Abstract
HIV infection can be effectively treated by lifelong administration of combination antiretroviral therapy, but an effective vaccine will likely be required to end the HIV epidemic. Although the majority of current vaccine strategies focus on the induction of neutralizing antibodies, there is substantial evidence that cellular immunity mediated by CD8+ T cells can sustain long-term disease-free and transmission-free HIV control and may be harnessed to induce both therapeutic and preventive antiviral effects. In this Review, we discuss the increasing evidence derived from individuals who spontaneously control infection without antiretroviral therapy as well as preclinical immunization studies that provide a clear rationale for renewed efforts to develop a CD8+ T cell-based HIV vaccine in conjunction with B cell vaccine efforts. Further, we outline the remaining challenges in translating these findings into viable HIV prevention, treatment and cure strategies.
Similar content being viewed by others
Introduction
Combination antiretroviral therapy (ART) has dramatically transformed HIV infection from a death sentence to a clinically manageable chronic disease. Strict adherence to ART effectively suppresses viraemia, halts progression to acquired immune deficiency syndrome (AIDS), thwarts HIV acquisition when used as pre-exposure or post-exposure prophylaxis, and prevents transmission1. However, limitations in medication access and adherence, together with emerging drug resistance, remain major obstacles to ending the epidemic with ART alone2. Moreover, the risks of disease progression and virus transmission rapidly re-emerge in the vast majority of individuals upon cessation of ART due to rebound from viral reservoirs that persist during treatment3, necessitating lifelong ART administration. These key limitations compel refocused efforts towards understanding the determinants of HIV immune control, and the failure of such control, in order to inform the development of preventive and therapeutic vaccine strategies.
A central challenge to immune containment and vaccine development is that HIV is among the most diverse and rapidly evolving human pathogens ever encountered. For perspective, a snapshot of HIV sequence diversity within a single individual is comparable to the global diversity of influenza virus over an entire year4. Perhaps unsurprisingly in light of such variation, vaccine strategies to date have failed to provide adequate protection from HIV infection5,6,7, and the path forwards to an effective neutralizing antibody-based vaccine remains uncertain. Considering the formidable challenges associated with a preventive vaccine, it is perhaps even more difficult to imagine immune-based elimination of HIV infection, which, in addition to combating viral diversity, would also need to eradicate persistent viral reservoirs. Indeed, the only two examples of apparent HIV reservoir clearance have been mediated not by immunological or pharmacological interventions but rather by HIV-resistant haematopoietic stem cell transplantation8,9.
In contrast to these cases of transplant-induced elimination of infection, approximately 1 in 300 infected individuals is able to maintain viraemia below thresholds associated with transmission and disease progression without the need for ART10. An extensive body of evidence indicates that this durable control is mediated not by antibodies but by effective HIV-specific CD8+ T cells. Given documented cases of spontaneous viral control for decades, such untreated HIV controllers represent a viable model for an effective vaccine and durable immune-mediated HIV remission. In this Review, we synthesize evidence that supports a translatable CD8+ T cell-mediated mechanism of durable HIV control, highlighting preclinical evidence suggesting that vaccine-induced HIV-specific CD8+ T cells are able to limit both the transmission and establishment of persistent viral reservoirs. Finally, we outline the remaining hurdles for T cell-based HIV prevention and cure strategies to complement and synergize with ongoing B cell vaccine approaches.
Spontaneous control of HIV infection
Whereas most infectious agents are effectively contained or eliminated by pathogen-specific immune responses, immune failure is a hallmark of HIV pathogenesis. In the early stages of HIV infection, the window of opportunity to mount an effective immune response is limited by profound CD4+ T cell depletion in blood and tissues11, mutational escape from early HIV-specific immune responses12 and immune exhaustion13,14,15. Consequently, most immune responses ultimately fail to contain HIV infection, resulting in progression to AIDS.
By contrast, a small subset of infected individuals, known as HIV controllers, can suppress viral replication without antiretroviral medications. These unique persons have been broadly defined by sustained levels of viraemia below 2,000 RNA copies per ml of plasma, a level at which disease progression and sexual transmission are markedly reduced16,17,18, and can be subdivided into elite controllers (often defined as those with a viral load <50 RNA copies per ml) and viraemic controllers (viral load 50–2,000 RNA copies per ml). An additional category has been termed long-term non-progressors, usually defined by the ability to maintain stable CD4+ T cell counts above 500 cells per µl for 10 or more years in the absence of ART19. Despite considerable clinical, genetic and phenotypical heterogeneity among these groups, common features have emerged indicating that HIV-specific CD8+ T cells mediate durable HIV control, offering critical insight for the design and implementation of approaches to induce similar immunity on a broader scale for HIV prevention and cure.
CD8+ T cells and HIV control: clues from host genetics
One of the early links implicating CD8+ T cells in modulating HIV control came not from studies of immune function but rather from cohort studies of host HLA class I alleles20, which present viral peptides on the surface of infected cells for recognition by HIV-specific CD8+ T cells. Unlike many features assessed in comparative studies between HIV controllers and chronic progressors, genetic polymorphisms are not influenced by HIV infection. Initial studies in long-term non-progressors identified a dramatic enrichment in HLA-B*57 (ref.21), and subsequent genome-wide association studies encompassing over 6,000 infected individuals across multiple large international cohorts have confirmed that the major genetic polymorphisms modulating HIV control reside almost entirely within the HLA-B and HLA-C loci on chromosome six22,23,24,25,26. In particular, these studies identified strong associations between HIV control and polymorphic amino acids lining the HLA class I peptide-binding groove, which likely influence the viral peptides that are presented for CD8+ T cell recognition of virally infected cells22. Variation at these amino acid positions define specific class I HLA alleles that previous studies have repeatedly shown to be associated with increased likelihood of control (for example, HLA-B*57, HLA-B*27, HLA-B*52 and HLA-B*14) and others with risk of progression (for example, HLA-B*07, HLA-B*08 and HLA-B*35).
However, genetic associations offer an incomplete explanation, as no HLA class I allele is necessary or sufficient for viral control and genetic associations defined by genome-wide association studies account for less than 25% of observed variance in host control22. Thus, additional non-genetic factors must influence control in individuals without protective alleles and progression in individuals with protective alleles. The HLA associations indicate a critical role for CD8+ T cell-mediated immunity in spontaneous HIV control, an inference that is most strongly supported by CD8+ T cell depletion studies leading to loss of simian immunodeficiency virus (SIV) control in a non-human primate (NHP) model27,28,29,30. The characteristics that define effective CD8+ T cell responses in infected persons are coming into ever greater focus, as are outlined below.
CD8+ T cell function in spontaneous HIV control
Nearly all individuals infected by HIV mount high-magnitude, virus-specific CD8+ T cell responses, including the vast majority who fail to control infection31,32,33. Interferon-γ (IFNγ) secretion by CD8+ T cells exposed to HIV antigens, a widely used metric for identifying antigen-specific responses, does not correlate with HIV control33,34,35. Additionally, the limitations of IFNγ assays are underscored by a study showing that HIV-specific CD8+ T cells that secrete IFNγ are rarely the same cells that kill infected target cells36. The ability of antigen-stimulated cells to secrete combinations of cytokines and effector molecules, termed polyfunctionality, is greater in controllers than progressors37,38,39. However, whether this enhanced functionality is a cause or consequence of lower viral load has been difficult to discern. Importantly, polyfunctionality is lowest in those with the lowest viral loads by ultrasensitive assays, the smallest viral reservoirs and the least culturable virus40,41.
In contrast to assays measuring IFNγ secretion or polyfunctionality, assays measuring in vitro expansion of HIV-specific CD8+ T cells following stimulation with HIV antigens revealed properties of these cells that consistently distinguished controllers from progressors42. HIV-specific CD8+ T cells from controllers, compared with those from progressors, have greater capacity to proliferate and develop cytolytic potential upon in vitro antigenic stimulation42,43; this is also the case for HIV-specific CD8+ T cells from elite controllers with undetectable responses following ex vivo antigen stimulation, as described above44. By contrast, CD8+ T cells from progressors often exhibit strong ex vivo activation but fail to proliferate or acquire cytolytic capacity due to exhaustion and necroptotic cell death42,45, and these deficiencies are not restored despite prolonged ART46,47. Moreover, the ability of in vitro-expanded CD8+ T cell populations to suppress HIV replication38,48 also distinguishes elite controllers from progressors, further implicating the role of CD8+ T cell function in HIV control.
Given that the functional ability of CD8+ T cells to proliferate differentiates controllers from progressors, the lack of detectable IFNγ responses or CD8+ T cell polyfunctionality by direct ex vivo assays in some elite controllers likely reflects differential in vivo antigen exposure rather than being evidence for alternative CD8+ T cell-independent mechanisms of control. Thus, combined analysis of both ex vivo and expanded CD8+ T cell responses in the context of antigen exposure provides a more accurate perspective on which qualities of CD8+ T cells are associated with spontaneous viral control (Fig. 1). In comparison with progressors, CD8+ T cell responses in HIV controllers show an overall increased ability to maintain long-term memory and effector potential, with the ability to kill infected cells before progeny virions are produced49,50,51, providing a clear rationale for exploiting these features for treatment, prevention and cure.
The graph illustrates correlations between in vivo antigen exposure and HIV reservoir size (x-axis), increasing levels of mutational escape and T cell exhaustion in elite controllers (EC), viraemic controllers (VC) and chronic progressors (CP), and HIV-specific CD8+ T cell function following ex vivo antigenic stimulation (y-axis) by different measurements, including interferon-γ (IFNγ) production in ELISPOT assays (solid blue line), immediate cytolytic capacity (dashed blue line) and proliferative capacity and cytolytic capacity of expanded T cells (red line).
CD8+ T cell specificity in spontaneous HIV control
In addition to the importance of functional features of HIV-specific CD8+ T cells in viral control, evidence also points to the importance of the specificity of these responses. Antigenic specificity of CD8+ T cell responses is influenced by the unique binding properties of expressed HLA class I alleles, each of which presents a restricted set of HIV-derived peptides 8–11 amino acids in length52. Polymorphic positions within the HLA peptide-binding groove determine differential peptide binding constraints among HLA alleles, including anchor residue preference and orientation of peptide presentation to T cell receptors (TCRs). Despite the strong association between certain HLA alleles and disease outcome, specific alleles are neither necessary nor sufficient for durable HIV control. The majority of infected individuals with protective HLA alleles are progressors and a significant proportion of HIV controllers lack protective HLA alleles22.
Comprehensive assessment of CD8+ T cell specificity in viral control is complicated by HIV diversity, as assays are typically confined to recognition of a single reference strain of the virus and longitudinal samples spanning from acute to chronic phases of infection are limited. The magnitude and breadth of CD8+ T cell responses measured by IFNγ ELISPOT targeting the relatively conserved HIV structural protein Gag but not the highly variable surface protein Env are associated with a lower viral load and preserved CD4+ T cell counts irrespective of HLA allele and epitope sequence conservation53,54,55. Moreover, comparative analysis of sequence conservation of targeted epitopes does not distinguish HLA-B*57/HLA-B*27-negative controllers and progressors56.
Although sequence conservation is a reasonable proxy for mutational constraint, immune targeting of conserved regions may be insufficient as only a subset of conserved regions exhibits fitness defects when mutated57. Computational modelling of HIV sequence diversity has revealed that Gag regions targeted by controller responses have a lower mutational tolerance than those targeted by progressors, implying a structural basis for the observed mutational constraints58,59,60. The importance of CD8+ T cell epitope specificity is further supported by application of network theory to HIV protein structure. Network analysis of crystallographic HIV protein structures using a novel algorithm to quantify the contribution of non-covalent amino acid side chain interactions to overall protein structure revealed that proliferative CD8+ T cell responses in HIV controllers, irrespective of expressed HLA alleles, preferentially target HIV epitopes containing amino acids derived from highly interconnected (‘networked’) regions of viral protein structure61. Mutation of these highly networked amino acids was also highly disruptive to viral fitness, making them ideal targets for immune pressure. Consistent with this, robust CD8+ T cell targeting of highly networked epitopes in controllers was associated with fewer mutations, especially at HLA anchor and TCR contact sites61. Perhaps most importantly, elite controllers lacking protective HLA alleles were also found to target epitopes containing highly networked amino acids that are not commonly targeted, suggesting that what has been considered a genetic barrier to control can be overcome.
TCR usage may also influence differences in CD8+ T cell-mediated HIV control. Viral control has been associated with more cross-reactive public TCR clonotypes62,63,64. HLA alleles associated with HIV control impart thymic selection for TCR repertoires with increased HIV recognition and variant cross-reactivity65. Although differential TCR usage alone is insufficient to explain spontaneous control or confer functional differences in CD8+ T cells66,67,68, cross-reactive TCRs have been shown to limit viral escape pathways in controllers, consistent with the findings that mutations within epitopes targeted predominantly by controllers are poorly tolerated due to impaired viral replication69,70 and that their recognition is maintained in controllers by cross-reactive TCRs and de novo responses71,72,73.
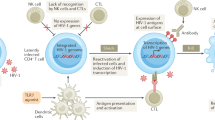
Taken together, the combination of functional CD8+ T cell responses and targeting of mutationally constrained epitopes offers a compelling mechanism to explain the observed genetic association between HIV control and expression of certain class I HLA alleles, and how individuals without protective HLA alleles can mediate effective immune control. The data indicate that specificity and function of CD8+ T cells are necessary for durable control, yet neither alone is sufficient, as mutational escape from functional responses and dysfunctional responses against non-mutated epitopes represent parallel pathways of immune evasion (Fig. 2). Induction of functional CD8+ T cell responses against highly networked epitopes restricted by common HLA alleles offers an opportunity for broad translation of these findings towards preventive and curative interventions74.
Diagram representing CD8+ T cell interactions with infected cells in HIV controllers (top) and progressors (bottom). HIV controller CD8+ T cells recognize structurally constrained HIV peptides presented via MHC class I (pMHC-I) on infected cells via T cell receptors (TCRs), inducing proliferation and perforin/granzyme-mediated cytolysis of infected cells. Mutation of HIV epitopes presented via MHC-I in HIV progressors promotes escape from recognition by functional CD8+ T cells, and dysfunctional CD8+ T cells recognize non-escaped viral epitopes through TCR–pMHC-I interactions and secrete interferon-γ (IFNγ) but fail to proliferate or kill infected cells due to inhibitory receptor–ligand interactions, such as PD1 and PDL1. Infected cells that evade killing via escape and/or exhaustion spread infection and promote further immune dysregulation in progressors.
CD8+ T cell localization in spontaneous HIV control
In order to control chronic infection, HIV-specific CD8+ T cells must be able to traffic to and function at anatomical sites where HIV persists, especially mucosal and gastrointestinal lymphoid tissues where infected CD4+ T cells are present in high concentrations75. Consistent with this, increased frequencies of functional CD8+ T cells have been observed in the rectal mucosa of HIV controllers76. HIV-infected follicular helper CD4+ T cells, which reside within B cell follicles in lymphoid tissue and direct humoral immune responses, constitute a significant fraction of the persistent viral reservoir during chronic infection77,78. Importantly, CD8+ T cells are largely excluded from B cell follicles, representing a significant obstacle to CD8+ T cell-mediated clearance of infected follicular helper T cell reservoirs79. Recent evidence demonstrates that a population of follicular CD8+ T cells that express the follicle-homing receptor CXCR5 can traffic to or reside in lymphoid tissues under certain circumstances80,81,82. Viral control is associated with a higher proportion and functionality of follicular CD8+ T cells83,84,85,86, but viral replication within follicles can persist even during elite control of HIV87,88, underscoring the need to develop strategies that promote the trafficking and function of HIV-specific follicular CD8+ T cells. Tissue-resident memory T (TRM) cells represent an additional subset of CD8+ T cells that control viral infection in various tissue sites89. Relative to peripheral blood CD8+ T cells, follicular CD8+ T cells and TRM cells in lymphoid tissues have a lower expression of perforin and granzymes, potentially due to tissue-specific microenvironmental regulation of T cell function as a means to limit tissue inflammation and damage90,91. However, their precise functional profiles and roles in HIV control remain unclear. Suppression of viral replication by lymphoid CD8+ T cells from elite controllers has been observed in the absence of detectable cytolytic activity, suggesting that non-cytolytic CD8+ T cell functions may also be important for ongoing control of HIV in lymphoid tissues92. Although, as with peripheral blood responses, ex vivo function of HIV-specific follicular CD8+ T cells and TRM cells in lymphoid tissue must be carefully interpreted in the context of recent local antigen exposure, epitope specificity and viral escape from recognition. Additional studies aimed at understanding the precise role and mechanisms of action of HIV-specific follicular CD8+ T cells and TRM cells in spontaneous control will be crucial to the development of strategies that harness these cells for the cure and prevention of HIV infection.
Additional host and viral contributions to CD8+ T cell-mediated HIV control
Factors that modulate the magnitude and duration of initial viraemia influence the ability of CD8+ T cells to control infection. Consistent with this, a role for natural killer cells in modulating HIV control through modest reductions in viral load has been well established by a large body of literature, including robust genetic associations93,94,95,96,97. Viral factors have also been implicated in modulating HIV control; however, it is difficult to distinguish causal from consequential associations due to CD8+ T cell-associated mutations that impair viral replication capacity98. Although it is clear that fully replication-competent viruses can be isolated from controllers99, control has also been associated with viruses harbouring mutations that impair viral replication fitness100,101,102. Studies of HIV transmission events between controllers and progressors demonstrate that viral properties are neither necessary nor sufficient to mediate spontaneous HIV control103,104. Nonetheless, many transmitted viruses are pre-adapted to immune responses in their prior hosts, which may impair fitness and/or blunt the ability of CD8+ T cells to achieve spontaneous control in recipients expressing the same HLA allele105,106.
In contrast to cellular immunity, there is little evidence to support a role for humoral immunity in maintaining spontaneous HIV control. Antibody neutralization breadth, which requires affinity maturation against continually evolving Env antigenic variants, is associated with the duration of uncontrolled chronic viraemia and is negatively correlated with spontaneous HIV control107,108,109,110. Moreover, broadly neutralizing monoclonal antibodies from HIV controllers rarely target autologous virus111. Studies of non-neutralizing antibody function in HIV controllers have not reported strong correlations with HIV control112,113. Although HIV-specific CD4+ T cells are required for effective adaptive immunity in HIV controllers and are affected by chronic infection, depletion of either CD4+ T cells or B cells in a NHP model of SIV control had no impact on viraemia114,115, suggesting that neither cell type is directly involved in durable spontaneous HIV control.
Spontaneous HIV control as a model for functional cure and vaccine development
The demonstrated antiviral efficacy of CD8+ T cells in mediating long-term spontaneous control of infection in many individuals offers a clear rationale for harnessing cellular immunity to combat HIV; however, there remains a lack of consensus as to whether this represents a viable model for the development of preventive and therapeutic HIV vaccines. On the one hand, a small fraction (1.2% per year) of HIV controllers ultimately lose control, experiencing rebound viraemia and CD4+ T cell depletion10. Furthermore, some controllers experience significant immune activation in the absence of detectable viraemia due to persistent viral replication in tissue sites and expression of antigen from defective proviruses88,116,117,118, which can be reduced by ART119. On the other hand, a subset of elite controllers is able to maintain a stable state of undetectable viraemia with no appreciable immune activation. Indeed, many elite controllers have extremely small replication-competent proviral reservoirs with low sequence diversity, suggesting functional reservoir suppression120,121,122,123. In some elite controllers, viral reservoirs are suppressed so profoundly that replication-competent virus cannot be detected or recovered102,124. Such cases of exceptional control, albeit rare, represent a natural model for immune-mediated functional cure approaches with the goal of durable remission, providing optimism that immune-mediated eradication of infection may be possible. Further research is warranted to determine whether individuals with the best reservoir containment represent an extreme on a continuum of spontaneous HIV control mediated by a common mechanism or whether additional mechanistic features distinguish this subset from other HIV controllers.
In addition to spontaneous HIV control, some individuals also control viraemia after ART interruption125,126. These persons, termed post-treatment controllers (PTCs), have smaller HIV reservoirs and are often treated early after acute infection125,126,127,128,129,130. Interestingly, although protective HLA alleles have been associated with delayed viral rebound following ART interruption131, such alleles were not enriched in PTCs125, raising the possibility of distinct mechanisms of viral control in these individuals, which warrant further investigation. The translation of findings from both spontaneous controllers and PTCs to the broader population for the development of effective HIV immunization and cure strategies remains an important goal.
Prospects for T cell-based HIV vaccines
Vaccines seeking to elicit sterilizing humoral immunity have a strong historical precedent. However, the induction of broadly neutralizing antibodies (bNAbs) against HIV has thus far proved challenging and will likely require serial immunization with a succession of variant Env immunogens to mobilize germline precursors and guide subsequent somatic hypermutation to achieve neutralization breadth132,133,134. Moreover, humoral immunity alone may be insufficient for protection against the transmission of cell-associated HIV135,136,137. By contrast, T cell-based vaccines enable targeting of viral antigens less permissive to viral escape than Env, do not require additional somatic hypermutation and may be able to augment humoral vaccine efforts. Although cellular immunity-based vaccines lack historical precedent from other infectious diseases and cells must become infected before CD8+ T cells can recognize and induce cytolysis, in vitro studies clearly show that HIV-specific CD8+ T cells can kill both activated and resting CD4+ T cells before progeny virus is produced49,50,51,138. This suggests the possibility that these responses may not only serve to control established infection but may also, in sufficient numbers, be able to clear the first infected cells rapidly before persistent reservoirs are established.
Human trials of T cell-based HIV vaccines to date have been disappointing. However, accumulating preclinical evidence demonstrates that emerging approaches for vaccine-induced CD8+ T cell-mediated immunity can provide sterilizing protection from retroviral infection and establish durable remission upon therapeutic vaccination (Fig. 3). Moreover, clear evidence in some persons of the ability of HLA class I-restricted HIV-specific CD8+ T cells to provide durable control following infection represents an important means to potentially enhance the impact of vaccine-induced humoral immunity that is not fully protective. Although efforts to induce protective T cell responses in clinical trials have not been successful, they have revealed the need for the rational design and selection of immunogens and vectors for improved next-generation T cell-based immunogens (Box 1).
Diagram representing relative HIV viral load (VL, green), relative total CD4+ T cell count (CD4, violet), and relative natural (nCD8) or vaccine-induced (vCD8) HIV-specific CD8+ T cell responses over time. In natural HIV infection (top panel), incomplete natural CD8+ T cell-mediated control of VL eventually leads to disease progression, marked by CD4+ T cell decline and uncontrolled VLs. Therapeutic vaccines that elicit effective vaccine-induced CD8+ T cell-mediated control of viraemia aim to protect against disease progression, lower VLs to below the transmission threshold and contain and reduce the residual HIV reservoir in the absence of antiretroviral therapy. Preventive CD8+ T cell-mediated vaccines aim to induce a rapid memory-recall, vaccine-induced CD8+ T cell response and clear the first infected cells to prevent reservoir establishment as well as to control and ideally clear residual infected cells.
CD8+ T cells in prophylactic immunization: moving beyond STEP and HVTN505
Much of the scepticism of CD8+ T cell-based vaccines for HIV prevention emerged from the disappointing results of the STEP and HVTN505 trials6,139. In STEP, a recombinant adenovirus serotype 5 (Ad5) vector encoding HIV Gag, Pol and Nef induced HIV-specific CD8+ T cells and showed evidence of T cell selection pressure, although responses were of low magnitude, targeted few epitopes, and did not improve the prevention of HIV acquisition or control of subsequent HIV infection6. Moreover, there was an increased risk of HIV acquisition in males lacking circumcision and with pre-existing anti-Ad5 antibodies. In HVTN505, these at-risk groups were excluded and a DNA prime/Ad5 boost was used to elicit antiviral T cell responses but, again, no protection from HIV infection was afforded139. Consequently, major concerns about the viability of CD8+ T cell-based vaccines for HIV prevention have persisted.
Upon further examination of both trials, an important consideration may be the use of full-length HIV antigens. These immunogens largely give rise to CD8+ T cell responses specific for immunodominant viral epitopes that are similar to those targeted in natural infection, where the majority of individuals fail to control infection due to the emergence of viral variants that evade immune recognition. Consistent with this, viral sequence analysis from STEP trial participants revealed a ‘sieving’ effect, whereby vaccine-induced CD8+ T cell responses led to an enrichment of viral species with mutations at common HLA class I restricted T cell epitope sites140. This also occurred in HVTN505, where CD8+ T cell responses were primarily directed against Env141, which is among the most variable viral proteins and whose targeting by CD8+ T cells is associated with higher viral loads53.
To induce immune responses that resemble those found in spontaneous HIV controllers, next-generation prophylactic T cell vaccines may benefit from the use of immunogens limited to mutationally intolerant regions within the HIV proteome. Preclinical studies of vaccine-induced CD8+ T cells targeting three epitopes presented by the protective Mamu-B*08 allele were able to suppress viral loads to less than 1,000 RNA copies per ml in six of eight vaccinated Mamu-B*08+ rhesus macaques compared with only one of eight in a Mamu-B*08+ control arm142, suggesting that narrowly targeted, vaccine-induced, virus-specific CD8+ T cell responses against specific epitopes can effectively control replication following SIV challenge to below the transmission threshold. Thus, new immunogens composed specifically of sequence-conserved regions of the viral proteome143,144, regions with a relative association with viral control in large patient cohorts (HIVACAT T cell immunogen)145 or structurally constrained ‘networked’ epitopes that are mutationally intolerant61 may portend a higher probability of success in future studies. Moreover, directing T cell responses to a limited number of epitopes may reduce antigenic interference of vaccine components146 and limit the induction of strong T helper 1-biased CD4+ T cell responses, which have been shown to diminish vaccine efficacy in NHPs147.
While immune focusing is likely to be crucial for prophylactic vaccine development, additional efforts have been dedicated to improving the breadth of T cell-based antigens in order to address possible vaccine–virus sequence mismatch between immunogen and circulating virus species. One promising strategy involves using a ‘mosaic’ of viral sequences, which are comprised of naturally occurring epitope sequences and common variants148,149. Correlates of protection in NHPs include Env-specific antibody responses measured by ELISA and T cell responses measured by IFNγ149. These antigens have been tested in human subjects and have demonstrated safety and tolerability as part of the APPROACH study150, with clinical efficacy assessment already underway. As part of a complementary strategy, immunogen designs have also aimed to induce broad cellular immune responses solely against sequence-conserved regions of the virus (tHIVConsvX)151, which integrates the concepts of protective specificity and breadth into a single immunogen and may afford even greater coverage of incoming viruses.
Beyond immunogen design, developments in vaccine delivery vectors have also yielded promising new directions for prophylactic T cell-based HIV vaccines. The reliance of many existing vaccine modalities on the induction of central memory CD8+ T cell responses has raised concern over whether immune recall will be sufficiently rapid to prevent the early phases of viral dissemination. Circumventing this issue are novel rhesus cytomegalovirus (RhCMV) vectors that are able to elicit persistently active effector memory CD8+ T cell responses in rhesus macaques due to the constitutive expression of SIV Gag antigen and CMV-induced memory inflation152,153. Remarkably, in numerous studies, these vectors have been able to consistently protect ~50% of animals from viral challenge154,155,156,157. Moreover, detailed evaluation at necropsy revealed that vaccinated animals lacked any detectable virus, indicating that continuous effector memory T cell responses were able to clear tissue-associated viral reservoirs and thus achieve sterilizing immunity155. This protection was also afforded by attenuated RhCMV, even when challenged 3 years post-vaccination156.
Follow-up studies of these animals have revealed that CD8+ T cell responses elicited by RhCMV vectors are exceptionally broad and restricted non-classically by HLA class II and the simian HLA-E orthologue Mamu-E157. Due to the limited polymorphism of HLA-E in humans (with only two known alleles)158, inducing such responses may be particularly advantageous for a prophylactic T cell-based HIV vaccine to broadly achieve both global coverage and sterilizing immunity. In conjunction with vector modification efforts to achieve these same kinds of responses in humans, additional studies to determine the immune parameters that differentiate protected from non-protected animals may also elucidate further improvements to this innovative vector system in order to achieve protection from infection in a larger percentage of those vaccinated. Nonetheless, an emerging spectrum of immunogens, vectors and delivery routes to improve the specificity, function and location of vaccine-induced CD8+ T cell responses highlights the promise of prophylactic T cell-based HIV vaccine approaches currently in development (Table 1), which are being actively pursued alongside humoral vaccines.
CD8+ T cells in therapeutic immunization: translating HIV control to the population at-large
In addition to their utility for HIV prevention, CD8+ T cells that can durably suppress viral replication as demonstrated by spontaneous HIV controllers represent a promising therapeutic modality towards the development of a functional HIV cure or remission. Reversal of immune exhaustion alone is unlikely to have durable clinical benefit as many of the epitopes targeted in chronic infection have mutated to escape recognition159. Indeed, although additional studies are warranted to test immunotherapies and other strategies being developed for immune oncology160, preclinical trials of immune checkpoint blockade during ART suppression did not significantly delay or reduce rebound viraemia upon treatment interruption161,162,163. Likewise, attempts to mobilize the latent reservoir with pharmacological interventions, such as histone deacetylase inhibitors, will require CD8+ T cell elimination of these cells, where again issues of pre-existing immune escape are prominent159. Together, these considerations suggest that vaccine-mediated induction of new HIV-specific CD8+ T cell responses to mutationally constrained epitopes will be required.
Early therapeutic immunization studies using antigen-pulsed dendritic cells have shown modest augmentation of immune responses and viral load reduction164,165,166,167,168, whereas other therapeutic vaccination strategies aimed at eliciting CD8+ T cell responses have yielded promising preclinical results. Delivery of rAd26 and modified vaccinia Ankara expressing full-length SIV Gag–Pol–Env along with Toll-like receptor 7 agonist GS-986 to ART-suppressed, SIV-infected rhesus macaques led to the induction of broad de novo cellular immune responses, which were strongly correlated with a reduction in median set point viral load to 1,000 SIV RNA copies per ml and a delay in viral rebound following ART cessation169. The impressive cellular immune breadth achieved by animals with more effective viral suppression is consistent with observations in HIV-infected humans, where broad and functional cellular immune responses to non-escaped CD8+ T cell epitopes have been implicated in a reduction of the latent HIV-1 reservoir159.
Similar to T cell-based preventive vaccines, such insights emphasize the need for therapeutic immunogens that are able to direct cellular immune responses to specific invariant sites within the HIV proteome. Initial efforts to accomplish this in HIV-infected individuals have primarily focused on sequence-conserved viral regions, with one of these immunogens (tHIVConsv) having been administered in a therapeutic vaccine setting to evaluate its safety and immunogenicity (BCN01)170. Vaccination with a chimpanzee adenovirus vector (ChAdOx) prime and a modified vaccinia Ankara boost of tHIVConsv led to a clear shift in CD8+ T cell immunodominance patterns towards conserved segments170. These data provide optimism that augmentation of cellular antiviral immunity and redirection towards protective epitopes is possible via therapeutic vaccination with focused HIV immunogens.
Because sequence conservation of targeted epitopes does not clearly differentiate HIV controllers from progressors56, alternative approaches to therapeutic immunogen design are warranted and being pursued. Higher order HIV sequence analysis of couplings between viral mutations within the conserved Gag protein using random matrix theory58 and quantitative fitness landscapes59 has revealed multidimensional constraints on viral evolution that predict regions of vulnerability. These mutationally intolerant amino acids are likely related to structural interdependencies that are important for viral fitness, and thus represent promising targets for CD8+ T cell pressure. Recent identification of structurally constrained epitopes restricted by common HLA alleles and strongly associated with spontaneous HIV control provides an additional avenue for re-directing CD8+ T cell responses, which may further increase the probability of inducing protective immunity61.
Challenges facing prophylactic and therapeutic vaccine development
Given the advantages and disadvantages of each approach, cellular and humoral HIV vaccine methods will likely be complementary in providing full protection from HIV infection (Box 1). However, eliciting each response will require different immunogens and optimization of distinct parameters, warranting separate but parallel development efforts171. In addition, the selection of vaccine immunogens, vectors and delivery routes will have important impacts on CD8+ T cell function, specificity and localization (Table 1). Optimal immunogen design may require an improved understanding of the effects of immunodominance and original antigenic sin on immunogenicity and protective efficacy. The selection of vectors and adjuvants to elicit the appropriate targeting of functional responses in the appropriate tissues against protective antigenic epitopes will require significant development and empirical testing, which should be derived by iterative, small-scale human immunogenicity studies before moving forwards to larger efficacy trials. The induction of CD4+ T cell help to assist the development of effective CD8+ T cell immunity will also be required, but activation of these CD4+ T cells will need to be carefully regulated given that this may cause increased susceptibility to HIV infection, diminished vaccine efficacy and more rapid disease progression147,172,173. The widespread availability and use of pre-exposure prophylactic ART poses unique challenges to the design of and recruitment for prophylactic vaccine efficacy trials, and the anticipated introduction of long-acting injectable ART may further complicate future studies174.
The development of therapeutic vaccines involves many unique challenges. Efficacy trials require monitored interruption of ART, including for individuals not receiving vaccination in controlled studies, which has raised important medical and ethical concerns175. Recent evidence that brief treatment interruption has no significant long-term impact on immune activation, drug resistance, reservoir size or composition indicates that such studies are safe176,177,178. Another challenge is that preventive vaccines may be ineffective in a therapeutic setting. For example, therapeutic immunization with the same RhCMV vector that afforded substantial protection and early clearance of SIV infection in a prophylactic setting was ineffective in reducing the SIV reservoir after therapeutic immunization despite early initiation of ART179. Pre-existing immune escape and dysfunctional immune responses in chronically infected individuals represent added challenges for therapeutic immunization approaches as immunization may preferentially expand these responses. Nonetheless, much of the preliminary clinical testing for cellular immune HIV vaccines will likely occur in a therapeutic setting owing to practical considerations in evaluating efficacy and managing risk, and where shifts in CD8+ T cell immunodominance to specific regions of the viral proteome have been previously demonstrated165,170. The development of optimal adjuvants, immunomodulatory agents and latency-reversing agents for coadministration with immunogens will also be important for the success of therapeutic HIV vaccines180. Intriguingly, the infusion of two potent bNAbs (3BNC117 and 10-1074) early during SHIV infection led to sustainable viral suppression that was lost upon subsequent delivery of a CD8β-depleting antibody, suggesting a possible vaccinal effect of these bNAbs that can improve the induction of antiviral CD8+ T cells181. Although NHP models have provided clear benefits for preclinical vaccine testing182, differences in viral diversity, duration of therapy, anti-vector immunity and pathogenesis may contribute towards the limited translation of efficacy from such models to humans.
Conclusions
Several remaining hurdles will need to be overcome to successfully harness T cells for prevention, treatment and cure. However, the above data provide ample support for renewed efforts to develop CD8+ T cell-based HIV vaccines in conjunction with ongoing B cell vaccine efforts. Persons with durable spontaneous control of HIV demonstrate that CD8+ T cells hold substantial promise to combat the HIV epidemic. Mechanistic studies in spontaneous HIV controllers highlight the function, specificity and localization of CD8+ T cells as important determinants of protection, and the ability to uncouple protection from specific HLA alleles represents a key step towards broad clinical translation. Preclinical and clinical vaccine trials have provided additional insights towards the development of improved T cell-based prevention and cure modalities. Further rational design of immunogens and careful selection of appropriate vectors to elicit functional effector-memory CD8+ T cell responses against protective epitopes in vulnerable viral regions represents a promising path forwards for preventive and therapeutic T cell-based HIV vaccines.
References
Eisinger, R. W., Dieffenbach, C. W. & Fauci, A. S. HIV viral load and transmissibility of HIV infection: undetectable equals untransmittable. JAMA 321, 451–452 (2019).
Katz, I. T., Ehrenkranz, P. & El-Sadr, W. The global HIV epidemic: what will it take to get to the finish line? JAMA 319, 1094–1095 (2018).
Siliciano, J. D. & Siliciano, R. F. Recent developments in the effort to cure HIV infection: going beyond N = 1. J. Clin. Invest. 126, 409–414 (2016).
Korber, B. et al. Evolutionary and immunological implications of contemporary HIV-1 variation. Br. Med. Bull. 58, 19–42 (2001).
Pitisuttithum, P. et al. Randomized, double-blind, placebo-controlled efficacy trial of a bivalent recombinant glycoprotein 120 HIV-1 vaccine among injection drug users in Bangkok, Thailand. J. Infect. Dis. 194, 1661–1671 (2006).
Buchbinder, S. P. et al. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the step study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet 372, 1881–1893 (2008).
Rerks-Ngarm, S. et al. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N. Engl. J. Med. 361, 2209–2220 (2009).
Hutter, G. et al. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. N. Engl. J. Med. 360, 692–698 (2009).
Gupta, R. K. et al. HIV-1 remission following CCR5Δ32/Δ32 haematopoietic stem-cell transplantation. Nature 568, 244–248 (2019).
Yang, O. O., Cumberland, W. G., Escobar, R., Liao, D. & Chew, K. W. Demographics and natural history of HIV-1-infected spontaneous controllers of viremia. AIDS 31, 1091–1098 (2017).
Mehandru, S. et al. Primary HIV-1 infection is associated with preferential depletion of CD4+ T lymphocytes from effector sites in the gastrointestinal tract. J. Exp. Med. 200, 761–770 (2004).
Salazar-Gonzalez, J. F. et al. Genetic identity, biological phenotype, and evolutionary pathways of transmitted/founder viruses in acute and early HIV-1 infection. J. Exp. Med. 206, 1273–1289 (2009).
Day, C. L. et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 443, 350–354 (2006).
Petrovas, C. et al. PD-1 is a regulator of virus-specific CD8+ T cell survival in HIV infection. J. Exp. Med. 203, 2281–2292 (2006).
Trautmann, L. et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat. Med. 12, 1198–1202 (2006).
O’Brien, T. R. et al. Serum HIV-1 RNA levels and time to development of AIDS in the multicenter hemophilia cohort study. JAMA 276, 105–110 (1996).
Quinn, T. C. et al. Viral load and heterosexual transmission of human immunodeficiency virus type 1. Rakai Project Study Group. N. Engl. J. Med. 342, 921–929 (2000).
Gray, R. H. et al. Probability of HIV-1 transmission per coital act in monogamous, heterosexual, HIV-1-discordant couples in Rakai, Uganda. Lancet 357, 1149–1153 (2001).
Gurdasani, D. et al. A systematic review of definitions of extreme phenotypes of HIV control and progression. AIDS 28, 149–162 (2014).
Kaslow, R. A. et al. Influence of combinations of human major histocompatibility complex genes on the course of HIV-1 infection. Nat. Med. 2, 405–411 (1996).
Migueles, S. A. et al. HLA B*5701 is highly associated with restriction of virus replication in a subgroup of HIV-infected long term nonprogressors. Proc. Natl Acad. Sci. USA 97, 2709–2714 (2000).
Pereyra, F. et al. The major genetic determinants of HIV-1 control affect HLA class I peptide presentation. Science 330, 1551–1557 (2010). Genome-wide association study of thousands of individuals confirmed that polymorphisms within the HLA class I binding pocket are strongly associated with spontaneous HIV control.
Fellay, J. et al. A whole-genome association study of major determinants for host control of HIV-1. Science 317, 944–947 (2007).
Dalmasso, C. et al. Distinct genetic loci control plasma HIV-RNA and cellular HIV-DNA levels in HIV-1 infection: the ANRS Genome wide association 01 study. PLoS One 3, e3907 (2008).
Limou, S. et al. Genomewide association study of an AIDS-nonprogression cohort emphasizes the role played by HLA genes (ANRS Genomewide Association Study 02). J. Infect. Dis. 199, 419–426 (2009).
McLaren, P. J. et al. Evaluating the impact of functional genetic variation on HIV-1 control. J. Infect. Dis. 216, 1063–1069 (2017).
Schmitz, J. E. et al. Control of viremia in simian immunodeficiency virus infection by CD8+ lymphocytes. Science 283, 857–860 (1999).
Jin, X. et al. Dramatic rise in plasma viremia after CD8+ T cell depletion in simian immunodeficiency virus-infected macaques. J. Exp. Med. 189, 991–998 (1999).
Friedrich, T. C. et al. Subdominant CD8+ T-cell responses are involved in durable control of AIDS virus replication. J. Virol. 81, 3465–3476 (2007).
Chowdhury, A. et al. Differential impact of in vivo CD8+ T lymphocyte depletion in controller versus progressor simian immunodeficiency virus-infected macaques. J. Virol. 89, 8677–8686 (2015).
Dalod, M. et al. Broad, intense anti-human immunodeficiency virus (HIV) ex vivo CD8+ responses in HIV type 1-infected patients: comparison with anti-Epstein-Barr virus responses and changes during antiretroviral therapy. J. Virol. 73, 7108–7116 (1999).
Gea-Banacloche, J. C. et al. Maintenance of large numbers of virus-specific CD8+ T cells in HIV-infected progressors and long-term nonprogressors. J. Immunol. 165, 1082–1092 (2000).
Betts, M. R. et al. Analysis of total human immunodeficiency virus (HIV)-specific CD4+ and CD8+ T-cell responses: relationship to viral load in untreated HIV infection. J. Virol. 75, 11983–11991 (2001).
Migueles, S. A. & Connors, M. Frequency and function of HIV-specific CD8+ T cells. Immunol. Lett. 79, 141–150 (2001).
Addo, M. M. et al. Comprehensive epitope analysis of human immunodeficiency virus type 1 (HIV-1)-specific T-cell responses directed against the entire expressed HIV-1 genome demonstrate broadly directed responses, but no correlation to viral load. J. Virol. 77, 2081–2092 (2003).
Varadarajan, N. et al. A high-throughput single-cell analysis of human CD8+ T cell functions reveals discordance for cytokine secretion and cytolysis. J. Clin. Invest. 121, 4322–4331 (2011).
Betts, M. R. et al. HIV nonprogressors preferentially maintain highly functional HIV-specific CD8+ T cells. Blood 107, 4781–4789 (2006).
Saez-Cirion, A. et al. HIV controllers exhibit potent CD8 T cell capacity to suppress HIV infection ex vivo and peculiar cytotoxic T lymphocyte activation phenotype. Proc. Natl Acad. Sci. USA 104, 6776–6781 (2007).
Hersperger, A. R. et al. Perforin expression directly ex vivo by HIV-specific CD8 T-cells is a correlate of HIV elite control. PLoS Pathog. 6, e1000917 (2010).
Lecuroux, C. et al. Both HLA-B*57 and plasma HIV RNA levels contribute to the HIV-specific CD8+ T cell response in HIV controllers. J. Virol. 88, 176–187 (2014).
Noel, N. et al. Long-term spontaneous control of HIV-1 is related to low frequency of infected cells and inefficient viral reactivation. J. Virol. 90, 6148–6158 (2016).
Migueles, S. A. et al. HIV-specific CD8+ T cell proliferation is coupled to perforin expression and is maintained in nonprogressors. Nat. Immunol. 3, 1061–1068 (2002). Maintenance of HIV-specific CD8 + T cell proliferation upon antigenic stimulation strongly differentiated spontaneous controllers from non-controllers and was associated with expression of the cytolytic effector perforin.
Migueles, S. A. et al. Lytic granule loading of CD8+ T cells is required for HIV-infected cell elimination associated with immune control. Immunity 29, 1009–1021 (2008). Cytolytic function of HIV-specific CD8 + T cells was enhanced in spontaneous controllers upon antigenic stimulation and expansion compared with non-escaped responses in non-controllers.
Ndhlovu, Z. M. et al. The breadth of expandable memory CD8+ T cells inversely correlates with residual viral loads in HIV elite controllers. J. Virol. 89, 10735–10747 (2015).
Gaiha, G. D. et al. Dysfunctional HIV-specific CD8+ T cell proliferation is associated with increased caspase-8 activity and mediated by necroptosis. Immunity 41, 1001–1012 (2014).
Migueles, S. A. et al. Defective human immunodeficiency virus-specific CD8+ T-cell polyfunctionality, proliferation, and cytotoxicity are not restored by antiretroviral therapy. J. Virol. 83, 11876–11889 (2009).
Rehr, M. et al. Emergence of polyfunctional CD8+ T cells after prolonged suppression of human immunodeficiency virus replication by antiretroviral therapy. J. Virol. 82, 3391–3404 (2008).
Shasha, D. et al. Elite controller CD8+ T cells exhibit comparable viral inhibition capacity, but better sustained effector properties compared to chronic progressors. J. Leukoc. Biol. 100, 1425–1433 (2016).
Yang, O. O. et al. Efficient lysis of human immunodeficiency virus type 1-infected cells by cytotoxic T lymphocytes. J. Virol. 70, 5799–5806 (1996).
Sacha, J. B. et al. Gag-specific CD8+ T lymphocytes recognize infected cells before AIDS-virus integration and viral protein expression. J. Immunol. 178, 2746–2754 (2007).
Monel, B. et al. HIV controllers exhibit effective CD8+ T cell recognition of HIV-1-infected non-activated CD4+ T cells. Cell Rep. 27, 142–153.e4 (2019).
Marsh, S. G. E., Parham, P. & Barber, L. D. The HLA FactsBook (Academic Press, 2000).
Kiepiela, P. et al. CD8+ T-cell responses to different HIV proteins have discordant associations with viral load. Nat. Med. 13, 46–53 (2007).
Zuniga, R. et al. Relative dominance of Gag p24-specific cytotoxic T lymphocytes is associated with human immunodeficiency virus control. J. Virol. 80, 3122–3125 (2006).
Edwards, B. H. et al. Magnitude of functional CD8+ T-cell responses to the gag protein of human immunodeficiency virus type 1 correlates inversely with viral load in plasma. J. Virol. 76, 2298–2305 (2002).
Migueles, S. A. et al. CD8+ T-cell cytotoxic capacity associated with human immunodeficiency virus-1 control can be mediated through various epitopes and human leukocyte antigen types. EBioMedicine 2, 46–58 (2014).
Rolland, M. et al. HIV-1 conserved-element vaccines: relationship between sequence conservation and replicative capacity. J. Virol. 87, 5461–5467 (2013).
Dahirel, V. et al. Coordinate linkage of HIV evolution reveals regions of immunological vulnerability. Proc. Natl Acad. Sci. USA 108, 11530–11535 (2011).
Ferguson, A. L. et al. Translating HIV sequences into quantitative fitness landscapes predicts viral vulnerabilities for rational immunogen design. Immunity 38, 606–617 (2013).
Barton, J. P. et al. Relative rate and location of intra-host HIV evolution to evade cellular immunity are predictable. Nat. Commun. 7, 11660 (2016).
Gaiha, G. D. et al. Structural topology defines protective CD8+ T cell epitopes in the HIV proteome. Science 364, 480–484 (2019). Network analysis of HIV protein structures identified CD8 + T cell epitopes derived from topologically important regions of the viral proteome that were constrained from mutation, targeted by proliferative responses in spontaneous HIV controllers and presented by common HLA alleles with broad representation in the global population.
Almeida, J. R. et al. Antigen sensitivity is a major determinant of CD8+ T-cell polyfunctionality and HIV-suppressive activity. Blood 113, 6351–6360 (2009).
Price, D. A. et al. Public clonotype usage identifies protective Gag-specific CD8+ T cell responses in SIV infection. J. Exp. Med. 206, 923–936 (2009).
Chen, H. et al. TCR clonotypes modulate the protective effect of HLA class I molecules in HIV-1 infection. Nat. Immunol. 13, 691–700 (2012).
Kosmrlj, A. et al. Effects of thymic selection of the T-cell repertoire on HLA class I-associated control of HIV infection. Nature 465, 350–354 (2010).
Mendoza, D. et al. HLA B*5701-positive long-term nonprogressors/elite controllers are not distinguished from progressors by the clonal composition of HIV-specific CD8+ T cells. J. Virol. 86, 4014–4018 (2012).
Flerin, N. C. et al. T-cell receptor (TCR) clonotype-specific differences in inhibitory activity of HIV-1 cytotoxic T-cell clones is not mediated by TCR alone. J. Virol. 91, e02412–16 (2017).
Joglekar, A. V. et al. T cell receptors for the HIV KK10 epitope from patients with differential immunologic control are functionally indistinguishable. Proc. Natl Acad. Sci. USA 115, 1877–1882 (2018).
Iglesias, M. C. et al. Escape from highly effective public CD8+ T-cell clonotypes by HIV. Blood 118, 2138–2149 (2011).
Gorin, A. M. et al. HIV-1 epitopes presented by MHC class I types associated with superior immune containment of viremia have highly constrained fitness landscapes. PLoS Pathog. 13, e1006541 (2017). Saturation mutagenesis of immunodominant CD8 + T cell epitopes revealed that those presented by protective HLA alleles (B*57, B*27) tolerated markedly fewer single and double mutations, implicating epitope mutational constraint as a contributing factor to HLA-mediated control of HIV.
Bailey, J. R., Williams, T. M., Siliciano, R. F. & Blankson, J. N. Maintenance of viral suppression in HIV-1-infected HLA-B*57+ elite suppressors despite CTL escape mutations. J. Exp. Med. 203, 1357–1369 (2006).
O’Connell, K. A. et al. Prolonged control of an HIV type 1 escape variant following treatment interruption in an HLA-B*27-positive patient. AIDS Res. Hum. Retroviruses 26, 1307–1311 (2010).
Pohlmeyer, C. W., Buckheit, R. W. 3rd, Siliciano, R. F. & Blankson, J. N. CD8+ T cells from HLA-B*57 elite suppressors effectively suppress replication of HIV-1 escape mutants. Retrovirology 10, 152 (2013).
McMichael, A. J. & Carrington, M. Topological perspective on HIV escape. Science 364, 438–439 (2019).
Brenchley, J. M. et al. CD4+ T cell depletion during all stages of HIV disease occurs predominantly in the gastrointestinal tract. J. Exp. Med. 200, 749–759 (2004).
Ferre, A. L. et al. Mucosal immune responses to HIV-1 in elite controllers: a potential correlate of immune control. Blood 113, 3978–3989 (2009).
Perreau, M. et al. Follicular helper T cells serve as the major CD4 T cell compartment for HIV-1 infection, replication, and production. J. Exp. Med. 210, 143–156 (2013).
Banga, R. et al. PD-1+ and follicular helper T cells are responsible for persistent HIV-1 transcription in treated aviremic individuals. Nat. Med. 22, 754–761 (2016).
Connick, E. et al. CTL fail to accumulate at sites of HIV-1 replication in lymphoid tissue. J. Immunol. 178, 6975–6983 (2007).
Quigley, M. F., Gonzalez, V. D., Granath, A., Andersson, J. & Sandberg, J. K. CXCR5+ CCR7- CD8 T cells are early effector memory cells that infiltrate tonsil B cell follicles. Eur. J. Immunol. 37, 3352–3362 (2007).
Connick, E. et al. Compartmentalization of simian immunodeficiency virus replication within secondary lymphoid tissues of rhesus macaques is linked to disease stage and inversely related to localization of virus-specific CTL. J. Immunol. 193, 5613–5625 (2014).
Petrovas, C. et al. Follicular CD8 T cells accumulate in HIV infection and can kill infected cells in vitro via bispecific antibodies. Sci. Transl Med. 9, eaag2285 (2017).
Leong, Y. A. et al. CXCR5+ follicular cytotoxic T cells control viral infection in B cell follicles. Nat. Immunol. 17, 1187–1196 (2016).
He, R. et al. Follicular CXCR5-expressing CD8+ T cells curtail chronic viral infection. Nature 537, 412–428 (2016).
Mylvaganam, G. H. et al. Dynamics of SIV-specific CXCR5+ CD8 T cells during chronic SIV infection. Proc. Natl Acad. Sci. USA 114, 1976–1981 (2017).
Li, S. et al. Simian immunodeficiency virus-producing cells in follicles are partially suppressed by CD8+ cells in vivo. J. Virol. 90, 11168–11180 (2016).
Fukazawa, Y. et al. B cell follicle sanctuary permits persistent productive simian immunodeficiency virus infection in elite controllers. Nat. Med. 21, 132–139 (2015).
Boritz, E. A. et al. Multiple origins of virus persistence during natural control of HIV infection. Cell 166, 1004–1015 (2016). Sequencing of HIV genomes, integration sites and T cell receptors revealed active HIV replication in lymph nodes of spontaneous HIV controllers despite undetectable viraemia, suggesting ongoing viral replication within lymphoid sanctuaries.
Mueller, S. N. & Mackay, L. K. Tissue-resident memory T cells: local specialists in immune defence. Nat. Rev. Immunol. 16, 79–89 (2016).
Reuter, M. A. et al. HIV-specific CD8+ T cells exhibit reduced and differentially regulated cytolytic activity in lymphoid tissue. Cell Rep. 21, 3458–3470 (2017).
Buggert, M. et al. Identification and characterization of HIV-specific resident memory CD8+ T cells in human lymphoid tissue. Sci. Immunol. 3, eaar4526 (2018). HIV-specific CD8 + T cells in lymphoid tissue of spontaneous controllers resembled tissue-resident memory cells, exhibited skewed TCR clonotypes and expressed more effector genes compared with circulating CD8 + T cells.
Nguyen, S. et al. Elite control of HIV is associated with distinct functional and transcriptional signatures in lymphoid tissue CD8+ T cells. Sci. Transl Med. 11, eaax4077 (2019).
Martin, M. P. et al. Epistatic interaction between KIR3DS1 and HLA-B delays the progression to AIDS. Nat. Genet. 31, 429–434 (2002).
Martin, M. P. et al. Innate partnership of HLA-B and KIR3DL1 subtypes against HIV-1. Nat. Genet. 39, 733–740 (2007).
Pelak, K. et al. Copy number variation of KIR genes influences HIV-1 control. PLoS Biol. 9, e1001208 (2011).
Apps, R. et al. Influence of HLA-C expression level on HIV control. Science 340, 87–91 (2013).
Ramsuran, V. et al. Elevated HLA-A expression impairs HIV control through inhibition of NKG2A-expressing cells. Science 359, 86–90 (2018).
Miura, T. et al. HLA-associated viral mutations are common in human immunodeficiency virus type 1 elite controllers. J. Virol. 83, 3407–3412 (2009).
Veenhuis, R. T. et al. Long-term remission despite clonal expansion of replication-competent HIV-1 isolates. JCI Insight 3, 122795 (2018).
Miura, T. et al. Impaired replication capacity of acute/early viruses in persons who become HIV controllers. J. Virol. 84, 7581–7591 (2010).
Casado, C. et al. Viral characteristics associated with the clinical nonprogressor phenotype are inherited by viruses from a cluster of HIV-1 elite controllers. mBio 9, e02338–17 (2018).
Zaunders, J. et al. Possible clearance of transfusion-acquired nef/LTR-deleted attenuated HIV-1 infection by an elite controller with CCR5 Δ32 heterozygous and HLA-B57 genotype. J. Virus Erad. 5, 73–83 (2019).
Bailey, J. R. et al. Transmission of human immunodeficiency virus type 1 from a patient who developed AIDS to an elite suppressor. J. Virol. 82, 7395–7410 (2008).
Buckheit, R. W. 3rd et al. Host factors dictate control of viral replication in two HIV-1 controller/chronic progressor transmission pairs. Nat. Commun. 3, 716 (2012).
Yue, L. et al. Transmitted virus fitness and host T cell responses collectively define divergent infection outcomes in two HIV-1 recipients. PLoS Pathog. 11, e1004565 (2015).
Carlson, J. M. et al. Impact of pre-adapted HIV transmission. Nat. Med. 22, 606–613 (2016).
Bailey, J. R. et al. Neutralizing antibodies do not mediate suppression of human immunodeficiency virus type 1 in elite suppressors or selection of plasma virus variants in patients on highly active antiretroviral therapy. J. Virol. 80, 4758–4770 (2006).
Lambotte, O. et al. Heterogeneous neutralizing antibody and antibody-dependent cell cytotoxicity responses in HIV-1 elite controllers. AIDS 23, 897–906 (2009).
Pereyra, F. et al. Persistent low-level viremia in HIV-1 elite controllers and relationship to immunologic parameters. J. Infect. Dis. 200, 984–990 (2009).
Doria-Rose, N. A. et al. Breadth of human immunodeficiency virus-specific neutralizing activity in sera: clustering analysis and association with clinical variables. J. Virol. 84, 1631–1636 (2010).
Freund, N. T. et al. Coexistence of potent HIV-1 broadly neutralizing antibodies and antibody-sensitive viruses in a viremic controller. Sci. Transl Med. 9, eaal2144 (2017).
Ackerman, M. E. et al. Polyfunctional HIV-specific antibody responses are associated with spontaneous HIV control. PLoS Pathog. 12, e1005315 (2016).
Smalls-Mantey, A. et al. Antibody-dependent cellular cytotoxicity against primary HIV-infected CD4+ T cells is directly associated with the magnitude of surface IgG binding. J. Virol. 86, 8672–8680 (2012).
Mudd, P. A. et al. Reduction of CD4+ T cells in vivo does not affect virus load in macaque elite controllers. J. Virol. 85, 7454–7459 (2011).
Schmitz, J. E. et al. Effect of humoral immune responses on controlling viremia during primary infection of rhesus monkeys with simian immunodeficiency virus. J. Virol. 77, 2165–2173 (2003).
Pollack, R. A. et al. Defective HIV-1 proviruses are expressed and can be recognized by cytotoxic T lymphocytes, which shape the proviral landscape. Cell Host Microbe 21, 494–506.e4 (2017).
de Azevedo, S. S. D. et al. Highly divergent patterns of genetic diversity and evolution in proviral quasispecies from HIV controllers. Retrovirology 14, 29 (2017).
Mens, H. et al. HIV-1 continues to replicate and evolve in patients with natural control of HIV infection. J. Virol. 84, 12971–12981 (2010).
Li, J. Z. et al. ART reduces T cell activation and immune exhaustion markers in HIV controllers. Clin. Infect. Dis. https://doi.org/10.1093/cid/ciz442 (2019).
Blankson, J. N. et al. Isolation and characterization of replication-competent human immunodeficiency virus type 1 from a subset of elite suppressors. J. Virol. 81, 2508–2518 (2007).
Julg, B. et al. Infrequent recovery of HIV from but robust exogenous infection of activated CD4+ T cells in HIV elite controllers. Clin. Infect. Dis. 51, 233–238 (2010).
Graf, E. H. et al. Elite suppressors harbor low levels of integrated HIV DNA and high levels of 2-LTR circular HIV DNA compared to HIV+ patients on and off HAART. PLoS Pathog. 7, e1001300 (2011).
Hatano, H. et al. Comparison of HIV DNA and RNA in gut-associated lymphoid tissue of HIV-infected controllers and noncontrollers. AIDS 27, 2255–2260 (2013).
Mendoza, D. et al. Comprehensive analysis of unique cases with extraordinary control over HIV replication. Blood 119, 4645–4655 (2012). Characterization of individuals with extraordinary CD8 + T cell-mediated spontaneous control of HIV infection to levels at which replication-competent virus was unable to be recovered, suggesting potential avenues for functional cure, remission or perhaps even clearance of replication-competent HIV reservoirs.
Saez-Cirion, A. et al. Post-treatment HIV-1 controllers with a long-term virological remission after the interruption of early initiated antiretroviral therapy ANRS VISCONTI study. PLoS Pathog. 9, e1003211 (2013).
Namazi, G. et al. The Control of HIV after Antiretroviral Medication Pause (CHAMP) study: posttreatment controllers identified from 14 clinical studies. J. Infect. Dis. 218, 1954–1963 (2018).
Williams, J. P. et al. HIV-1 DNA predicts disease progression and post-treatment virological control. eLife 3, e03821 (2014).
Lodi, S. et al. Immunovirologic control 24 months after interruption of antiretroviral therapy initiated close to HIV seroconversion. Arch. Intern. Med. 172, 1252–1255 (2012).
Goujard, C. et al. HIV-1 control after transient antiretroviral treatment initiated in primary infection: role of patient characteristics and effect of therapy. Antivir. Ther. 17, 1001–1009 (2012).
Sharaf, R. et al. HIV-1 proviral landscapes distinguish posttreatment controllers from noncontrollers. J. Clin. Invest. 128, 4074–4085 (2018).
Park, Y. J. et al. Impact of HLA class I alleles on timing of HIV rebound after antiretroviral treatment interruption. Pathog. Immun. 2, 431–445 (2017).
Briney, B. et al. Tailored immunogens direct affinity maturation toward HIV neutralizing antibodies. Cell 166, 1459–1470.e11 (2016).
Escolano, A. et al. Sequential immunization elicits broadly neutralizing anti-HIV-1 antibodies in Ig knockin mice. Cell 166, 1445–1458.e12 (2016).
Steichen, J. M. et al. A generalized HIV vaccine design strategy for priming of broadly neutralizing antibody responses. Science 366, eaax4380 (2019).
Kolodkin-Gal, D. et al. Efficiency of cell-free and cell-associated virus in mucosal transmission of human immunodeficiency virus type 1 and simian immunodeficiency virus. J. Virol. 87, 13589–13597 (2013).
Parsons, M. S. et al. Partial efficacy of a broadly neutralizing antibody against cell-associated SHIV infection. Sci. Transl Med. 9, eaaf1483 (2017).
Abela, I. A. et al. Cell-cell transmission enables HIV-1 to evade inhibition by potent CD4bs directed antibodies. PLoS Pathog. 8, e1002634 (2012).
Buckheit, R. W. III, Siliciano, R. F. & Blankson, J. N. Primary CD8+ T cells from elite suppressors effectively eliminate non-productively HIV-1 infected resting and activated CD4+ T cells. Retrovirology 10, 68 (2013).
Hammer, S. M. et al. Efficacy trial of a DNA/rAd5 HIV-1 preventive vaccine. N. Engl. J. Med. 369, 2083–2092 (2013).
Rolland, M. et al. Genetic impact of vaccination on breakthrough HIV-1 sequences from the STEP trial. Nat. Med. 17, 366–371 (2011). HIV sequence analysis of breakthrough infections from 68 participants of the STEP trial revealed a ‘sieving’ effect at commonly targeted immunodominant epitopes, suggesting emergence of viral escape from vaccine-induced CD8 + T cell responses.
Janes, H. E. et al. Higher T-cell responses induced by DNA/rAd5 HIV-1 preventive vaccine are associated with lower HIV-1 infection risk in an efficacy trial. J. Infect. Dis. 215, 1376–1385 (2017).
Mudd, P. A. et al. Vaccine-induced CD8+ T cells control AIDS virus replication. Nature 491, 129–133 (2012). Vaccine-induced CD8 + T cell epitopes directed towards specific epitopes achieved durable viral control after challenge in six of eight animals, providing proof of principle that narrowly targeted vaccine-induced CD8 + T cell responses can control viral replication.
Letourneau, S. et al. Design and pre-clinical evaluation of a universal HIV-1 vaccine. PLoS One 2, e984 (2007).
Rolland, M., Nickle, D. C. & Mullins, J. I. HIV-1 group M conserved elements vaccine. PLoS Pathog. 3, e157 (2007).
Mothe, B. et al. Definition of the viral targets of protective HIV-1-specific T cell responses. J. Transl Med. 9, 208 (2011).
Kallas, E. G. et al. Antigenic competition in CD4+ T cell responses in a randomized, multicenter, double-blind clinical HIV vaccine trial. Sci. Transl Med. 11, eaaw1673 (2019).
Chamcha, V. et al. Strong TH1-biased CD4 T cell responses are associated with diminished SIV vaccine efficacy. Sci. Transl Med. 11, eaav1800 (2019).
Santra, S. et al. Mosaic vaccines elicit CD8+ T lymphocyte responses that confer enhanced immune coverage of diverse HIV strains in monkeys. Nat. Med. 16, 324–328 (2010).
Barouch, D. H. et al. Mosaic HIV-1 vaccines expand the breadth and depth of cellular immune responses in rhesus monkeys. Nat. Med. 16, 319–323 (2010).
Barouch, D. H. et al. Evaluation of a mosaic HIV-1 vaccine in a multicentre, randomised, double-blind, placebo-controlled, phase 1/2a clinical trial (APPROACH) and in rhesus monkeys (NHP 13-19). Lancet 392, 232–243 (2018).
Ondondo, B. et al. Novel conserved-region T-cell mosaic vaccine with high global HIV-1 coverage is recognized by protective responses in untreated infection. Mol. Ther. 24, 832–842 (2016).
Hansen, S. G. et al. Effector memory T cell responses are associated with protection of rhesus monkeys from mucosal simian immunodeficiency virus challenge. Nat. Med. 15, 293–299 (2009).
Kim, J., Kim, A. R. & Shin, E. C. Cytomegalovirus infection and memory T cell inflation. Immune Netw. 15, 186–190 (2015).
Hansen, S. G. et al. Profound early control of highly pathogenic SIV by an effector memory T-cell vaccine. Nature 473, 523–527 (2011).
Hansen, S. G. et al. Immune clearance of highly pathogenic SIV infection. Nature 502, 100–104 (2013). Progressive clearance of SIV from ~50% of RhCMV-vectored SIV-vaccinated rhesus macaques suggests that vaccine-induced effector-memory CD8 + T cells are capable of preventing the establishment of persistent viral reservoirs.
Hansen, S. G. et al. A live-attenuated RhCMV/SIV vaccine shows long-term efficacy against heterologous SIV challenge. Sci. Transl Med. 11, eaaw2607 (2019).
Hansen, S. G. et al. Broadly targeted CD8+ T cell responses restricted by major histocompatibility complex E. Science 351, 714–720 (2016).
Geraghty, D. E., Stockschleader, M., Ishitani, A. & Hansen, J. A. Polymorphism at the HLA-E locus predates most HLA-A and -B polymorphism. Hum. Immunol. 33, 174–184 (1992).
Deng, K. et al. Broad CTL response is required to clear latent HIV-1 due to dominance of escape mutations. Nature 517, 381–385 (2015).
Mylvaganam, G., Yanez, A. G., Maus, M. & Walker, B. D. Toward T cell-mediated control or elimination of HIV reservoirs: lessons from cancer immunology. Front. Immunol. 10, 2109 (2019).
Amancha, P. K., Hong, J. J., Rogers, K., Ansari, A. A. & Villinger, F. In vivo blockade of the programmed cell death-1 pathway using soluble recombinant PD-1-Fc enhances CD4+ and CD8+ T cell responses but has limited clinical benefit. J. Immunol. 191, 6060–6070 (2013).
Mylvaganam, G. H. et al. Combination anti-PD-1 and antiretroviral therapy provides therapeutic benefit against SIV. JCI Insight 3, 122940 (2018).
Bekerman, E. et al. PD-1 blockade and TLR7 activation lack therapeutic benefit in chronic simian immunodeficiency virus-infected macaques on antiretroviral therapy. Antimicrob. Agents Chemother. 63, e01163–19 (2019).
Lu, W., Arraes, L. C., Ferreira, W. T. & Andrieu, J. M. Therapeutic dendritic-cell vaccine for chronic HIV-1 infection. Nat. Med. 10, 1359–1365 (2004).
Kloverpris, H. et al. Induction of novel CD8+ T-cell responses during chronic untreated HIV-1 infection by immunization with subdominant cytotoxic T-lymphocyte epitopes. AIDS 23, 1329–1340 (2009).
Garcia, F. et al. A dendritic cell-based vaccine elicits T cell responses associated with control of HIV-1 replication. Sci. Transl Med. 5, 166ra2 (2013).
Levy, Y. et al. Dendritic cell-based therapeutic vaccine elicits polyfunctional HIV-specific T-cell immunity associated with control of viral load. Eur. J. Immunol. 44, 2802–2810 (2014).
Macatangay, B. J. et al. Therapeutic vaccination with dendritic cells loaded with autologous HIV type 1-infected apoptotic cells. J. Infect. Dis. 213, 1400–1409 (2016).
Borducchi, E. N. et al. Ad26/MVA therapeutic vaccination with TLR7 stimulation in SIV-infected rhesus monkeys. Nature 540, 284–287 (2016). rAd26 and modified vaccinia Ankara-vectored SIV therapeutic vaccine with TLR7 agonist induced broad CD8 + T cell responses, delayed viral rebound following ART interruption and reduced set point viral loads in immunized rhesus macaques.
Mothe, B. et al. Therapeutic vaccination refocuses T-cell responses towards conserved regions of HIV-1 in early treated individuals (BCN 01 study). EClinicalMedicine 11, 65–80 (2019). Immunogenicity trial of conserved HIV elements vectored by ChAdV and modified vaccinia Ankara in therapeutic vaccine setting elicited strong responses against vaccine epitopes that would otherwise be subdominant in natural infection, providing proof of principle that CD8 + T cell responses in chronically infected individuals can be redirected towards specific regions of the viral proteome.
Shapiro, S. Z. Lessons for general vaccinology research from attempts to develop an HIV vaccine. Vaccine 37, 3400–3408 (2019).
Fauci, A. S., Marovich, M. A., Dieffenbach, C. W., Hunter, E. & Buchbinder, S. P. Immunology. Immune activation with HIV vaccines. Science 344, 49–51 (2014).
Penaloza-MacMaster, P. et al. Vaccine-elicited CD4 T cells induce immunopathology after chronic LCMV infection. Science 347, 278–282 (2015).
Landovitz, R. J. et al. Safety, tolerability, and pharmacokinetics of long-acting injectable cabotegravir in low-risk HIV-uninfected individuals: HPTN 077, a phase 2a randomized controlled trial. PLoS Med. 15, e1002690 (2018).
Henderson, G. E. et al. Ethics of treatment interruption trials in HIV cure research: addressing the conundrum of risk/benefit assessment. J. Med. Ethics 44, 270–276 (2018).
Clarridge, K. E. et al. Effect of analytical treatment interruption and reinitiation of antiretroviral therapy on HIV reservoirs and immunologic parameters in infected individuals. PLoS Pathog. 14, e1006792 (2018). HIV reservoir size and markers of immune activation and exhaustion were not elevated 6 to 12 months after analytical ART interruption, demonstrating the safety of treatment interruptions for evaluating therapeutic vaccines.
Strongin, Z. et al. Effect of short-term antiretroviral therapy interruption on levels of integrated HIV DNA. J. Virol. 92, e00285–18 (2018).
Salantes, D. B. et al. HIV-1 latent reservoir size and diversity are stable following brief treatment interruption. J. Clin. Invest. 128, 3102–3115 (2018).
Okoye, A. A. et al. Early antiretroviral therapy limits SIV reservoir establishment to delay or prevent post-treatment viral rebound. Nat. Med. 24, 1430–1440 (2018).
Kamphorst, A. O., Araki, K. & Ahmed, R. Beyond adjuvants: immunomodulation strategies to enhance T cell immunity. Vaccine 33, B21–B28 (2015).
Nishimura, Y. et al. Early antibody therapy can induce long-lasting immunity to SHIV. Nature 543, 559–563 (2017). Passive infusion of broadly neutralizing antibodies, but not ART, initiated early after SHIV infection, resulted in long-term control of viraemia in rhesus macaques due to a proposed vaccinal effect of antibody–antigen complexes that may facilitate induction of de novo antiviral CD8 + T cell responses.
Rahman, M. A. & Robert-Guroff, M. Accelerating HIV vaccine development using non-human primate models. Expert. Rev. Vaccines 18, 61–73 (2019).
Kalams, S. A. et al. Safety and comparative immunogenicity of an HIV-1 DNA vaccine in combination with plasmid interleukin 12 and impact of intramuscular electroporation for delivery. J. Infect. Dis. 208, 818–829 (2013).
Leal, L. et al. Phase I clinical trial of an intranodally administered mRNA-based therapeutic vaccine against HIV-1 infection. AIDS 32, 2533–2545 (2018).
Brekke, K. et al. Intranasal administration of a therapeutic HIV vaccine (Vacc-4×) induces dose-dependent systemic and mucosal immune responses in a randomized controlled trial. PLoS One 9, e112556 (2014).
Stephenson, K. E. et al. First-in-human randomized controlled trial of an oral, replicating adenovirus 26 vector vaccine for HIV-1. PLoS One 13, e0205139 (2018).
Tan, H. X. et al. Induction of vaginal-resident HIV-specific CD8 T cells with mucosal prime-boost immunization. Mucosal Immunol. 11, 994–1007 (2018).
Shin, H. & Iwasaki, A. A vaccine strategy that protects against genital herpes by establishing local memory T cells. Nature 491, 463–467 (2012).
Acknowledgements
We are grateful to the participants of all cited studies. We apologize to the many authors whose work was not cited in this review owing to space limitations. This work was supported by the US National Institutes of Health (NIH) grant UM1AI100663 and R37AI067073.
Author information
Authors and Affiliations
Contributions
All listed authors contributed to the writing, editing and preparation of this review article.
Corresponding author
Ethics declarations
Competing interests
G.D.G. and B.D.W. have filed a provisional patent application (62/817,094) related to HIV vaccine design. D.R.C. declares no competing interests.
Additional information
Peer review information
Nature Reviews Immunology thanks P. Goulder, S. Lewin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Collins, D.R., Gaiha, G.D. & Walker, B.D. CD8+ T cells in HIV control, cure and prevention. Nat Rev Immunol 20, 471–482 (2020). https://doi.org/10.1038/s41577-020-0274-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41577-020-0274-9
This article is cited by
-
Mucosal T-cell responses to chronic viral infections: Implications for vaccine design
Cellular & Molecular Immunology (2024)
-
Prevention, treatment and cure of HIV infection
Nature Reviews Microbiology (2023)
-
HIV infection
Nature Reviews Disease Primers (2023)
-
Strategies for HIV-1 vaccines that induce broadly neutralizing antibodies
Nature Reviews Immunology (2023)
-
High monoclonal neutralization titers reduced breakthrough HIV-1 viral loads in the Antibody Mediated Prevention trials
Nature Communications (2023)