Abstract
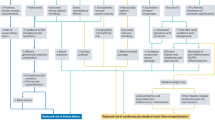
Management of fluid overload is one of the most challenging problems in the care of critically ill patients with oliguric acute kidney injury. Various clinical practice guidelines support fluid removal using ultrafiltration during kidney replacement therapy. However, ultrafiltration is associated with considerable risks. Emerging evidence from observational studies suggests that both slow and fast rates of net fluid removal (that is, net ultrafiltration (UFNET)) during continuous kidney replacement therapy are associated with increased mortality compared with moderate UFNET rates. In addition, fast UFNET rates are associated with an increased risk of cardiac arrhythmias. Experimental studies in patients with kidney failure who were treated with intermittent haemodialysis suggest that fast UFNET rates are also associated with ischaemic injury to the heart, brain, kidney and gut. The UFNET rate should be prescribed based on patient body weight in millilitres per kilogramme per hour with close monitoring of patient haemodynamics and fluid balance. Dialysate cooling and sodium modelling may prevent haemodynamic instability and facilitate large volumes of fluid removal in patients with kidney failure who are treated with intermittent haemodialysis, but the effects of this strategy on organ injury are less well studied in critically ill patients treated with continuous kidney replacement therapy. Randomized trials are required to examine whether moderate UFNET rates are associated with a reduced risk of haemodynamic instability, organ injury and improved outcomes in critically ill patients.
Key points
-
The rate of net ultrafiltration should be based on patient body weight (ml/kg/h) rather than on absolute volumes (ml/h).
-
Epidemiological studies in critically ill patients with acute kidney injury and in patients with kidney failure suggest that high and low net ultrafiltration (UFNET) rates are associated with increased mortality compared with moderate UFNET rates.
-
Intradialytic hypotension during UFNET is common with both intermittent haemodialysis and continuous kidney replacement therapy.
-
During UFNET, careful attention should be paid to patient haemodynamics, with frequent assessment of end organ perfusion and function.
-
Randomized clinical trials are required to determine whether moderate UFNET rates are associated with improved patient outcomes compared with slow or faster UFNET rates.
-
Further research is required to investigate the feasibility and efficacy of interventions such as dialysate cooling, sodium profiling, remote ischaemic preconditioning and passive intradialytic exercise for organ protection during ultrafiltration in critically ill patients.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Alwall, N. On the artificial kidney; apparatus for dialysis of the blood in vivo. Acta Med. Scand. 128, 317–325 (1947).
Balakumar, V. et al. Both positive and negative fluid balance may be associated with reduced long-term survival in the critically Ill. Crit. Care Med. 45, e749–e757 (2017).
Vaara, S. T. et al. Fluid overload is associated with an increased risk for 90-day mortality in critically ill patients with renal replacement therapy: data from the prospective FINNAKI study. Crit. Care 16, R197 (2012).
Mitchell, K. H. et al. Volume overload: prevalence, risk factors, and functional outcome in survivors of septic shock. Ann. Am. Thorac. Soc. 12, 1837–1844 (2015).
Woodward, C. W. et al. Fluid overload associates with major adverse kidney events in critically Ill patients with acute kidney injury requiring continuous renal replacement therapy. Crit. Care Med. 47, e753–e760 (2019).
Bellomo, R. et al. An observational study fluid balance and patient outcomes in the randomized evaluation of normal vs. augmented level of replacement therapy trial. Crit. Care Med. 40, 1753–1760 (2012).
Bouchard, J. et al. Fluid accumulation, survival and recovery of kidney function in critically ill patients with acute kidney injury. Kidney Int. 76, 422–427 (2009).
Rosner, M. H. et al. Indications and management of mechanical fluid removal in critical illness. Br. J. Anaesth. 113, 764–771 (2014).
Sutherland, S. M. et al. Fluid overload and mortality in children receiving continuous renal replacement therapy: the prospective pediatric continuous renal replacement therapy registry. Am. J. Kidney Dis. 55, 316–325 (2010).
Goldstein, S. L. et al. Pediatric patients with multi-organ dysfunction syndrome receiving continuous renal replacement therapy. Kidney Int. 67, 653–658 (2005).
Bellomo, R. et al. Intensity of continuous renal-replacement therapy in critically ill patients. N. Engl. J. Med. 361, 1627–1638 (2009).
Palevsky, P. M. et al. Intensity of renal support in critically ill patients with acute kidney injury. N. Engl. J. Med. 359, 7–20 (2008).
Murugan, R. et al. Net ultrafiltration intensity and mortality in critically ill patients with fluid overload. Crit. Care 22, 223 (2018).
Murugan, R. et al. Net ultrafiltration prescription and practice among critically ill patients receiving renal replacement therapy: a multinational survey of critical care practitioners. Crit. Care Med. 48, e87–e97 (2020).
Kidney Disease Improving Global Outcomes (KDIGO) Workgroup. Clinical practice guideline for acute kidney injury. Kidney Int. Suppl. 2, 1–138 (2012).
Modem, V., Thompson, M., Gollhofer, D., Dhar, A. V. & Quigley, R. Timing of continuous renal replacement therapy and mortality in critically ill children*. Crit. Care Med. 42, 943–953 (2014).
Costanzo, M. R. et al. Extracorporeal ultrafiltration for fluid overload in heart failure: current status and prospects for further research. J. Am. Coll. Cardiol. 69, 2428–2445 (2017).
Kazory, A. More efficient sodium removal by ultrafiltration compared to diuretics in acute heart failure; underexplored and overstated. Blood Purif. 42, 279–281 (2016).
Kazory, A. Ultrafiltration therapy for heart failure: balancing likely benefits against possible risks. Clin. J. Am. Soc. Nephrol. 11, 1463–1471 (2016).
Ronco, C., Ricci, Z., Bellomo, R. & Bedogni, F. Extracorporeal ultrafiltration for the treatment of overhydration and congestive heart failure. Cardiology 96, 155–168 (2001).
Neri, M. et al. Nomenclature for renal replacement therapy in acute kidney injury: basic principles. Crit. Care 20, 318 (2016).
Ronco, C., Ghezzi, P. M., Brendolan, A., Crepaldi, C. & La Greca, G. The haemodialysis system: basic mechanisms of water and solute transport in extracorporeal renal replacement therapies. Nephrol. Dial. Transpl. 13, 3–9 (1998).
Clark, W. R., Macias, W. L., Molitoris, B. A. & Wang, N. H. Plasma protein adsorption to highly permeable hemodialysis membranes. Kidney Int. 48, 481–488 (1995).
Claure-Del Granado, R. et al. Effluent volume in continuous renal replacement therapy overestimates the delivered dose of dialysis. Clin. J. Am. Soc. Nephrol. 6, 467–475 (2011).
Feldhoff, P., Turnham, T. & Klein, E. Effect of plasma proteins on the sieving spectra of hemofilters. Artif. Organs 8, 186–192 (1984).
Joannidis, M. & Oudemans-van Straaten, H. M. Clinical review: patency of the circuit in continuous renal replacement therapy. Crit. Care 11, 218 (2007).
Ficheux, A., Ronco, C., Brunet, P. & Argiles, A. The ultrafiltration coefficient: this old ‘grand inconnu’ in dialysis. Nephrol. Dial. Transpl. 30, 204–208 (2015).
Ronco, C. et al. A new scintigraphic method to characterize ultrafiltration in hollow fiber dialyzers. Kidney Int. 41, 1383–1393 (1992).
Lauer, A. et al. Continuous arteriovenous hemofiltration in the critically ill patient. clinical use and operational characteristics. Ann. Intern. Med. 99, 455–460 (1983).
Koomans, H. A., Geers, A. B. & Mees, E. J. Plasma volume recovery after ultrafiltration in patients with chronic renal failure. Kidney Int. 26, 848–854 (1984).
Douvris, A. et al. Mechanisms for hemodynamic instability related to renal replacement therapy: a narrative review. Intensive Care Med. 45, 1333–1346 (2019).
Douvris, A. et al. Interventions to prevent hemodynamic instability during renal replacement therapy in critically ill patients: a systematic review. Crit. Care 22, 41 (2018).
Akhoundi, A. et al. Incidence of adverse events during continuous renal replacement therapy. Blood Purif. 39, 333–339 (2015).
Shawwa, K. et al. Hypotension within one-hour from starting CRRT is associated with in-hospital mortality. J. Crit. Care 54, 7–13 (2019).
Uchino, S. et al. Continuous renal replacement therapy: a worldwide practice survey. The beginning and ending supportive therapy for the kidney (B.E.S.T. kidney) investigators. Intensive Care Med. 33, 1563–1570 (2007).
Vinsonneau, C. et al. Continuous venovenous haemodiafiltration versus intermittent haemodialysis for acute renal failure in patients with multiple-organ dysfunction syndrome: a multicentre randomised trial. Lancet 368, 379–385 (2006).
Bitker, L. et al. Prevalence and risk factors of hypotension associated with preload-dependence during intermittent hemodialysis in critically ill patients. Crit. Care 20, 44 (2016).
Schortgen, F. et al. Hemodynamic tolerance of intermittent hemodialysis in critically ill patients: usefulness of practice guidelines. Am. J. Respir. Crit. Care Med. 162, 197–202 (2000).
Tanguay, T. A., Jensen, L. & Johnston, C. Predicting episodes of hypotension by continuous blood volume monitoring among critically ill patients in acute renal failure on intermittent hemodialysis. Dynamics 18, 19–24 (2007).
Tonelli, M. et al. Blood volume monitoring in intermittent hemodialysis for acute renal failure. Kidney Int. 62, 1075–1080 (2002).
Silversides, J. A. et al. Fluid balance, intradialytic hypotension, and outcomes in critically ill patients undergoing renal replacement therapy: a cohort study. Crit. Care 18, 624 (2014).
Murugan, R. et al. Precision fluid management in continuous renal replacement therapy. Blood Purif. 42, 266–278 (2016).
Pirkle, J. L. Jr. et al. Effects of weight-based ultrafiltration rate limits on intradialytic hypotension in hemodialysis. Hemodial. Int. 22, 270–278 (2018).
Mottes, T. A., Goldstein, S. L. & Basu, R. K. Process based quality improvement using a continuous renal replacement therapy dashboard. BMC Nephrol. 20, 17 (2019).
Rewa, O. G. et al. Quality of care and safety measures of acute renal replacement therapy: workgroup statements from the 22nd acute disease quality initiative (ADQI) consensus conference. J. Crit. Care 54, 52–57 (2019).
Hoste, E. A. et al. Four phases of intravenous fluid therapy: a conceptual model. Br. J. Anaesth. 113, 740–747 (2014).
Murugan, R. et al. Association of net ultrafiltration rate with mortality among critically ill adults with acute kidney injury receiving continuous venovenous hemodiafiltration: a secondary analysis of the randomized evaluation of normal vs augmented level (RENAL) of renal replacement therapy trial. JAMA Netw. Open 2, e195418 (2019).
Naorungroj, T. et al. Early net ultrafiltration rate and mortality in critically ill patients receiving continuous renal replacement therapy. Nephrol. Dial. Transpl. https://doi.org/10.1093/ndt/gfaa032 (2020).
Edrees, F. Y., Katari, S., Baty, J. D. & Vijayan, A. A Pilot study evaluating the effect of cooler dialysate temperature on hemodynamic stability during prolonged intermittent renal replacement therapy in acute kidney injury. Crit. Care Med. 47, e74–e80 (2019).
Robert, R. et al. Benefits of an early cooling phase in continuous renal replacement therapy for ICU patients. Ann. Intensive Care 2, 40 (2012).
Oliver, M. J., Edwards, L. J. & Churchill, D. N. Impact of sodium and ultrafiltration profiling on hemodialysis-related symptoms. J. Am. Soc. Nephrol. 12, 151–156 (2001).
Song, J. H., Lee, S. W., Suh, C. K. & Kim, M. J. Time-averaged concentration of dialysate sodium relates with sodium load and interdialytic weight gain during sodium-profiling hemodialysis. Am. J. Kidney Dis. 40, 291–301 (2002).
Paganini, E. P., Sandy, D., Moreno, L., Kozlowski, L. & Sakai, K. The effect of sodium and ultrafiltration modelling on plasma volume changes and haemodynamic stability in intensive care patients receiving haemodialysis for acute renal failure: a prospective, stratified, randomized, cross-over study. Nephrol. Dial. Transpl. 11, 32–37 (1996).
Lynch, K. E. et al. Sodium modelling to reduce intradialytic hypotension during haemodialysis for acute kidney injury in the intensive care unit. Nephrology 21, 870–877 (2016).
Osman, D. et al. Cardiac filling pressures are not appropriate to predict hemodynamic response to volume challenge. Crit. Care Med. 35, 64–68 (2007).
Marik, P. E., Monnet, X. & Teboul, J. L. Hemodynamic parameters to guide fluid therapy. Ann. Intensive Care 1, 1 (2011).
Santoro, A. et al. Blood volume controlled hemodialysis in hypotension-prone patients: a randomized, multicenter controlled trial. Kidney Int. 62, 1034–1045 (2002).
Piccoli, A., Rossi, B., Pillon, L. & Bucciante, G. A new method for monitoring body fluid variation by bioimpedance analysis: the RXc graph. Kidney Int. 46, 534–539 (1994).
Kalantari, K., Chang, J. N., Ronco, C. & Rosner, M. H. Assessment of intravascular volume status and volume responsiveness in critically ill patients. Kidney Int. 83, 1017–1028 (2013).
Myatchin, I., Abraham, P. & Malbrain, M. Bio-electrical impedance analysis in critically ill patients: are we ready for prime time? J. Clin. Monit. Comput. 34, 401–410 (2019).
Basso, F. et al. Fluid management in the intensive care unit: bioelectrical impedance vector analysis as a tool to assess hydration status and optimal fluid balance in critically ill patients. Blood Purif. 36, 192–199 (2013).
Hise, A. & Gonzalez, M. C. Assessment of hydration status using bioelectrical impedance vector analysis in critical patients with acute kidney injury. Clin. Nutr. 37, 695–700 (2018).
Jones, S. L. et al. Bioelectrical impedance vector analysis in critically ill patients: a prospective, clinician-blinded investigation. Crit. Care 19, 290 (2015).
Razzera, E. L. et al. Parameters of bioelectrical impedance are good predictors of nutrition risk, length of stay, and mortality in critically Ill patients: a prospective cohort study. JPEN 44, 849–854 (2019).
Rochwerg, B. et al. Assessment of postresuscitation volume status by bioimpedance analysis in patients with sepsis in the intensive care unit: a pilot observational study. Can. Respir. J. 2016, 8671742 (2016).
Samoni, S. et al. Impact of hyperhydration on the mortality risk in critically ill patients admitted in intensive care units: comparison between bioelectrical impedance vector analysis and cumulative fluid balance recording. Crit. Care 20, 95 (2016).
Chen, H., Wu, B., Gong, D. & Liu, Z. Fluid overload at start of continuous renal replacement therapy is associated with poorer clinical condition and outcome: a prospective observational study on the combined use of bioimpedance vector analysis and serum N-terminal pro-B-type natriuretic peptide measurement. Crit. Care 19, 135 (2015).
Park, K. H., Shin, J. H., Hwang, J. H. & Kim, S. H. Utility of volume assessment using bioelectrical impedance analysis in critically Ill patients receiving continuous renal replacement therapy: a prospective observational study. Korean J. Crit. Care Med. 32, 256–264 (2017).
Rhee, H. et al. Use of multifrequency bioimpedance analysis in male patients with acute kidney injury who are undergoing continuous veno-venous hemodiafiltration. PLoS One 10, e0133199 (2015).
Recommended methods for measurement of red-cell and plasma volume. International Committee for Standardization in Haematology. J. Nucl. Med. 21, 793–800 (1980).
Dworkin, H. J., Premo, M. & Dees, S. Comparison of red cell and whole blood volume as performed using both chromium-51-tagged red cells and iodine-125-tagged albumin and using I-131-tagged albumin and extrapolated red cell volume. Am. J. Med. Sci. 334, 37–40 (2007).
Hocking, K. M. et al. Peripheral i.v. analysis (PIVA) of venous waveforms for volume assessment in patients undergoing haemodialysis. Br. J. Anaesth. 119, 1135–1140 (2017).
Miles, M. et al. Peripheral intravenous volume analysis (PIVA) for quantitating volume overload in patients hospitalized with acute decompensated heart failure — a pilot study. J. Card. Fail. 24, 525–532 (2018).
Sileshi, B. et al. Peripheral venous waveform analysis for detecting early hemorrhage: a pilot study. Intensive Care Med. 41, 1147–1148 (2015).
Hocking, K. M. et al. Peripheral venous waveform analysis for detecting hemorrhage and iatrogenic volume overload in a porcine model. Shock 46, 447–452 (2016).
Tehranian, S., Shawwa, K., Kashani, K.B. Net ultrafiltration rate and its impact on mortality in patients with acute kidney injury receiving continuous renal replacement therapy. Clin. Kidney J. https://doi.org/10.1093/ckj/sfz179 (2019).
Naorungroj, T. et al. Mediators of the impact of hourly net ultrafiltration rate on mortality in critically ill patients receiving continuous renal replacement therapy. Crit. Care Med. 48, e934–e942 (2020).
Flythe, J. E., Curhan, G. C. & Brunelli, S. M. Shorter length dialysis sessions are associated with increased mortality, independent of body weight. Kidney Int. 83, 104–113 (2013).
Flythe, J. E., Kimmel, S. E. & Brunelli, S. M. Rapid fluid removal during dialysis is associated with cardiovascular morbidity and mortality. Kidney Int. 79, 250–257 (2011).
Kim, T. W. et al. Association of ultrafiltration rate with mortality in incident hemodialysis patients. Nephron 139, 13–22 (2018).
Movilli, E. et al. Association between high ultrafiltration rates and mortality in uraemic patients on regular haemodialysis. A 5-year prospective observational multicentre study. Nephrol. Dial. Transpl. 22, 3547–3552 (2007).
Saran, R. et al. Longer treatment time and slower ultrafiltration in hemodialysis: associations with reduced mortality in the DOPPS. Kidney Int. 69, 1222–1228 (2006).
Kramer, H. et al. Ultrafiltration rate thresholds in maintenance hemodialysis: an NKF-KDOQI controversies report. Am. J. Kidney Dis. 68, 522–532 (2016).
Mahmoud, H., Forni, L. G., McIntyre, C. W. & Selby, N. M. Myocardial stunning occurs during intermittent haemodialysis for acute kidney injury. Intensive Care Med. 43, 942–944 (2017).
Slessarev, M., Salerno, F., Ball, I. M. & McIntyre, C. W. Continuous renal replacement therapy is associated with acute cardiac stunning in critically ill patients. Hemodial. Int. 23, 325–332 (2019).
Vieillard-Baron, A. et al. Actual incidence of global left ventricular hypokinesia in adult septic shock. Crit. Care Med. 36, 1701–1706 (2008).
Buchanan, C. et al. Intradialytic cardiac magnetic resonance imaging to assess cardiovascular responses in a short-term trial of hemodiafiltration and hemodialysis. J. Am. Soc. Nephrol. 28, 1269–1277 (2017).
Veenstra, G. et al. Ultrafiltration rate is an important determinant of microcirculatory alterations during chronic renal replacement therapy. BMC Nephrol. 18, 71 (2017).
Bemelmans, R. H. et al. Changes in the volume status of haemodialysis patients are reflected in sublingual microvascular perfusion. Nephrol. Dial. Transpl. 24, 3487–3492 (2009).
Burton, J. O., Jefferies, H. J., Selby, N. M. & McIntyre, C. W. Hemodialysis-induced repetitive myocardial injury results in global and segmental reduction in systolic cardiac function. Clin. J. Am. Soc. Nephrol. 4, 1925–1931 (2009).
Burton, J. O., Jefferies, H. J., Selby, N. M. & McIntyre, C. W. Hemodialysis-induced cardiac injury: determinants and associated outcomes. Clin. J. Am. Soc. Nephrol. 4, 914–920 (2009).
McIntyre, C. W. Effects of hemodialysis on cardiac function. Kidney Int. 76, 371–375 (2009).
Polinder-Bos, H. A. et al. Hemodialysis induces an acute decline in cerebral blood flow in elderly patients. J. Am. Soc. Nephrol. 29, 1317–1325 (2018).
Findlay, M. D. et al. Investigating the relationship between cerebral blood flow and cognitive function in hemodialysis patients. J. Am. Soc. Nephrol. 30, 147–158 (2019).
Marants, R., Qirjazi, E., Grant, C. J., Lee, T. Y. & McIntyre, C. W. Renal perfusion during hemodialysis: intradialytic blood flow decline and effects of dialysate cooling. J. Am. Soc. Nephrol. 30, 1086–1095 (2019).
McIntyre, C. W. et al. Circulating endotoxemia: a novel factor in systemic inflammation and cardiovascular disease in chronic kidney disease. Clin. J. Am. Soc. Nephrol. 6, 133–141 (2011).
Harrison, L. E., Selby, N. M. & McIntyre, C. W. Central venous oxygen saturation: a potential new marker for circulatory stress in haemodialysis patients? Nephron Clin. Pract. 128, 57–60 (2014).
Selby, N. M., Lambie, S. H., Camici, P. G., Baker, C. S. & McIntyre, C. W. Occurrence of regional left ventricular dysfunction in patients undergoing standard and biofeedback dialysis. Am. J. Kidney Dis. 47, 830–841 (2006).
Burton, J. O., Korsheed, S., Grundy, B. J. & McIntyre, C. W. Hemodialysis-induced left ventricular dysfunction is associated with an increase in ventricular arrhythmias. Ren. Fail. 30, 701–709 (2008).
Eldehni, M. T. & McIntyre, C. W. Are there neurological consequences of recurrent intradialytic hypotension? Semin. Dial. 25, 253–256 (2012).
Eldehni, M. T., Odudu, A. & McIntyre, C. W. Randomized clinical trial of dialysate cooling and effects on brain white matter. J. Am. Soc. Nephrol. 26, 957–965 (2015).
Gleeson, P. J. C. et al. Determinants of renal recovery and mortality in patients undergoing continuous renal replacement therapy in the ICU. Intensive Care Med. Exp. 3, A54 (2015).
Sjolund, J. et al. Diuretics, limited ultrafiltration, and residual renal function in incident hemodialysis patients: a case series. Semin. Dial. 29, 410–415 (2016).
Jakob, S. M., Ruokonen, E., Vuolteenaho, O., Lampainen, E. & Takala, J. Splanchnic perfusion during hemodialysis: evidence for marginal tissue perfusion. Crit. Care Med. 29, 1393–1398 (2001).
Ribitsch, W. et al. Increased hepato-splanchnic vasoconstriction in diabetics during regular hemodialysis. PLoS One 10, e0145411 (2015).
Yu, A. W. et al. Splanchnic erythrocyte content decreases during hemodialysis: a new compensatory mechanism for hypovolemia. Kidney Int. 51, 1986–1990 (1997).
Assa, S. et al. Effect of isolated ultrafiltration and isovolemic dialysis on myocardial perfusion and left ventricular function assessed with (13)N-NH3 positron emission tomography and echocardiography. Am. J. Physiol. Ren. Physiol 314, F445–F452 (2018).
van der Sande, F. M., Cheriex, E. C., van Kuijk, W. H. & Leunissen, K. M. Effect of dialysate calcium concentrations on intradialytic blood pressure course in cardiac-compromised patients. Am. J. Kidney Dis. 32, 125–131 (1998).
van Kuijk, W. H., Mulder, A. W., Peels, C. H., Harff, G. A. & Leunissen, K. M. Influence of changes in ionized calcium on cardiovascular reactivity during hemodialysis. Clin. Nephrol. 47, 190–196 (1997).
Bersin, R. M., Chatterjee, K. & Arieff, A. I. Metabolic and hemodynamic consequences of sodium bicarbonate administration in patients with heart disease. Am. J. Med. 87, 7–14 (1989).
Assa, S. et al. Hemodialysis-induced regional left ventricular systolic dysfunction and inflammation: a cross-sectional study. Am. J. Kidney Dis. 64, 265–273 (2014).
Friedrich, B. et al. Acute effects of hemodialysis on cytokine transcription profiles: evidence for C-reactive protein-dependency of mediator induction. Kidney Int. 70, 2124–2130 (2006).
Barbar, S. D. et al. Timing of renal-replacement therapy in patients with acute kidney injury and sepsis. N. Engl. J. Med. 379, 1431–1442 (2018).
Gaudry, S. et al. Initiation strategies for renal-replacement therapy in the intensive care unit. N. Engl. J. Med. 375, 122–133 (2016).
Zarbock, A. et al. Effect of early vs delayed initiation of renal replacement therapy on mortality in critically ill patients with acute kidney injury: the ELAIN randomized clinical trial. JAMA 315, 2190–2199 (2016).
Canaud, B. & Cohen, E. P. Initiation of renal-replacement therapy in the intensive care unit. N. Engl. J. Med. 375, 1901 (2016).
National Kidney Foundation. KDOQI clinical practice guideline for hemodialysis adequacy: 2015 update. Am. J. Kidney Dis. 66, 884–930 (2015).
Di Maggio, A., Basile, C. & Scatizzi, A. Plasma volume changes induced by sequential ultrafiltration-hemodialysis and sequential hemodialysis-ultrafiltration. Int. J. Artif. Organs 10, 291–294 (1987).
Crowley, L. E. & McIntyre, C. W. Remote ischaemic conditioning-therapeutic opportunities in renal medicine. Nat. Rev. Nephrol. 9, 739–746 (2013).
Salerno, F. R., Crowley, L. E., Odudu, A. & McIntyre, C. W. Remote ischemic preconditioning protects against hemodialysis-induced cardiac injury. Kidney Int. Rep. 5, 99–103 (2020).
Penny, J. D. et al. Intradialytic exercise preconditioning: an exploratory study on the effect on myocardial stunning. Nephrol. Dial. Transpl. 34, 1917–1923 (2019).
Pajewski, R., Gipson, P. & Heung, M. Predictors of post-hospitalization recovery of renal function among patients with acute kidney injury requiring dialysis. Hemodial. Int. 22, 66–73 (2018).
Assimon, M. M., Wenger, J. B., Wang, L. & Flythe, J. E. Ultrafiltration rate and mortality in maintenance hemodialysis patients. Am. J. Kidney Dis. 68, 911–922 (2016).
Lee, Y. et al. Ultrafiltration rate effects declines in residual kidney function in hemodialysis patients. Am. J. Nephrol. 50, 481–488 (2019).
Bos, W. J. et al. Cardiac and hemodynamic effects of hemodialysis and ultrafiltration. Am. J. Kidney Dis. 35, 819–826 (2000).
Stiller, S. et al. Less symptomatic hypotension using blood volume controlled ultrafiltration. ASAIO Trans. 37, M139–141 (1991).
Yartsev, A. Ultrafiltration as a mechanism for fluid removal (Deranged Physiology, 2015).
McIntyre, C. W. et al. Hemodialysis-induced cardiac dysfunction is associated with an acute reduction in global and segmental myocardial blood flow. Clin. J. Am. Soc. Nephrol. 3, 19–26 (2008).
Acknowledgements
Partial support for R.M.’s work was provided by the National Institute of Diabetes and Digestive and Kidney Diseases (5R01DK106256).
Author information
Authors and Affiliations
Contributions
R.M. was responsible for the concept of the review and the design and drafting of the manuscript. J.A.K., R.B. and P.M.P. contributed to critical revision of the manuscript and important intellectual content.
Corresponding author
Ethics declarations
Competing interests
R.M. reports receiving grants and personal fees from La Jolla Inc; grants from Bioporto, Inc, and the National Institute of Diabetes and Digestive and Kidney Diseases; and personal fees from Beckman Coulter and AM Pharma, Inc, outside the submitted work. R.B. reports receiving grants from Baxter International outside the submitted work. P.M.P. reported receiving personal fees from Novartis, GE Healthcare, HealthSpan Dx, and Baxter International and grants from Dascena outside the submitted work. J.A.K. reports receiving personal fees from NxStage and grants and personal fees from Baxter International.
Additional information
Peer review information
Nature Reviews Nephrology thanks Bernard Canaud, Stuart Goldstein and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Glossary
- Ultrafiltration
-
The process of filtration of plasma water from blood using a medium such as a haemofiltration membrane that is fine enough to retain colloidal particles and large molecules.
- Net ultrafiltration (UFNET) rate
-
The rate at which extracellular fluid volume is removed from the patient per unit of time during ultrafiltration. The UFNET rate is calculated by excluding the replacement fluid volume from the ultrafiltrate and so reflects the true volume of fluid removed from the patient during continuous kidney replacement therapy. During intermittent haemodialysis, replacement fluids are not administered, so the ultrafiltration rate is the same as the UFNET rate.
- Ultrafiltration rate
-
The rate at which plasma water is removed from blood per unit of time. During continuous kidney replacement therapy, the ultrafiltrate typically includes the volume of replacement fluids.
- Haemofiltration
-
The process of removing solutes from the blood using convection. Solutes are passively removed by solvent drag that occurs during ultrafiltration owing to increased transmembrane pressure. Replacement fluids are used to increase transmembrane pressure during convection. Dialysate is not used during haemofiltration.
- Haemodiafiltration
-
The process of removing solutes from the blood using a combination of convection and diffusion. Diffusive clearance is achieved with the use of a dialysate in which solutes are transported across the haemofiltration membrane owing to the concentration gradient. Both replacement fluids and dialysate are used.
- Gibbs–Donnan effect
-
Unequal distribution of permeant charged ions (such as chloride) on either side of a semipermeable membrane occurring because of the presence of impermeant charged ions (for example, plasma proteins). The electrochemical gradients caused by the unequal distribution of charged ions produces a transmembrane potential difference.
- Starling forces
-
Forces governing the passive exchange of water between the capillary microcirculation and the interstitial fluid. These forces not only determine the directionality of net water movement between two different compartments but also the rate at which water exchange occurs.
- Tonicity
-
The ability of an extracellular solution to make water move into or out of a cell by osmosis. Tonicity is a measure of the effective osmotic pressure gradient.
- Osmolarity
-
A measure of the total concentration of all solutes present in the solution.
- Stabilization
-
The stabilization phase reflects the point at which the patient is in a steady state without shock. In this phase, fluid therapy is used only for ongoing maintenance in the setting of normal fluid losses.
- De-escalation
-
The de-escalation (or de-resuscitation) phase reflects the point at which the patient is in a steady state without shock or ongoing fluid losses. Fluid can be removed from patients in the de-escalation phase.
- Fast Fourier transformation
-
A mathematical method for transforming a function of time into a function of frequency. Fast Fourier transformation is used for analysis of time-dependent phenomena.
Rights and permissions
About this article
Cite this article
Murugan, R., Bellomo, R., Palevsky, P.M. et al. Ultrafiltration in critically ill patients treated with kidney replacement therapy. Nat Rev Nephrol 17, 262–276 (2021). https://doi.org/10.1038/s41581-020-00358-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-020-00358-3
This article is cited by
-
Response Letter to: Correspondence regarding the article by Murugan et al on Precision net ultrafiltration dosing in continuous kidney replacement therapy: a practical approach
Intensive Care Medicine Experimental (2024)
-
Knowledge, attitude, practice, needs, and implementation status of intensive care unit staff toward continuous renal replacement therapy: a survey of 66 hospitals in central and South China
BMC Nursing (2024)
-
Factors associated with successful liberation from continuous renal replacement therapy in children and young adults: analysis of the worldwide exploration of renal replacement outcomes collaborative in Kidney Disease Registry
Intensive Care Medicine (2024)
-
Fluid removal tolerance during the de-escalation phase: is preload unresponsiveness the best guiding candidate?
Critical Care (2023)
-
Precision net ultrafiltration dosing in continuous kidney replacement therapy: a practical approach
Intensive Care Medicine Experimental (2023)