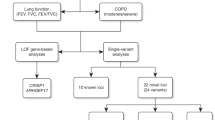
Abstract
Chronic obstructive pulmonary disease (COPD) is the leading cause of respiratory mortality worldwide. Genetic risk loci provide new insights into disease pathogenesis. We performed a genome-wide association study in 35,735 cases and 222,076 controls from the UK Biobank and additional studies from the International COPD Genetics Consortium. We identified 82 loci associated with P < 5 × 10−8; 47 of these were previously described in association with either COPD or population-based measures of lung function. Of the remaining 35 new loci, 13 were associated with lung function in 79,055 individuals from the SpiroMeta consortium. Using gene expression and regulation data, we identified functional enrichment of COPD risk loci in lung tissue, smooth muscle, and several lung cell types. We found 14 COPD loci shared with either asthma or pulmonary fibrosis. COPD genetic risk loci clustered into groups based on associations with quantitative imaging features and comorbidities. Our analyses provide further support for the genetic susceptibility and heterogeneity of COPD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Genome-wide association summary statistics are available at the database of Genotypes and Phenotypes (dbGaP) under accession phs000179.v5.p2 and via the UK Biobank. Derived phenotypic data for COPD case–control status are also available from UK Biobank.
References
GBD 2015 Chronic Respiratory Disease Collaborators. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir. Med. 5, 691–706 (2017).
World Health Organization. Global Health Estimates 2016: Disease burden by Cause, Age, Sex, by Country and by Region, 2000–2016. https://www.who.int/healthinfo/global_burden_disease/estimates/en/index1.html (2018).
Fuchsberger, C. et al. The genetic architecture of type 2 diabetes. Nature 536, 41–47 (2016).
Zhou, J. J. et al. Heritability of chronic obstructive pulmonary disease and related phenotypes in smokers. Am. J. Respir. Crit. Care Med. 188, 941–947 (2013).
Hobbs, B. D. et al. Genetic loci associated with chronic obstructive pulmonary disease overlap with loci for lung function and pulmonary fibrosis. Nat. Genet. 49, 426–432 (2017).
Jiang, Z. et al. A chronic obstructive pulmonary disease susceptibility gene, FAM13A, regulates protein stability of β-catenin. Am. J. Respir. Crit. Care Med. 194, 185–197 (2016).
Lao, T. et al. Hhip haploinsufficiency sensitizes mice to age-related emphysema. Proc. Natl. Acad. Sci. USA 113, E4681–E4687 (2016).
Sudlow, C. et al. UK Biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Vogelmeier, C. F. et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease 2017 Report. Gold Executive Summary. Am. J. Respir. Crit. Care Med. 195, 557–582 (2017).
Bulik-Sullivan, B. K. et al. LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291–295 (2015).
Cho, M. H. et al. Risk loci for chronic obstructive pulmonary disease: a genome-wide association study and meta-analysis. Lancet Respir. Med. 2, 214–225 (2014).
Wilk, J. B. et al. A genome-wide association study of pulmonary function measures in the Framingham Heart Study. PLoS Genet. 5, e1000429 (2009).
Repapi, E. et al. Genome-wide association study identifies five loci associated with lung function. Nat. Genet. 42, 36–44 (2010).
Hancock, D. B. et al. Meta-analyses of genome-wide association studies identify multiple loci associated with pulmonary function. Nat. Genet. 42, 45–52 (2010).
Soler Artigas, M. et al. Genome-wide association and large-scale follow up identifies 16 new loci influencing lung function. Nat. Genet. 43, 1082–1090 (2011).
Wain, L. V. et al. Novel insights into the genetics of smoking behaviour, lung function, and chronic obstructive pulmonary disease (UK BiLEVE): a genetic association study in UK Biobank. Lancet Respir. Med. 3, 769–781 (2015).
Soler Artigas, M. et al. Sixteen new lung function signals identified through 1000 Genomes Project reference panel imputation. Nat. Commun. 6, 8658 (2015).
Wain, L. V. et al. Genome-wide association analyses for lung function and chronic obstructive pulmonary disease identify new loci and potential druggable targets. Nat. Genet. 49, 416–425 (2017).
Wyss, A. B. et al. Multiethnic meta-analysis identifies ancestry-specific and cross-ancestry loci for pulmonary function. Nat. Commun. 9, 2976 (2018).
Jackson, V. E. et al. Meta-analysis of exome array data identifies six novel genetic loci for lung function. Wellcome Open Res. 3, 4 (2018).
Shrine, N. et al. New genetic signals for lung function highlight pathways and pleiotropy, and chronic obstructive pulmonary disease associations across multiple ancestries. Preprint at https://www.biorxiv.org/content/early/2018/06/12/343293 (2018).
Yang, J. et al. Conditional and joint multiple-SNP analysis of GWAS summary statistics identifies additional variants influencing complex traits. Nat. Genet. 44, 369–375 (2012). S1-3.
Agusti, A. & Soriano, J. B. COPD as a systemic disease. COPD 5, 133–138 (2008).
Barnes, P. J. & Celli, B. R. Systemic manifestations and comorbidities of COPD. Eur. Respir. J. 33, 1165–1185 (2009).
Lu, Q. et al. Systematic tissue-specific functional annotation of the human genome highlights immune-related DNA elements for late-onset Alzheimer’s disease. PLoS Genet. 13, e1006933 (2017).
Finucane, H. K. et al. Partitioning heritability by functional annotation using genome-wide association summary statistics. Nat. Genet. 47, 1228–1235 (2015).
Cusanovich, D. A. et al. A single-cell atlas of in vivo mammalian chromatin accessibility. Cell 174, 1309–1324 (2018).
Xu, Y. et al. Single-cell RNA sequencing identifies diverse roles of epithelial cells in idiopathic pulmonary fibrosis. JCI Insight 1, e90558 (2016).
Jacob, A. et al. Differentiation of human pluripotent stem cells into functional lung alveolar epithelial cells. Cell Stem Cell 21, 472–488 (2017).
Ardini-Poleske, M. E. et al. LungMAP: the molecular atlas of lung development program. Am. J. Physiol. Lung Cell. Mol. Physiol. 313, L733–L740 (2017).
Finucane, H. K. et al. Heritability enrichment of specifically expressed genes identifies disease-relevant tissues and cell types. Nat. Genet. 50, 621–629 (2018).
Slowikowski, K., Hu, X. & Raychaudhuri, S. SNPsea: an algorithm to identify cell types, tissues and pathways affected by risk loci. Bioinformatics 30, 2496–2497 (2014).
Wakefield, J. A Bayesian measure of the probability of false discovery in genetic epidemiology studies. Am. J. Hum. Genet. 81, 208–227 (2007).
Visscher, P. M. et al. 10 years of GWAS discovery: biology, function, and translation. Am. J. Hum. Genet. 101, 5–22 (2017).
Zhou, X. et al. Identification of a chronic obstructive pulmonary disease genetic determinant that regulates HHIP. Hum. Mol. Genet. 21, 1325–1335 (2012).
Claussnitzer, M., Hui, C.-C. & Kellis, M. FTO obesity variant and adipocyte browning in humans. N. Engl. J. Med. 374, 192–193 (2016).
Barbeira, A. N. et al. Exploring the phenotypic consequences of tissue specific gene expression variation inferred from GWAS summary statistics. Nat. Commun. 9, 1825 (2018).
Lamontagne, M. et al. Leveraging lung tissue transcriptome to uncover candidate causal genes in COPD genetic associations. Hum. Mol. Genet. 27, 1819–1829 (2018).
Morrow, J. D. et al. Human lung DNA methylation quantitative trait loci colocalize with COPD genome-wide association loci. Am. J. Respir. Crit. Care Med. 197, 1275–1284 (2018).
Schmitt, A. D. et al. A compendium of chromatin contact maps reveals spatially active regions in the human genome. Cell Rep. 17, 2042–2059 (2016).
Shooshtari, P., Huang, H. & Cotsapas, C. Integrative genetic and epigenetic analysis uncovers regulatory mechanisms of autoimmune disease. Am. J. Hum. Genet. 101, 75–86 (2017).
Pers, T. H. et al. Biological interpretation of genome-wide association studies using predicted gene functions. Nat. Commun. 6, 5890 (2015).
Qiao, D. et al. Whole exome sequencing analysis in severe chronic obstructive pulmonary disease. Hum. Mol. Genet. 27, 3801–3812 (2018).
Sanseau, P. et al. Use of genome-wide association studies for drug repositioning. Nat. Biotechnol. 30, 317–320 (2012).
Lencz, T. & Malhotra, A. K. Targeting the schizophrenia genome: a fast track strategy from GWAS to clinic. Mol. Psychiatry 20, 820–826 (2015).
Lamb, J. et al. The Connectivity Map: using gene-expression signatures to connect small molecules, genes, and disease. Science 313, 1929–1935 (2006).
Sirota, M. et al. Discovery and preclinical validation of drug indications using compendia of public gene expression data. Sci. Transl. Med. 3, 96ra77 (2011).
Corsello, S. M. et al. The Drug Repurposing Hub: a next-generation drug library and information resource. Nat. Med. 23, 405–408 (2017).
Subramanian, A. et al. A next generation connectivity map: l1000 platform and the first 1,000,000 profiles. Cell 171, 1437–1452 (2017).
Cho, M. H. et al. A genome-wide association study of emphysema and airway quantitative imaging phenotypes. Am. J. Respir. Crit. Care Med. 192, 559–569 (2015).
MacArthur, J. et al. The new NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog). Nucleic Acids Res. 45, D896–D901 (2017).
Zheng, J. et al. LD Hub: a centralized database and web interface to perform LD score regression that maximizes the potential of summary level GWAS data for SNP heritability and genetic correlation analysis. Bioinformatics 33, 272–279 (2017).
Pickrell, J. K. et al. Detection and interpretation of shared genetic influences on 42 human traits. Nat. Genet. 48, 709–717 (2016).
Demenais, F. et al. Multiancestry association study identifies new asthma risk loci that colocalize with immune-cell enhancer marks. Nat. Genet. 50, 42–53 (2018).
Fingerlin, T. E. et al. Genome-wide imputation study identifies novel HLA locus for pulmonary fibrosis and potential role for auto-immunity in fibrotic idiopathic interstitial pneumonia. BMC Genet. 17, 74 (2016).
Skronska-Wasek, W. et al. Reduced frizzled receptor 4 expression prevents WNT/β-catenin-driven alveolar lung repair in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 196, 172–185 (2017).
Sakornsakolpat, P. et al. Integrative genomics identifies new genes associated with severe COPD and emphysema. Respir. Res. 19, 46 (2018).
Bui, D. S. et al. Childhood predictors of lung function trajectories and future COPD risk: a prospective cohort study from the first to the sixth decade of life. Lancet Respir. Med. 6, 535–544 (2018).
McGeachie, M. J. et al. Patterns of growth and decline in lung function in persistent childhood asthma. N. Engl. J. Med. 374, 1842–1852 (2016).
Ross, J. C. et al. Longitudinal modeling of lung function trajectories in smokers with and without COPD. Am. J. Respir. Crit. Care Med. 198, 1033–1042 (2018).
Boucherat, O., Morissette, M. C., Provencher, S., Bonnet, S. & Maltais, F. Bridging lung development with chronic obstructive pulmonary disease. Relevance of developmental pathways in chronic obstructive pulmonary disease pathogenesis. Am. J. Respir. Crit. Care Med. 193, 362–375 (2016).
Nelson, M. R. et al. The support of human genetic evidence for approved drug indications. Nat. Genet. 47, 856–860 (2015).
Miossec, P. & Kolls, J. K. Targeting IL-17 and TH17 cells in chronic inflammation. Nat. Rev. Drug Discov. 11, 763–776 (2012).
Mellett, M. et al. Orphan receptor IL-17RD tunes IL-17A signalling and is required for neutrophilia. Nat. Commun. 3, 1119 (2012).
O’Leary, N. A. et al. Reference Sequence (RefSeq) database at NCBI: current status, taxonomic expansion, and functional annotation. Nucleic Acids Res. 44, D733–D745 (2016).
Fagerberg, L. et al. Analysis of the human tissue-specific expression by genome-wide integration of transcriptomics and antibody-based proteomics. Mol. Cell. Proteomics 13, 397–406 (2014).
Saito, A., Ozaki, K., Fujiwara, T., Nakamura, Y. & Tanigami, A. Isolation and mapping of a human lung-specific gene, TSA1902, encoding a novel chitinase family member. Gene 239, 325–331 (1999).
Aminuddin, F. et al. Genetic association between human chitinases and lung function in COPD. Hum. Genet. 131, 1105–1114 (2012).
Birben, E. et al. The effects of an insertion in the 5′UTR of the AMCase on gene expression and pulmonary functions. Respir. Med. 105, 1160–1169 (2011).
Chatterjee, R., Batra, J., Das, S., Sharma, S. K. & Ghosh, B. Genetic association of acidic mammalian chitinase with atopic asthma and serum total IgE levels. J. Allergy Clin. Immunol. 122, 202–208 (2008).
Ober, C. & Chupp, G. L. The chitinase and chitinase-like proteins: a review of genetic and functional studies in asthma and immune-mediated diseases. Curr. Opin. Allergy Clin. Immunol. 9, 401–408 (2009).
Heinzmann, A. et al. Joint influences of acidic-mammalian-chitinase with interleukin-4 and Toll-like receptor-10 with interleukin-13 in the genetics of asthma. Pediatr. Allergy Immunol. 21, e679–e686 (2010).
Okawa, K. et al. Loss and gain of human acidic mammalian chitinase activity by nonsynonymous SNPs. Mol. Biol. Evol. 33, 3183–3193 (2016).
Yang, J. et al. Rootletin, a novel coiled-coil protein, is a structural component of the ciliary rootlet. J. Cell Biol. 159, 431–440 (2002).
Gibson, M. A., Hughes, J. L., Fanning, J. C. & Cleary, E. G. The major antigen of elastin-associated microfibrils is a 31-kDa glycoprotein. J. Biol. Chem. 261, 11429–11436 (1986).
Massaro, G. D. et al. Retinoic acid receptor-β: an endogenous inhibitor of the perinatal formation of pulmonary alveoli. Physiol. Genomics 4, 51–57 (2000).
Markovics, J. A. et al. Interleukin-1β induces increased transcriptional activation of the transforming growth factor-β-activating integrin subunit β8 through altering chromatin architecture. J. Biol. Chem. 286, 36864–36874 (2011).
Kitamura, H. et al. Mouse and human lung fibroblasts regulate dendritic cell trafficking, airway inflammation, and fibrosis through integrin αvβ8-mediated activation of TGF-β. J. Clin. Invest. 121, 2863–2875 (2011).
Araya, J. et al. Squamous metaplasia amplifies pathologic epithelial–mesenchymal interactions in COPD patients. J. Clin. Invest. 117, 3551–3562 (2007).
Zeltz, C. & Gullberg, D. The integrin–collagen connection: a glue for tissue repair? J. Cell. Sci. 129, 1284 (2016).
Hall, N. G., Klenotic, P., Anand-Apte, B. & Apte, S. S. ADAMTSL-3/punctin-2, a novel glycoprotein in extracellular matrix related to the ADAMTS family of metalloproteases. Matrix Biol. 22, 501–510 (2003).
Apte, S. S. A disintegrin-like and metalloprotease (reprolysin-type) with thrombospondin type 1 motif (ADAMTS) superfamily: functions and mechanisms. J. Biol. Chem. 284, 31493–31497 (2009).
Kutz, W. E. et al. ADAMTS10 protein interacts with fibrillin-1 and promotes its deposition in extracellular matrix of cultured fibroblasts. J. Biol. Chem. 286, 17156–17167 (2011).
Gabriel, L. A. R. et al. ADAMTSL4, a secreted glycoprotein widely distributed in the eye, binds fibrillin-1 microfibrils and accelerates microfibril biogenesis. Invest. Ophthalmol. Vis. Sci. 53, 461–469 (2012).
Tsutsui, K. et al. ADAMTSL-6 is a novel extracellular matrix protein that binds to fibrillin-1 and promotes fibrillin-1 fibril formation. J. Biol. Chem. 285, 4870–4882 (2010).
Ghosh, M. et al. Exhaustion of airway basal progenitor cells in early and established chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 197, 885–896 (2018).
Crystal, R. G. Airway basal cells. The ‘smoking gun’ of chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 190, 1355–1362 (2014).
Giordano, R. J. et al. Targeted induction of lung endothelial cell apoptosis causes emphysema-like changes in the mouse. J. Biol. Chem. 283, 29447–29460 (2008).
Franks, T. J. et al. Resident cellular components of the human lung: current knowledge and goals for research on cell phenotyping and function. Proc. Am. Thorac. Soc. 5, 763–766 (2008).
Boschetto, P. et al. Predominant emphysema phenotype in chronic obstructive pulmonary. Eur. Respir. J. 21, 450–454 (2003).
Castaldi, P. J. et al. Cluster analysis in the COPDGene study identifies subtypes of smokers with distinct patterns of airway disease and emphysema. Thorax 69, 415–422 (2014).
Cerveri, I. et al. The rapid FEV1 decline in chronic obstructive pulmonary disease is associated with predominant emphysema: a longitudinal study. COPD 10, 55–61 (2013).
Bonàs-Guarch, S. et al. Re-analysis of public genetic data reveals a rare X-chromosomal variant associated with type 2 diabetes. Nat. Commun. 9, 321 (2018).
Dehghan, A. et al. Meta-analysis of genome-wide association studies in >80 000 subjects identifies multiple loci for C-reactive protein levels. Circulation 123, 731–738 (2011).
Hersh, C. P. et al. Non-emphysematous chronic obstructive pulmonary disease is associated with diabetes mellitus. BMC Pulm. Med. 14, 164 (2014).
Higami, Y. et al. Increased epicardial adipose tissue is associated with the airway dominant phenotype of chronic obstructive pulmonary disease. PLoS One 11, e0148794 (2016).
Chung, K. F. & Barnes, P. J. Cytokines in asthma. Thorax 54, 825–857 (1999).
Kroegel, C., Julius, P., Matthys, H., Virchow, J. C. & Luttmann, W. Endobronchial secretion of interleukin-13 following local allergen challenge in atopic asthma: relationship to interleukin-4 and eosinophil counts. Eur. Respir. J. 9, 899–904 (1996).
Boutten, A. et al. Decreased expression of interleukin 13 in human lung emphysema. Thorax 59, 850–854 (2004).
Washko, G. R. et al. Lung volumes and emphysema in smokers with interstitial lung abnormalities. N. Engl. J. Med. 364, 897–906 (2011).
Chilosi, M., Poletti, V. & Rossi, A. The pathogenesis of COPD and IPF: distinct horns of the same devil? Respir. Res. 13, 3 (2012).
Kulkarni, T., O’Reilly, P., Antony, V. B., Gaggar, A. & Thannickal, V. J. Matrix remodeling in pulmonary fibrosis and emphysema. Am. J. Respir. Cell. Mol. Biol. 54, 751–760 (2016).
Wan, M., Qin, J., Songyang, Z. & Liu, D. OB fold–containing protein 1 (OBFC1), a human homolog of yeast Stn1, associates with TPP1 and is implicated in telomere length regulation. J. Biol. Chem. 284, 26725–26731 (2009).
Albrecht, E. et al. Telomere length in circulating leukocytes is associated with lung function and disease. Eur. Respir. J. 43, 983–992 (2014).
Armanios, M. Telomerase and idiopathic pulmonary fibrosis. Mutat. Res. 730, 52–58 (2012).
Stanley, S. E. et al. Telomerase mutations in smokers with severe emphysema. J. Clin. Invest. 125, 563–570 (2015).
Tinkelman, D. G., Price, D. B., Nordyke, R. J. & Halbert, R. J. Misdiagnosis of COPD and asthma in primary care patients 40 years of age and over. J. Asthma 43, 75–80 (2006).
Foreman, M. G. et al. Alpha-1 antitrypsin PiMZ genotype is associated with chronic obstructive pulmonary disease in two racial groups. Ann. Am. Thorac. Soc. 14, 1280–1287 (2017).
Han, M. K. et al. Gender and chronic obstructive pulmonary disease: why it matters. Am. J. Respir. Crit. Care Med. 176, 1179–1184 (2007).
Miller, M. R. et al. Standardisation of spirometry. Eur. Respir. J. 26, 319–338 (2005).
Hankinson, J. L., Odencrantz, J. R. & Fedan, K. B. Spirometric reference values from a sample of the general U.S. population. Am. J. Respir. Crit. Care Med. 159, 179–187 (1999).
Regan, E. A. et al. Genetic epidemiology of COPD (COPDGene) study design. COPD 7, 32–43 (2010).
McCarthy, S. et al. A reference panel of 64,976 haplotypes for genotype imputation. Nat. Genet. 48, 1279–1283 (2016).
Chang, C. C. et al. Second-generation PLINK: rising to the challenge of larger and richer datasets. Gigascience 4, 7 (2015).
Willer, C. J., Li, Y. & Abecasis, G. R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191 (2010).
Lutz, S. M. et al. A genome-wide association study identifies risk loci for spirometric measures among smokers of European and African ancestry. BMC Genet. 16, 138 (2015).
Loth, D. W. et al. Genome-wide association analysis identifies six new loci associated with forced vital capacity. Nat. Genet. 46, 669–677 (2014).
Hobbs, B. D. et al. Exome array analysis identifies a common variant in IL27 associated with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 194, 48–57 (2016).
Berisa, T. & Pickrell, J. K. Approximately independent linkage disequilibrium blocks in human populations. Bioinformatics 32, 283–285 (2016).
Tobacco and Genetics Consortium. Genome-wide meta-analyses identify multiple loci associated with smoking behavior. Nat. Genet. 42, 441–447 (2010).
Pruim, R. J. et al. LocusZoom: regional visualization of genome-wide association scan results. Bioinformatics 26, 2336–2337 (2010).
Hormozdiari, F. et al. Colocalization of GWAS and eQTL signals detects target genes. Am. J. Hum. Genet. 99, 1245–1260 (2016).
Martin, J. S. et al. HUGIn: Hi-C unifying genomic interrogator. Bioinformatics 33, 3793–3795 (2017).
Durinck, S., Spellman, P. T., Birney, E. & Huber, W. Mapping identifiers for the integration of genomic datasets with the R/Bioconductor package biomaRt. Nat. Protoc. 4, 1184–1191 (2009).
Machiela, M. J. & Chanock, S. J. LDlink: a web-based application for exploring population-specific haplotype structure and linking correlated alleles of possible functional variants. Bioinformatics 31, 3555–3557 (2015).
Acknowledgements
This work was supported by the Prince Mahidol Award Youth Program Scholarship (P. Sakornsakolpat); NHLBI R01HL084323, R01HL113264, R01HL089856, and P01HL105339 (E.K.S.); K08HL136928 (B.D.H.), the Parker B. Francis Research Opportunity Award (B.D.H.); and R01HL113264, R01HL137927, P01HL105339, and P01HL132825 (M.H.C.). This research was conducted by using the UK Biobank resource under application numbers 20915 (M.H.C.) and 648 (M.D.T.). Please refer to the Supplementary Note for full acknowledgements. Funding bodies had no role in the design of the study, the collection, analysis, or interpretation of the data, or the writing of the manuscript.
Author information
Authors and Affiliations
Consortia
Contributions
P. Sakornsakolpat contributed to study concept and design, data analysis, and manuscript writing. D.P., B.D.H., and M.H.C. contributed to study concept and design, data analysis, statistical support, and manuscript writing. A.B.W., K.d.J., S.J.L., and D.P.S. contributed to study concept and design and to data analysis. P.B., R.G.B., J.D.C., A.G., D.A.M., G.T.O’C., S.I.R., D.A.S., R.T.-S., Y.T., and E.K.S. contributed to study concept and design and to data collection. T.H.B. and J.E.H. contributed to study concept and design and to statistical support. I.P.H., H.M.B., L.V.W., and M.D.T. contributed to study concept and design. All authors, including those whose initials are not listed above, contributed to critical review and editing of the manuscript and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
M.H.C., E.K.S., L.V.W., M.D.T., D.A.L., and I.P.H. have received grant funding from GlaxoSmithKline (GSK). E.K.S. has received honoraria from Novartis for continuing medical education seminars and travel support from GSK. I.P.H. has received grant support from BI. R.T.-S. is an employee and shareholder of GSK. J.V. has received personal fees from GSK, Chiesi Pharmaceuticals, BI, Novartis, and AstraZeneca. D.L.D. has received grants from the National Institutes of Health for research on COPD and personal fees from Novartis. D.A.L. has received honoraria from GSK and chaired the Respiratory Therapy Area Board from 2012 to 2015. Outside the submitted work, L.L. reports expert consultation for Boehringer Ingelheim and Novartis and unrestricted grants from AstraZeneca and Chiesi.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Note
Supplementary Tables
Supplementary Tables 1–22
Rights and permissions
About this article
Cite this article
Sakornsakolpat, P., Prokopenko, D., Lamontagne, M. et al. Genetic landscape of chronic obstructive pulmonary disease identifies heterogeneous cell-type and phenotype associations. Nat Genet 51, 494–505 (2019). https://doi.org/10.1038/s41588-018-0342-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-018-0342-2
This article is cited by
-
Human genetic associations of the airway microbiome in chronic obstructive pulmonary disease
Respiratory Research (2024)
-
Genetics of chronic respiratory disease
Nature Reviews Genetics (2024)
-
PRANA: an R package for differential co-expression network analysis with the presence of additional covariates
BMC Genomics (2023)
-
X chromosome associations with chronic obstructive pulmonary disease and related phenotypes: an X chromosome-wide association study
Respiratory Research (2023)
-
A pseudo-value regression approach for differential network analysis of co-expression data
BMC Bioinformatics (2023)