleic Acids Res. https://doi.org/10.1093/nar/gks1268 (2013).
Wunderlich, S. et al. Primate iPS cells as tools for evolutionary analyses. Stem Cell Res. https://doi.org/10.1016/j.scr.2014.02.001 (2014).
Briscoe, J. & Small, S. Morphogen rules: design principles of gradient-mediated embryo patterning. Development https://doi.org/10.1242/dev.129452 (2015).
Young, N. M., Chong, H. J., Hu, D., Hallgrímsson, B. & Marcucio, R. S. Quantitative analyses link modulation of Sonic hedgehog signaling to continuous variation in facial growth and shape. Development https://doi.org/10.1242/dev.052340 (2010).
Hu, D. & Helms, J. A. The role of Sonic hedgehog in normal and abnormal craniofacial morphogenesis. Development 126, 4873–4884 (1999).
Pan, A., Chang, L., Nguyen, A. & James, A. W. A review of hedgehog signaling in cranial bone development. Front. Physiol. https://doi.org/10.3389/fphys.2013.00061 (2013).
Jeong, J., Mao, J., Tenzen, T., Kottmann, A. H. & McMahon, A. P. Hedgehog signaling in the neural crest cells regulates the patterning and growth of facial primordia. Genes Dev. https://doi.org/10.1101/gad.1190304 (2004).
Zhang, H. et al. Generation of Evc2/Limbin global and conditional KO mice and its roles during mineralized tissue formation. Genesis https://doi.org/10.1002/dvg.22879 (2015).
Badri, M. K. et al. Expression of Evc2 in craniofacial tissues and craniofacial bone defects in Evc2 knockout mouse. Arch. Oral Biol. https://doi.org/10.1016/j.archoralbio.2016.05.002 (2016).
Badri, M. K. et al. Ellis van Creveld2 is required for postnatal craniofacial bone development. Anat. Rec. https://doi.org/10.1002/ar.23353 (2016).
Takeda, H. et al. Positional cloning of the gene LIMBIN responsible for bovine chondrodysplastic dwarfism. Proc. Natl Acad. Sci. USA https://doi.org/10.1073/pnas.152337899 (2002).
Caparrós-Martín, J. A. et al. The ciliary EVC/EVC2 complex interacts with Smo and controls hedgehog pathway activity in chondrocytes by regulating Sufu/Gli3 dissociation and Gli3 trafficking in primary cilia. Hum. Mol. Genet. https://doi.org/10.1093/hmg/dds409 (2013).
Kulkarni, A. K. et al. A ciliary protein EVC2/LIMBIN plays a critical role in the skull base for mid-facial development. Front. Physiol. 9, 1484 (2018).
Pusapati, G. V. et al. EFCAB7 and IQCE regulate hedgehog signaling by tethering the EVC-EVC2 complex to the base of primary cilia. Dev. Cell https://doi.org/10.1016/j.devcel.2014.01.021 (2014).
Li, X. et al. Genome-wide linkage study suggests a susceptibility locus for isolated bilateral microtia on 4p15.32-4p16.2. PLoS ONE https://doi.org/10.1371/journal.pone.0101152 (2014).
Claes, P. et al. Modeling 3D facial shape from DNA. PLoS Genet. 10, e1004224 (2014).
Lieberman, D. E. & McCarthy, R. C. The ontogeny of cranial base angulation in humans and chimpanzees and its implications for reconstructing pharyngeal dimensions. J. Hum. Evol. https://doi.org/10.1006/jhev.1998.0287 (1999).
Pilot, M. et al. Diversifying selection between pure-breed and free-breeding dogs inferred from genome-wide SNP analysis. G3 (Bethesda) https://doi.org/10.1534/g3.116.029678 (2016).
Hu, Y. & Albertson, R. C. Hedgehog signaling mediates adaptive variation in a dynamic functional system in the cichlid feeding apparatus. Proc. Natl Acad. Sci. USA https://doi.org/10.1073/pnas.1323154111 (2014).
Burga, A. et al. A genetic signature of the evolution of loss of flight in the Galapagos cormorant. Science https://doi.org/10.1126/science.aal3345 (2017).
Dorus, S. et al. Sonic hedgehog, a key development gene, experienced intensified molecular evolution in primates. Hum. Mol. Genet. https://doi.org/10.1093/hmg/ddl123 (2006).
Claes, P. et al. Genome-wide mapping of global-to-local genetic effects on human facial shape. Nat. Genet. 50, 414–423 (2018).
Lieberman, P. The evolution of human speech: its anatomical and neural bases. Curr. Anthropol. 48, 39–66 (2007).
Boë, L.-J. et al. Which way to the dawn of speech?: Reanalyzing half a century of debates and data in light of speech science. Sci. Adv. 5, eaaw3916 (2019).
Rada-Iglesias, A. et al. Epigenomic annotation of enhancers predicts transcriptional regulators of human neural crest. Cell Stem Cell https://doi.org/10.1016/j.stem.2012.07.006 (2012).
Bajpai, V. K. et al. Reprogramming postnatal human epidermal keratinocytes toward functional neural crest fates. Stem Cells https://doi.org/10.1002/stem.2583 (2017).
Ward, M. C. et al. Silencing of transposable elements may not be a major driver of regulatory evolution in primate iPSCs. eLife https://doi.org/10.7554/eLife.33084 (2018).
Marchetto, M. C. N. et al. Differential L1 regulation in pluripotent stem cells of humans and apes. Nature https://doi.org/10.1038/nature12686 (2013).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics https://doi.org/10.1093/bioinformatics/bts635 (2013).
Tehranchi, A. et al. Fine-mapping cis-regulatory variants in diverse human populations. eLife https://doi.org/10.7554/elife.39595 (2019).
Romero, I. G. et al. A panel of induced pluripotent stem cells from chimpanzees: a resource for comparative functional genomics. eLife https://doi.org/10.7554/eLife.07103.001 (2015).
Weissbein, U., Plotnik, O., Vershkov, D. & Benvenisty, N. Culture-induced recurrent epigenetic aberrations in human pluripotent stem cells. PLoS Genet. https://doi.org/10.1371/journal.pgen.1006979 (2017).
Piñero, J. et al. DisGeNET: a discovery platform for the dynamical exploration of human diseases and their genes. Database 2015, bav028 (2015).
Hamosh, A., Scott, A. F., Amberger, J. S., Bocchini, C. A. & McKusick, V. A. Online Mendelian Inheritance in Man (OMIM), a knowledgebase of human genes and genetic disorders. Nucleic Acids Res. 33, D514–D517 (2005).
Pizzollo, J. et al. Comparative serum challenges show divergent patterns of gene expression and open chromatin in human and chimpanzee. Genome Biol. Evol. https://doi.org/10.1093/gbe/evy041 (2018).
Acknowledgements
We thank S. Bar, L. Carmel and members of the Fraser, Petrov and Pritchard laboratories for critical comments, and the Gilad laboratory (Chicago University) and the You laboratory (University of Pennsylvania) for sharing data and cells. D.G. was funded by the Human Frontier, Rothschild and Zuckerman fellowships. H.B.F. is supported by National Institutes of Health (NIH) grant no. 2R01GM097171-05A1. The cells used in this study were derived from the iPSCs generated by Gallego Romero et al.86, whose study was supported by the NIH, Office of Research Infrastructure Programs/OD (grant no. P51OD011132).
Author information
Authors and Affiliations
Contributions
D.G. and H.B.F. designed experiments and analyses and wrote the manuscript with input from all authors. D.G. conducted the analyses. R.M.A. designed the ASE pipeline and generated RNA-seq data. M.K. designed and performed the EVC2 and Hedgehog signaling experiments and was supervised by R.R. W.G. designed and performed the reporter assay experiments and was supervised by N.A. D.S. generated the hybrid cells. V.K.B. and S.N. differentiated the cells and were supervised by J.W. Coral Chen, A.C. and Chider Chen contributed human and chimpanzee DPSCs. D.A.P. cosupervised D.G. H.Z. and Y.M. generated the mouse KO. H.B.F. devised the original idea and supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Genetics thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Identifying human-chimpanzee expression changes using hybrid cells.
a, Immunostaining for CNCC markers NR2F1 and PAX3 was performed to confirm CNCC differentiation. b, Expression levels of positive and negative markers in the parental and hybrid CNCCs. c, Heatmap and dendrogram of total gene expression across iPSC and CNCC samples. d,e, Fold-change per gene for hybrid iPSCs and hybrid CNCCs when aligned to the human (GRCh38) vs chimpanzee (panTro5) genomes. Grey points are genes where the absolute difference in log2(fold-change) when aligned to the human vs. chimpanzee genome is greater than 1 (that is, genes with potential alignment bias that were excluded from the analysis). Genes with no observable alignment bias are marked with blue (significant ASE: q-value < 0.05) or yellow (non-significant ASE). f, Venn diagram of genes with significant human-chimpanzee expression changes in parental and hybrid samples. g. Parental vs hybrid iPSC expression changes. See Fig. 1d legend.
Extended Data Fig. 2 Differentially expressed genes are associated with divergent chromatin and phenotypes.
a, Overlap of ASE genes in CNCCs with loci showing divergent regulatory marks. Each of the datasets was examined twice: (1) against Ch > Hu genes (red), and (2) against Hu > Ch genes (blue). In 14 out of 16 datasets, expression differences reflect regulatory differences, that is, Hu > Ch regulatory marks show more overlap with Hu > Ch genes than with Ch > Hu genes, and vice versa. P-value shows one-tailed paired t-test for overall overlap (see Methods). Asterisks mark significant randomization test overlap (FDR < 0.05). See Supplementary Table 8. b, Mean fraction of divergent phenotypes for groups of genes with increasingly higher fold-change thresholds. c, Violin plots showing phenotype assignment accuracy in groups of genes with increasingly more divergent differential expression in parental cells. Randomization test P-values are shown for overall accuracy compared to random (PAUC), and accuracy increase compared to random (Pslope), as shown in d. See Fig. 2c legend. d, Randomization output for the phenotype assignment pipeline. Genes associated with each phenotype were randomly assigned a direction of expression change, while keeping their absolute fold-change. Randomization test P-values are shown for overall accuracy compared to random (PAUC), and accuracy increase compared to random (Pslope). e, Phenotype assignment accuracy before and after applying unidirectionality filtering, for ASE and parental differential expression with cis-contribution ≥ 90%. See Fig. 2d legend. In the unidirectionality filter, only phenotypes where all genes point in the same phenotypic direction (that is, complete agreement) are analyzed48.
Extended Data Fig. 3 EVC2 down-regulation in humans.
a, The down:up ratio of Hh signaling genes across increasingly more stringent FDR and fold-change thresholds. b, Differential expression along all of the exons of EVC2 in a CNCC hybrid (Hy1_30_rep1), showing that the majority of reads come from the chimpanzee alleles. Introns are not shown to scale. c, Violin plots of EVC2 expression across iPSC and CNCC non-hybrid samples from various sources, showing consistent EVC2 down-regulation in humans compared to chimpanzees. Diamonds show mean expression levels. DESeq2 FDR-adjusted P-values are presented for cell type. The observation that the human-chimpanzee ratios are similar to the ones observed within the hybrid cells suggests that the majority of differential expression is driven by cis changes. d, EVC2 expression across nine additional tissues for which both human and chimpanzee data are available56, showing that EVC2 down-regulation is not restricted to iPSCs and CNCCs. Dashed line shows mean expression. One-sided t-test P-values are shown. e, Gorilla vs human EVC2 expression. One-sided t-test P-values are shown. f, Western blot of EVC2 protein levels in human and chimpanzee DPSCs. The samples derive from the same experiment and blots were processed in parallel. For gel source data, see Source Data.
Extended Data Fig. 4 Differentially regulated regions in EVC2.
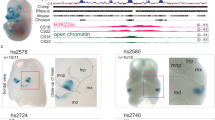
ATAC-seq read pileup along EVC2 and for the three loci showing species-biased peaks within EVC2. Arrows mark peaks. b,c, NR2F1 and TFAP2A ChIP-seq read pileup for loci <10 kb away from the ATAC-seq peaks. d, MUSCLE103 sequence alignment of rhesus, gorilla, chimp and human sequences. Regions with a high proportion of mismatches are colored in red. e, Reporter assay comparing relative firefly/Renilla luciferase activity for chimpanzee and human EVC2 sequences following transient transfection in human DPSCs. Empty vector (pGL4.11b) was used as negative control. Box plots show mean (center), 2nd and 3rd quartiles (box boundaries), and minima and maxima (whiskers). One-tailed t-test P-values in two independent experiments of quadruplet measurements (n = 8) are shown.
Extended Data Fig. 5 Reduced levels of EVC2 result in reduced Hedgehog signaling output and affect craniofacial phenotypes.
a, Western blot of Gli1 protein levels (a measure of Hh signaling output induced by Shh) at different Evc2 and Hh signaling input levels. EvcC2 was introduced at various levels into Evc2−/− mouse NIH/3T3 fibroblasts through retroviral infection. Cells with higher levels of Evc2 show higher Hh signaling output. p38 served as positive control. Pearson’s R and P-value are shown for 40 nM SHH. The samples derive from the same experiment and blots were processed in parallel. For gel source data, see Source Data. b, Micro-CT radiographic images of the palate bone, enamel (extra bright) and roots of the first mandibular molar in Evc2 control and Evc2 KO mice at P28. c, Diagram of the mandible indicating the landmarks for the parameters measured. d, Mean skull and mandible measurements from Evc2 control and Evc2 KO mice at P28. (n = 5 for each group, FDR-adjusted two-tailed t-test P-values are shown). Whiskers show one standard deviation in each direction. Landmarks used are shown in the titles.
Extended Data Fig. 6 No aneuploidies observed in CNCC hybrid samples.
Figure shows ASE (top), sliding window ASE median over 20 genes (middle), and Wilcoxon rank sum test P-values for each sliding window against the entire genome (bottom). Dashed line shows mean for ASE and sliding window ASE, and shows Bonferroni P-value cutoff for the Wilcoxon rank sum test. An example of data is presented for autosomal chromosomes 1 and 20, and for chromosome X from the CNCC Hy1_30_rep1 sample. No significant deviations were detected in any of the CNCC hybrid samples. See Agoglia et al. for iPSC aneuploidy analyses12.
Extended Data Fig. 7 No chromosomal duplications or losses observed in the CNCC hybrid samples.
Density plots of percentage of human-aligned reads per gene per chromosome for each of the CNCC hybrid samples. Vertical dashed lines show mean per sample.
Supplementary information
Supplementary Information
Supplementary Methods, Tables 24 and 25, and Figures
Supplementary Tables
Supplementary Tables 1–23 and 26–28
Source data
Source Data Fig. 4
Unprocessed western blots.
Source Data Extended Data Fig. 3
Unprocessed western blots.
Source Data Extended Data Fig. 5
Unprocessed western blots.
Rights and permissions
About this article
Cite this article
Gokhman, D., Agoglia, R.M., Kinnebrew, M. et al. Human–chimpanzee fused cells reveal cis-regulatory divergence underlying skeletal evolution. Nat Genet 53, 467–476 (2021). https://doi.org/10.1038/s41588-021-00804-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-021-00804-3
This article is cited by
-
Accounting for cis-regulatory constraint prioritizes genes likely to affect species-specific traits
Genome Biology (2023)
-
The relationship between regulatory changes in cis and trans and the evolution of gene expression in humans and chimpanzees
Genome Biology (2023)
-
Functional bioengineered models of the central nervous system
Nature Reviews Bioengineering (2023)
-
Current advances in primate genomics: novel approaches for understanding evolution and disease
Nature Reviews Genetics (2023)
-
Shaping faces: genetic and epigenetic control of craniofacial morphogenesis
Nature Reviews Genetics (2023)