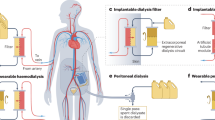
Abstract
Haemodialysis is an extracorporeal process in which the blood is cleansed via removal of uraemic retention products by a semipermeable membrane. Traditionally, dialysis membranes have been broadly classified on the basis of their composition (cellulosic or noncellulosic) and water permeability (low flux or high flux). However, advances in materials technology and polymer chemistry have led to the development of membranes with specific characteristics and refined properties that mandate a reconsideration of traditional membrane classification systems. For adequate characterization of these newer types of membranes, additional parameters are now relevant, including new permeability indices, the hydrophilic or hydrophobic nature of membranes, adsorption capacity and electrical potential. In this Review, we provide clinicians with an updated analysis of dialysis membranes and dialysers. We discuss the basic mechanisms that underlie solute and water removal in dialysis (that is, diffusion, convection, adsorption and ultrafiltration) in the context of treatments that use highly permeable membranes. Specifically, we highlight online haemodiafiltration and new therapies (for example, expanded haemodialysis) that utilize membranes designed to produce a high degree of internal filtration. Finally, we discuss the considerations that govern the clinically acceptable balance between large-solute clearance and albumin loss for extracorporeal therapies.
Key points
-
Traditional schemes for the classification of dialysis membranes, based simply on composition and water permeability, are outdated and new approaches are needed.
-
Dialyser utilization in clinical practice has evolved over time and is now dominated by devices with synthetic high-flux membranes.
-
Rational treatment prescription by clinicians requires an understanding of the basic mechanisms underlying solute and water removal in dialysis — namely, diffusion, convection, adsorption and ultrafiltration.
-
New therapies (including expanded haemodialysis) that utilize membranes designed to produce a high degree of internal filtration are undergoing clinical evaluation as potential alternatives to convective therapies, such as on-line haemodiafilitration.
-
The clinically acceptable amount of albumin loss for extracorporeal therapies remains to be defined.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Clark, W. R., Hamburger, R. J. & Lysaght, M. J. Effect of membrane structure and composition on performance and biocompatibility in hemodialysis. Kidney Int. 56, 2005–2015 (1999).
Ronco, C., Neri, M., Lorenzin, A., Garzotto, F. & Clark, W. R. Multidimensional classification of dialysis membranes. Contrib. Nephrol. 191, 115–126 (2017).
Bowry, S. K., Gatti, E. & Vienken, J. Contribution of polysulfone membranes to the success of convective dialysis therapies. Contrib. Nephrol. 173, 110–118 (2011).
Islam, M. S. et al. Vitamin E-coated and heparin-coated dialyzer membranes for heparin-free hemodialysis: a multicenter, randomized, crossover trial. Am. J. Kidney Dis. 68, 752–762 (2016).
Zweigart, C. et al. Medium cut-off membranes — closer to the natural kidney removal function. Int. J. Artif. Organs 40, 328–334 (2017).
Boschetti-de-Fierro, A. et al. Membrane innovation in dialysis. Contrib. Nephrol. 191, 100–114 (2017).
Clark, W. R., Macias, W. L., Molitoris, B. A. & Wang, N. H. Membrane adsorption of ß2-microglobulin: equilibrium and kinetic characterization. Kidney Int. 46, 1140–1146 (1994).
Désormeaux, A., Moreau, M. E., Lepage, Y., Chanard, J. & Adam, A. The effect of electronegativity and angiotensin-converting enzyme inhibition on the kinin-forming capacity of polyacrylonitrile dialysis membranes. Biomaterials 29, 1139–1146 (2008).
Haas, G. Dialysis of flowing blood in the patient. Klin. Wochenschr. 70, 1888 (1923).
Clark, W. R. Hemodialyzer membranes and configurations: a historical perspective. Semin. Dial. 13, 309–311 (2000).
Kolff, W. & Berk, H. The artificial kidney: a dialyzer with a great area. Acta Med. Scand. 117, 121–134 (1944).
Alwall, N. On the artificial kidney. I. Apparatus or dialysis of blood in vivo. Acta Med. Scand. 128, 317–325 (1947).
Kolff, W., Watschinger, B. & Vertes, B. Results in patients treated with the coil kidney. JAMA 161, 1433–1437 (1956).
Vertes, B., Aoyama, S. & Kolff, W. The twin-coil disposable artificial kidney. Trans. Am. Soc. Artif. Intern. Organs 3, 119–121 (1958).
Kiil, F. Development of a parallel-flow artificial kidney in plastics. Acta Chir. Scand. Suppl. 253, 142–150 (1960).
Cole, J., Pollard, T. & Murray, J. Studies on the modified polypropylene Kiil dialyzer. Trans. Am. Soc. Artif. Intern. Organs 9, 67–70 (1963).
Funck-Bretano, J. et al. A new disposable plate-kidney. Trans. Am. Soc. Artif. Intern. Organs 15, 127–130 (1969).
Lipps, B. et al. The hollow fiber artificial kidney. Trans. Am. Soc. Artif. Intern. Organs 13, 200–207 (1967).
Lysaght, M. J. Hemodialysis membranes in transition. Contrib. Nephrol. 61, 1–17 (1988).
Lysaght, M. J. Evolution of hemodialysis membranes. Contrib. Nephrol. 113, 1–10 (1995).
Craddock, P., Fehr, J., Dalmasso, A., Brigham, K. & Jacob, H. Hemodialysis leukopenia: pulmonary vascular leukostasis resulting from complement activation by dialyzer cellophane membranes. J. Clin. Invest. 59, 879–888 (1977).
Hakim, R., Fearon, D. & Lazarus, J. M. Biocompatibility of dialysis membranes: effects of chronic complement activation. Kidney Int. 26, 194–200 (1984).
Hakim, R. M. Clinical implications of hemodialysis membrane bioincompatibility. Kidney Int. 44, 484–494 (1993).
Takeyama, T. & Sakai, Y. Polymethylmethacrylate: one biomaterial for a series of membranes. Contrib. Nephrol. 125, 9–24 (1998).
Thomas, M., Moriyama, K. & Ledebo, I. AN69: evolution of the world’s first high permeability membrane. Contrib. Nephrol. 173, 119–129 (2011).
Clark, W. R. & Gao, D. Membranes for dialysis: composition, structure, and function. Contrib. Nephrol. 137, 70–77 (2002).
Zweigart, C. et al. in Comprehensive Membrane Science and Engineering 2nd edn (eds Drioli, E., Giorno, L. & Fontana, E.) 215–247 (Elsevier, 2017).
Chung, T. S. N. in Advanced Membrane Technology and Applications Ch. 31 (eds Li, N. N., Fane, A. G., Ho, W. S. W. & Matsuura, T.) (John Wiley and Sons, 2008).
Clark, W. R. Quantitative characterization of hemodialyzer solute and water transport. Semin. Dial. 14, 32–36 (2001).
Michaels, A. S. Operating parameters and performance criteria for hemodialyzers and other membrane-separation devices. Trans. Am. Soc. Artif. Intern. Organs 12, 387–392 (1966).
Kim, J. C. et al. Effects of arterial port design on blood flow distribution in hemodialyzers. Blood Purif. 28, 260–267 (2009).
Ronco, C. et al. Flow distribution analysis by helical scanning in polysulfone hemodialyzers: effects of fiber structure and design on flow patterns and solute clearances. Hemodial. Int. 10, 380–388 (2006).
Brendolan, A. et al. Dialytic performance evaluation of Rexeed: a new polysulfone-based dialyzer with improved flow distributions. Int. J. Artif. Organs. 28, 966–975 (2005).
Gastaldon, F. et al. Effects of novel manufacturing technology on blood and dialysate flow distribution in a new low flux “alpha Polysulfone” hemodialyzer. Int. J. Artif. Organs. 26, 105–112 (2003).
Ronco, C., Brendolan, A., Crepaldi, C., Gastaldon, F. & Levin, N. W. Flow distribution and cross filtration in hollow fiber hemodialyzers. Contrib. Nephrol. 137, 120–128 (2002).
Ronco, C. et al. Dialysate flow distribution in hollow fiber hemodialyzers with different dialysate pathway configurations. Int. J. Artif. Organs 23, 601–609 (2000).
Ronco, C. et al. Performance of DIAPES filters in CRRT. Contrib. Nephrol. 138, 144–152 (2003).
Brendolan, A. et al. Flow dynamic characteristics of DIAPES hemodialyzers. Contrib. Nephrol. 138, 27–36 (2003).
Ronco, C. et al. Hemodialyzer: from macro-design to membrane nanostructure; the case of the FX-class of hemodialyzers. Kidney Int. Suppl. 80, 126–142 (2002).
Ronco, C. et al. Effects of hematocrit and blood flow distribution on solute clearance in hollow-fiber hemodialyzers. Nephron 89, 243–250 (2001).
Ronco, C., Ballestri, M. & Brendolan, A. New developments in hemodialyzers. Blood Purif. 18, 267–275 (2000).
Ronco, C. et al. In vitro and in vivo evaluation of a new polysulfone membrane for hemodialysis. Reference methodology and clinical results. (Part. 2: in vivo study). Int. J. Artif. Organs 22, 616–624 (1999).
Ronco, C. et al. In vitro and in vivo evaluation of a new polysulfone membrane for hemodialysis. Reference methodology and clinical results. (Part 1: in vitro study). Int. J. Artif. Organs 22, 604–615 (1999).
Brendolan, A., Ronco, C., Ghezzi, P. M., Scabardi, M. & La Greca, G. Hydraulic and flow dynamic characteristics of PMMA dialyzers. Contrib. Nephrol. 125, 41–52 (1999).
Barzin, J. et al. Characterization of polyethersulfone hemodialysis membrane by ultrafiltration and atomic force microscopy. J. Memb. Sci. 237, 77–85 (2004).
Miyata, M., Konishi, S., Shimamoto, Y., Kamada, A. & Umimoto, K. Influence of sterilization and storage period on elution of polyvinylpyrollidone from wet-type polysulfone membrane dialyzers. ASAIO J. 61, 468–473 (2015).
Murakami, J., Kaneko, I., Kimata, N., Mineshima, M. & Akiba, T. Problems in the evaluation of polyvinylpyrollidone elution from polysulfone membranes sterilized by gamma-ray radiation. Ren. Replac. Ther. 2, 36 (2016).
Marques, I., Pinheiro, K., Carmo, L., Costa, M. & Abensur, H. Anaphylactic reaction induced by polysulfone/polyvinylpyrrolidone membrane in the 10th session of hemodialysis with the same dialyzer. Hemodial. Int. 15, 399–403 (2011).
Cheung, A. K. et al. Effects of hemodialyzer reuse on clearances of urea and β2-microglobulin. J. Am. Soc. Nephrol. 10, 117–127 (1999).
Kuwahara, T., Markert, M. & Wauters, J. Proteins adsorbed on hemodialysis membranes modulate neutrophil activation. Artif. Organs 13, 427–431 (1989).
Eberhart, R. et al. Influence of endogenous albumin binding on blood-material interactions. Ann. NY Acad. Sci. 516, 78–95 (1987).
Lambrecht, L. et al. The influence of pre-adsorbed canine von Willenbrand factor, fibronectin, and fibrinogen on ex vivo artificial surface-induced thrombogenesis. Thromb. Res. 41, 99–117 (1986).
Casa, L., Deaton, D. H. & Ku, D. N. Role of high shear rate in thrombosis. J. Vasc. Surg. 61, 1068–1080 (2015).
Clark, W. R., Gao, D., Neri, M. & Ronco, C. Solute transport in hemodialysis: advances and limitations of current membrane technology. Contrib. Nephrol. 191, 84–99 (2017).
Ronco, C., Ghezzi, P. M., Brendolan, A., Crepaldi, C. & La Greca, G. The haemodialysis system: basic mechanisms of water and solute transport in extracorporeal renal replacement therapies. Nephrol. Dial. Transplant. 13 (Suppl. 6), 3–9 (1998).
Huang, Z., Clark, W. R. & Gao, D. Determinants of small solute clearance in hemodialysis. Semin. Dial. 18, 30–35 (2005).
Colton, C. K. & Lowrie, E. G. in The Kidney 2nd edn (eds Brenner, B. M. & Rector, F. C.) 2425–2489 (WB Saunders, 1981).
Bird, R. B., Stewart, W. E. & Lightfoot, E. N. in Transport Phenomena 1st edn (eds Bird, R. B., Stewart, W. E. & Lightfoot, E. N.) 34–70 (John Wiley and Sons, 1960).
Merrill, E. W. Rheology of blood. Physiol. Rev. 49, 863–888 (1949).
Chapdelaine, I. et al. Optimization of the convection volume in online post-dilution haemodiafiltration: practical and technical issues. Clin. Kidney J. 8, 191–198 (2015).
Kim, J. C. et al. Effects of dialysate flow configurations in continuous renal replacement therapy on solute removal: computational modeling. Blood Purif. 35, 106–111 (2013).
Kim, J. C. et al. Computational modeling of effects of mechanical shaking on hemodynamics in hollow fibers. Int. J. Artif. Organs 35, 301–307 (2012).
Kim, J. C. et al. Enhancement of solute removal in a hollow-fiber hemodialyzer by mechanical vibration. Blood Purif. 31, 227–234 (2011).
Ronco, C. Fluid mechanics and crossfiltration in hollow-fiber hemodialyzers. Contrib. Nephrol. 158, 34–49 (2007).
Ronco, C. & Levin, N. W. Mechanisms of solute transport in extracorporeal therapies. Contrib. Nephrol. 149, 10–17 (2005).
Villarroel, F., Klein, E. & Holland, F. Solute flux in hemodialysis and hemofiltration membranes. Trans. Am. Soc. Artif. Organs 23, 225–232 (1977).
Zydney, A. L. Bulk mass transport limitations during high-flux hemodialysis. Artif. Organs 17, 919–924 (1993).
Ofsthun, N. J. & Zydney, A. L. Importance of convection in artificial kidney treatment. Contrib. Nephrol. 108, 53–70 (1994).
Huang, Z., Gao, D., Letteri, J. J. & Clark, W. R. Blood-membrane interactions during dialysis. Semin. Dial. 22, 623–628 (2009).
Langsdorf, L. J. & Zydney, A. L. Effect of blood contact on the transport properties of hemodialysis membranes: a two-layer model. Blood Purif. 12, 292–307 (1994).
Morti, S. M. & Zydney, A. L. Protein-membrane interactions during hemodialysis: effects on solute transport. ASAIO J. 44, 319–326 (1998).
Rockel, A. et al. Permeability and secondary membrane formation of a high flux polysulfone hemofilter. Kidney Int. 30, 429–432 (1986).
Clark, W. R., Macias, W. L., Molitoris, B. A. & Wang, N. H. L. Plasma protein adsorption to highly permeable dialysis membranes. Kidney Int. 48, 481–487 (1995).
Gachon, A., Mallet, J., Trideon, A. & Deteix, P. Analysis of proteins eluted from hemodialysis membranes. J. Biomater. Sci. Polym. Ed. 2, 263–276 (1991).
Colton, C. K., Henderson, L. W., Ford, C. A. & Lysaght, M. J. Kinetics of hemodiafiltration. I. In vitro transport characteristics of a hollow-fiber blood ultrafilter. J. Lab. Clin. Med. 85, 355–371 (1975).
Fiore, G. B., Guadagni, G., Lupi, A., Ricci, Z. & Ronco, C. A new semiempirical mathematical model for prediction of internal filtration in hollow fiber hemodialyzers. Blood Purif. 24, 555–568 (2006).
Fiore, G. B. & Ronco, C. Principles and practice of internal hemodiafiltration. Contrib. Nephrol. 158, 177–184 (2007).
Rangel, A. V. et al. Backfiltration: past, present, and future. Contrib. Nephrol. 175, 35–45 (2011).
Lorenzin, A., Neri, M., Clark, W. R. & Ronco, C. Experimental measurement of internal filtration rate for a new medium cut-off dialyzer. Contrib. Nephrol. 191, 127–141 (2017).
Ronco, C., Brendolan, A., Lupi, A., Bettini, M. C. & La Greca, G. Enhancement of convective transport by internal filtration in a modified experimental dialyzer. Kidney Int. 54, 979–985 (1998).
Ronco, C., Brendolan, A., Lupi, A., Metry, G. & Levin, N. W. Effects of reduced inner diameter of hollow fibers in hemodialyzers. Kidney Int. 58, 809–817 (2000).
Ronco, C., Brendolan, A., Crepaldi, C., Rodighiero, M. & Scabardi, M. Blood and dialysate flow distributions in hollow-fiber hemodialyzers analysed by computerized helical scanning technique. J. Am. Soc. Nephrol. 13, S53–S61 (2002).
Mineshima, M. New trends in HDF: validity of internal filtration-enhanced hemodialysis. Blood Purif. 22 (Suppl. 2), 60–66 (2004).
Ronco, C. & La Manna, G. Expanded hemodialysis: a new therapy for a new class of membranes. Contrib. Nephrol. 190, 124–133 (2017).
Ronco, C. The rise of expanded hemodialysis. Blood Purif. 44, I–VIII (2017).
Ronco, C. & Bowry, S. Nanoscale modulation of the pore dimensions, size distribution and structure of a new polysulfone-based high-flux dialysis membrane. Int. J. Artif. Organs 24, 726–735 (2001).
Goldman, M. et al. Adsorption of beta2-microglobulin on dialysis membranes: comparison of dialyzers and effect of reuse. Int. J. Artif. Organs 12, 373–378 (1989).
Mares, J., Thongboonkerd, V., Tuma, Z., Moravec, J. & Matejovic, M. Specific adsorption of some complement activation proteins to polysulfone dialysis membranes during hemodialysis. Kidney Int. 76, 404–413 (2009).
Clark, W. R. & Gao, D. Low-molecular weight proteins in end-stage renal disease: potential toxicity and dialytic removal mechanisms. J. Am. Soc. Nephrol. 13, S41–S47 (2002).
Massy, Z. & Liabeuf, S. Middle-molecule uremic toxins and outcomes in chronic kidney disease. Contrib. Nephrol. 191, 8–17 (2017).
Barreto, F. C., Barreto, D. V. & Canziani, M. E. F. Uremia retention molecules and clinical outcomes. Contrib. Nephrol. 191, 18–31 (2017).
Eloot, S. et al. Protein-bound uremic toxin profiling as a tool to optimize hemodialysis. PLoS ONE 11, e0147159 (2016).
Kneis, C. et al. Elimination of middle-sized uremic solutes with high-flux and high-cut-off membranes: a randomized in vivo study. Blood Purif. 36, 287–294 (2013).
Yu, X. The evolving patterns of uremia: unmet clinical needs in dialysis. Contrib. Nephrol. 191, 1–7 (2017).
Ward, R. A. Protein-leaking membranes for hemodialysis: a new class of membranes in search of an application? J. Am. Soc. Nephrol. 16, 2421–2430 (2005).
US Food and Drug Administration. Guidance for the content of premarket notifications for conventional and high permeability hemodialyzers. FDA https://www.fda.gov/downloads/medicaldevices/deviceregulationandguidance/guidancedocuments/ucm080166.pdf (1998).
Keshaviah, P., Luehmann, D., Ilstrup, K. & Collins, A. Technical requirements for rapid high-efficiency therapies. Artif. Organs 10, 189–194 (1986).
Eknoyan, G. et al. Effect of dialysis dose and membrane flux in maintenance hemodialysis. N. Engl. J. Med. 347, 2010–2019 (2002).
Tattersall, J. E. & Ward, R. A., EUDIAL group. Online hemodiafiltration: definition, dose quantification, and safety revisited. Nephrol. Dial. Transplant. 28, 542–550 (2013).
Boschetti-de-Fierro, A., Voigt, M., Storr, M. & Krause, B. Extended characterization of a new class of membranes for blood purification: the high cut-off membranes. Int. J. Artif. Organs 36, 455–463 (2013).
Rousseau-Gagnon, M., Agharazii, M., De Serres, S. A. & Desmeules, S. Effectiveness of haemodiafiltration with heat sterilized high-flux polyphenylene HF dialyzer in reducing free light chains in patients with myeloma cast nephropathy. PLoS ONE 10, e0140463 (2015).
Hutchison, C. A. et al. Immunoglobulin free light chain levels and recovery from myeloma kidney on treatment with chemotherapy and high cut-off haemodialysis. Nephrol. Dial. Transplant. 27, 3823–3828 (2012).
Hutchison, C. A. et al. Treatment of acute renal failure secondary to multiple myeloma with chemotherapy and extended high cut-off hemodialysis. Clin. J. Am. Soc. Nephrol. 4, 745–754 (2009).
Hutchison, C. A. et al. Serum free-light chain removal by high cutoff hemodialysis: optimizing removal and supportive care. Artif. Organs 32, 910–917 (2008).
Atan, R. et al. High cut-off hemofiltration versus standard hemofiltration: effect on plasma cytokines. Int. J. Artif. Organs 39, 479–486 (2016).
Chelazzi, C. et al. Hemodialysis with high cut-off hemodialyzers in patients with multi-drug resistant gram-negative sepsis and acute kidney injury: a retrospective, case-control study. Blood Purif. 42, 186–193 (2016).
Villa, G. et al. Organ dysfunction during continuous veno-venous high cut-off hemodialysis in patients with septic acute kidney injury: a prospective observational study. PLoS ONE 12, e0172039 (2017).
Boschetti-de-Fierro, A., Voigt, M., Storr, M. & Krause, B. MCO membranes: enhanced selectivity in high-flux class. Sci. Rep. 5, 18448 (2015).
Streicher, E. & Schneider, H. The development of a polysulfone membrane. A new perspective in dialysis? Contrib. Nephrol. 46, 1–13 (1985).
Schneider, H. & Streicher, E. Clinical observations of the polyamide hollow-fiber hemofilter in hemofiltration systems. J. Dial. 1, 737–744 (1977).
Ota, K. et al. Short-time hemodiafiltration using polymethylmethacrylate hemodiafilter. Trans. Am. Soc. Artif. Intern. Organs 24, 454–457 (1978).
Kaiser, J., Hagemann, J., Von Herrath, D. & Schaefer, K. Different handling of beta2-microglobulin during hemodialysis and hemofiltration. Nephron 48, 132–135 (1988).
Jorstad, S., Smeby, L., Balstad, T. & Wideroe, T. E. Removal, generation, and adsorption of beta-2-microglobulin during hemofiltration with five different membranes. Blood Purif. 6, 96–105 (1988).
Jindal, K. K., McDougall, J., Woods, B., Nowakowski, L. & Goldstein, M. B. A study of the basic principles determining the performance of several high-flux dialyzers. Am. J. Kidney Dis. 14, 507–511 (1989).
Zingraff, J. et al. Influence of haemodialysis membranes on beta2-microglobulin kinetics: in vivo and in vitro studies. Nephrol. Dial. Transplant. 3, 284–290 (1988).
Klinke, B., Rockel, A., Abdelhamid, S., Fiegel, P. & Walb, D. Transmembrane transport and adsorption of beta2-microglobulin during hemodialysis using polysulfone, polyacrylonitrile, polymethylmethacrylate, and cuprammonium rayon membranes. Int. J. Artif. Organs 12, 697–702 (1989).
Floege, J. et al. High flux synthetic versus cellulosic membranes for beta2-microglobulin removal during hemodialysis, hemodiafiltration, and hemofiltration. Nephrol. Dial. Transplant. 4, 653–657 (1989).
Naitoh, A., Tatsuguchi, T., Okada, M., Ohmura, T. & Sakai, K. Removal of beta2-microglobulin by diffusion is feasible using highly permeable dialysis membranes. Trans. Am. Soc. Artif. Intern. Organs 34, 630–634 (1988).
Mineshima, M. et al. Difference in beta2-microglobulin removal between cellulosic and synthetic polymer membrane dialylzers. Trans. Am. Soc. Artif. Intern. Organs 36, M643–M646 (1990).
Ronco, C. et al. Beta2-microglobulin removal by synthetic dialysis membranes: mechanisms and kinetics of the molecule. Int. J. Artif. Organs 20, 136–143 (1997).
Floege, J. et al. Beta2-microglobulin kinetics during hemodialysis and hemofiltration. Nephrol. Dial. Transplant. 1, 223–228 (1987).
Maeda, N. et al. Performance and mechanism of beta-2-microglobulin elimination with a new PAN hollow fiber membrane. Jap. J. Artif. Organs 17, 3–9 (1988).
Kachel, H., Altmeyer, P., Baldamus, C. & Koch, K. Deposition of an amyloid-like substance as a possible complication of regular dialysis treatment. Contrib. Nephrol. 36, 127–132 (1983).
Gejyo, F. et al. A new form of amyloid protein associated with chronic hemodialysis was identified as β2-microglobulin. Biochem. Biophys. Res. Commun. 129, 701–706 (1985).
Ahrenholz, P., Winkler, R. E., Ramlow, W., Tiess, M. & Muller, W. On-line hemodiafiltration with pre- and post-dilution: a comparison of efficacy. Int. J. Artif. Organs 20, 81–90 (1997).
Ono, M., Taoka, M., Takagi, T., Ogawa, H. & Saito, A. Comparison of type of on-line hemodiafiltration from the standpoint of low-molecular weight protein removal. Contrib. Nephrol. 108, 38–45 (1994).
Colton, C. K. Analysis of membrane processes for blood purification. Blood Purif. 5, 202–251 (1987).
Ward, R. A., Schmidt, B., Hullin, J., Hillebrand, G. F. & Samtleben, W. A comparison of on-line hemodiafiltration and high-flux hemodialylsis: a prospective clinical study. J. Am. Soc. Nephrol. 11, 2344–2350 (2000).
Ahrenholz, P. G., Winkler, R. E., Michelsen, A., Lang, D. A. & Bowry, S. K. Dialysis membrane-dependent removal of middle molecules during hemodiafiltration: the beta2-microglobulin/albumin relationship. Clin. Nephrol. 62, 21–28 (2004).
Maduell, F. et al. Elimination of large uremic toxins by a dialyzer specifically designed for high-volume convective therapies. Blood Purif. 37, 125–130 (2014).
Maduell, F. et al. High-efficiency postdilution online hemodiafiltration reduces all-cause mortality in hemodialysis patients. J. Am. Soc. Nephrol. 24, 487–497 (2013).
Canaud, B. & Bowry, S. K. Emerging clinical evidence on online hemodialfiltration: does volume of ultrafiltration matter? Blood Purif. 35, 55–62 (2013).
Ok, E. et al. Mortality and cardiovascular events in online haemodiafiltration (OL-HDF) compared with high-flux dialysis: results from the Turkish OL-HDF Study. Nephrol. Dial. Transplant. 28, 192–202 (2013).
Grooteman, M. P. et al. Effect of online hemodiafiltration on all-cause mortality and cardiovascular outcomes. J. Am. Soc. Nephrol. 23, 1087–1096 (2012).
Fournier, A., Birmele, B., Francois, M., Prat, L. & Halimi, J. M. Factors associated with albumin loss in post-dilution hemodiafiltration. Int. J. Artif. Organs 38, 76–82 (2015).
Ronco, C. Hemodiafiltration: technical and clinical issues. Blood Purif. 40 (Suppl. 1), 2–11 (2015).
Panichi, V. et al. Divert to ULTRA: differences in infused volumes and clearance in two on-line hemodiafiltration treatments. Int. J. Artif. Organs 35, 435–443 (2012).
Pedrini, L. A. et al. Transmembrane pressure modulation in high-volume mixed hemodiafiltration to optimize efficiency and minimize protein loss. Kidney Int. 69, 573–579 (2006).
Gayrard, N. et al. Consequences of increasing convection onto patient care and protein removal in hemodialysis. PLoS ONE 12, e0171179 (2017).
Ficheux, A., Ronco, C., Brunet, P. & Argiles, A. The ultrafiltration coefficient: this old ‘grand inconnu’ in dialyisis. Nephrol. Dial. Transplant. 30, 204–208 (2015).
Maduell, F. et al. Mid-dilution hemodiafiltration: a comparison with pre- and postdilution modes using the same polyphenylene membrane. Blood Purif. 28, 268–274 (2009).
Krieter, D. H. et al. Clinical cross-over comparison of mid-dilution hemodiafiltration using a novel dialyzer concept and post-dilution hemodiafiltration. Kidney Int. 67, 349–356 (2005).
Shinzato, T. & Maeda, K. Push/pull hemodiafiltration. Contrib. Nephrol. 158, 169–176 (2007).
Kirsch, A. H. et al. Performance of hemodialysis with novel medium cut-off dialyzers. Nephrol. Dial. Transplant. 32, 165–172 (2017).
Kirsch, A. H., Rosenkranz, A. R., Lyko, R. & Krieter, D. H. Effects of hemodialysis therapy using medium cut-off membranes on middle molecules. Contrib. Nephrol. 191, 158–167 (2017).
Heyne, N. Expanded hemodialysis therapy: prescription and delivery. Contrib. Nephrol. 191, 153–157 (2017).
Florens, N. & Julliard, L. Large middle molecular and albumin removal: why should we not rest on our laurels? Contrib. Nephrol. 191, 178–187 (2017).
Nagai, K. et al. Implications of albumin leakage for survival in maintenance hemodialysis patients: a 7-year observational study. Ther. Apher. Dial. 21, 378–386 (2017).
Balafa, O., Halbesma, N., Struijk, D. G., Dekker, F. W. & Krediet, R. T. Peritoneal albumin and protein losses do not predict outcome in peritoneal dialysis patients. Clin. J. Am. Soc. Nephrol. 6, 561–566 (2011).
Lavaud, S. et al. Optimal anticoagulation strategy in haemodialysis with heparin-coated polyacrylonitrile membrane. Nephrol. Dial. Transplant. 18, 2097–2104 (2003).
Thomas, M., Valette, P., Mausset, A. L. & Déjardin, P. High molecular weight kininogen adsorption on hemodialysis membranes: influence of pH and relationship with contact phase activation of blood plasma. influence of pre-treatment with poly(ethyleneimine). Int. J. Artif. Organs 23, 20–26 (2000).
Panagiotou, A. et al. Antioxidant dialytic approach with vitamin E-coated membranes. Contrib. Nephrol. 171, 101–106 (2011).
Cruz, D. N. et al. Effect of vitamin E-coated dialysis membranes on anemia in patients with chronic kidney disease: an Italian multicenter study. Int. J. Artif. Organs 31, 545–552 (2008).
Ghezzi, P. M. & Ronco, C. Excebrane: hemocompatibility studies by the intradialytic monitoring of oxygen saturation. Contrib. Nephrol. 127, 177–191 (1999).
Kim, S. et al. Diffusive silicon nanopore membranes for hemodialysis applications. PLoS ONE 11, e0159526 (2016).
Canaud, B., Bosc, J. Y., Leray, H. & Stec, F. Microbiological purity of dialysate for on-line substitution fluid preparation. Nephrol. Dial. Transplant. 15 (Suppl. 2), 21–30 (2000).
Acknowledgements
The authors thank M. Storr (Baxter International), S. Bowry (Fresenius Medical Care), R. Baldini (B. Braun Medical), L. Fecondini (Medica), L. Frattini (Medtronic), W. Oshihara (Toray Medical), A. Simionato (Asahi Kasei Medical) and S. Takashi (Nipro Corporation) for their invaluable comments and the generous provision of membrane images. The authors recognize the seminal contributions to end-stage renal disease therapy made by L. Henderson, who passed away in 2017. He was a source of inspiration for many of us, and we owe him a debt of gratitude for his exemplary leadership in the field.
Author information
Authors and Affiliations
Contributions
Both authors contributed to researching data for the article and writing, reviewing and editing the article before submission.
Corresponding author
Ethics declarations
Competing interests
C.R. has received consultant or honoraria fees from Astute Medical, Ortho Clinical Diagnostics, Baxter International, Asahi Kasei Medical, General Electric, Jafron Biomedical, Estor Medical and Toray Medical. W.R.C. was formerly employed by Baxter International, has received consulting fees from Baxter International and owns Baxter International stock; he is also a consultant with Medtronic, Nikkiso America and Astute Medical.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ronco, C., Clark, W.R. Haemodialysis membranes. Nat Rev Nephrol 14, 394–410 (2018). https://doi.org/10.1038/s41581-018-0002-x
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41581-018-0002-x
This article is cited by
-
Innovating dialysis through computational modelling of hollow-fibre haemodialysers
Nature Reviews Nephrology (2024)
-
Effect of Membrane Permeance and System Parameters on the Removal of Protein-Bound Uremic Toxins in Hemodialysis
Annals of Biomedical Engineering (2024)
-
On the total albumin losses during haemocatharsis
Journal of Artificial Organs (2024)
-
Advances in Enhancing Hemocompatibility of Hemodialysis Hollow-Fiber Membranes
Advanced Fiber Materials (2023)
-
A study on the effects of thermal treatments and gamma rays irradiation on expired “Helixone” membranes
Macromolecular Research (2023)