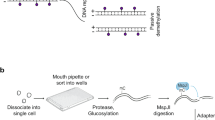
Abstract
DNA methylation is a crucial layer of epigenetic regulation during mammalian embryonic development1,2,3. Although the DNA methylome of early human embryos has been analyzed4,5,6, some of the key features have not been addressed thus far. Here we performed single-cell DNA methylome sequencing for human preimplantation embryos and found that tens of thousands of genomic loci exhibited de novo DNA methylation. This finding indicates that genome-wide DNA methylation reprogramming during preimplantation development is a dynamic balance between strong global demethylation and drastic focused remethylation. Furthermore, demethylation of the paternal genome is much faster and thorough than that of the maternal genome. From the two-cell to the postimplantation stage, methylation of the paternal genome is consistently lower than that of the maternal genome. We also show that the genetic lineage of early blastomeres can be traced by DNA methylation analysis. Our work paves the way for deciphering the secrets of DNA methylation reprogramming in early human embryos.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bird, A. DNA methylation patterns and epigenetic memory. Genes Dev. 16, 6–21 (2002).
Smith, Z. D. & Meissner, A. DNA methylation: roles in mammalian development. Nat. Rev. Genet. 14, 204–220 (2013).
Hackett, J. A. & Surani, M. A. DNA methylation dynamics during the mammalian life cycle. Phil. Trans. R. Soc. Lond. B 368, 20110328 (2013).
Guo, H. et al. The DNA methylation landscape of human early embryos. Nature 511, 606–610 (2014).
Smith, Z. D. et al. DNA methylation dynamics of the human preimplantation embryo. Nature 511, 611–615 (2014).
Okae, H. et al. Genome-wide analysis of DNA methylation dynamics during early human development. PLoS Genet. 10, e1004868 (2014).
Fulka, H., Mrazek, M., Tepla, O. & Fulka, J. Jr. DNA methylation pattern in human zygotes and developing embryos. Reproduction 128, 703–708 (2004).
Molaro, A. et al. Sperm methylation profiles reveal features of epigenetic inheritance and evolution in primates. Cell 146, 1029–1041 (2011).
Vanneste, E. et al. Chromosome instability is common in human cleavage-stage embryos. Nat. Med. 15, 577–583 (2009).
Ambartsumyan, G. & Clark, A. T. Aneuploidy and early human embryo development. Hum. Mol. Genet. 17R1, R10–R15 (2008).
Fang, F. et al. Genomic landscape of human allele-specific DNA methylation. Proc. Natl. Acad. Sci. USA 109, 7332–7337 (2012).
Smallwood, S. A. et al. Single-cell genome-wide bisulfite sequencing for assessing epigenetic heterogeneity. Nat. Methods 11, 817–820 (2014).
Farlik, M. et al. Single-cell DNA methylome sequencing and bioinformatic inference of epigenomic cell-state dynamics. Cell Rep. 10, 1386–1397 (2015).
Guo, H. et al. Single-cell methylome landscapes of mouse embryonic stem cells and early embryos analyzed using reduced representation bisulfite sequencing. Genome Res. 23, 2126–2135 (2013).
Miura, F., Enomoto, Y., Dairiki, R. & Ito, T. Amplification-free whole-genome bisulfite sequencing by post-bisulfite adaptor tagging. Nucleic Acids Res. 40, e136 (2012).
Gao, F. et al. De novo DNA methylation during monkey pre-implantation embryogenesis. Cell Res. 27, 526–539 (2017).
Batzer, M. A. & Deininger, P. L. Alu repeats and human genomic diversity. Nat. Rev. Genet. 3, 370–379 (2002).
Hamada, H. et al. Allele-specific methylome and transcriptome analysis reveals widespread imprinting in the human placenta. Am. J. Hum. Genet. 99, 1045–1058 (2016).
Court, F. et al. Genome-wide parent-of-origin DNA methylation analysis reveals the intricacies of human imprinting and suggests a germline methylation-independent mechanism of establishment. Genome Res. 24, 554–569 (2014).
Mooijman, D., Dey, S. S., Boisset, J.-C., Crosetto, N. & van Oudenaarden, A. Single-cell 5hmC sequencing reveals chromosome-wide cell-to-cell variability and enables lineage reconstruction. Nat. Biotechnol. 34, 852–856 (2016).
Hanna, C. W. et al. Pervasive polymorphic imprinted methylation in the human placenta. Genome Res. 26, 756–767 (2016).
Hardarson, T. et al. A morphological and chromosomal study of blastocysts developing from morphologically suboptimal human pre-embryos compared with control blastocysts. Hum. Reprod. 18, 399–407 (2003).
Sathananthan, A. H. & Osianlis, T. Human embryo culture and assessment for the derivation of embryonic stem cells (ESC). Methods Mol. Biol. 584, 1–20 (2010).
Chian, R.-C., Lim, J.-H. & Tan, S.-L. State of the art in in-vitro oocyte maturation. Curr. Opin. Obstet. Gynecol. 16, 211–219 (2004).
Li, R., Qiao, J., Wang, L., Zhen, X. & Lu, Y. Serum progesterone concentration on day of HCG administration and IVF outcome. Reprod. Biomed. Online 16, 627–631 (2008).
Baltaci, V. et al. Relationship between embryo quality and aneuploidies. Reprod. Biomed. Online 12, 77–82 (2006).
Yan, L. et al. Single-cell RNA-Seq profiling of human preimplantation embryos and embryonic stem cells. Nat. Struct. Mol. Biol. 20, 1131–1139 (2013).
Li, R. et al. Retain singleton or twins? Multifetal pregnancy reduction strategies in triplet pregnancies with monochorionic twins. Eur. J. Obstet. Gynecol. Reprod. Biol. 167, 146–148 (2013).
Krueger, F. & Andrews, S. R. Bismark: a flexible aligner and methylation caller for Bisulfite-Seq applications. Bioinformatics 27, 1571–1572 (2011).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Trapnell, C., Pachter, L. & Salzberg, S. L. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 25, 1105–1111 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Borgel, J. et al. Targets and dynamics of promoter DNA methylation during early mouse development. Nat. Genet. 42, 1093–1100 (2010).
Xie, W. et al. Epigenomic analysis of multilineage differentiation of human embryonic stem cells. Cell 153, 1134–1148 (2013).
Orozco, L. D. et al. Epigenome-wide association of liver methylation patterns and complex metabolic traits in mice. Cell Metab. 21, 905–917 (2015).
Guo, W. et al. BS-Seeker2: a versatile aligning pipeline for bisulfite sequencing data. BMC Genomics 14, 774 (2013).
Liu, Y., Siegmund, K. D., Laird, P. W. & Berman, B. P. Bis-SNP: combined DNA methylation and SNP calling for Bisulfite-seq data. Genome Biol. 13, R61 (2012).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Acknowledgements
We thank W. Guo for his insightful discussion. L.Y., J.Q., and F.T. were supported by grants from the National Natural Science Foundation of China (81561138005, 31230047, 31522034, 31571544, 81521002, and 31625018) and the National Basic Research Program of China (2014CB943200 and 2017YFA0102702). J.Q. and F.T. were also supported by a grant from the Beijing Municipal Science and Technology Commission (D151100002415000). L.Y. was supported by a grant from the National High-Technology Research and Development Program (2015AA020407). The work was supported by the Beijing Advanced Innovation Center for Genomics at Peking University.
Author information
Authors and Affiliations
Contributions
F.T., J.Q., and L.Y. conceived the project. H.G., Y.R., Y.H., R.L., Y.L., X.F., Y.G., X.W., Y.W., P.L., J.Y., X.R., P.Y., Y.Y., Z.Y. and L.W. performed the experiments. P.Z., J.D., B.H. and H.G. conducted the bioinformatic analyses. F.T., J.Q., L.Y., H.G., P.Z., Y.R. and Y.H. wrote the manuscript with help from all of the authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Isolation of human early embryos for single-cell DNA methylome analysis.
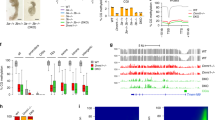
a, Morphologies of oocytes and human early embryos at different developmental stages. Scale bar, 100 μm. b–f, The polar body and nuclear region of MII oocytes were biopsied by laser-assisted micromanipulation. Scale bar, 100 μm. b, Bright-field image of an MII oocyte. c, Staining with Hoechst 33342 dye to confirm the absence of genomic contaminants and reveal the nuclear region (arrow) and the first polar body (triangle) of the MII oocyte. d, Bright-field image of the aspiration of a polar body of an MII oocyte (triangle). e, Bright-field image of aspiration of the nuclear region of an MII oocyte (arrow). f, Hoechst 33342 staining showing aspiration of the nuclear region of an MII oocyte (arrow). g, Heat map showing the pairwise correlations of euploid single cells from different developmental stages. Unsupervised hierarchical clustering indicated that there were at least three major clusters, representing the sperm and male pronuclei (cluster I), GV and MII oocytes and female pronuclei (cluster II), and the cells from cleavage-stage embryos (cluster III), corresponding to the hypermethylation, intermediate methylation, and hypomethylation features of the different cell types.
Supplementary Figure 2 General quality control and sequencing statistics of the single-cell DNA methylome sequencing samples.
a, The number of CpG sites with at least one-, three-, and fivefold coverage across the single-cell samples. The “Mixed” label represents blastocyst cells that we did not separate manually into the ICM and TE (but directly dissociated the whole blastocyst into single-cell suspension and randomly picked single cells from them). The box plot represents the median, 25% and 75% quantiles; whiskers indicate 1.5 times the IQR (interquartile range). b, The CpG coverage (1×) at each developmental stage and the CpG coverage of single-cell and bulk samples at the same stages were merged together. c, Box plot showing the percentage of the digitized CpG methylation output in the single-cell DNA methylome sequencing samples; greater than 90% of CpG sites at each developmental stage had a methylation level of either fully methylated or unmethylated. The box plot represents the median, 25% and 75% quantiles; whiskers indicate 1.5 times the IQR. d, The general quality controls of all samples across different developmental stages; aneuploid samples and samples with low sequencing quality were excluded from subsequent analyses. Low-quality samples were determined by performing pairwise correlation analysis of all single cells at the same developmental stage. e, Pairwise comparison at different developmental stages showing the heterogeneity across intra- and inter-embryos; eight-cell stage embryo and morula were excluded from this analysis owing to the inadequate number of samples; n.s., not significant (Student's t test). The box plot represents the median, 25% and 75% quantiles; whiskers indicate 1.5 times the IQR.
Supplementary Figure 3 The copy number variation landscapes of human early embryos deduced from the DNA methylome dataset.
a, The normal sate (light blue), gain (red), and loss (cyan) of autosomes at different developmental stages. The “Mixed” label represents blastocyst cells that we did not separate manually into the ICM and TE (but directly dissociated the whole blastocyst into single-cell suspension and randomly picked single cells from them). b, Several representative examples of euploid and aneuploid single-cell samples; chromosomes with an aberrant copy number are highlighted in the purple boxes. c,d, Histograms of the statistics regarding the number of embryos (c) or single cells (d) with CNVs on each chromosome.
Supplementary Figure 4 Effects of chromosome copy number variation on DNA methylation.
a, Density distribution of DNA methylation for ICM samples grouped by chromosome copy number. b, Density distribution of DNA methylation for TE samples grouped by chromosome copy number. c, Box plots show the DNA methylation distribution of 35 individual TE cells isolated from the same embryo and grouped by chromosome copy number; P values were calculated by t test. The box plot represents the median, 25% and 75% quantiles; whiskers indicate 1.5 times the IQR.
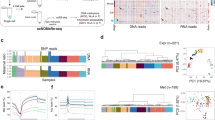
Supplementary Figure 5 Main features of de novo DNA methylation in human early embryos.
a, The representative regions that were de novo methylated from the earlier stage to the later stage. White open circles represent unmethylated CpG dinucleotides, whereas black filled circles represent methylated CpG dinucleotides. b, Histogram showing the numbers of increasing (magenta) and decreasing (cyan) CpG sites across consecutive stages. c, Enrichment analysis of de novo–methylated tiles on different genomic elements from the early male to mid-pronuclear stage and the four- to eight-cell stage (hypergeometric test).
Supplementary Figure 6 Select representative loci showing the hypermethylated maternal genome and hypomethylated paternal genome at different developmental stages.
White open circles represent unmethylated CpG dinucleotides, whereas black filled circles represent methylated CpG dinucleotides. Heterozygous SNPs were used to trace the parental genome.
Supplementary Figure 7 Parental-specific methylation and allele-specific expression in blastocyst-stage embryos.
Scatterplot showing ASE (x axis) and parental-specific methylation at the promoter regions of the corresponding genes (y axis), both presented as maternal scores minus paternal scores. The histogram shows the frequency distribution of methylation (M – P) indicating hypermethylation of the maternal genome. Genes showed both parental-specific methylation at promoters and ASE was labeled in blue. Genes labeled more than once were presented by different embryos (n = 4). ICM samples were displayed in a, and TE samples are displayed in b.
Supplementary Figure 8 The main features of the gamete-specific methylation regions (DMRs).
a, Length distributions of sperm-specific (red) and MII oocyte–specific (blue) DMRs. b, Gene ontology analysis of the genes with promoters located in the gamete-specific DMRs. c,d, Two representative regions showing the longest sperm-specific DMRs (c) and one representative maternal imprinting region, PEG3 (d) (red, CpG sites with methylation greater than 50%; blue, CpG sites with methylation level less than 50%). e,f, Enrichment analysis of the sperm-specific (e) and MII oocyte–specific (f) DMRs on different genomic elements (hypergeometric test).
Supplementary Figure 9 DNA methylation of all 31 known imprinting regions in preimplantation euploid cells and postimplantation tissues Heat map showing the DNA methylation of all 31 known imprinting regions in both preimplantation euploid cells and postimplantation tissues.
Colors indicate methylation levels from low (cyan) to high (red). White indicates missing (undetected) values. The “Mixed” label represents blastocyst cells that we did not separate manually into the ICM and TE (but directly dissociated the whole blastocyst into single-cell suspension and randomly picked single cells from them).
Supplementary Figure 10 Schematic of the chromosome-level DNA methylation discrepancies from the zygotes to four-cell embryos.
Three distribution types of DNA methylation for one genomic region in each cell from the zygote to four-cell embryos, indicating either “high–low” or “intermediate–intermediate” DNA methylation patterns for the two sibling cells in a four-cell embryo from a mother's cell at the two-cell stage.
Supplementary information
General statistics of all samples analyzed in this study
Supplementary Table 1.
Sequencing statistics of all samples used in this study
Supplementary Table 2.
CpG coverage information for all bisulfite sequencing samples with 1×, 3× and 5× read depth
Supplementary Table 3.
Parental specifically methylated regions determined at the blastocyst stage
Supplementary Table 4.
Oligonucleotide sequences used in this study
Supplementary Table 5.
Rights and permissions
About this article
Cite this article
Zhu, P., Guo, H., Ren, Y. et al. Single-cell DNA methylome sequencing of human preimplantation embryos. Nat Genet 50, 12–19 (2018). https://doi.org/10.1038/s41588-017-0007-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-017-0007-6
This article is cited by
-
Single-cell multi-omics profiling of human preimplantation embryos identifies cytoskeletal defects during embryonic arrest
Nature Cell Biology (2024)
-
Novel MEI1 mutations cause chromosomal and DNA methylation abnormalities leading to embryonic arrest and implantation failure
Molecular Genetics and Genomics (2024)
-
A genomic mutational constraint map using variation in 76,156 human genomes
Nature (2024)
-
Whole-genome transcriptome and DNA methylation dynamics of pre-implantation embryos reveal progression of embryonic genome activation in buffaloes
Journal of Animal Science and Biotechnology (2023)
-
Methylomics and cancer: the current state of methylation profiling and marker development for clinical care
Cancer Cell International (2023)