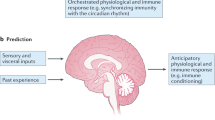
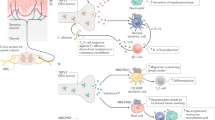
Abstract
Interactions between the immune system and the nervous system have been described mostly in the context of diseases. More recent studies have begun to reveal how certain immune cell-derived soluble effectors, the cytokines, can influence host behaviour even in the absence of infection. In this Review, we contemplate how the immune system shapes nervous system function and how it controls the manifestation of host behaviour. Interactions between these two highly complex systems are discussed here also in the context of evolution, as both may have evolved to maximize an organism’s ability to respond to environmental threats in order to survive. We describe how the immune system relays information to the nervous system and how cytokine signalling occurs in neurons. We also speculate on how the brain may be hardwired to receive and process information from the immune system. Finally, we propose a unified theory depicting a co-evolution of the immune system and host behaviour in response to the evolutionary pressure of pathogens.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Medzhitov, R. Toll-like receptors and innate immunity. Nat. Rev. Immunol. 1, 135–145 (2001).
Basbaum, A. I., Bautista, D. M., Scherrer, G. & Julius, D. Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009).
Pinho-Ribeiro, F. A., Verri, W. A. & Chiu, I. M. Nociceptor sensory neuron-immune interactions in pain and inflammation. Trends Immunol. 38, 5–19 (2017).
Baral, P. et al. Nociceptor sensory neurons suppress neutrophil and γδ T cell responses in bacterial lung infections and lethal pneumonia. Nat. Med. 24, 417–426 (2018).
Pinho-Ribeiro, F. A. et al. Blocking neuronal signaling to immune cells treats streptococcal invasive infection. Cell 173, 1083–1097.e1022 (2018).
Diaz-Salazar, C. et al. Cell-intrinsic adrenergic signaling controls the adaptive NK cell response to viral infection. J. Exp. Med. 217, e20190549 (2020).
Wieduwild, E. et al. β2-adrenergic signals downregulate the innate immune response and reduce host resistance to viral infection. J. Exp. Med. 217, e20190554 (2020).
Takenaka, M. C., Guereschi, M. G. & Basso, A. S. Neuroimmune interactions: dendritic cell modulation by the sympathetic nervous system. Semin. Immunopathol. 39, 165–176 (2017).
Cardoso, V. et al. Neuronal regulation of type 2 innate lymphoid cells via neuromedin U. Nature 549, 277–281 (2017).
Wallrapp, A. et al. The neuropeptide NMU amplifies ILC2-driven allergic lung inflammation. Nature 549, 351–356 (2017).
Klose, C. S. N. et al. The neuropeptide neuromedin U stimulates innate lymphoid cells and type 2 inflammation. Nature 549, 282–286 (2017).
Diogenes, A., Ferraz, C. C. R., Akopian, A. N., Henry, M. A. & Hargreaves, K. M. LPS sensitizes TRPV1 via activation of TLR4 in trigeminal sensory neurons. J. Dent. Res. 90, 759–764 (2011).
Liu, T., Gao, Y.-J. & Ji, R.-R. Emerging role of Toll-like receptors in the control of pain and itch. Neurosci. Bull. 28, 131–144 (2012).
Wang, T. & Ma, C. Peripheral nociceptors as immune sensors in the development of pain and itch. Adv. Exp. Med. Biol. 904, 77–85 (2016).
Oetjen, L. K. et al. Sensory neurons co-opt classical immune signaling pathways to mediate chronic itch. Cell 171, 217–228.e213 (2017). This articles provides evidence for how type 2 cytokines, IL-4 and IL-13, can activate sensory neurons to further chronic itch.
Steinberg, B. E. et al. Cytokine-specific neurograms in the sensory vagus nerve. Bioelectron. Med. 3, 7–17 (2016). This article demonstrates how the peripheral vagus nerve can be directly activated by pro-inflammatory cytokines.
Zhang, X.-C., Kainz, V., Burstein, R. & Levy, D. Tumor necrosis factor-α induces sensitization of meningeal nociceptors mediated via local COX and p38 MAP kinase actions. Pain 152, 140–149 (2011).
Kioussis, D. & Pachnis, V. Immune and nervous systems: more than just a superficial similarity? Immunity 31, 705–710 (2009).
Bergner, R. M. What is behavior? And so what? New Ideas Psychol. 29, 147–155 (2011).
Medzhitov, R., Schneider, D. S. & Soares, M. P. Disease tolerance as a defense strategy. Science 335, 936–941 (2012).
Matzinger, P. The danger model: a renewed sense of self. Science 296, 301–305 (2002).
Derecki, N. C. et al. Regulation of learning and memory by meningeal immunity: a key role for IL-4. J. Exp. Med. 207, 1067–1080 (2010). This is one of the first studies demonstrating how T cell-derived cytokines, particularly IL-4, in physiological conditions can influence learning and the acquisition of spatial memory in mice.
Filiano, A. J. et al. Unexpected role of interferon-γ in regulating neuronal connectivity and social behaviour. Nature 535, 425–429 (2016). This article implicates direct neuronal signalling of T cell-derived IFNγ in regulating an aspect of social behaviour in mice.
Alves de Lima, K. et al. Meningeal γδ T cells regulate anxiety-like behavior via IL-17a signaling in neurons. Nat. Immunol. https://doi.org/10.1038/s41590-020-0776-4 (2020). This article illustrates how meningeal γδ T cells produce IL-17A at homeostasis to signal to neurons in the prefrontal cortex and maintain basal anxiety levels in mice.
Alves de Lima, K., Rustenhoven, J. & Kipnis, J. Meningeal immunity and its function in maintenance of the central nervous system in health and disease. Annu. Rev. Immunol. 38, 597–620 (2020).
Ribeiro, M. et al. Meningeal γδ T cell-derived IL-17 controls synaptic plasticity and short-term memory. Sci. Immunol. 4, eaay5199 (2019).
Swanson, L. W. Cerebral hemisphere regulation of motivated behavior. Brain Res. 886, 113–164 (2000).
Kupari, J., Häring, M., Agirre, E., Castelo-Branco, G. & Ernfors, P. An atlas of vagal sensory neurons and their molecular specialization. Cell Rep. 27, 2508–2523.e2504 (2019).
Chiu, I. M. et al. Transcriptional profiling at whole population and single cell levels reveals somatosensory neuron molecular diversity. eLife 3, e04660 (2014).
Fleischer, J., Breer, H. & Strotmann, J. Mammalian olfactory receptors. Front. Cell Neurosci. 3, 9 (2009).
Roper, S. D. & Chaudhari, N. Taste buds: cells, signals and synapses. Nat. Rev. Neurosci. 18, 485–497 (2017).
Cisek, P. & Kalaska, J. F. Neural mechanisms for interacting with a world full of action choices. Annu. Rev. Neurosci. 33, 269–298 (2010).
Chandrashekar, J. et al. T2Rs function as bitter taste receptors. Cell 100, 703–711 (2000).
Peng, Y. et al. Sweet and bitter taste in the brain of awake behaving animals. Nature 527, 512–515 (2015).
Tinbergen, N. The Study of Instinct (Clarendon Press/Oxford University Press, 1951).
Sokolowski, K. & Corbin, J. G. Wired for behaviors: from development to function of innate limbic system circuitry. Front. Mol. Neurosci. 5, 55 (2012).
Ball, G. F. & Balthazart, J. How useful is the appetitive and consummatory distinction for our understanding of the neuroendocrine control of sexual behavior? Horm. Behav. 53, 307–311 (2008).
Inglis, I. R., Langton, S., Forkman, B. & Lazarus, J. An information primacy model of exploratory and foraging behaviour. Anim. Behav. 62, 543–557 (2001).
Li, Y. et al. Neuronal representation of social information in the medial amygdala of awake behaving mice. Cell 171, 1176–1190.e1117 (2017).
Remedios, R. et al. Social behaviour shapes hypothalamic neural ensemble representations of conspecific sex. Nature 550, 388–392 (2017).
Tierney, A. J. The evolution of learned and innate behavior: contributions from genetics and neurobiology to a theory of behavioral evolution. Anim. Learn. Behav. 14, 339–348 (1986).
Allen, W. E. et al. Global representations of goal-directed behavior in distinct cell types of mouse Neocortex. Neuron 94, 891–907.e896 (2017).
Riceberg, J. S. & Shapiro, M. L. Reward stability determines the contribution of orbitofrontal cortex to adaptive behavior. J. Neurosci. 32, 16402–16409 (2012).
Sutton, R. S. & Barto, A. G. Reinforcement learning: an introduction. IEEE Trans. Neural Netw. 9, 1054–1054 (1998).
Janak, P. H. & Tye, K. M. From circuits to behaviour in the amygdala. Nature 517, 284–292 (2015).
Chen, P. & Hong, W. Neural circuit mechanisms of social behavior. Neuron 98, 16–30 (2018).
LeDoux, J. & Daw, N. D. Surviving threats: neural circuit and computational implications of a new taxonomy of defensive behaviour. Nat. Rev. Neurosci. 19, 269–282 (2018).
Kim, J., Zhang, X., Muralidhar, S., LeBlanc, S. A. & Tonegawa, S. Basolateral to central amygdala neural circuits for appetitive behaviors. Neuron 93, 1464–1479.e1465 (2017).
Cai, H., Haubensak, W., Anthony, T. E. & Anderson, D. J. Central amygdala PKC-δ+ neurons mediate the influence of multiple anorexigenic signals. Nat. Neurosci. 17, 1240–1248 (2014).
Kohl, J. et al. Functional circuit architecture underlying parental behaviour. Nature 556, 326–331 (2018).
Beier, K. T. et al. Circuit architecture of VTA dopamine neurons revealed by systematic input-output mapping. Cell 162, 622–634 (2015).
Schwarz, L. A. et al. Viral-genetic tracing of the input-output organization of a central noradrenaline circuit. Nature 524, 88–92 (2015).
Marder, E. Neuromodulation of neuronal circuits: back to the future. Neuron 76, 1–11 (2012). This article provides a detailed overview of the principles of neuromodulation.
McCormick, D. A. & Nusbaum, M. P. Editorial overview: neuromodulation: tuning the properties of neurons, networks and behavior. Curr. Opin. Neurobiol. 29, iv–vii (2014).
Nadim, F. & Bucher, D. Neuromodulation of neurons and synapses. Curr. Opin. Neurobiol. 29, 48–56 (2014).
Katz, P. Beyond Neurotransmissionneuromodulation and Its Importance for Information Processing (Oxford University Press, 1999).
Katz, P. S. Neural mechanisms underlying the evolvability of behaviour. Philos. Trans. R. Soc. Lond. B Biol. Sci. 366, 2086–2099 (2011).
Katz, P. S. & Lillvis, J. L. Reconciling the deep homology of neuromodulation with the evolution of behavior. Curr. Opin. Neurobiol. 29, 39–47 (2014).
Bucher, D. & Marder, E. SnapShot: neuromodulation. Cell 155, 482–482.e481 (2013).
Bargmann, C. I. Beyond the connectome: how neuromodulators shape neural circuits. Bioessays 34, 458–465 (2012).
Janeway, C. A. Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb. Symp. Quant. Biol. 54, 1–13 (1989).
Matzinger, P. Tolerance, danger, and the extended family. Annu. Rev. Immunol. 12, 991–1045 (1994).
Kono, H. & Rock, K. L. How dying cells alert the immune system to danger. Nat. Rev. Immunol. 8, 279–289 (2008).
Janeway, C. A. & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 20, 197–216 (2002).
Cooper, M. D. & Alder, M. N. The evolution of adaptive immune systems. Cell 124, 815–822 (2006).
Boehm, T. & Swann, J. B. Origin and evolution of adaptive immunity. Annu. Rev. Anim. Biosci. 2, 259–283 (2014).
Flajnik, M. F. & Kasahara, M. Origin and evolution of the adaptive immune system: genetic events and selective pressures. Nat. Rev. Genet. 11, 47–59 (2010).
Zhang, J.-M. & An, J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 45, 27–37 (2007).
Akdis, M. et al. Interleukins (from IL-1 to IL-38), interferons, transforming growth factor β, and TNF-α: receptors, functions, and roles in diseases. J. Allergy Clin. Immunol. 138, 984–1010 (2016).
DeVries, M. E. et al. Defining the origins and evolution of the chemokine/chemokine receptor system. J. Immunol. 176, 401–415 (2006).
Ozaki, K. & Leonard, W. J. Cytokine and cytokine receptor pleiotropy and redundancy. J. Biol. Chem. 277, 29355–29358 (2002).
Becher, B., Spath, S. & Goverman, J. Cytokine networks in neuroinflammation. Nat. Rev. Immunol. 17, 49–59 (2017).
Kipnis, J. Multifaceted interactions between adaptive immunity and the central nervous system. Science 353, 766–771 (2016).
Kipnis, J. Immune system: the “seventh sense”. J. Exp. Med. 215, 397–398 (2018).
Dantzer, R., O’Connor, J. C., Freund, G. G., Johnson, R. W. & Kelley, K. W. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat. Rev. Neurosci. 9, 46–56 (2008).
McKim, D. B. et al. Microglial recruitment of IL-1β-producing monocytes to brain endothelium causes stress-induced anxiety. Mol. Psychiatry 23, 1421–1431 (2018).
Dantzer, R., Konsman, J. P., Bluthé, R. M. & Kelley, K. W. Neural and humoral pathways of communication from the immune system to the brain: parallel or convergent? Auton. Neurosci. 85, 60–65 (2000).
Baral, P., Udit, S. & Chiu, I. M. Pain and immunity: implications for host defence. Nat. Rev. Immunol. 19, 433–447 (2019).
Berthoud, H. R. & Neuhuber, W. L. Functional and chemical anatomy of the afferent vagal system. Auton. Neurosci. 85, 1–17 (2000).
Bonaz, B., Sinniger, V. & Pellissier, S. The vagus nerve in the neuro-immune axis: implications in the pathology of the gastrointestinal tract. Front. Immunol. 8, 1452 (2017).
von Banchet, G. S., Kiehl, M. & Schaible, H.-G. Acute and long-term effects of IL-6 on cultured dorsal root ganglion neurones from adult rat. J. Neurochem. 94, 238–248 (2005).
Jin, X. & Gereau, R. W. Acute p38-mediated modulation of tetrodotoxin-resistant sodium channels in mouse sensory neurons by tumor necrosis factor-alpha. J. Neurosci. 26, 246–255 (2006).
Shen, K.-F. et al. Interleukin-10 down-regulates voltage gated sodium channels in rat dorsal root ganglion neurons. Exp. Neurol. 247, 466–475 (2013).
Stemkowski, P. L., Noh, M.-C., Chen, Y. & Smith, P. A. Increased excitability of medium-sized dorsal root ganglion neurons by prolonged interleukin-1β exposure is K+ channel dependent and reversible. J. Physiol. 593, 3739–3755 (2015).
Chiu, I. M. et al. Bacteria activate sensory neurons that modulate pain and inflammation. Nature 501, 52–57 (2013). This article describes how bacterial products directly activate nociceptor sensory neurons to influence pain.
Da Mesquita, S., Fu, Z. & Kipnis, J. The meningeal lymphatic system: a new player in neurophysiology. Neuron 100, 375–388 (2018).
Wrona, D. Neural-immune interactions: an integrative view of the bidirectional relationship between the brain and immune systems. J. Neuroimmunol. 172, 38–58 (2006).
Blatteis, C. M. The afferent signalling of fever. J. Physiol. 526, 470 (2000).
Dantzer, R. Cytokine, sickness behavior, and depression. Immunol. Allergy Clin. North. Am. 29, 247–264 (2009).
Vichaya, E. G. et al. Microglia depletion fails to abrogate inflammation-induced sickness in mice and rats. J. Neuroinflammation 17, 172 (2020).
Blank, T. et al. Brain endothelial- and epithelial-specific interferon receptor chain 1 drives virus-induced sickness behavior and cognitive impairment. Immunity 44, 901–912 (2016). This article illustrates how type I interferons induced on viral infection can signal to brain endothelial and epithelial cells to inhibit hippocampal plasticity and impair cognition.
Van Dam, A. M., Brouns, M., Man-A-Hing, W. & Berkenbosch, F. Immunocytochemical detection of prostaglandin E2 in microvasculature and in neurons of rat brain after administration of bacterial endotoxin. Brain Res. 613, 331–336 (1993).
Blomqvist, A. & Engblom, D. Neural mechanisms of inflammation-induced fever. Neuroscientist 24, 381–399 (2018).
Ching, S. et al. Endothelial-specific knockdown of interleukin-1 (IL-1) type 1 receptor differentially alters CNS responses to IL-1 depending on its route of administration. J. Neurosci. 27, 10476–10486 (2007).
Liu, X. et al. Cell-type-specific interleukin 1 receptor 1 signaling in the brain regulates distinct neuroimmune activities. Immunity 50, 317–333.e316 (2019). This article demonstrates how endothelial-specific IL-1R1 is necessary and sufficient to promote sickness behaviour in mice, and how expression of IL-1R1 in other cells can contribute to other aspects of CNS inflammation.
Duan, L. et al. PDGFRβ cells rapidly relay inflammatory signal from the circulatory system to neurons via chemokine CCL2. Neuron 100, 183–200.e188 (2018). This article shows how blood vessel-associated mural cells function as intermediaries conveying systemic inflammation information to CNS neurons through chemokine signalling.
Aubert, A., Vega, C., Dantzer, R. & Goodall, G. Pyrogens specifically disrupt the acquisition of a task involving cognitive processing in the rat. Brain Behav. Immun. 9, 129–148 (1995).
Kent, S., Bluthé, R. M., Kelley, K. W. & Dantzer, R. Sickness behavior as a new target for drug development. Trends Pharmacol. Sci. 13, 24–28 (1992).
Konsman, J. P., Parnet, P. & Dantzer, R. Cytokine-induced sickness behaviour: mechanisms and implications. Trends Neurosci. 25, 154–159 (2002).
Hart, B. L. Biological basis of the behavior of sick animals. Neurosci. Biobehav. Rev. 12, 123–137 (1988).
Erickson, M. A. & Banks, W. A. Cytokine and chemokine responses in serum and brain after single and repeated injections of lipopolysaccharide: multiplex quantification with path analysis. Brain Behav. Immun. 25, 1637–1648 (2011).
Garlanda, C., Dinarello, C. A. & Mantovani, A. The interleukin-1 family: back to the future. Immunity 39, 1003–1018 (2013).
Krueger, J. M., Walter, J., Dinarello, C. A., Wolff, S. M. & Chedid, L. Sleep-promoting effects of endogenous pyrogen (interleukin-1). Am. J. Physiol. 246, R994–R999 (1984). This article provides one of the first examples of how IL-1β can alter sleep, which has since been associated with sickness behaviour.
Anforth, H. R. et al. Biological activity and brain actions of recombinant rat interleukin-1alpha and interleukin-1beta. Eur. Cytokine Netw. 9, 279–288 (1998).
Cibelli, M. et al. Role of interleukin-1beta in postoperative cognitive dysfunction. Ann. Neurol. 68, 360–368 (2010).
Skelly, D. T. et al. Acute transient cognitive dysfunction and acute brain injury induced by systemic inflammation occur by dissociable IL-1-dependent mechanisms. Mol. Psychiatry 24, 1533–1548 (2019).
Fang, J., Wang, Y. & Krueger, J. M. Effects of interleukin-1 beta on sleep are mediated by the type I receptor. Am. J. Physiol. 274, R655–R660 (1998).
Takahashi, S., Fang, J., Kapás, L., Wang, Y. & Krueger, J. M. Inhibition of brain interleukin-1 attenuates sleep rebound after sleep deprivation in rabbits. Am. J. Physiol. 273, R677–R682 (1997).
Opp, M. R. & Krueger, J. M. Anti-interleukin-1 beta reduces sleep and sleep rebound after sleep deprivation in rats. Am. J. Physiol. 266, R688–R695 (1994).
Cremona, S., Goujon, E., Kelley, K. W., Dantzer, R. & Parnet, P. Brain type I but not type II IL-1 receptors mediate the effects of IL-1 beta on behavior in mice. Am. J. Physiol. 274, R735–R740 (1998).
Cunningham, E. T. et al. In situ histochemical localization of type I interleukin-1 receptor messenger RNA in the central nervous system, pituitary, and adrenal gland of the mouse. J. Neurosci. 12, 1101–1114 (1992).
Deyerle, K. L., Sims, J. E., Dower, S. K. & Bothwell, M. A. Pattern of IL-1 receptor gene expression suggests role in noninflammatory processes. J. Immunol. 149, 1657–1665 (1992).
Zhang, Y. et al. An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. J. Neurosci. 34, 11929–11947 (2014).
Bluthé, R. M. et al. Role of interleukin-1beta and tumour necrosis factor-alpha in lipopolysaccharide-induced sickness behaviour: a study with interleukin-1 type I receptor-deficient mice. Eur. J. Neurosci. 12, 4447–4456 (2000).
Dantzer, R. Cytokine-induced sickness behavior: where do we stand? Brain Behav. Immun. 15, 7–24 (2001).
Fang, J., Wang, Y. & Krueger, J. M. Mice lacking the TNF 55 kDa receptor fail to sleep more after TNFalpha treatment. J. Neurosci. 17, 5949–5955 (1997).
Krueger, J. M. et al. Sleep. A physiologic role for IL-1 beta and TNF-alpha. Ann. N. Y. Acad. Sci. 856, 148–159 (1998).
Vitkovic, L., Bockaert, J. & Jacque, C. “Inflammatory” cytokines: neuromodulators in normal brain? J. Neurochem. 74, 457–471 (2000).
Lenczowski, M. J. et al. Central administration of rat IL-6 induces HPA activation and fever but not sickness behavior in rats. Am. J. Physiol. 276, R652–R658 (1999).
Bluthé, R. M., Michaud, B., Poli, V. & Dantzer, R. Role of IL-6 in cytokine-induced sickness behavior: a study with IL-6 deficient mice. Physiol. Behav. 70, 367–373 (2000).
Chai, Z., Gatti, S., Toniatti, C., Poli, V. & Bartfai, T. Interleukin (IL)-6 gene expression in the central nervous system is necessary for fever response to lipopolysaccharide or IL-1 beta: a study on IL-6-deficient mice. J. Exp. Med. 183, 311–316 (1996).
Ruzek, M. C., Miller, A. H., Opal, S. M., Pearce, B. D. & Biron, C. A. Characterization of early cytokine responses and an interleukin (IL)-6-dependent pathway of endogenous glucocorticoid induction during murine cytomegalovirus infection. J. Exp. Med. 185, 1185–1192 (1997).
Schöbitz, B., Voorhuis, D. A. & De Kloet, E. R. Localization of interleukin 6 mRNA and interleukin 6 receptor mRNA in rat brain. Neurosci. Lett. 136, 189–192 (1992).
Schöbitz, B., de Kloet, E. R., Sutanto, W. & Holsboer, F. Cellular localization of interleukin 6 mRNA and interleukin 6 receptor mRNA in rat brain. Eur. J. Neurosci. 5, 1426–1435 (1993).
Harden, L. M., du Plessis, I., Poole, S. & Laburn, H. P. Interleukin (IL)-6 and IL-1 beta act synergistically within the brain to induce sickness behavior and fever in rats. Brain Behav. Immun. 22, 838–849 (2008). This article illustrates how two inflammatory cytokines, IL-6 and IL-1β, act in synergy to promote different aspects of sickness behaviour in rodents.
Renault, P. F. et al. Psychiatric complications of long-term interferon alfa therapy. Arch. Intern. Med. 147, 1577–1580 (1987).
Capuron, L. et al. Neurobehavioral effects of interferon-alpha in cancer patients: phenomenology and paroxetine responsiveness of symptom dimensions. Neuropsychopharmacology 26, 643–652 (2002).
Capuron, L. & Miller, A. H. Cytokines and psychopathology: lessons from interferon-alpha. Biol. Psychiatry 56, 819–824 (2004).
Mostafavi, S. et al. Type I interferon signaling genes in recurrent major depression: increased expression detected by whole-blood RNA sequencing. Mol. Psychiatry 19, 1267–1274 (2014).
Miller, A. H. Norman Cousins Lecture. Mechanisms of cytokine-induced behavioral changes: psychoneuroimmunology at the translational interface. Brain Behav. Immun. 23, 149–158 (2009).
Capuron, L. et al. Association between decreased serum tryptophan concentrations and depressive symptoms in cancer patients undergoing cytokine therapy. Mol. Psychiatry 7, 468–473 (2002).
Steiner, J. et al. Severe depression is associated with increased microglial quinolinic acid in subregions of the anterior cingulate gyrus: evidence for an immune-modulated glutamatergic neurotransmission? J. Neuroinflammation 8, 94 (2011).
Capuron, L. et al. Association of exaggerated HPA axis response to the initial injection of interferon-alpha with development of depression during interferon-alpha therapy. Am. J. Psychiatry 160, 1342–1345 (2003).
Juengling, F. D. et al. Prefrontal cortical hypometabolism during low-dose interferon alpha treatment. Psychopharmacology 152, 383–389 (2000).
Capuron, L. et al. Anterior cingulate activation and error processing during interferon-alpha treatment. Biol. Psychiatry 58, 190–196 (2005).
Raison, C. L., Capuron, L. & Miller, A. H. Cytokines sing the blues: inflammation and the pathogenesis of depression. Trends Immunol. 27, 24–31 (2006).
Plata-Salamán, C. R. Cytokine-induced anorexia. Behavioral, cellular, and molecular mechanisms. Ann. N. Y. Acad. Sci. 856, 160–170 (1998).
Prieto, G. A. & Cotman, C. W. Cytokines and cytokine networks target neurons to modulate long-term potentiation. Cytokine Growth Factor Rev. 34, 27–33 (2017).
Donzis, E. J. & Tronson, N. C. Modulation of learning and memory by cytokines: signaling mechanisms and long term consequences. Neurobiol. Learn. Mem. 115, 68–77 (2014).
Yirmiya, R. & Goshen, I. Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain Behav. Immun. 25, 181–213 (2011).
Bellinger, F. P., Madamba, S. & Siggins, G. R. Interleukin 1 beta inhibits synaptic strength and long-term potentiation in the rat CA1 hippocampus. Brain Res. 628, 227–234 (1993).
Tong, L. et al. Brain-derived neurotrophic factor-dependent synaptic plasticity is suppressed by interleukin-1β via p38 mitogen-activated protein kinase. J. Neurosci. 32, 17714–17724 (2012).
Vereker, E., O’Donnell, E. & Lynch, M. A. The inhibitory effect of interleukin-1beta on long-term potentiation is coupled with increased activity of stress-activated protein kinases. J. Neurosci. 20, 6811–6819 (2000).
Farrar, W. L., Kilian, P. L., Ruff, M. R., Hill, J. M. & Pert, C. B. Visualization and characterization of interleukin 1 receptors in brain. J. Immunol. 139, 459–463 (1987).
Smith, D. E. et al. A central nervous system-restricted isoform of the interleukin-1 receptor accessory protein modulates neuronal responses to interleukin-1. Immunity 30, 817–831 (2009).
Imamura, Y. et al. Interleukin-1β causes long-term potentiation deficiency in a mouse model of septic encephalopathy. Neuroscience 187, 63–69 (2011).
Kitazawa, M. et al. Blocking IL-1 signaling rescues cognition, attenuates tau pathology, and restores neuronal β-catenin pathway function in an Alzheimer’s disease model. J. Immunol. 187, 6539–6549 (2011).
Habbas, S. et al. Neuroinflammatory tnfα impairs memory via astrocyte signaling. Cell 163, 1730–1741 (2015). This article illustrates how TNF produced during neuroinflammation can signal to astrocytes, which in turn alters synaptic function and learning.
Garré, J. M., Silva, H. M., Lafaille, J. J. & Yang, G. CX3CR1+ monocytes modulate learning and learning-dependent dendritic spine remodeling via TNF-α. Nat. Med. 23, 714–722 (2017).
Patterson, P. H. Maternal infection: window on neuroimmune interactions in fetal brain development and mental illness. Curr. Opin. Neurobiol. 12, 115–118 (2002).
Brown, A. S. et al. Serologic evidence of prenatal influenza in the etiology of schizophrenia. Arch. Gen. Psychiatry 61, 774–780 (2004).
Brown, A. S. Prenatal infection as a risk factor for schizophrenia. Schizophr. Bull. 32, 200–202 (2006).
Knuesel, I. et al. Maternal immune activation and abnormal brain development across CNS disorders. Nat. Rev. Neurol. 10, 643–660 (2014).
Choi, G. B. et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 351, 933–939 (2016). This article demonstrates how injection of IL-17A into pregnant mice or into the fetal brain can lead to anatomical brain changes and behavioural abnormalities in adult offspring.
Shin Yim, Y. et al. Reversing behavioural abnormalities in mice exposed to maternal inflammation. Nature 549, 482–487 (2017). This article shows how ablation of IL-17RA in neural cells of adult offspring exposed to maternal immune activation can rescue the associated behavioural deficits.
Lammert, C. R. et al. Cutting edge: critical roles for microbiota-mediated regulation of the immune system in a prenatal immune activation model of autism. J. Immunol. 201, 845–850 (2018).
Kim, S. et al. Maternal gut bacteria promote neurodevelopmental abnormalities in mouse offspring. Nature 549, 528–532 (2017).
Smith, S. E. P., Li, J., Garbett, K., Mirnics, K. & Patterson, P. H. Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. 27, 10695–10702 (2007). This seminal article demonstrates how injection of IL-6 into pregnant mice can result in lasting autism-like or schizophrenia-related behavioural changes in the offspring.
Swerdlow, N. R., Braff, D. L., Hartston, H., Perry, W. & Geyer, M. A. Latent inhibition in schizophrenia. Schizophr. Res. 20, 91–103 (1996).
Wynn, J. K. et al. Prepulse facilitation and prepulse inhibition in schizophrenia patients and their unaffected siblings. Biol. Psychiatry 55, 518–523 (2004).
Reed, M. D. et al. IL-17a promotes sociability in mouse models of neurodevelopmental disorders. Nature 577, 249–253 (2020).
Cervero, F. Sensory innervation of the viscera: peripheral basis of visceral pain. Physiol. Rev. 74, 95–138 (1994).
Binshtok, A. M. et al. Nociceptors are interleukin-1beta sensors. J. Neurosci. 28, 14062–14073 (2008).
Richter, F. et al. Interleukin-17 sensitizes joint nociceptors to mechanical stimuli and contributes to arthritic pain through neuronal interleukin-17 receptors in rodents. Arthritis Rheum. 64, 4125–4134 (2012).
Thakur, M. et al. Defining the nociceptor transcriptome. Front. Mol. Neurosci. 7, 87 (2014).
Hess, A. et al. Blockade of TNF-α rapidly inhibits pain responses in the central nervous system. Proc. Natl Acad. Sci. USA 108, 3731–3736 (2011).
Cevikbas, F. et al. A sensory neuron-expressed IL-31 receptor mediates T helper cell-dependent itch: Involvement of TRPV1 and TRPA1. J. Allergy Clin. Immunol. 133, 448–460 (2014).
Xu, J. et al. The cytokine TGF-β induces interleukin-31 expression from dermal dendritic cells to activate sensory neurons and stimulate wound itching. Immunity https://doi.org/10.1016/j.immuni.2020.06.023 (2020).
Wilson, S. R. et al. The epithelial cell-derived atopic dermatitis cytokine TSLP activates neurons to induce itch. Cell 155, 285–295 (2013).
Cook, A. D., Christensen, A. D., Tewari, D., McMahon, S. B. & Hamilton, J. A. Immune cytokines and their receptors in inflammatory pain. Trends Immunol. 39, 240–255 (2018).
Avital, A. et al. Impaired interleukin-1 signaling is associated with deficits in hippocampal memory processes and neural plasticity. Hippocampus 13, 826–834 (2003).
Hryniewicz, A., Bialuk, I., Kamiński, K. A. & Winnicka, M. M. Impairment of recognition memory in interleukin-6 knock-out mice. Eur. J. Pharmacol. 577, 219–220 (2007).
Naude, P. J. W. et al. Analysis of cognition, motor performance and anxiety in young and aged tumor necrosis factor alpha receptor 1 and 2 deficient mice. Behav. Brain Res. 258, 43–51 (2014).
Stellwagen, D. & Malenka, R. C. Synaptic scaling mediated by glial TNF-alpha. Nature 440, 1054–1059 (2006).
Beattie, E. C. et al. Control of synaptic strength by glial TNFalpha. Science 295, 2282–2285 (2002).
Ross, F. M., Allan, S. M., Rothwell, N. J. & Verkhratsky, A. A dual role for interleukin-1 in LTP in mouse hippocampal slices. J. Neuroimmunol. 144, 61–67 (2003).
Goshen, I. et al. A dual role for interleukin-1 in hippocampal-dependent memory processes. Psychoneuroendocrinology 32, 1106–1115 (2007).
Kipnis, J., Cohen, H., Cardon, M., Ziv, Y. & Schwartz, M. T cell deficiency leads to cognitive dysfunction: implications for therapeutic vaccination for schizophrenia and other psychiatric conditions. Proc. Natl Acad. Sci. USA 101, 8180–8185 (2004).
Brynskikh, A., Warren, T., Zhu, J. & Kipnis, J. Adaptive immunity affects learning behavior in mice. Brain Behav. Immun. 22, 861–869 (2008).
Rattazzi, L. et al. CD4+ but not CD8+ T cells revert the impaired emotional behavior of immunocompromised RAG-1-deficient mice. Transl Psychiatry 3, e280 (2013).
Quinnies, K. M., Cox, K. H. & Rissman, E. F. Immune deficiency influences juvenile social behavior and maternal behavior. Behav. Neurosci. 129, 331–338 (2015).
Cohen, H. et al. Maladaptation to mental stress mitigated by the adaptive immune system via depletion of naturally occurring regulatory CD4+CD25+ cells. J. Neurobiol. 66, 552–563 (2006).
Filiano, A. J., Gadani, S. P. & Kipnis, J. How and why do T cells and their derived cytokines affect the injured and healthy brain? Nat. Rev. Neurosci. 18, 375–384 (2017).
Brombacher, T. M. et al. IL-13-mediated regulation of learning and memory. J. Immunol. 198, 2681–2688 (2017).
Vogelaar, C. F. et al. Fast direct neuronal signaling via the IL-4 receptor as therapeutic target in neuroinflammation. Sci. Transl Med. 10, eaao2304 (2018).
Zhu, P. J. et al. Suppression of PKR promotes network excitability and enhanced cognition by interferon-γ-mediated disinhibition. Cell 147, 1384–1396 (2011).
Lebwohl, M. et al. Phase 3 studies comparing brodalumab with ustekinumab in psoriasis. N. Engl. J. Med. 373, 1318–1328 (2015).
Chiricozzi, A., Romanelli, M., Saraceno, R. & Torres, T. No meaningful association between suicidal behavior and the use of IL-17A-neutralizing or IL-17RA-blocking agents. Expert Opin. Drug Saf. 15, 1653–1659 (2016).
Kabitzke, P. A., Barr, G. A., Chan, T., Shair, H. N. & Wiedenmayer, C. P. Medial prefrontal cortex processes threatening stimuli in juvenile rats. Neuropsychopharmacology 39, 1924–1932 (2014).
Fernández-Guasti, A. & López-Rubalcava, C. Modification of the anxiolytic action of 5-HT1A compounds by GABA–benzodiazepine agents in rats. Pharmacol. Biochem. Behav. 60, 27–32 (1998).
Mesquita, A. R. et al. IL-10 modulates depressive-like behavior. J. Psychiatr. Res. 43, 89–97 (2008).
Zalcman, S. S. & Siegel, A. The neurobiology of aggression and rage: role of cytokines. Brain Behav. Immun. 20, 507–514 (2006).
Jaehne, E. J. & Baune, B. T. Effects of chemokine receptor signalling on cognition-like, emotion-like and sociability behaviours of CCR6 and CCR7 knockout mice. Behav. Brain Res. 261, 31–39 (2014).
Milenkovic, V. M., Stanton, E. H., Nothdurfter, C., Rupprecht, R. & Wetzel, C. H. The role of chemokines in the pathophysiology of major depressive disorder. Int. J. Mol. Sci. 20, 2283 (2019).
de Vries, H. E. et al. Effect of endotoxin on permeability of bovine cerebral endothelial cell layers in vitro. J. Pharmacol. Exp. Ther. 277, 1418–1423 (1996).
Tunkel, A. R., Rosser, S. W., Hansen, E. J. & Scheld, W. M. Blood-brain barrier alterations in bacterial meningitis: development of an in vitro model and observations on the effects of lipopolysaccharide. In Vitro Cell Dev. Biol. 27A, 113–120 (1991).
Korin, B. et al. High-dimensional, single-cell characterization of the brain’s immune compartment. Nat. Neurosci. 20, 1300–1309 (2017).
Mrdjen, D. et al. High-dimensional single-cell mapping of central nervous system immune cells reveals distinct myeloid subsets in health, aging, and disease. Immunity 48, 380–395.e386 (2018).
Van Hove, H. et al. A single-cell atlas of mouse brain macrophages reveals unique transcriptional identities shaped by ontogeny and tissue environment. Nat. Neurosci. 22, 1021–1035 (2019).
Louveau, A. et al. Structural and functional features of central nervous system lymphatic vessels. Nature 523, 337–341 (2015).
Norris, G. T. & Kipnis, J. Immune cells and CNS physiology: microglia and beyond. J Exp. Med. 216, 60–70 (2019).
Rustenhoven, J. et al. Functional characterization of the dural sinuses as a neuroimmune interface. Cell 184, 1000–1016 (2021).
Iliff, J. J. et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci. Transl Med. 4, 147ra111 (2012).
Yang, N. J. & Chiu, I. M. Bacterial signaling to the nervous system through toxins and metabolites. J. Mol. Biol. 429, 587–605 (2017).
Mukai, K., Tsai, M., Saito, H. & Galli, S. J. Mast cells as sources of cytokines, chemokines, and growth factors. Immunol. Rev. 282, 121–150 (2018).
Sayed, B. A., Christy, A. L., Walker, M. E. & Brown, M. A. Meningeal mast cells affect early T cell central nervous system infiltration and blood-brain barrier integrity through TNF: a role for neutrophil recruitment? J. Immunol. 184, 6891–6900 (2010).
Nagasawa, M., Spits, H. & Ros, X. R. Innate lymphoid cells (ilcs): cytokine hubs regulating immunity and tissue homeostasis. Cold Spring Harb. Perspect. Biol. 10, a030304 (2018).
Brocker, C., Thompson, D., Matsumoto, A., Nebert, D. W. & Vasiliou, V. Evolutionary divergence and functions of the human interleukin (IL) gene family. Hum. Genomics 5, 30–55 (2010).
Boulay, J.-L., O’Shea, J. J. & Paul, W. E. Molecular phylogeny within type I cytokines and their cognate receptors. Immunity 19, 159–163 (2003).
Secombes, C. J., Wang, T. & Bird, S. in The Evolution of the Immune System 87–150 (Elsevier, 2016).
Kirsten, K., Soares, S. M., Koakoski, G., Carlos Kreutz, L. & Barcellos, L. J. G. Characterization of sickness behavior in zebrafish. Brain Behav. Immun. 73, 596–602 (2018).
Kirsten, K., Fior, D., Kreutz, L. C. & Barcellos, L. J. G. First description of behavior and immune system relationship in fish. Sci. Rep. 8, 846 (2018).
Chen, C. et al. IL-17 is a neuromodulator of Caenorhabditis elegans sensory responses. Nature 542, 43–48 (2017). This article describes an orthologue of IL-17A in C. elegans and how its direct signalling to peripheral neurons can modify the response to oxygen input and subsequently aggregation behaviour.
Masuzzo, A., Montanari, M., Kurz, L. & Royet, J. How bacteria impact host nervous system and behaviors: lessons from flies and worms. Trends Neurosci. 43, 998–1010 (2020).
Arbouzova, N. I. & Zeidler, M. P. JAK/STAT signalling in Drosophila: insights into conserved regulatory and cellular functions. Development 133, 2605–2616 (2006).
Copf, T., Goguel, V., Lampin-Saint-Amaux, A., Scaplehorn, N. & Preat, T. Cytokine signaling through the JAK/STAT pathway is required for long-term memory in Drosophila. Proc. Natl Acad. Sci. USA 108, 8059–8064 (2011).
Masuzzo, A. et al. Peptidoglycan-dependent NF-κB activation in a small subset of brain octopaminergic neurons controls female oviposition. eLife 8, e50559 (2019).
Barajas-Azpeleta, R. et al. Antimicrobial peptides modulate long-term memory. PLoS Genet. 14, e1007440 (2018).
Toda, H., Williams, J. A., Gulledge, M. & Sehgal, A. A sleep-inducing gene, nemuri, links sleep and immune function in Drosophila. Science 363, 509–515 (2019).
Zeisel, A. et al. Molecular architecture of the mouse nervous system. Cell 174, 999–1014.e1022 (2018).
Saunders, A. et al. Molecular diversity and specializations among the cells of the adult mouse brain. Cell 174, 1015–1030.e1016 (2018).
Deverman, B. E. & Patterson, P. H. Cytokines and CNS development. Neuron 64, 61–78 (2009).
Altan-Bonnet, G. & Mukherjee, R. Cytokine-mediated communication: a quantitative appraisal of immune complexity. Nat. Rev. Immunol. 19, 205–217 (2019).
Dunkelberger, J. R. & Song, W.-C. Complement and its role in innate and adaptive immune responses. Cell Res. 20, 34–50 (2010).
Stephan, A. H., Barres, B. A. & Stevens, B. The complement system: an unexpected role in synaptic pruning during development and disease. Annu. Rev. Neurosci. 35, 369–389 (2012).
Schafer, D. P. et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 74, 691–705 (2012).
McDevitt, H. O. Discovering the role of the major histocompatibility complex in the immune response. Annu. Rev. Immunol. 18, 1–17 (2000).
Shatz, C. J. MHC class I: an unexpected role in neuronal plasticity. Neuron 64, 40–45 (2009).
Ek, M., Kurosawa, M., Lundeberg, T. & Ericsson, A. Activation of vagal afferents after intravenous injection of interleukin-1beta: role of endogenous prostaglandins. J. Neurosci. 18, 9471–9479 (1998).
Goehler, L. E. et al. Interleukin-1beta in immune cells of the abdominal vagus nerve: a link between the immune and nervous systems? J. Neurosci. 19, 2799–2806 (1999).
Pavlov, V. A., Chavan, S. S. & Tracey, K. J. Molecular and functional neuroscience in immunity. Annu. Rev. Immunol. 36, 783–812 (2018).
Litman, G. W., Rast, J. P. & Fugmann, S. D. The origins of vertebrate adaptive immunity. Nat. Rev. Immunol. 10, 543–553 (2010).
Thompson, C. B. New insights into V(D)J recombination and its role in the evolution of the immune system. Immunity 3, 531–539 (1995).
Acknowledgements
The authors thank S. Smith for editing the manuscript, A. Ampagliazzo for assistance with the figures and M. Lemieux for helping with the discussion of Fig. 4. This work was supported by grants from the US National Institutes of Health (MH108156, AT010416 and AG034113) to J.K.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
J.K. is a member of scientific advisory group for Puretech Health. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Immunology thanks R. Medzhitov and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Autonomic
-
Autonomic behaviours are generally characterized as reflexive or unconscious behavioural responses to stimuli.
- Proprioception
-
The perception of movement, position and force of limbs. Fast-conducting Aα fibres receive and transmit proprioceptive signals.
- Interoception
-
The sensations or afferents that arise from visceral organs such as the gastrointestinal, urinary, circulatory or respiratory systems.
- Neurotransmitters
-
Small molecules released from chemical synapses upon depolarization or in response to an increase in intracellular calcium concentrations in neurons. They are stored in small synaptic vesicles in the presynaptic terminal along with the enzymes crucial for its synthesis. They bind to receptors in the postsynaptic cell to influence gene expression and resting potential.
- Neuropeptides
-
Small proteins produced with neurotransmitters in the presynaptic neuron but that are located in different cell compartments. They are present in larger granular vesicles and are released in response to specific stimuli. Their effect is of slow onset and longer lasting compared with neurotransmitters, and they act on G-protein-coupled receptors on the postsynaptic cell.
- Dorsal root ganglia
-
A group of cell bodies or somas of sensory afferent fibres located along the spine. They carry information from the periphery to the central nervous system.
- Synaptic transmission
-
The process by which neurons communicate; it can occur in two ways. Chemical transmission occurs via the release of neurotransmitters from the presynaptic terminal on depolarization to the postsynaptic cell. Electrical transmission, on the other hand, is bidirectional and occurs through clusters of gap junctions between adjacent cells. The gap junction channels allow the passage of electrical currents and small molecules.
- Long-term potentiation
-
(LTP). The strengthening of synaptic connections between neurons that occurs after repeated high-frequency electrical stimulation.
- Hypothalamic–pituitary–adrenal (HPA) axis
-
The major anatomical structures responsible for regulating stress responses. The HPA axis results in the release of cortisol, which promotes vascular activity, reduces immune responses, restricts inflammation and maximizes resources to protect the organism from damaging effects of stress.
- Synaptic scaling
-
The process of calibrating the neuronal synapses to maintain stable or homeostatic firing rates.
- Morris water maze
-
Used to test the hippocampal-dependent learning and memory of rodents. It comprises two parts: spatial acquisition and reversal. In spatial acquisition, the animal is allowed to navigate a swimming arena with opaque water where a hidden platform is located. It must use distal, spatial cues to find the hidden platform. The animals are tested daily after being placed in random start locations. The time it takes for the animal to find the platform is then recorded across the testing days. In reversal, the hidden platform is relocated to the opposite quadrant, and the animal’s ability to find the platform is tested.
- Inhibitory neurons
-
Typically release inhibitory neurotransmitters such as GABA, which leads to the hyperpolarization of the postsynaptic cell.
- Three-chamber sociability assay
-
Tests the sociability of rodents. The test animal is placed in a three-chamber box, and, because rodents are typically social animals, they will display preference for a novel mouse compared with a novel object. The time spent in each chamber either with the novel mouse or the novel object is then recorded.
- Dural meninges
-
The outermost layer of the meninges, which is a membranous structure that lines the skull and surrounds the central nervous system. Lymphatic vessels that drain the cerebrospinal fluid are located in the dural meninges.
- Elevated plus maze
-
A behavioural task that assesses anxiety-like behaviour in rodents. It uses a plus-shaped elevated apparatus with two open arms and two closed arms. Rodents typically show a tendency for dark, enclosed spaces as they hide from predators, and they have an unconditioned fear of heights and open spaces. Increased time spent on the open arms suggests a reduction in anxiety-like behaviour.
- Open field test
-
Measures the activity and exploratory behaviour of rodents. It can also be used to test for anxiety-related behaviour. The typical behaviour of rodents is to spend more time in close proximity to the walls compared with the open area. Increased time spent in the open area indicates less anxiety-like behaviour.
- Excitatory glutamatergic neurons
-
Release glutamate, an excitatory neurotransmitter, to the postsynaptic cell, which causes its depolarization.
- Y-maze test
-
Used to assess the natural predisposition of rodents to explore novel environments. The test animal is allowed to freely move across the three arms of the maze, and the number of new arm entries is measured as the animal does not prefer to enter the arm that has been visited recently.
- Glymphatic system
-
The astrocyte-dependent system of movement of cerebrospinal fluid and interstitial fluid across the brain parenchyma to clear metabolic waste.
- Subarachnoid space
-
The space between the arachnoid mater and the pia mater, which are two of the layers of the meninges. This space is filled with cerebrospinal fluid.
Rights and permissions
About this article
Cite this article
Salvador, A.F., de Lima, K.A. & Kipnis, J. Neuromodulation by the immune system: a focus on cytokines. Nat Rev Immunol 21, 526–541 (2021). https://doi.org/10.1038/s41577-021-00508-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41577-021-00508-z
This article is cited by
-
Early life stress moderates the relation between systemic inflammation and neural activation to reward in adolescents both cross-sectionally and longitudinally
Neuropsychopharmacology (2024)
-
Neuroimmunology of Cardiovascular Disease
Current Hypertension Reports (2024)
-
Chronic social defeat alters brain vascular-associated cell gene expression patterns leading to vascular dysfunction and immune system activation
Journal of Neuroinflammation (2023)
-
Inducible co-stimulatory molecule (ICOS) alleviates paclitaxel-induced neuropathic pain via an IL-10-mediated mechanism in female mice
Journal of Neuroinflammation (2023)
-
The neuroimmune axis of Alzheimer’s disease
Genome Medicine (2023)