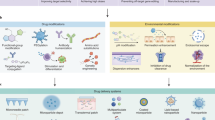
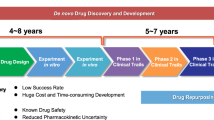
Abstract
Progress in the field of precision medicine has changed the landscape of cancer therapy. Precision medicine is propelled by technologies that enable molecular profiling, genomic analysis and optimized drug design to tailor treatments for individual patients. Although precision medicines have resulted in some clinical successes, the use of many potential therapeutics has been hindered by pharmacological issues, including toxicities and drug resistance. Drug delivery materials and approaches have now advanced to a point where they can enable the modulation of a drug’s pharmacological parameters, without compromising the desired effect on molecular targets. Specifically, they can modulate a drug’s pharmacokinetics, stability, absorption and exposure to tumours and healthy tissues, and facilitate the administration of synergistic drug combinations. This Review highlights recent progress in precision therapeutics and drug delivery, and identifies opportunities for strategies to improve the therapeutic index of cancer drugs and, consequently, clinical outcomes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tran, S., DeGiovanni, P. J., Piel, B. & Rai, P. Cancer nanomedicine: a review of recent success in drug delivery. Clin. Transl. Med. 6, 44 (2017).
Dugger, S. A., Platt, A. & Goldstein, D. B. Drug development in the era of precision medicine. Nat. Rev. Drug Discov. 17, 183–196 (2017).
Blanco, E., Shen, H. & Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 33, 941–951 (2015).
Bhullar, K. S. et al. Kinase-targeted cancer therapies: progress, challenges and future directions. Mol. Cancer 17, 48 (2018).
Ferguson, F. M. & Gray, N. S. Kinase inhibitors: the road ahead. Nat. Rev. Drug Discov. 17, 353–377 (2018). Review of kinase inhibitors as a leading modality in personalized medicine.
Roskoski, R. Properties of FDA-approved small molecule protein kinase inhibitors. Pharmacol. Res. 144, 19–50 (2019).
Smalley, I. & Smalley, K. S. M. ERK inhibition: a new front in the war against MAPK pathway–driven cancers? Cancer Discov. 8, 140–142 (2018).
Yaeger, R. & Corcoran, R. B. Targeting alterations in the RAF–MEK pathway. Cancer Discov. 9, 329–341 (2019).
Gabizon, A., Shmeeda, H. & Barenholz, Y. Pharmacokinetics of pegylated liposomal doxorubicin. Clin. Pharmacokinet. 42, 419–436 (2003).
Wilhelm, S. et al. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 1, 16014 (2016). Major challenges of nanomedicines in oncology, with an emphasis on drug loading.
Song, Q. et al. Reduction responsive self-assembled nanoparticles based on disulfide-linked drug–drug conjugate with high drug loading and antitumor efficacy. Mol. Pharm. 13, 190–201 (2016).
Shamay, Y. et al. Quantitative self-assembly prediction yields targeted nanomedicines. Nat. Mater. 17, 361–368 (2018).
Bamrungsap, S. et al. Nanotechnology in therapeutics: a focus on nanoparticles as a drug delivery system. Nanomedicine 7, 1253–1271 (2012).
Hong, D. et al. Phase I study of LY2606368, a checkpoint kinase 1 inhibitor, in patients with advanced cancer. J. Clin. Oncol. 34, 1764–1771 (2016).
Mak, G. et al. A phase Ib dose-finding, pharmacokinetic study of the focal adhesion kinase inhibitor GSK2256098 and trametinib in patients with advanced solid tumours. Br. J. Cancer 120, 975–981 (2019).
Postel-Vinay, S. et al. Clinical benefit in Phase-I trials of novel molecularly targeted agents: does dose matter? Br. J. Cancer 100, 1373–1378 (2009).
Wong, H. H., Barton, C., Acton, G., McLeod, R. & Halford, S. Trends in the characteristics, dose-limiting toxicities and efficacy of phase I oncology trials: The Cancer Research UK experience. Eur. J. Cancer 66, 9–16 (2016).
Hanker, A. B., Kaklamani, V. & Arteaga, C. L. Challenges for the clinical development of PI3K inhibitors: strategies to improve their impact in solid tumors. Cancer Discov. 9, 482–491 (2019).
Bullock, J. M., Rahman, A. & Liu, Q. Lessons learned: dose selection of small molecule-targeted oncology drugs. Clin. Cancer Res. 22, 2630–2638 (2016).
Waldner, M., Fantus, D., Solari, M. & Thomson, A. W. New perspectives on mTOR inhibitors (rapamycin, rapalogs and TORKinibs) in transplantation. Br. J. Clin. Pharmacol. 82, 1158–1170 (2016).
Infante, J. R. et al. Safety, pharmacokinetic, pharmacodynamic, and efficacy data for the oral MEK inhibitor trametinib: a phase 1 dose-escalation trial. Lancet Oncol. 13, 773–781 (2012).
Tyagi, P. & Santiago, C. New features in MEK retinopathy. BMC Ophthalmol. 18, 221–221 (2018).
Khan, K. H. et al. Hyperglycemia and phosphatidylinositol 3-kinase/protein kinase B/mammalian target of rapamycin (PI3K/AKT/mTOR) inhibitors in phase I trials: incidence, predictive factors, and management. Oncologist 21, 855–860 (2016).
Touyz, R. M., Herrmann, S. M. S. & Herrmann, J. Vascular toxicities with VEGF inhibitor therapies–focus on hypertension and arterial thrombotic events. J. Am. Soc. Hypertens. 12, 409–425 (2018).
Touyz, R. M. & Herrmann, J. Cardiotoxicity with vascular endothelial growth factor inhibitor therapy. NPJ Precis. Oncol. 2, 13 (2018).
Massey, P. R., Okman, J. S., Wilkerson, J. & Cowen, E. W. Tyrosine kinase inhibitors directed against the vascular endothelial growth factor receptor (VEGFR) have distinct cutaneous toxicity profiles: a meta-analysis and review of the literature. Support. Care Cancer 23, 1827–1835 (2015).
Thill, M. & Schmidt, M. Management of adverse events during cyclin-dependent kinase 4/6 (CDK4/6) inhibitor-based treatment in breast cancer. Ther. Adv. Med. Oncol. https://doi.org/10.1177/175883591879332 (2018).
Hantschel, O. Unexpected off-targets and paradoxical pathway activation by kinase Inhibitors. ACS Chem. Biol. 10, 234–245 (2015). A description of on-target toxicity of PI3K/AKT/mTOR pathway inhibitors.
Force, T., Krause, D. S. & Van Etten, R. A. Molecular mechanisms of cardiotoxicity of tyrosine kinase inhibition. Nat. Rev. Cancer 7, 332–344 (2007).
Miyazawa, K. Encountering unpredicted off-target effects of pharmacological inhibitors. J. Biochem. 150, 1–3 (2011).
de Groot, C. O. et al. A cell biologist’s field guide to aurora kinase inhibitors. Front. Oncol. 5, 285 (2015).
Roberts, A. W. et al. Phase 1 study of the safety, pharmacokinetics, and antitumour activity of the BCL2 inhibitor navitoclax in combination with rituximab in patients with relapsed or refractory CD20+ lymphoid malignancies. Br. J. Haematol. 170, 669–678 (2015).
Cang, S., Iragavarapu, C., Savooji, J., Song, Y. & Liu, D. ABT-199 (venetoclax) and BCL-2 inhibitors in clinical development. J. Hematol. Oncol. 8, 129 (2015).
Bazak, R., Houri, M., El Achy, S., Kamel, S. & Refaat, T. Cancer active targeting by nanoparticles: a comprehensive review of literature. J. Cancer Res. Clin. Oncol. 141, 769–784 (2015).
Kantarjian, H. M. et al. Stage I of a phase 2 study assessing the efficacy, safety, and tolerability of barasertib (AZD1152) versus low-dose cytosine arabinoside in elderly patients with acute myeloid leukemia. Cancer 119, 2611–2619 (2013).
Ashton, S. et al. Aurora kinase inhibitor nanoparticles target tumors with favorable therapeutic index in vivo. Sci. Transl. Med. 8, 325ra17 (2016).
Keen, N. & Taylor, S. Mitotic drivers — inhibitors of the Aurora B Kinase. Cancer Metastasis Rev. 28, 185–195 (2009).
Burris, H. A. et al. A phase I, open-label, first-time-in-patient dose escalation and expansion study to assess the safety, tolerability, and pharmacokinetics of nanoparticle encapsulated Aurora B kinase inhibitor AZD2811 in patients with advanced solid tumours. J. Clin. Oncol. 35, 15 (2017).
Shamay, Y. et al. P-selectin is a nanotherapeutic delivery target in the tumor microenvironment. Sci. Transl. Med. 8, 345ra87 (2016).
Bachelet, L. et al. Affinity of low molecular weight fucoidan for P-selectin triggers its binding to activated human platelets. Biochim. Biophys. Acta 1790, 141–146 (2009).
Mizrachi, A. et al. Tumour-specific PI3K inhibition via nanoparticle-targeted delivery in head and neck squamous cell carcinoma. Nat. Commun. 8, 14292 (2017).
Zumsteg, Z. S. et al. Taselisib (GDC-0032), a potent β-sparing small molecule inhibitor of PI3K, radiosensitizes head and neck squamous carcinomas containing activating PIK3CA alterations. Clin. Cancer Res. 22, 2009–2019 (2016).
Horn, D., Hess, J., Freier, K., Hoffmann, J. & Freudlsperger, C. Targeting EGFR-PI3K-AKT-mTOR signaling enhances radiosensitivity in head and neck squamous cell carcinoma. Expert Opin. Ther. Targets 19, 795–805 (2015).
Josephs, D. H., Fisher, D. S., Spicer, J. & Flanagan, R. J. Clinical pharmacokinetics of tyrosine kinase inhibitors: implications for therapeutic drug monitoring. Ther. Drug Monit. 35, 562–587 (2013).
van Erp, N. P., Gelderblom, H. & Guchelaar, H. J. Clinical pharmacokinetics of tyrosine kinase inhibitors. Cancer Treat. Rev. 35, 692–706 (2009).
Wells, S. A. et al. Vandetanib in patients with locally advanced or metastatic medullary thyroid cancer: a randomized, double-blind phase III trial. J. Clin. Oncol. 30, 134–141 (2012).
Poggi, L. & Kolesar, J. M. Vismodegib for the treatment of basal cell skin cancer. Am. J. Health Syst. Pharm. 70, 1033–1038 (2013).
Goel, V. et al. Population pharmacokinetics of sonidegib (LDE225), an oral inhibitor of hedgehog pathway signaling, in healthy subjects and in patients with advanced solid tumors. Cancer Chemother. Pharmacol. 77, 745–755 (2016).
Burris, H. A. 3rd et al. Phase I safety, pharmacokinetics, and clinical activity study of lapatinib (GW572016), a reversible dual inhibitor of epidermal growth factor receptor tyrosine kinases, in heavily pretreated patients with metastatic carcinomas. J. Clin. Oncol. 23, 5305–5313 (2005).
Kramkimel, N. et al. Vemurafenib pharmacokinetics and its correlation with efficacy and safety in outpatients with advanced BRAF-mutated melanoma. Target. Oncol. 11, 59–69 (2016).
Wagner, M. C. et al. Nilotinib shows prolonged intracellular accumulation upon pulse-exposure: a novel mechanism for induction of apoptosis in CML cells. Leukemia 27, 1567–1570 (2013).
Lipka, D. B., Wagner, M. C., Dziadosz, M. & Fischer, T. Prolonged cellular midostaurin retention suggests potential alternative dosing strategies for FLT3-ITD-positive leukemias. Leukemia 30, 2090–2093 (2016).
Schafranek, L. et al. Sustained inhibition of STAT5, but not JAK2, is essential for TKI-induced cell death in chronic myeloid leukemia. Leukemia 29, 76–85 (2015).
Vasconcelos, T., Sarmento, B. & Costa, P. Solid dispersions as strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov. Today 12, 1068–1075 (2007).
Farouk, F. & Shamma, R. Chemical structure modifications and nano-technology applications for improving ADME-Tox properties, a review. Arch. Pharm. 352, 1800213 (2019).
Chow, E. K. & Ho, D. Cancer nanomedicine: from drug delivery to imaging. Sci. Transl. Med. 5, 216rv14 (2013).
Muhamad, N., Plengsuriyakarn, T. & Na-Bangchang, K. Application of active targeting nanoparticle delivery system for chemotherapeutic drugs and traditional/herbal medicines in cancer therapy: a systematic review. Int. J. Nanomed. 13, 3921–3935 (2018).
Sykes, E. A. et al. Tailoring nanoparticle designs to target cancer based on tumor pathophysiology. Proc. Natl Acad. Sci. USA 113, E1142–E1151 (2016).
Carmeliet, P. & Jain, R. K. Principles and mechanisms of vessel normalization for cancer and other angiogenic diseases. Nat. Rev. Drug Discov. 10, 417–427 (2011).
Peng, F. et al. Nanoparticles promote in vivo breast cancer cell intravasation and extravasation by inducing endothelial leakiness. Nat. Nanotechnol. 14, 279–286 (2019).
Pérez-Medina, C. et al. Nanoreporter PET predicts the efficacy of anti-cancer nanotherapy. Nat. Commun. 7, 11838 (2016).
Boulikas, T. Clinical overview on Lipoplatin: a successful liposomal formulation of cisplatin. Expert Opin. Investig. Drugs 18, 1197–1218 (2009).
Bartelink, I. H. et al. Tumor drug penetration measurements could be the neglected piece of the personalized cancer treatment puzzle. Clin. Pharmacol. Ther. 106, 148–163 (2019). A road map for the clinical implementation of precision dosing.
Reddy, L. H. & Couvreur, P. Nanotechnology for therapy and imaging of liver diseases. J. Hepatol. 55, 1461–1466 (2011).
In, G. K. & Nieva, J. Emerging chemotherapy agents in lung cancer: nanoparticles therapeutics for non-small cell lung cancer. Transl. Cancer Res. 4, 340–355 (2015).
Harisinghani, M. G. et al. Noninvasive detection of clinically occult lymph-node metastases in prostate cancer. N. Engl. J. Med. 348, 2491–2499 (2003).
Heller, D. A. et al. Modular ‘click-in-emulsion’ bone-targeted nanogels. Adv. Mater. 25, 1449–1454 (2013).
Sago, C. D. et al. Nanoparticles that deliver RNA to bone marrow identified by in vivo directed evolution. J. Am. Chem. Soc. 140, 17095–17105 (2018).
Alidori, S. et al. Targeted fibrillar nanocarbon RNAi treatment of acute kidney injury. Sci. Transl. Med. 8, 331ra39 (2016).
Williams, R. M. et al. Selective nanoparticle targeting of the renal tubules. Hypertension 71, 87–94 (2018).
Williams, R. M. et al. Mesoscale nanoparticles selectively target the renal proximal tubule epithelium. Nano Lett. 15, 2358–2364 (2015).
Choi, C. H. J., Zuckerman, J. E., Webster, P. & Davis, M. E. Targeting kidney mesangium by nanoparticles of defined size. Proc. Natl Acad. Sci. USA 108, 6656–6661 (2011).
Zuckerman, J. E., Choi, C. H. J., Han, H. & Davis, M. E. Polycation-siRNA nanoparticles can disassemble at the kidney glomerular basement membrane. Proc. Natl Acad. Sci. USA 109, 3137–3142 (2012).
Lee, H. et al. An endoscope with integrated transparent bioelectronics and theranostic nanoparticles for colon cancer treatment. Nat. Commun. 6, 10059 (2015).
Elci, S. G. et al. Surface charge controls the suborgan biodistributions of gold nanoparticles. ACS Nano 10, 5536–5542 (2016).
Hirn, S. et al. Particle size-dependent and surface charge-dependent biodistribution of gold nanoparticles after intravenous administration. Eur. J. Pharm. Biopharm. 77, 407–416 (2011).
Thapa, B., Kumar, P., Zeng, H. B. & Narain, R. Asialoglycoprotein receptor-mediated gene delivery to hepatocytes using galactosylated polymers. Biomacromolecules 16, 3008–3020 (2015).
Han, J. et al. Acute and chronic shear stress differently regulate endothelial internalization of nanocarriers targeted to platelet-endothelial cell adhesion molecule-1. ACS Nano 6, 8824–8836 (2012).
Parhiz, H. et al. PECAM-1 directed re-targeting of exogenous mRNA providing two orders of magnitude enhancement of vascular delivery and expression in lungs independent of apolipoprotein E-mediated uptake. J. Control. Rel. 291, 106–115 (2018).
Dan, M., Cochran, D. B., Yokel, R. A. & Dziubla, T. D. Binding, transcytosis and biodistribution of anti-PECAM-1 iron oxide nanoparticles for brain-targeted delivery. PLoS ONE 8, e81051 (2013).
Reychler, G. & Michotte, J. B. Development challenges and opportunities in aerosol drug delivery systems in non-invasive ventilation in adults. Expert Opin. Drug Deliv. 16, 153–162 (2019).
Mainprize, T. et al. Blood-brain barrier opening in primary brain tumors with non-invasive MR-guided focused ultrasound: a clinical safety and feasibility study. Sci. Rep. 9, 321 (2019).
Wang, C., Ye, Y., Hochu, G. M., Sadeghifar, H. & Gu, Z. Enhanced cancer immunotherapy by microneedle patch-assisted delivery of anti-PD1 antibody. Nano Lett. 16, 2334–2340 (2016).
Shi, J., Kantoff, P. W., Wooster, R. & Farokhzad, O. C. Cancer nanomedicine: progress, challenges and opportunities. Nat. Rev. Cancer 17, 20–37 (2017).
Hrkach, J. et al. Preclinical development and clinical translation of a PSMA-targeted docetaxel nanoparticle with a differentiated pharmacological profile. Sci. Transl. Med. 4, 128ra39 (2012).
Wang, Y., Huang, H. Y., Yang, L., Zhang, Z. & Ji, H. Cetuximab-modified mesoporous silica nano-medicine specifically targets EGFR-mutant lung cancer and overcomes drug resistance. Sci. Rep. 6, 25468 (2016).
Shamay, Y., Golan, M., Tyomkin, D. & David, A. Assessing the therapeutic efficacy of VEGFR-1-targeted polymer drug conjugates in mouse tumor models. J. Control. Rel. 229, 192–199 (2016).
Oh, P. et al. In vivo proteomic imaging analysis of caveolae reveals pumping system to penetrate solid tumors. Nat. Med. 20, 1062–1068 (2014).
Shamay, Y., Paulin, D., Ashkenasy, G. & David, A. E-selectin binding peptide–polymer–drug conjugates and their selective cytotoxicity against vascular endothelial cells. Biomaterials 30, 6460–6468 (2009).
Shamay, Y. et al. Inhibition of primary and metastatic tumors in mice by E-selectin-targeted polymer-drug conjugates. J. Control. Rel. 217, 102–112 (2015).
Seguin, L., Desgrosellier, J. S., Weis, S. M. & Cheresh, D. A. Integrins and cancer: regulators of cancer stemness, metastasis, and drug resistance. Trends Cell Biol. 25, 234–240 (2015).
Hallahan, D. E. et al. Targeting drug delivery to radiation-induced neoantigens in tumor microvasculature. J. Control. Rel. 74, 183–191 (2001).
Temming, K. et al. Delivery of the p38 MAPkinase inhibitor SB202190 to angiogenic endothelial cells: development of novel RGD-equipped and PEGylated drug–albumin conjugates using platinum(II)-based drug linker technology. Bioconjug. Chem. 17, 1246–1255 (2006).
Li, X. et al. Synthesis and biological evaluation of RGD-conjugated MEK1/2 kinase inhibitors for integrin-targeted cancer therapy. Molecules 18, 13957–13978 (2013).
Chandarlapaty, S. Negative feedback and adaptive resistance to the targeted therapy of cancer. Cancer Discov. 2, 311–319 (2012).
Carracedo, A. et al. Inhibition of mTORC1 leads to MAPK pathway activation through a PI3K-dependent feedback loop in human cancer. J. Clin. Invest. 118, 3065–3074 (2008).
O’Reilly, K. E. et al. mTOR inhibition induces upstream receptor tyrosine kinase signaling and activates Akt. Cancer Res. 66, 1500–1508 (2006).
Sergina, N. V. et al. Escape from HER-family tyrosine kinase inhibitor therapy by the kinase-inactive HER3. Nature 445, 437–441 (2007).
Coffee, E. M. et al. Concomitant BRAF and PI3K/mTOR blockade is required for effective treatment of BRAF(V600E) colorectal cancer. Clin. Cancer Res. 19, 2688–2698 (2013).
Wee, S. et al. PI3K pathway activation mediates resistance to MEK inhibitors in KRAS mutant cancers. Cancer Res. 69, 4286–4293 (2009).
Molina-Arcas, M., Hancock, D. C., Sheridan, C., Kumar, M. S. & Downward, J. Coordinate direct input of both KRAS and IGF1 receptor to activation of PI3 kinase in KRAS-mutant lung cancer. Cancer Discov. 3, 548–563 (2013).
Murillo, M. M. et al. RAS interaction with PI3K p110alpha is required for tumor-induced angiogenesis. J. Clin. Invest. 124, 3601–3611 (2014).
Manchado, E. et al. A combinatorial strategy for treating KRAS-mutant lung cancer. Nature 534, 647–651 (2016).
Chandarlapaty, S. et al. AKT inhibition relieves feedback suppression of receptor tyrosine kinase expression and activity. Cancer Cell 19, 58–71 (2011).
Serra, V. et al. PI3K inhibition results in enhanced HER signaling and acquired ERK dependency in HER2-overexpressing breast cancer. Oncogene 30, 2547–2557 (2011).
Chakrabarty, A., Sanchez, V., Kuba, M. G., Rinehart, C. & Arteaga, C. L. Feedback upregulation of HER3 (ErbB3) expression and activity attenuates antitumor effect of PI3K inhibitors. Proc. Natl Acad. Sci. USA 109, 2718–2723 (2012).
Garrett, J. T. et al. Transcriptional and posttranslational up-regulation of HER3 (ErbB3) compensates for inhibition of the HER2 tyrosine kinase. Proc. Natl Acad. Sci. USA 108, 5021–5026 (2011).
Tao, J. J. et al. Antagonism of EGFR and HER3 enhances the response to inhibitors of the PI3K-Akt pathway in triple-negative breast cancer. Sci. Signal. 7, ra29 (2014).
Carver, B. S. et al. Reciprocal feedback regulation of PI3K and androgen receptor signaling in PTEN-deficient prostate cancer. Cancer Cell 19, 575–586 (2011).
Bosch, A. et al. PI3K inhibition results in enhanced estrogen receptor function and dependence in hormone receptor–positive breast cancer. Sci. Transl. Med. 7, 283ra51 (2015).
Toska, E. et al. PI3K pathway regulates ER-dependent transcription in breast cancer through the epigenetic regulator KMT2D. Science 355, 1324–1330 (2017).
Dickler, M. N. et al. A phase II study of the PI3K inhibitor taselisib (GDC-0032) combined with fulvestrant (F) in patients (pts) with HER2-negative (HER2-), hormone receptor-positive (HR+) advanced breast cancer (BC). J. Clin. Oncol. 34, 520 (2016).
Saura, C. et al. Primary results of LORELEI: a phase II randomized, double-blind study of neoadjuvant letrozole (LET) plus taselisib versus LET plus placebo (PLA) in postmenopausal patients (pts) with ER+/HER2-negative early breast cancer (EBC). Ann. Oncol. https://doi.org/10.1093/annonc/mdx440.001 (2017).
Juvekar, A. et al. Combining a PI3K inhibitor with a PARP inhibitor provides an effective therapy for BRCA1-related breast cancer. Cancer Discov. 2, 1048–1063 (2012).
Sun, C. et al. Rational combination therapy with PARP and MEK inhibitors capitalizes on therapeutic liabilities in RAS mutant cancers. Sci. Transl. Med. 9, eaal5148 (2017).
Lee, H.-Y. et al. Response of non-small cell lung cancer cells to the inhibitors of phosphatidylinositol 3-kinase/Akt- and MAPK kinase 4/c-Jun NH2-terminal kinase pathways: an effective therapeutic strategy for lung cancer. Clin. Cancer Res. 11, 6065–6074 (2005).
Duong, H.-Q., Kim, H. J., Kang, H. J., Seong, Y.-S. & Bae, I. ZSTK474, a PI3K inhibitor, suppresses proliferation and sensitizes to gemcitabine in human pancreatic adenocarcinoma cells. Oncol. Rep. 27, 182–188 (2012).
Dreaden, E. C. et al. Tumor-targeted synergistic blockade of MAPK and PI3K from a layer-by-layer nanoparticle. Clin. Cancer Res. 21, 4410–4419 (2015). A notable example of the use of nanomedicine to enable a therapeutic combination to combat drug resistance and avoid toxicity of kinase inhibitors.
Pisarevsky, E. et al. Rational design of polyglutamic acid delivering an optimized combination of drugs targeting mutated BRAF and MEK in melanoma. Adv. Ther. https://doi.org/10.1002/adtp.202000028 (2020).
Morton, S. W. et al. A nanoparticle-based combination chemotherapy delivery system for enhanced tumor killing by dynamic rewiring of signaling pathways. Sci. Signal. 7, ra44 (2014).
Ibrahim, Y. H. et al. PI3K inhibition impairs BRCA1/2 expression and sensitizes BRCA-proficient triple-negative breast cancer to PARP inhibition. Cancer Discov. 2, 1036–1047 (2012).
Patel, S. et al. Boosting intracellular delivery of lipid nanoparticle-encapsulated mRNA. Nano Lett. 17, 5711–5718 (2017).
Fire, A. et al. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391, 806–811 (1998).
Ting, A. H., Schuebel, K. E., Herman, J. G. & Baylin, S. B. Short double-stranded RNA induces transcriptional gene silencing in human cancer cells in the absence of DNA methylation. Nat. Genet. 37, 906–910 (2005).
Xu, C.-f & Wang, J. Delivery systems for siRNA drug development in cancer therapy. Asian J. Pharm. Sci. 10, 1–12 (2015).
Jackson, A. L. & Linsley, P. S. Recognizing and avoiding siRNA off-target effects for target identification and therapeutic application. Nat. Rev. Drug Discov. 9, 57–67 (2010).
Wang, J., Lu, Z., Wientjes, M. G. & Au, J. L. S. Delivery of siRNA therapeutics: barriers and carriers. AAPS J. 12, 492–503 (2010).
Wong, A. W., Baginski, T. K. & Reilly, D. E. Enhancement of DNA uptake in FUT8-deleted CHO cells for transient production of afucosylated antibodies. Biotechnol. Bioeng. 106, 751–763 (2010).
Kim, Y. G., Cha, J. & Chandrasegaran, S. Hybrid restriction enzymes: zinc finger fusions to Fok I cleavage domain. Proc. Natl Acad. Sci. USA 93, 1156–1160 (1996).
Chou, S. T., Leng, Q. & Mixson, A. J. Zinc finger nucleases: tailor-made for gene therapy. Drugs Future 37, 183–196 (2012).
LaFountaine, J. S., Fathe, K. & Smyth, H. D. C. Delivery and therapeutic applications of gene editing technologies ZFNs, TALENs, and CRISPR/Cas9. Int. J. Pharm. 494, 180–194 (2015).
Perez, E. E. et al. Establishment of HIV-1 resistance in CD4(+) T cells by genome editing using zinc-finger nucleases. Nat. Biotechnol. 26, 808–816 (2008).
Mussolino, C. et al. A novel TALE nuclease scaffold enables high genome editing activity in combination with low toxicity. Nucleic Acids Res. 39, 9283–9293 (2011).
Zhu, H. et al. Baculoviral transduction facilitates TALEN-mediated targeted transgene integration and Cre/LoxP cassette exchange in human-induced pluripotent stem cells. Nucleic Acids Res. 41, e180 (2013).
Hu, Z. et al. TALEN-mediated targeting of HPV oncogenes ameliorates HPV-related cervical malignancy. J. Clin. Invest. 125, 425–436 (2015).
Ru, R. et al. Targeted genome engineering in human induced pluripotent stem cells by penetrating TALENs. Cell Regen. 2, 5 (2013).
Liu, J., Gaj, T., Patterson, J. T., Sirk, S. J. & Barbas III, C. F. Cell-penetrating peptide-mediated delivery of TALEN proteins via bioconjugation for genome engineering. PLoS ONE 9, e85755 (2014).
Wang, J. & Quake, S. R. RNA-guided endonuclease provides a therapeutic strategy to cure latent herpesviridae infection. Proc. Natl Acad. Sci. USA 111, 13157–13162 (2014).
Cheng, R. et al. Efficient gene editing in adult mouse livers via adenoviral delivery of CRISPR/Cas9. FEBS Lett. 588, 3954–3958 (2014).
Mintzer, M. A. & Simanek, E. E. Nonviral vectors for gene delivery. Chem. Rev. 109, 259–302 (2009).
Putnam, D. Polymers for gene delivery across length scales. Nat. Mater. 5, 439–451 (2006).
Davis, M. E. The first targeted delivery of siRNA in humans via a self-assembling, cyclodextrin polymer-based nanoparticle: from concept to clinic. Mol. Pharm. 6, 659–668 (2009).
Semple, S. C. et al. Rational design of cationic lipids for siRNA delivery. Nat. Biotechnol. 28, 172–176 (2010).
Love, K. T. et al. Lipid-like materials for low-dose, in vivo gene silencing. Proc. Natl Acad. Sci. USA 107, 1864–1869 (2010).
Monopoli, M. P., Aberg, C., Salvati, A. & Dawson, K. A. Biomolecular coronas provide the biological identity of nanosized materials. Nat. Nano 7, 779–786 (2012).
Ando, H. et al. Polycation liposomes as a vector for potential intracellular delivery of microRNA. J. Gene Med. 15, 375–383 (2013).
Kim, J. S., Oh, M. H., Park, J. Y., Park, T. G. & Nam, Y. S. Protein-resistant, reductively dissociable polyplexes for in vivo systemic delivery and tumor-targeting of siRNA. Biomaterials 34, 2370–2379 (2013).
Guo, J. et al. Systemic delivery of therapeutic small interfering RNA using a pH-triggered amphiphilic poly-l-lysine nanocarrier to suppress prostate cancer growth in mice. Eur. J. Pharm. Sci. 45, 521–532 (2012).
Weinstein, S. et al. Harnessing RNAi-based nanomedicines for therapeutic gene silencing in B-cell malignancies. Proc. Natl Acad. Sci. USA 113, E16–E22 (2016).
Tabernero, J. et al. First-in-humans trial of an RNA interference therapeutic targeting VEGF and KSP in cancer patients with liver involvement. Cancer Discov. 3, 406–417 (2013).
Liang, X. et al. Rapid and highly efficient mammalian cell engineering via Cas9 protein transfection. J. Biotechnol. 208, 44–53 (2015).
Zuris, J. A. et al. Efficient delivery of genome-editing proteins in vitro and in vivo. Nat. Biotechnol. 33, 73–80 (2015).
Wang, M. et al. Efficient delivery of genome-editing proteins using bioreducible lipid nanoparticles. Proc. Natl Acad. Sci. USA 113, 2868–2873 (2016).
Yin, H. et al. Therapeutic genome editing by combined viral and non-viral delivery of CRISPR system components in vivo. Nat. Biotechnol. 34, 328–333 (2016).
Miller, J. B. et al. Non-viral CRISPR/Cas gene editing in vitro and in vivo enabled by synthetic nanoparticle co-delivery of Cas9 mRNA and sgRNA. Angew. Chem. Int. Ed. Engl. 56, 1059–1063 (2017).
Ramakrishna, S. et al. Gene disruption by cell-penetrating peptide-mediated delivery of Cas9 protein and guide RNA. Genome Res. 24, 1020–1027 (2014).
Platt, R. J. et al. CRISPR-Cas9 knockin mice for genome editing and cancer modeling. Cell 159, 440–455 (2014).
Wei, T., Cheng, Q., Min, Y.-L., Olson, E. N. & Siegwart, D. J. Systemic nanoparticle delivery of CRISPR-Cas9 ribonucleoproteins for effective tissue specific genome editing. Nat. Commun. 11, 3232 (2020).
Hu, Q. et al. Conjugation of haematopoietic stem cells and platelets decorated with anti-PD-1 antibodies augments anti-leukaemia efficacy. Nat. Biomed. Eng. 2, 831–840 (2018).
Robert, C. et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 372, 320–330 (2014).
Chen, Q. et al. Photothermal therapy promotes tumor infiltration and antitumor activity of CAR T cells. Adv. Mater. 31, 1900192 (2019).
Larkin, J. et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N. Engl. J. Med. 373, 23–34 (2015).
Menon, S., Shin, S. & Dy, G. Advances in cancer immunotherapy in solid tumors. Cancers 8, 106 (2016).
de Charette, M., Marabelle, A. & Houot, R. Turning tumour cells into antigen presenting cells: The next step to improve cancer immunotherapy? Eur. J. Cancer 68, 134–147 (2016).
Santomasso, B., Bachier, C., Westin, J., Rezvani, K. & Shpall, E. J. The other side of CAR T-cell therapy: cytokine release syndrome, neurologic toxicity, and financial burden. Am. Soc. Clin. Oncol. Educ. Book 39, 433–444 (2019).
Jo, S. D., Nam, G.-H., Kwak, G., Yang, Y. & Kwon, I. Harnessing designed nanoparticles: Current strategies and future perspectives in cancer immunotherapy. Nano Today 17, 23–37 (2017).
Nam, J. et al. Cancer nanomedicine for combination cancer immunotherapy. Nat. Rev. Mater. 4, 398–414 (2019). Opportunities for combination immunotherapy based on nanoparticle platforms designed for chemotherapy, photothermal therapy, photodynamic therapy, radiotherapy and gene therapy.
Liu, Z., Jiang, W., Nam, J., Moon, J. J. & Kim, B. Y. S. Immunomodulating nanomedicine for cancer therapy. Nano Lett. 18, 6655–6659 (2018).
Zhang, L.-x, Xie, X.-x, Liu, D.-q, Xu, Z. P. & Liu, R.-t Efficient co-delivery of neo-epitopes using dispersion-stable layered double hydroxide nanoparticles for enhanced melanoma immunotherapy. Biomaterials 174, 54–66 (2018).
Alskar, L. C., Porter, C. J. & Bergstrom, C. A. Tools for early prediction of drug loading in lipid-based formulations. Mol. Pharm. 13, 251–261 (2016).
Kuai, R., Ochyl, L. J., Bahjat, K. S., Schwendeman, A. & Moon, J. J. Designer vaccine nanodiscs for personalized cancer immunotherapy. Nat. Mater. 16, 489–496 (2016).
Jeanbart, L. et al. Enhancing efficacy of anticancer vaccines by targeted delivery to tumor-draining lymph nodes. Cancer Immunol. Res. 2, 436–447 (2014).
Kim, J. et al. Injectable, spontaneously assembling, inorganic scaffolds modulate immune cells in vivo and increase vaccine efficacy. Nat. Biotechnol. 33, 64–72 (2015).
Li, A. W. et al. A facile approach to enhance antigen response for personalized cancer vaccination. Nat. Mater. 17, 528–534 (2018).
Lu, K. et al. Low-dose X-ray radiotherapy–radiodynamic therapy via nanoscale metal–organic frameworks enhances checkpoint blockade immunotherapy. Nat. Biomed. Eng. 2, 600–610 (2018).
Sau, S. et al. Multifunctional nanoparticles for cancer immunotherapy: A groundbreaking approach for reprogramming malfunctioned tumor environment. J. Control. Rel. 274, 24–34 (2018).
Lu, C. et al. Phase I clinical trial of systemically administered TUSC2(FUS1)-nanoparticles mediating functional gene transfer in humans. PLoS ONE 7, e34833 (2012).
Ji, L. et al. Expression of several genes in the human chromosome 3p21.3 homozygous deletion region by an adenovirus vector results in tumor suppressor activities in vitro and in vivo. Cancer Res. 62, 2715–2720 (2002).
Demeure, M. J. et al. A phase I/II study of TKM-080301, a PLK1-targeted RNAi in patients with adrenocortical cancer (ACC). J. Clin. Oncol. 34, 2547–2547 (2016).
M.D. Anderson Cancer Center https://ClinicalTrials.gov/show/NCT01591356 (2015).
Sarker, D. et al. First-in-human, first-in-class phase I study of MTL-CEBPA, a small activating RNA (saRNA) targeting the transcription factor C/EBP-α in patients with advanced liver cancer. J. Clin. Oncol. 35, TPS2612 (2017).
Gonzalez-Angulo, A. M. et al. Weekly nab-rapamycin in patients with advanced nonhematologic malignancies: final results of a phase I trial. Clin. Cancer Res. 19, 5474–5484 (2013).
Plummer, R. et al. A Phase I clinical study of cisplatin-incorporated polymeric micelles (NC-6004) in patients with solid tumours. Br. J. Cancer 104, 593–598 (2011).
Shamay, Y. Quantitative prediction of self assembly yields tumor targeted nanoparticles. Nat. Mater. 17, 361–368 (2018). Predicting drug encapsulation into nanocarriers based on drug molecular structure.
Tuomela, A., Hirvonen, J. & Peltonen, L. Stabilizing agents for drug nanocrystals: effect on bioavailability. Pharmaceutics 8, 16 (2016).
Guo, S. & Huang, L. Nanoparticles containing insoluble drug for cancer therapy. Biotechnol. Adv. 32, 778–788 (2014).
Woodhead, J. L. & Hall, C. K. Encapsulation efficiency and micellar structure of solute-carrying block copolymer nanoparticles. Macromolecules 44, 5443–5451 (2011).
Dinarvand, R., Sepehri, N., Manoochehri, S., Rouhani, H. & Atyabi, F. Polylactide-co-glycolide nanoparticles for controlled delivery of anticancer agents. Int. J. Nanomed. 6, 877–895 (2011).
Maojo, V. et al. Nanoinformatics: a new area of research in nanomedicine. Int. J. Nanomed. 7, 3867–3890 (2012).
Seidler, J., McGovern, S. L., Doman, T. N. & Shoichet, B. K. Identification and prediction of promiscuous aggregating inhibitors among known drugs. J. Med. Chem. 46, 4477–4486 (2003).
Irwin, J. J. et al. An aggregation advisor for ligand discovery. J. Med. Chem. 58, 7076–7087 (2015).
Feng, B. Y., Shelat, A., Doman, T. N., Guy, R. K. & Shoichet, B. K. High-throughput assays for promiscuous inhibitors. Nat. Chem. Biol. 1, 146–148 (2005).
Puzyn, T. et al. Using nano-QSAR to predict the cytotoxicity of metal oxide nanoparticles. Nat. Nanotechnol. 6, 175–178 (2011).
Zhang, Y. et al. Lipid-modified aminoglycoside derivatives for in vivo siRNA delivery. Adv. Mater. 25, 4641–4645 (2013).
Guo, C., Luo, Y., Zhou, R. & Wei, G. Probing the self-assembly mechanism of diphenylalanine-based peptide nanovesicles and nanotubes. ACS Nano 6, 3907–3918 (2012).
Guo, C., Luo, Y., Zhou, R. & Wei, G. Triphenylalanine peptides self-assemble into nanospheres and nanorods that are different from the nanovesicles and nanotubes formed by diphenylalanine peptides. Nanoscale 6, 2800–2811 (2014).
Lee, O. S., Stupp, S. I. & Schatz, G. C. Atomistic molecular dynamics simulations of peptide amphiphile self-assembly into cylindrical nanofibers. J. Am. Chem. Soc. 133, 3677–3683 (2011).
Frederix, P. W. et al. Exploring the sequence space for (tri-)peptide self-assembly to design and discover new hydrogels. Nat. Chem. 7, 30–37 (2015).
Shi, C. et al. A drug-specific nanocarrier design for efficient anticancer therapy. Nat. Commun. 6, 7449 (2015). De novo nanocarrier design based on drug structure to enhance activity.
Yaari, Z. et al. Theranostic barcoded nanoparticles for personalized cancer medicine. Nat. Commun. 7, 13325 (2016).
Gebhart, G. et al. Molecular imaging as a tool to investigate heterogeneity of advanced HER2-positive breast cancer and to predict patient outcome under trastuzumab emtansine (T-DM1): the ZEPHIR trial. Ann. Oncol. 27, 619–624 (2016).
Perez-Gracia, J. L. et al. Strategies to design clinical studies to identify predictive biomarkers in cancer research. Cancer Treat. Rev. 53, 79–97 (2017).
Arai, N., Sasaki, H., Tamura, R., Ohara, K. & Yoshida, K. Unusual magnetic resonance imaging findings of a glioblastoma arising during treatment with lenvatinib for thyroid cancer. World Neurosurg. 107, 1047.e9–1047.e15 (2017).
Man, F., Lammers, T. & de Rosales, R. T. M. Imaging nanomedicine-based drug delivery: a review of clinical studies. Mol. Imaging Biol. 20, 683–695 (2018). A review of clinical data on the imaging of nanoparticle uptake into tumours to predict efficacy of drug delivery in tumours.
Weickhardt, A. J. et al. Rapid-onset hypogonadism secondary to crizotinib use in men with metastatic nonsmall cell lung cancer. Cancer 118, 5302–5309 (2012).
Chapman, P. B. et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 364, 2507–2516 (2011).
Lee, H. et al. (64)Cu-MM-302 positron emission tomography quantifies variability of enhanced permeability and retention of nanoparticles in relation to treatment response in patients with metastatic breast cancer. Clin. Cancer Res. 23, 4190–4202 (2017).
Golombek, S. K. et al. Tumor targeting via EPR: Strategies to enhance patient responses. Adv. Drug. Deliv. Rev. 130, 17–38 (2018).
Lammers, T. et al. Cancer nanomedicine: is targeting our target? Nat. Rev. Mater. 1, 16069 (2016).
Schmid, D. et al. T cell-targeting nanoparticles focus delivery of immunotherapy to improve antitumor immunity. Nat. Commun. 8, 1747 (2017).
Lucky, S. S., Soo, K. C. & Zhang, Y. Nanoparticles in photodynamic therapy. Chem. Rev. 115, 1990–2042 (2015).
Tinkle, S. et al. Nanomedicines: addressing the scientific and regulatory gap. Ann. N. Y. Acad. Sci. 1313, 35–56 (2014).
Crist, R. M. et al. Common pitfalls in nanotechnology: lessons learned from NCI’s Nanotechnology Characterization Laboratory. Integr. Biol. 5, 66–73 (2013). Trends in nanoparticle safety and biocompatibility, and common mistakes regarding nanoparticle development for translation.
Piret, J.-P. et al. Pan-European inter-laboratory studies on a panel of in vitro cytotoxicity and pro-inflammation assays for nanoparticles. Arch. Toxicol. 91, 2315–2330 (2017).
Kantarjian, H. M. et al. Bosutinib safety and management of toxicity in leukemia patients with resistance or intolerance to imatinib and other tyrosine kinase inhibitors. Blood 9, 1309–1318 (2014).
Shaw, A. T. et al. Ceritinib in ALK-rearranged non–small-cell lung cancer. N. Engl. J. Med. 370, 1189–1197 (2014).
Hauschild, A. et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, open-label, phase 3 randomised controlled trial. Lancet 380, 358–365 (2012).
Robert, C. et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N. Engl. J. Med. 372, 30–39 (2015).
Welsh, S. J. & Corrie, P. G. Management of BRAF and MEK inhibitor toxicities in patients with metastatic melanoma. Ther. Adv. Med. Oncol. 7, 122–136 (2015).
Sequist, L. V. et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J. Clin. Oncol. 31, 3327–3334 (2013).
Flaherty, K. T. et al. Improved survival with MEK inhibition in BRAF-mutated melanoma. N. Engl. J. Med. 367, 107–114 (2012).
Wells, E. M., Rao, A. A. N., Scafidi, J. & Packer, R. J. Neurotoxicity of biologically targeted agents in pediatric cancer trials. Pediatr. Neurol. 46, 212–221 (2012).
Dreyling, M. et al. Phase 2A study of copanlisib, aNnovel PI3K Inhibitor, in patients with indolent lymphoma. Blood 124, 1701 (2014).
Younes, A. et al. An open-label phase II study of buparlisib (BKM120) in patients with relapsed and refractory diffuse large B-cell lymphoma, mantle cell lymphoma or follicular lymphoma. Blood 124, 1718 (2014).
Younes, A. et al. An open-label phase II study of buparlisib (BKM120) in patients with relapsed and refractory diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma (MCL) and follicular lymphoma (FL). Blood 126, 1493 (2015).
Coutré, S. E. et al. Management of adverse events associated with idelalisib treatment: expert panel opinion. Leuk. Lymphoma 56, 2779–2786 (2015).
Cheah, C. Y. & Fowler, N. H. Idelalisib in the management of lymphoma. Blood 128, 331–336 (2016).
Robinson, G. W. et al. Irreversible growth plate fusions in children with medulloblastoma treated with a targeted hedgehog pathway inhibitor. Oncotarget 8, 69295–69302 (2017).
Seet, R. C. S., Rabinstein, A. A., Lindell, P. E., Uhm, J. H. & Wijdicks, E. F. Cerebrovascular events after bevacizumab treatment: an early and severe complication. Neurocrit. Care 15, 421–427 (2011).
Seet, R. C. S. & Rabinstein, A. A. Clinical features and outcomes of posterior reversible encephalopathy syndrome following bevacizumab treatment. QJM 105, 69–75 (2012).
Grothey, A. et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381, 303–312 (2013).
Jhaveri, K. D., Wanchoo, R., Sakhiya, V., Ross, D. W. & Fishbane, S. Adverse renal effects of novel molecular oncologic targeted therapies: a narrative review. Kidney Int. Rep. 2, 108–123 (2017).
Elisei, R. et al. Cabozantinib in progressive medullary thyroid cancer. J. Clin. Oncol. 31, 3639–3646 (2013).
Schlumberger, M. et al. Lenvatinib versus placebo in radioiodine-refractory thyroid caner. N. Engl. J. Med. 372, 621–630 (2015).
Qi, W.-X., Shen, Z., Tang, L.-N. & Yao, Y. Congestive heart failure risk in cancer patients treated with vascular endothelial growth factor tyrosine kinase inhibitors: a systematic review and meta-analysis of 36 clinical trials. Br. J. Clin. Pharmacol. 78, 748–762 (2014).
Alibolandi, M. et al. The chemotherapeutic potential of doxorubicin-loaded PEG-b-PLGA nanopolymersomes in mouse breast cancer model. Eur. J. Pharm. Biopharm. 94, 521–531 (2015).
Sirsi, S. R., Williams, J. H. & Lutz, G. J. Poly(ethylene imine)–poly(ethylene glycol) copolymers facilitate efficient delivery of antisense oligonucleotides to nuclei of mature muscle cells of mdx mice. Hum. Gene Ther. 16, 1307–1317 (2005).
Tian, H. Y. et al. Biodegradable cationic PEG-PEI-PBLG hyperbranched block copolymer: synthesis and micelle characterization. Biomaterials 26, 4209–4217 (2005).
Kulkarni, S. B., Betageri, G. V. & Singh, M. Factors affecting microencapsulation of drugs in liposomes. J. Microencapsul. 12, 229–246 (1995).
Allen, T. M., Sapra, P. & Moase, E. Use of the post-insertion method for the formation of ligand-coupled liposomes. Cell. Mol. Biol. Lett. 7, 889–894 (2002).
Nii, T. & Ishii, F. Encapsulation efficiency of water-soluble and insoluble drugs in liposomes prepared by the microencapsulation vesicle method. Int. J. Pharm. 298, 198–205 (2005).
Muller, R. H. & Keck, C. M. Challenges and solutions for the delivery of biotech drugs–a review of drug nanocrystal technology and lipid nanoparticles. J. Biotechnol. 113, 151–170 (2004).
Lv, P. P. et al. Porous quaternized chitosan nanoparticles containing paclitaxel nanocrystals improved therapeutic efficacy in non-small-cell lung cancer after oral administration. Biomacromolecules 12, 4230–4239 (2011).
Junghanns, J. U. & Muller, R. H. Nanocrystal technology, drug delivery and clinical applications. Int. J. Nanomed. 3, 295–309 (2008).
Peterson, C. M. et al. HPMA copolymer delivery of chemotherapy and photodynamic therapy in ovarian cancer. Adv. Exp. Med. Biol. 519, 101–123 (2003).
Veronese, F. M. et al. PEG-doxorubicin conjugates: influence of polymer structure on drug release, in vitro cytotoxicity, biodistribution, and antitumor activity. Bioconjug. Chem. 16, 775–784 (2005).
Fox, M. E., Szoka, F. C. & Frechet, J. M. Soluble polymer carriers for the treatment of cancer: the importance of molecular architecture. Acc. Chem. Res. 42, 1141–1151 (2009).
Desai, N. et al. Increased antitumor activity, intratumor paclitaxel concentrations, and endothelial cell transport of cremophor-free, albumin-bound paclitaxel, ABI-007, compared with cremophor-based paclitaxel. Clin. Cancer Res. 12, 1317–1324 (2006).
Altintas, I. et al. Nanobody-albumin nanoparticles (NANAPs) for the delivery of a multikinase inhibitor 17864 to EGFR overexpressing tumor cells. J. Control. Rel. 165, 110–118 (2013).
Dong, C., Li, B., Li, Z., Shetty, S. & Fu, J. Dasatinib-loaded albumin nanoparticles possess diminished endothelial cell barrier disruption and retain potent anti-leukemia cell activity. Oncotarget 7, 49699–49709 (2016).
Gu, J., Fan, W., Shimojima, A. & Okubo, T. Organic–inorganic mesoporous nanocarriers integrated with biogenic ligands. Small 3, 1740–1744 (2007).
Loh, X. J. & Lee, T. C. Gene delivery by functional inorganic nanocarriers. Recent Pat. DNA Gene Seq. 6, 108–114 (2012).
Lin, G., Mi, P., Chu, C., Zhang, J. & Liu, G. Inorganic nanocarriers overcoming multidrug resistance for cancer theranostics. Adv. Sci. 3, 1600134 (2016).
Nissinen, T. et al. Tailored dual PEGylation of inorganic porous nanocarriers for extremely long blood circulation in vivo. ACS Appl. Mater. Interfaces 8, 32723–32731 (2016).
Pan, G. et al. Mesoporous silica nanoparticles (MSNs)-based organic/inorganic hybrid nanocarriers loading 5-Fluorouracil for the treatment of colon cancer with improved anticancer efficacy. Colloids Surf. B Biointerfaces 159, 375–385 (2017).
Levina, M. & Rajabi-Siahboomi, A. R. The influence of excipients on drug release from hydroxypropyl methylcellulose matrices. J. Pharm. Sci. 93, 2746–2754 (2004).
Jeong, B., Bae, Y. H. & Kim, S. W. Drug release from biodegradable injectable thermosensitive hydrogel of PEG–PLGA–PEG triblock copolymers. J. Control. Rel. 63, 155–163 (2000).
Kamaly, N., Yameen, B., Wu, J. & Farokhzad, O. C. Degradable controlled-release polymers and polymeric nanoparticles: mechanisms of controlling drug release. Chem. Rev. 116, 2602–2663 (2016).
Manzari, M. T. et al. Genomically informed small-molecule drugs overcome resistance to a sustained-release formulation of an engineered death receptor agonist in patient-derived tumor models. Sci. Adv. 5, eaaw9162 (2019).
Tanner, T. & Marks, R. Delivering drugs by the transdermal route: review and comment. Skin Res. Technol. 14, 249–260 (2008).
Ma, G. & Wu, C. Microneedle, bio-microneedle and bio-inspired microneedle: a review. J. Control. Rel. 251, 11–23 (2017).
Prausnitz, M. R. Microneedles for transdermal drug delivery. Adv. Drug Deliv. Rev. 56, 581–587 (2004).
Ruel-Gariépy, E. & Leroux, J.-C. In situ-forming hydrogels—review of temperature-sensitive systems. Eur. J. Pharm. Biopharm. 58, 409–426 (2004).
Murdan, S. Electro-responsive drug delivery from hydrogels. J. Control. Rel. 92, 1–17 (2003).
Lin, C.-C. & Metters, A. T. Hydrogels in controlled release formulations: Network design and mathematical modeling. Adv. Drug Deliv. Rev. 58, 1379–1408 (2006).
Sham, J. O. H., Zhang, Y., Finlay, W. H., Roa, W. H. & Löbenberg, R. Formulation and characterization of spray-dried powders containing nanoparticles for aerosol delivery to the lung. Int. J. Pharm. 269, 457–467 (2004).
Sung, J. C., Pulliam, B. L. & Edwards, D. A. Nanoparticles for drug delivery to the lungs. Trends Biotechnol. 25, 563–570 (2007).
Mansour, H. M., Rhee, Y.-S. & Wu, X. Nanomedicine in pulmonary delivery. Int. J. Nanomed. 4, 299–319 (2009).
Liu, Z. et al. Drug delivery with carbon nanotubes for in vivo cancer treatment. Cancer Res. 68, 6652–6660 (2008).
Gillies, E. R. & Fréchet, J. M. J. Dendrimers and dendritic polymers in drug delivery. Drug Discov. Today 10, 35–43 (2005).
Liu, M. & Fréchet, J. M. Designing dendrimers for drug delivery. Pharm. Sci. Technol. Today 2, 393–401 (1999).
Patri, A. K., Majoros, I. J. & Baker, J. R. Dendritic polymer macromolecular carriers for drug delivery. Curr. Opin. Chem. Biol. 6, 466–471 (2002).
Leto, S. M. & Trusolino, L. Primary and acquired resistance to EGFR-targeted therapies in colorectal cancer: impact on future treatment strategies. J. Mol. Med. 92, 709–722 (2014).
Sato, H. et al. Combined inhibition of MEK and PI3K pathways overcomes acquired resistance to EGFR-TKIs in non-small cell lung cancer. Cancer Sci. 109, 3183–3196 (2018).
Wilson, F. H. et al. A functional landscape of resistance to ALK inhibition in lung cancer. Cancer Cell 27, 397–408 (2015).
Masoud, V. & Pagès, G. Targeted therapies in breast cancer: New challenges to fight against resistance. World J. Clin. Oncol. 8, 120–134 (2017).
Higgins, M. J. & Baselga, J. Targeted therapies for breast cancer. J. Clin. Investig. 121, 3797–3803 (2011).
Villanueva, J. et al. Acquired resistance to BRAF inhibitors mediated by a RAF kinase switch in melanoma can be overcome by cotargeting MEK and IGF-1R/PI3K. Cancer Cell 18, 683–695 (2010).
McCubrey, J. A. et al. Targeting survival cascades induced by activation of Ras/Raf/MEK/ERK, PI3K/PTEN/Akt/mTOR and Jak/STAT pathways for effective leukemia therapy. Leukemia 22, 708–722 (2008).
McLornan, D., Percy, M. & McMullin, M. F. JAK2 V617F: a single mutation in the myeloproliferative group of disorders. Ulster Med. J. 75, 112–119 (2006).
Piloto, O. et al. Prolonged exposure to FLT3 inhibitors leads to resistance via activation of parallel signaling pathways. Blood 109, 1643–1652 (2007).
Chu, S. et al. Detection of BCR-ABL kinase mutations in CD34+ cells from chronic myelogenous leukemia patients in complete cytogenetic remission on imatinib mesylate treatment. Blood 105, 2093–2098 (2005).
Jiang, Z.-M. et al. Crizotinib-loaded polymeric nanoparticles in lung cancer chemotherapy. Med. Oncol. 32, 193 (2015).
Hazkani, I. et al. Can molecular profiling enhance radiotherapy? Impact of personalized targeted gold nanoparticles on radiosensitivity and imaging of adenoid cystic carcinoma. Theranostics 7, 3962–3971 (2017).
Cortese, B., D’Amone, S. & Palamà, I. E. Wool-like hollow polymeric nanoparticles for CML chemo-combinatorial therapy. Pharmaceutics 10, 52 (2018).
James, A. R. et al. Computational and mechanistic studies on the effect of galactoxyloglucan: Imatinib nanoconjugate in imatinib resistant K562 cells. Tumor Biol. 39, 1010428317695946 (2017).
Theochari, I. et al. Drug nanocarriers for cancer chemotherapy based on microemulsions: The case of Vemurafenib analog PLX4720. Colloids Surf. B. Biointerfaces 154, 350–356 (2017).
Savla, R., Garbuzenko, O. B., Chen, S., Rodriguez-Rodriguez, L. & Minko, T. Tumor-targeted responsive nanoparticle-based systems for magnetic resonance imaging and therapy. Pharm. Res. 31, 3487–3502 (2014).
Liu, C. et al. Tumor-targeted nanoparticles deliver a vitamin D-based drug payload for the treatment of EGFR tyrosine kinase inhibitor-resistant lung cancer. Mol. Pharm. 15, 3216–3226 (2018).
He, Y., Su, Z., Xue, L., Xu, H. & Zhang, C. Co-delivery of erlotinib and doxorubicin by pH-sensitive charge conversion nanocarrier for synergistic therapy. J. Control. Rel. 229, 80–92 (2016).
Ali, A. A. A. et al. Erlotinib-conjugated iron oxide nanoparticles as a smart cancer-targeted theranostic probe for MRI. Sci. Rep. 6, 36650 (2016).
Lam, A. T. N. et al. Colloidal gold nanoparticle conjugates of gefitinib. Colloids Surf. B. Biointerfaces 123, 61–67 (2014).
Shende, P., Patil, S. & Gaud, R. S. A combinatorial approach of inclusion complexation and dendrimer synthesization for effective targeting EGFR-TK. Mater. Sci. Eng. C 76, 959–965 (2017).
Liu, Y., Zhao, Y., Luo, H., Liu, F. & Wu, Y. Construction of EGFR peptide gefitinib/quantum dots long circulating polymeric liposomes for treatment and detection of nasopharyngeal carcinoma. Biochem. Biophys. Res. Commun. 490, 141–146 (2017).
Zhou, Z. et al. Delayed sequential co-delivery of gefitinib and doxorubicin for targeted combination chemotherapy. Mol. Pharm. 14, 4551–4559 (2017).
Wang, J. et al. iRGD-decorated polymeric nanoparticles for the efficient delivery of vandetanib to hepatocellular carcinoma: preparation and in vitro and in vivo evaluation. ACS Appl. Mater. Interfaces 8, 19228–19237 (2016).
Kallus, S. et al. Nanoformulations of anticancer FGFR inhibitors with improved therapeutic index. Nanomedicine 14, 2632–2643 (2018).
Simon, T. et al. Design of FLT3 inhibitor - gold nanoparticle conjugates as potential therapeutic agents for the treatment of acute myeloid leukemia. Nanoscale Res. Lett. 10, 466 (2015).
Suarasan, S., Simon, T., Boca, S., Tomuleasa, C. & Astilean, S. Gelatin-coated gold nanoparticles as carriers of FLT3 Inhibitors for acute myeloid leukemia treatment. Chem. Biol. Drug Des. 87, 927–935 (2016).
Segura-Ibarra, V. et al. Rapamycin nanoparticles localize in diseased lung vasculature and prevent pulmonary arterial hypertension. Int. J. Pharm. 524, 257–267 (2017).
Rizwan, H., Mohanta, J., Si, S. & Pal, A. Gold nanoparticles reduce high glucose-induced oxidative-nitrosative stress regulated inflammation and apoptosis via tuberin-mTOR/NF-κB pathways in macrophages. Int. J. Nanomed. 12, 5841–5862 (2017).
Li, H., Teng, Y., Sun, J. & Liu, J. Inhibition of hemangioma growth using polymer–lipid hybrid nanoparticles for delivery of rapamycin. Biomed. Pharmacother. 95, 875–884 (2017).
Haeri, A., Osouli, M., Bayat, F., Alavi, S. & Dadashzadeh, S. Nanomedicine approaches for sirolimus delivery: a review of pharmaceutical properties and preclinical studies. Artif. Cell Nanomed. Biotechnol. 46, 1–14 (2018).
Kasper, M. et al. Novel everolimus-loaded nanocarriers for topical treatment of murine experimental autoimmune uveoretinitis (EAU). Exp. Eye Res. 168, 49–56 (2018).
Kabary, D. M. et al. Inhalable multi-compartmental phospholipid enveloped lipid core nanocomposites for localized mTOR inhibitor/herbal combined therapy of lung carcinoma. Eur. J. Pharm. Biopharm. 130, 152–164 (2018).
Lunova, M. et al. Targeting the mTOR signaling pathway utilizing nanoparticles: a critical overview. Cancers 11, 82 (2019).
Dasa, S. S. K. et al. Plectin-targeted liposomes enhance the therapeutic efficacy of a PARP inhibitor in the treatment of ovarian cancer. Theranostics 8, 2782–2798 (2018).
Novohradsky, V., Zajac, J., Vrana, O., Kasparkova, J. & Brabec, V. Simultaneous delivery of olaparib and carboplatin in PEGylated liposomes imparts this drug combination hypersensitivity and selectivity for breast tumor cells. Oncotarget 9, 28456–28473 (2018).
Du, C. et al. Epidermal growth factor receptor-targeting peptide nanoparticles simultaneously deliver gemcitabine and olaparib to treat pancreatic cancer with breast cancer 2 (BRCA2) mutation. ACS Nano 12, 10785–10796 (2018).
Duan, W. & Liu, Y. Targeted and synergistic therapy for hepatocellular carcinoma: monosaccharide modified lipid nanoparticles for the co-delivery of doxorubicin and sorafenib. Drug Des. Devel. Ther. 12, 2149–2161 (2018).
Ke, Y. & Xiang, C. Transferrin receptor-targeted HMSN for sorafenib delivery in refractory differentiated thyroid cancer therapy. Int. J. Nanomed. 13, 8339–8354 (2018).
Tang, S. & Li, Y. Sorafenib-loaded ligand-functionalized polymer-lipid hybrid nanoparticles for enhanced therapeutic effect against liver cancer. J. Nanosci. Nanotechnol. 19, 6866–6871 (2019).
Hamarat Şanlıer, Ş. et al. Development of ultrasound-triggered and magnetic-targeted nanobubble system for dual-drug delivery. J. Pharm. Sci. 108, 1272–1283 (2019).
Chakravarty, R. et al. Hollow mesoporous silica nanoparticles for tumor vasculature targeting and PET image-guided drug delivery. Nanomedicine 10, 1233–1246 (2015).
Shi, J.-F. et al. A combination of targeted sunitinib liposomes and targeted vinorelbine liposomes for treating invasive breast cancer. J. Biomed. Nanotechnol. 11, 1568–1582 (2015).
Huo, M. et al. Tumor-targeted delivery of sunitinib base enhances vaccine therapy for advanced melanoma by remodeling the tumor microenvironment. J. Control. Rel. 245, 81–94 (2017).
Saber, M. M., Bahrainian, S., Dinarvand, R. & Atyabi, F. Targeted drug delivery of Sunitinib Malate to tumor blood vessels by cRGD-chiotosan-gold nanoparticles. Int. J. Pharm. 517, 269–278 (2017).
Nazari-Vanani, R., Azarpira, N., Heli, H., Karimian, K. & Sattarahmady, N. A novel self-nanoemulsifying formulation for sunitinib: Evaluation of anticancer efficacy. Colloids Surf. B. Biointerfaces 160, 65–72 (2017).
Kim, H. et al. Combination of sunitinib and PD-L1 blockade enhances anticancer efficacy of TLR7/8 agonist-based nanovaccine. Mol. Pharm. 16, 1200–1210 (2019).
Fang, S. et al. Dimeric camptothecin derived phospholipid assembled liposomes with high drug loading for cancer therapy. Colloids Surf. B. Biointerfaces 166, 235–244 (2018).
Wu, Y. et al. Co-delivery of dual chemo-drugs with precisely controlled, high drug loading polymeric micelles for synergistic anti-cancer therapy. Biomater. Sci. 8, 949–959 (2020).
Ménard, M. et al. Mesoporous silica templated-albumin nanoparticles with high doxorubicin payload for drug delivery assessed with a 3-D tumor cell model. Biochim. Biophys. Acta Biomembr. 1863, 332–341 (2019).
Kang, S. J., Durairaj, C., Kompella, U. B., O’Brien, J. M. & Grossniklaus, H. E. Subconjunctival nanoparticle carboplatin in the treatment of murine retinoblastoma. Arch. Ophthalmol. 127, 1043–1047 (2009).
You, Y., He, L., Ma, B. & Chen, T. High-drug-loading mesoporous silica nanorods with reduced toxicity for precise cancer therapy against nasopharyngeal carcinoma. Adv. Funct. Mater. 27, 1703313 (2017).
Kather, J. N. et al. Predicting survival from colorectal cancer histology slides using deep learning: A retrospective multicenter study. PLoS Med. 16, e1002730 (2019).
Bradbury, M. et al. Clinical translation of an ultrasmall inorganic optical-PET imaging nanoparticle probe. Sci. Transl. Med. 6, 260ra149 (2014).
Acknowledgements
This work is supported, in part, by the NIH Director’s New Innovator Award (DP2-HD075698) and NIH grants R01-CA215719, R01-DK114321, R01-NS116353, R01-CA190642, UL1-TR002384, T32-CA062948 and P30-CA008748, the National Science Foundation (CAREER 1752506 and PHY 1545832), the American Cancer Society Research Scholar Grant (GC230452), the Pershing Square Sohn Cancer Research Alliance, the Breast Cancer Foundation, the Stand Up to Cancer Foundation, the Geoffrey Beene Cancer Research Center, the Expect Miracles Foundation - Financial Services Against Cancer, the Anna Fuller Fund, the Louis V. Gerstner Jr. Young Investigator’s Fund, the Frank A. Howard Scholars Program, the Honorable Tina Brozman Foundation for Ovarian Cancer Research, Cycle for Survival, the Alan and Sandra Gerry Metastasis and Tumor Ecosystems Center, Mr. William H. Goodwin and Mrs. Alice Goodwin and the Commonwealth Foundation for Cancer Research, the Center for Experimental Therapeutics, the Imaging & Radiation Sciences Program and the Center for Molecular Imaging and Nanotechnology of Memorial Sloan Kettering Cancer. Y.S. is a Jack Klein fellow. The opinions, results, findings and/or interpretations of data contained therein are the responsibility of the contractor and do not necessarily represent the opinions, interpretations or policy of the state or, if funded with federal funds, the applicable federal funding agency. Special thanks to S. D’Angelo for helpful discussions.
Author information
Authors and Affiliations
Contributions
M.T.M., Y.S. and H.K. contributed equally to the literature research for the article, creating figures, writing the manuscript and contributing to the discussion of the content. D.A.H., N.R. and M.S. discussed the content of the article and wrote perspective content relevant to their respective fields of expertise. D.A.H. conceived the article. All authors contributed to reviewing and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
D.A.H. is a co-founder and officer with equity interest in Goldilocks Therapeutics, Inc., LipidSense, Inc. and Nirova BioSense, Inc., as well as a member of the scientific advisory boards of Concarlo Holdings, LLC and Nanorobotics, Inc. M.S. is a full-time employee at AstraZeneca, has received research funds from Puma Biotechnology, AstraZeneca, Daiichi Sankyo, Immunomedics, Targimmune and Menarini Ricerche, is a co-founder of Medendi.org and is on the advisory board of Menarini Ricerche. N.R. is on the scientific advisory board (SAB) of Chugai, BeiGene, Fortress Biotech, Daiichi Sankyo, AstraZeneca, F-Prime, Zai Lab, Arvinas and Array BioPharma, and is a past SAB member of Millennium Takeda, Kadmon, Kura Oncology and Araxes. N.R. is also a consultant to Novartis Biomedical, Boehringer Ingelheim, Tarveda, Foresite Capital, Array BioPharma and Revolution Medicines, and, in recent years, has also consulted with Eli Lilly, Merrimack, Kura Oncology, Araxes and Kadmon. N.R. owns equity in BeiGene, Zai Lab, Fortress Biotech, Kura Oncology, Araxes, Kadmon and Effector. N.R. is a full-time employee at National Resilience Inc., collaborates with Plexxikon and receives research support from Chugai.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Manzari, M.T., Shamay, Y., Kiguchi, H. et al. Targeted drug delivery strategies for precision medicines. Nat Rev Mater 6, 351–370 (2021). https://doi.org/10.1038/s41578-020-00269-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41578-020-00269-6
This article is cited by
-
Dual drug-loaded cubosome nanoparticles for hepatocellular carcinoma: a design of experiment approach for optimization and in vitro evaluation
Future Journal of Pharmaceutical Sciences (2024)
-
Ultrasound-activated prodrug-loaded liposome for efficient cancer targeting therapy without chemotherapy-induced side effects
Journal of Nanobiotechnology (2024)
-
RBM45 reprograms lipid metabolism promoting hepatocellular carcinoma via Rictor and ACSL1/ACSL4
Oncogene (2024)
-
DNA mechanocapsules for programmable piconewton responsive drug delivery
Nature Communications (2024)
-
Recent advances in small-scale hydrogel-based robots for adaptive biomedical applications
Nano Research (2024)