Abstract
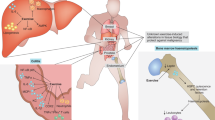
Remodelling of the immune system with age — immunosenescence — is a substantial contributor to poor health in older adults, with increasing risk of infections, cancer and chronic inflammatory disease contributing to age-related multi-morbidity. What is seldom considered when examining the immune response of an aged individual is that the immune system is profoundly influenced by physical activity. Habitual physical activity levels decline with age, with significant consequences for muscle mass and function. Skeletal muscle is a major immune regulatory organ and generates a range of proteins, termed myokines, which have anti-inflammatory and immunoprotective effects. Several studies indicate that maintaining physical activity has immune benefits in older adults, for example, it reduces the systemic inflammation associated with chronic age-related diseases. Here, we discuss how physical activity can prevent or ameliorate age-related multi-morbidity by boosting immune function, and we consider whether physical activity could improve immunotherapy outcomes in age-related conditions such as cancer.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kirkwood, T. B. L. Why and how are we living longer? Exp. Physiol. 102, 1067–1074 (2017).
Salomon, J. A. et al. Healthy life expectancy for 187 countries, 1990–2010: a systematic analysis for the Global Burden Disease Study 2010. Lancet 380, 2144–2162 (2012).
Rocca, W. A. et al. Prevalence of multimorbidity in a geographically defined American population: patterns by age, sex, and race/ethnicity. Mayo Clin. Proc. 89, 1336–1349 (2014).
Colchero, F. et al. The emergence of longevous populations. Proc. Natl Acad. Sci. USA 113, E7681–E7690 (2016).
Booth, F. W., Chakravarthy, M. V. & Spangenburg, E. E. Exercise and gene expression: physiological regulation of the human genome through physical activity. J. Physiol. 543, 399–411 (2002).
Harridge, S. D. & Lazarus, N. R. Physical activity, aging, and physiological function. Physiology 32, 152–161 (2017).
Gopinath, B., Kifley, A., Flood, V. M. & Mitchell, P. Physical activity as a determinant of successful aging over ten years. Sci. Rep. 8, 10522 (2018).
Harber, M. P. et al. Impact of cardiorespiratory fitness on all-cause and disease-specific mortality: advances since 2009. Prog. Cardiovasc. Dis. 60, 11–20 (2017).
Barry, V. W. et al. Fitness versus fatness on all-cause mortality: a meta-analysis. Prog. Cardiovasc. Dis. 56, 382–390 (2014).
Bouchard, C., Blair, S. N. & Katzmarzyk, P. T. Less sitting, more physical activity, or higher fitness? Mayo Clin. Proc. 90, 1533–1540 (2015). This paper reviews the evidence for physical activity level and sedentary time as independent variables influencing health, proposing there should be recommendations made for both.
Cabanas-Sanchez, V. et al. Physical activity, sitting time, and mortality from inflammatory diseases in older adults. Front. Physiol. 9, 898 (2018).
Gleeson, M. et al. The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat. Rev. Immunol. 11, 607–615 (2011).
Dhalwani, N. N. et al. Long terms trends of multimorbidity and association with physical activity in older English population. Int. J. Behav. Nutr. Phys. Act. 13, 8 (2016). This longitudinal study describes population-level data suggesting an inverse association between physical activity level and multi-morbidity in older adults.
Vancampfort, D. et al. Chronic physical conditions, multimorbidity and physical activity across 46 low- and middle-income countries. Int. J. Behav. Nutr. Phys. Act. 14, 6 (2017).
Andersen, Z. J. et al. A study of the combined effects of physical activity and air pollution on mortality in elderly urban residents: the Danish Diet, Cancer, and Health Cohort. Environ. Health Perspect. 123, 557–563 (2015).
Pape, K. et al. Leisure-time physical activity and the risk of suspected bacterial infections. Med. Sci. Sports Exerc. 48, 1737–1744 (2016).
Baik, I. et al. A prospective study of age and lifestyle factors in relation to community-acquired pneumonia in US men and women. Arch. Intern. Med. 160, 3082–3088 (2000).
Simpson, R. J. et al. Cardiorespiratory fitness is associated with better control of latent herpesvirus infections in a large ethnically diverse community sample: evidence from the Texas City Stress and Health Study. Brain Behav. Immun. 66, e35 (2017). This paper suggests that involvement in regular physical activity improves antiviral immunity to prevalent latent viral infections such as CMV, which are thought to be one driver of immunosenescence.
Leitzmann, M. et al. European code against cancer 4th edition: physical activity and cancer. Cancer Epidemiol. 39 (Suppl. 1), 46–55 (2015).
Higueras-Fresnillo, S. et al. Physical activity and association between frailty and all-cause and cardiovascular mortality in older adults: population-based prospective cohort study. J. Am. Geriatr. Soc. 66, 2097–2103 (2018).
Morris, J. K. et al. Aerobic exercise for Alzheimer’s disease: a randomized controlled pilot trial. PLOS ONE 12, e0170547 (2017).
Lowder, T., Padgett, D. A. & Woods, J. A. Moderate exercise protects mice from death due to influenza virus. Brain Behav. Immun. 19, 377–380 (2005).
Lowder, T., Padgett, D. A. & Woods, J. A. Moderate exercise early after influenza virus infection reduces the Th1 inflammatory response in lungs of mice. Exerc. Immunol. Rev. 12, 97–111 (2006).
Andrew, M. K. et al. The importance of frailty in the assessment of influenza vaccine effectiveness against influenza-related hospitalization in elderly people. J. Infect. Dis. 216, 405–414 (2017).
Pascoe, A. R., Fiatarone Singh, M. A. & Edwards, K. M. The effects of exercise on vaccination responses: a review of chronic and acute exercise interventions in humans. Brain Behav. Immun. 39, 33–41 (2014).
Woods, J. A. et al. Cardiovascular exercise training extends influenza vaccine seroprotection in sedentary older adults: the immune function intervention trial. J. Am. Geriatr. Soc. 57, 2183–2191 (2009).
Kohut, M. L. et al. Moderate exercise improves antibody response to influenza immunization in older adults. Vaccine 22, 2298–2306 (2004). This is one of the first papers to show that an extended period of increased physical activity increases vaccine responses in older adults.
Metsios, G. S. et al. Individualised exercise improves endothelial function in patients with rheumatoid arthritis. Ann. Rheum. Dis. 73, 748–751 (2014).
Manning, V. L. et al. Education, self-management, and upper extremity exercise training in people with rheumatoid arthritis: a randomized controlled trial. Arthritis Care Res. 66, 217–227 (2014).
McLean, H. Q. et al. Influenza vaccine effectiveness in the United States during 2012–2013: variable protection by age and virus type. J. Infect. Dis. 211, 1529–1540 (2015).
Foxman, B. Urinary tract infection syndromes: occurrence, recurrence, bacteriology, risk factors, and disease burden. Infect. Dis. Clin. North Am. 28, 1–13 (2014).
Kawai, K., Gebremeskel, B. G. & Acosta, C. J. Systematic review of incidence and complications of herpes zoster: towards a global perspective. BMJ Open 4, e004833 (2014).
Trintinaglia, L. et al. Features of immunosenescence in women newly diagnosed with breast cancer. Front. Immunol. 9, 1651 (2018).
Franceschi, C. & Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. A Biol. Sci. Med. Sci. 69 (Suppl. 1), 4–9 (2014). This paper presents an update on the evidence suggesting that the age-related increase in systemic inflammation is a contributor to diseases of old age.
Fuggle, N. R. et al. Relationships between markers of inflammation and bone density: findings from the Hertfordshire Cohort Study. Osteoporos. Int. 29, 1581–1589 (2018).
Turner, J. E. Is immunosenescence influenced by our lifetime “dose” of exercise? Biogerontology 17, 581–602 (2016).
Flynn, M. G., Markofski, M. M. & Carrillo, A. E. Elevated inflammatory status and increased risk of chronic disease in chronological aging — inflammaging or inflamm-inactivity? Aging Dis. 10, 147–156 (2019).
Callender, L. A. et al. Human CD8+ EMRA T cells display a senescence-associated secretory phenotype regulated by p38 MAPK. Aging Cell 17, e12675 (2018). Together with Frasca et al. (2017), this paper shows that highly differentiated lymphocytes have the same pro-inflammatory secretory phenotype seen in non-immune cells and may thus contribute to inflammageing.
Frasca, D., Diaz, A., Romero, M. & Blomberg, B. B. Human peripheral late/exhausted memory B cells express a senescent-associated secretory phenotype and preferentially utilize metabolic signaling pathways. Exp. Gerontol. 87, 113–120 (2017).
Martinez de Toda, I. et al. Immune function parameters as markers of biological age and predictors of longevity. Aging 8, 3110–3119 (2016).
Alpert, A. et al. High resolution longitudinal immune profiling reveals a clinically meaningful Metric 2 from dynamics of healthy immune-aging towards an older adult homeostasis. Nat. Med. (in the press).
Koetz, K. et al. T cell homeostasis in patients with rheumatoid arthritis. Proc. Natl Acad. Sci. USA 97, 9203–9208 (2000).
Thewissen, M. et al. Analyses of immunosenescent markers in patients with autoimmune disease. Clin. Immunol. 123, 209–218 (2007).
Pollock, R. D. et al. An investigation into the relationship between age and physiological function in highly active older adults. J. Physiol. 593, 657–680 (2015). This study assesses a group of highly active older adults and reveals that many classic features of physiological ageing, such as sarcopenia, were ameliorated by maintained physical activity.
Duggal, N. A., Pollock, R. D., Lazarus, N. R., Harridge, S. & Lord, J. M. Major features of immunesenescence, including reduced thymic output, are ameliorated by high levels of physical activity in adulthood. Aging Cell 17, 12750 (2018). This paper, based on the same cohort as in Pollock et al. (2015), reveals that many but not all features of immunosenescence were prevented by maintained physical activity.
Pereira, B. I. & Akbar, A. N. Convergence of innate and adaptive immunity during human aging. Front. Immunol. 7, 445 (2016).
Duggal, N. A., Upton, J., Phillips, A. C., Sapey, E. & Lord, J. M. An age-related numerical and functional deficit in CD19+ CD24hi CD38hi B cells is associated with an increase in systemic autoimmunity. Aging Cell 12, 873–881 (2013).
Spielmann, G. et al. Aerobic fitness is associated with lower proportions of senescent blood T cells in man. Brain Behav. Immun. 25, 1521–1529 (2011).
Kruger, K. et al. Apoptosis of T cell subsets after acute high-intensity interval exercise. Med. Sci. Sports Exerc. 48, 2021–2029 (2016).
Mooren, F. C. & Kruger, K. Apoptotic lymphocytes induce progenitor cell mobilization after exercise. J. Appl. Physiol. 119, 135–139 (2015).
de Araujo, A. L. et al. Elderly men with moderate and intense training lifestyle present sustained higher antibody responses to influenza vaccine. Age 37, 105 (2015). This paper reports that maintained physical activity in old age results in increased functional immunity, namely, improved vaccine responses.
Pedersen, B. K. & Febbraio, M. A. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 8, 457–465 (2012).
Whitham, M. et al. Contraction-induced interleukin-6 gene transcription in skeletal muscle is regulated by c-Jun terminal kinase/activator protein-1. J. Biol. Chem. 287, 10771–10779 (2012).
Munoz-Canoves, P., Scheele, C., Pedersen, B. K. & Serrano, A. L. Interleukin-6 myokine signaling in skeletal muscle: a double-edged sword? FEBS J. 280, 4131–4148 (2013).
Bethin, K. E., Vogt, S. K. & Muglia, L. J. Interleukin-6 is an essential, corticotropin-releasing hormone-independent stimulator of the adrenal axis during immune system activation. Proc. Natl Acad. Sci. USA 97, 9317–9322 (2000).
Starkie, R., Ostrowski, S. R., Jauffred, S., Febbraio, M. & Pedersen, B. K. Exercise and IL-6 infusion inhibit endotoxin-induced TNF-α production in humans. FASEB J. 17, 884–886 (2003).
Rao, R. R. et al. Meteorin-like is a hormone that regulates immune-adipose interactions to increase beige fat thermogenesis. Cell 157, 1279–1291 (2014).
Nilsson, A., Bergens, O. & Kadi, F. Physical activity alters inflammation in older adults by different intensity levels. Med. Sci. Sports Exerc. 50, 1502–1507 (2018).
Haugen, F. et al. IL-7 is expressed and secreted by human skeletal muscle cells. Am. J. Physiol. Cell Physiol. 298, C807–C816 (2010). Together with Rinnov et al. (2014), this paper shows how active skeletal muscle could support the function, survival and proliferation of immune cells through IL-7 and IL-15 production.
Rinnov, A. et al. Endurance training enhances skeletal muscle interleukin-15 in human male subjects. Endocrine 45, 271–278 (2014).
Shitara, S. et al. IL-7 produced by thymic epithelial cells plays a major role in the development of thymocytes and TCRγδ+ intraepithelial lymphocytes. J. Immunol. 190, 6173–6179 (2013).
Wallace, D. L. et al. Prolonged exposure of naive CD8+ T cells to interleukin-7 or interleukin-15 stimulates proliferation without differentiation or loss of telomere length. Immunology 119, 243–253 (2006).
Nielsen, A. R. et al. Association between interleukin-15 and obesity: interleukin-15 as a potential regulator of fat mass. J. Clin. Endocrinol. Metab. 93, 4486–4493 (2008).
Yang, H., Youm, Y. H. & Dixit, V. D. Inhibition of thymic adipogenesis by caloric restriction is coupled with reduction in age-related thymic involution. J. Immunol. 183, 3040–3052 (2009).
Al-Attar, A. et al. Human body composition and immunity: visceral adipose tissue produces IL-15 and muscle strength inversely correlates with NK cell function in elderly humans. Front. Immunol. 9, 440 (2018).
Whitham, M. et al. Extracellular vesicles provide a means for tissue crosstalk during exercise. Cell Metab. 27, 237–251 (2018). This paper shows that the endocrine role of skeletal muscle may be mediated through the release of exosomes.
Nieman, D. C. et al. Physical activity and immune function in elderly women. Med. Sci. Sports. Exerc. 25, 823–831 (1993).
Bartlett, D. B. et al. Habitual physical activity is associated with the maintenance of neutrophil migratory dynamics in healthy older adults. Brain Behav. Immun. 56, 12–20 (2016).
Timmerman, K. L., Flynn, M. G., Coen, P. M., Markofski, M. M. & Pence, B. D. Exercise training-induced lowering of inflammatory (CD14+CD16+) monocytes: a role in the anti-inflammatory influence of exercise? J. Leukoc. Biol. 84, 1271–1278 (2008).
Bartlett, D. B. et al. Ten weeks of high-intensity interval walk training is associated with reduced disease activity and improved innate immune function in older adults with rheumatoid arthritis: a pilot study. Arthritis Res. Ther. 20, 127 (2018).
Auerbach, P. et al. Differential effects of endurance training and weight loss on plasma adiponectin multimers and adipose tissue macrophages, in younger moderately overweight men. Am. J. Physiol. Regul. Integr. Comp. Physiol. 305, R490–R498 (2013).
Nikolich-Zugich, J. & van Lier, R. A. W. Cytomegalovirus (CMV) research in immune senescence comes of age: overview of the 6th International Workshop on CMV and Immunosenescence. Geroscience 39, 245–249 (2017).
Griffiths, P. D. & Mahungu, T. Why CMV is a candidate for elimination and then eradication. J. Virus Erad. 2, 131–135 (2016).
Kunz, H. E. et al. A single exercise bout augments adenovirus-specific T cell mobilization and function. Physiol. Behav. 194, 56–65 (2018).
Spielmann, G., Bollard, C. M., Kunz, H., Hanley, P. J. & Simpson, R. J. A single exercise bout enhances the manufacture of viral-specific T cells from healthy donors: implications for allogeneic adoptive transfer immunotherapy. Sci. Rep. 6, 25852 (2016).
Simpson, R. J., Bigley, A. B., Agha, N., Hanley, P. J. & Bollard, C. M. Mobilizing immune cells with exercise for cancer immunotherapy. Exerc. Sports Sci. Rev. 45, 163–172 (2017).
Rooney, B. V. et al. Lymphocytes and monocytes egress peripheral blood within minutes after cessation of steady state exercise: a detailed temporal analysis of leukocyte extravasation. Physiol. Behav. 194, 260–267 (2018).
Graff, R. M. et al. β2-Adrenergic receptor signaling mediates the preferential mobilization of differentiated subsets of CD8+ T cells, NK-cells and non-classical monocytes in response to acute exercise in humans. Brain Behav. Immun. 74, 143–153 (2018).
Pedersen, L. et al. Voluntary running suppresses tumor growth through epinephrine- and IL-6-dependent NK cell mobilization and redistribution. Cell Metab. 23, 554–562 (2016).
LaVoy, E. C. et al. A single bout of dynamic exercise enhances the expansion of MAGE-A4 and PRAME-specific cytotoxic T cells from healthy adults. Exerc. Immunol. Rev. 21, 144–153 (2015).
Dethlefsen, C. et al. Exercise-induced catecholamines activate the Hippo tumor suppressor pathway to reduce risks of breast cancer development. Cancer Res. 77, 4894–4904 (2017).
Spielmann, G. et al. The effects of age and latent cytomegalovirus infection on the redeployment of CD8+ T cell subsets in response to acute exercise in humans. Brain Behav. Immun. 39, 142–151 (2014).
O’Hara, N., Daul, A. E., Fesel, R., Siekmann, U. & Brodde, O. E. Different mechanisms underlying reduced β2-adrenoceptor responsiveness in lymphocytes from neonates and old subjects. Mech. Ageing Dev. 31, 115–122 (1985).
Bigley, A. B., Spielmann, G., Agha, N. & Simpson, R. J. The effects of age and latent cytomegalovirus infection on NK-cell phenotype and exercise responsiveness in man. Oxid. Med. Cell. Longev. 2015, 979645 (2015).
O’Toole, P. W. & Jeffery, I. B. Gut microbiota and aging. Science 350, 1214–1215 (2015).
An, R. et al. Age-dependent changes in GI physiology and microbiota: time to reconsider? Gut 67, 2213–2222 (2018).
Biagi, E. et al. Gut microbiota and extreme longevity. Curr. Biol. 26, 1480–1485 (2016).
Biagi, E. et al. Ageing and gut microbes: perspectives for health maintenance and longevity. Pharmacol. Res. 69, 11–20 (2013).
Claesson, M. J. et al. Gut microbiota composition correlates with diet and health in the elderly. Nature 488, 178–184 (2012).
Conley, M. N. et al. Aging and serum MCP-1 are associated with gut microbiome composition in a murine model. PeerJ 4, e1854 (2016).
Thevaranjan, N. et al. Age-associated microbial dysbiosis promotes intestinal permeability, systemic inflammation and macrophage dysfunction. Cell Host Microbe 21, 455–466 (2017).
Matsumoto, M. et al. Voluntary running exercise alters microbiota composition and increases n-butyrate concentration in the rat cecum. Biosci. Biotechnol. Biochem. 72, 572–576 (2008).
Clarke, S. F. et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 63, 1913–1920 (2014).
Allen, J. M. et al. Exercise alters gut microbiota composition and function in lean and obese humans. Med. Sci. Sports Exerc. 50, 747–757 (2018).
Long, J. E. et al. Vaccination response following aerobic exercise: can a brisk walk enhance antibody response to pneumococcal and influenza vaccinations? Brain Behav. Immun. 26, 680–687 (2012).
Ranadive, S. M. et al. Effect of acute aerobic exercise on vaccine efficacy in older adults. Med. Sci. Sports Exerc. 46, 455–461 (2014).
Poropatich, K., Fontanarosa, J., Samant, S., Sosman, J. A. & Zhang, B. Cancer immunotherapies: are they as effective in the elderly? Drugs Aging 34, 567–581 (2017).
Turner, J. E. & Brum, P. C. Does regular exercise counter T cell immunosenescence reducing the risk of developing cancer and promoting successful treatment of malignancies? Oxid. Med. Cell. Longev. 2017, 4234765 (2017).
Minnella, E. M. et al. Effect of exercise and nutrition prehabilitation on functional capacity in esophagogastric cancer surgery: a randomized clinical trial. JAMA Surg. 153, 1081–1089 (2018).
Gillis, C. et al. Prehabilitation versus rehabilitation: a randomized control trial in patients undergoing colorectal resection for cancer. Anesthesiology 121, 937–947 (2014).
Wood, W. A. et al. Cardiopulmonary fitness in patients undergoing hematopoietic SCT: a pilot study. Bone Marrow Transplant. 48, 1342–1349 (2013).
Agha, N. H. et al. Vigorous exercise mobilizes CD34+ hematopoietic stem cells to peripheral blood via the β2-adrenergic receptor. Brain Behav. Immun. 68, 66–75 (2018).
Baker, D. J. et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479, 232–236 (2011). The paper describes a progeroid mouse engineered to delete senescent cells as they arise, providing the first evidence that the accumulation of senescent cells with age is a driver of ageing and age-related disease.
Kirkland, J. L., Tchkonia, T., Zhu, Y., Niedernhofer, L. J. & Robbins, P. D. The clinical potential of senolytic drugs. J. Am. Geriatr. Soc. 65, 2297–2301 (2017).
Horvath, S. DNA methylation age of human tissues and cell types. Genome Biol. 14, R115 (2013).
Quach, A. et al. Epigenetic clock analysis of diet, exercise, education, and lifestyle factors. Aging 9, 419–446 (2017).
Gale, C. R. et al. The epigenetic clock and objectively measured sedentary and walking behavior in older adults: the Lothian Birth Cohort 1936. Clin. Epigenet. 10, 4 (2018).
Marioni, R. E. et al. The epigenetic clock is correlated with physical and cognitive fitness in the Lothian Birth Cohort 1936. Int. J. Epidemiol. 44, 1388–1396 (2015).
Lopez-Otin, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217 (2013).
Du, M. et al. Physical activity, sedentary behavior and leukocyte telomere length in women. Am. J. Epidemiol. 175, 414–422 (2012).
Denham, J., O’Brien, B. J. & Charchar, F. J. Telomere length maintenance and cardio-metabolic disease prevention through exercise training. Sports Med. 46, 1213–1237 (2016).
Ornish, D. et al. Effect of comprehensive lifestyle changes on telomerase activity and telomere length in men with biopsy-proven low-risk prostate cancer: 5-year follow-up of a descriptive pilot study. Lancet Oncol. 14, 1112–1120 (2013).
Friedenreich, C. M. et al. Effect of a 12-month exercise intervention on leukocyte telomere length: results from the ALPHA trial. Cancer Epidemiol. 56, 67–74 (2018).
Schafer, M. J. et al. Exercise prevents diet-induced cellular senescence in adipose tissue. Diabetes 65, 1606–1615 (2016).
He, C. et al. Exercise-induced BCL2-regulated autophagy is required for muscle glucose homeostasis. Nature 481, 511–515 (2012).
Akbar, A. N. & Fletcher, J. M. Memory T cell homeostasis and senescence during aging. Curr. Opin. Immunol. 17, 480–485 (2005).
Zhang, H., Puleston, D. J. & Simon, A. K. Autophagy in immune senescence. Trends Mol. Med. 22, 671–686 (2016).
Mannick, J. B. et al. TORC1 inhibition enhances immune function and reduces infections in the elderly. Sci. Transl Med. 10, eaaq1564 (2018). This is the first report of the use of drugs, rapamycin analogues, that target a core ageing process and improve functional immunity in older adults.
Drew, W. et al. Inflammation and neutrophil immunosenescence in health and disease: targeted treatment to improve clinical outcomes in the elderly. Exp. Gerontol. 105, 70–77 (2018).
Sapey, E. et al. Phosphoinositide 3 kinase inhibition restores neutrophil accuracy in the elderly: towards targeted treatments for immunesenescence. Blood 123, 239–248 (2014).
Qian, F. et al. Age-associated elevation in TLR5 leads to increased inflammatory responses in the elderly. Aging Cell 11, 104–110 (2012).
Hearps, A. C. et al. Aging is associated with chronic innate immune activation and dysregulation of monocyte phenotype and function. Aging Cell 11, 867–875 (2012).
Hazeldine, J. et al. Reduced release and binding of perforin at the immunological synapse underlies the age-related decline in natural killer cell cytotoxicity. Aging Cell 11, 751–759 (2012).
Peralbo, E. et al. Invariant NKT and NKT-like lymphocytes: two different T cell subsets that are differentially affected by ageing. Exp. Gerontol. 42, 703–708 (2007).
Agrawal, A. et al. Role of dendritic cells in inflammation and loss of tolerance in the elderly. Front. Immunol. 8, 896 (2017).
Chougnet, C. A. et al. Loss of phagocytic and antigen cross-presenting capacity in aging dendritic cells is associated with mitochondrial dysfunction. J. Immunol. 195, 2624–2632 (2015).
Majumdar, S. et al. Thymic atrophy: experimental studies and therapeutic interventions. Scand. J. Immunol. 87, 4–14 (2018).
Ventevogel, M. S. & Sempowski, G. D. Thymic rejuvenation and aging. Curr. Opin. Immunol. 25, 516–522 (2013).
Jergovic, M. et al. Intrinsic and extrinsic contributors to defective CD8+ T cell responses with aging. Exp. Gerontol. 105, 140–145 (2018).
Jagger, A., Shimojima, Y., Goronzy, J. J. & Weyand, C. M. Regulatory T cells and the immune aging process: a mini-review. Gerontology 60, 130–137 (2014).
Cancro, M. P. et al. B cells and aging: molecules and mechanisms. Trends Immunol. 30, 313–318 (2009).
Kogut, I. et al. B cell maintenance and function in aging. Semin. Immunol. 24, 342–349 (2012).
Acknowledgements
The authors thank M. Whitham and A. Skulas-Ray for their careful reading of the manuscript.
Reviewer information
Nature Reviews Immunology thanks M. Phillips, K. Timmerman and J. Turner for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.M.L., N.A.D., S.D.R.H., R.J.S. and G.N. were all involved in researching the data, discussing the content, and writing, reviewing and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Healthy life expectancy
-
Life expectancy is the predicted total number of years an individual is likely to live, and the proportion of life that will be spent in good health is termed the healthy life expectancy or health span.
- Sarcopenia
-
A condition of low muscle mass and function (strength) that commonly occurs with age or chronic illness. The European Working Group on Sarcopenia in Older People has defined low muscle mass as >2 standard deviations from the mean value for young adults, and low strength as a walking speed of less than 0.8 m s–1 and a hand grip strength of <30 kg in males or <20 kg in females.
- Inflammageing
-
The twofold to fourfold increase in systemic levels of inflammatory cytokines (for example, tumour necrosis factor, IL-1β and IL-6) and reduced levels of anti-inflammatory cytokines (for example, IL-10) seen with advanced age. The degree of inflammageing is associated with increased risk of a range of age-related diseases, including cardiovascular disease, osteoporosis, cancer and dementia.
- Senescence-associated secretory phenotype
-
(SASP). Senescent cells are classically proliferatively quiescent but highly active metabolically. They have a rich secretory output termed the SASP, which contains pro-inflammatory cytokines and chemokines, matrix metalloproteinases and growth factors, such as vascular endothelial growth factor. The SASP is thought to be a key mediator of the ageing process.
- VO2 max
-
The maximum rate of oxygen consumption measured during incremental exercise. The value is a measure of an individual’s cardiorespiratory fitness, as it represents the maximum rate at which the heart, lungs and muscles can use oxygen during exercise.
- M2-like macrophages
-
‘M1’ and ‘M2’ are classifications historically used to define macrophages activated in vitro as either pro-inflammatory (when ‘classically’ activated with interferon and lipopolysaccharide) or anti-inflammatory (when ‘alternatively’ activated with IL-4 or IL-10), respectively. However, in vivo macrophages are highly specialized, transcriptomically dynamic and extremely heterogeneous with regard to their phenotypes and functions, which are continuously shaped by their tissue microenvironment. Therefore, the M1 or M2 classification is too simplistic to describe the true nature of in vivo macrophages, although these terms are still often used to indicate whether the macrophages in question are more pro-inflammatory or anti-inflammatory.
- Biological mechanisms driving ageing
-
In many species, these mechanisms have been proposed to consist of various responses to cell and organelle damage. They include the accumulation of senescent cells, altered nutrient sensing and reduced mitochondrial fitness and stem cell function. Inflammation is one of the key downstream mediators, as senescent cells release pro-inflammatory cytokines.
Rights and permissions
About this article
Cite this article
Duggal, N.A., Niemiro, G., Harridge, S.D.R. et al. Can physical activity ameliorate immunosenescence and thereby reduce age-related multi-morbidity?. Nat Rev Immunol 19, 563–572 (2019). https://doi.org/10.1038/s41577-019-0177-9
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41577-019-0177-9
This article is cited by
-
Physical activity is associated with a lower risk of contracting and dying in infection and sepsis: a Swedish population-based cohort study
Critical Care (2024)
-
The ENJOY Seniors Exercise Park IMP-ACT project: IMProving older people’s health through physical ACTivity: a hybrid II implementation design study protocol
Archives of Public Health (2024)
-
Memory B cells
Nature Reviews Immunology (2024)
-
Association of Lifestyle Factors with Multimorbidity Risk in China: A National Representative Study
Applied Research in Quality of Life (2024)
-
Genetically predicted effects of physical activity and sedentary behavior on myasthenia gravis: evidence from mendelian randomization study
BMC Neurology (2023)