Abstract
The management of advanced-stage renal cell carcinoma (RCC) has been transformed by the development of immune-checkpoint inhibitors (ICIs). Nonetheless, most patients do not derive durable clinical benefit from these agents. Importantly, unlike other immunotherapy-responsive solid tumours, most RCCs have only a moderate mutational burden, and paradoxically, high levels of tumour CD8+ T cell infiltration are associated with a worse prognosis in patients with this disease. Building on the successes of antibodies targeting the PD-1 and CTLA4 immune checkpoints, multiple innovative immunotherapies are now in clinical development for the treatment of patients with RCC, including ICIs with novel targets, co-stimulatory pathway agonists, modified cytokines, metabolic pathway modulators, cell therapies and therapeutic vaccines. However, the successful development of such novel immune-based treatments and of immunotherapy-based combinations will require a disease-specific framework that incorporates a deep understanding of RCC immunobiology. In this Review, using the structure provided by the well-described cancer–immunity cycle, we outline the key steps required for a successful antitumour immune response in the context of RCC, and describe the development of promising new immunotherapies within the context of this framework. With this approach, we summarize and analyse the most encouraging targets of novel immune-based therapies within the RCC microenvironment, and review the landscape of emerging antigen-directed therapies for this disease.
Key points
-
The biology of renal cell carcinoma (RCC) differs substantially from that of other immunotherapy-responsive solid tumours; therefore, the successful development of novel immune treatments requires an understanding of disease-specific biology.
-
RCCs are highly infiltrated with CD8+ T cells and consequently many therapeutic approaches focus on reinvigorating T cells present in the tumour immune microenvironment.
-
Novel immune-checkpoint inhibitors, co-stimulatory pathway agonists, modified cytokine therapies and metabolic pathway modulators are all promising approaches that have the potential to remodel the RCC microenvironment and improve antitumour T cell responses.
-
Precision immunotherapies aim to target RCC-specific antigens, thereby ‘steering’ the immune response towards malignant cells.
-
Monoclonal antibodies, adoptive cell therapies and therapeutic vaccines are all promising approaches that act in an antigen-directed manner and might improve the efficacy of current immunotherapies in patients with RCC.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mckay, R. R., Boss, D. & Choueiri, T. K. Evolving systemic treatment landscape for patients with advanced renal cell carcinoma. J. Clin. Oncol. 36, 3615–3623 (2018).
Motzer, R. J. et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N. Engl. J. Med. 380, 1103–1115 (2019).
Rini, B. I. et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N. Engl. J. Med. 380, 1116–1127 (2019).
Motzer, R. J. et al. Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N. Engl. J. Med. 378, 1277–1290 (2018).
McDermott, D. F. et al. The high-dose aldesleukin “select” trial: a trial to prospectively validate predictive models of response to treatment in patients with metastatic renal cell carcinoma. Clin. Cancer Res. 21, 561–568 (2015).
Maruschke, M., Anastasiadis, A. G. & Hakenberg, O. W. Spontaneous regression of renal cell carcinoma: reality or myth? World J. Clin. Urol. 3, 201–208 (2014).
Janiszewska, A. D., Poletajew, S. & Wasiutyński, A. Spontaneous regression of renal cell carcinoma. Contemp. Oncol. 17, 123–127 (2013).
Takahashi, Y. et al. Regression of human kidney cancer following allogeneic stem cell transplantation is associated with recognition of an HERV-E antigen by T cells. J. Clin. Invest. 118, 1099–1109 (2008); correction 118, 1584 (2008).
Signoretti, S., Flaifel, A., Chen, Y. B. & Reuter, V. E. Renal cell carcinoma in the era of precision medicine: from molecular pathology to tissue-based biomarkers. J. Clin. Oncol. 36, 3553–3559 (2018).
Albiges, L., Flippot, R., Rioux-Leclercq, N. & Choueiri, T. K. Non-clear cell renal cell carcinomas: from shadow to light. J. Clin. Oncol. 36, 3624–3631 (2018).
Chen, D. S. & Mellman, I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 39, 1–10 (2013).
Vigneron, N. Human tumor antigens and cancer immunotherapy. Biomed. Res. Int. 2015, 948501 (2015).
Samstein, R. M. et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat. Genet. 51, 202–206 (2019).
FDA. FDA approves pembrolizumab for adults and children with TMB-H solid tumors https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-pembrolizumab-adults-and-children-tmb-h-solid-tumors (2020).
Yarchoan, M., Hopkins, A. & Jaffee, E. M. Tumor mutational burden and response rate to PD-1 inhibition. N. Engl. J. Med. 377, 2500–2501 (2017).
McDermott, D. F. et al. Clinical activity and molecular correlates of response to atezolizumab alone or in combination with bevacizumab versus sunitinib in renal cell carcinoma. Nat. Med. 24, 749–757 (2018).
Braun, D. A. et al. Interplay of somatic alterations and immune infiltration modulates response to PD-1 blockade in advanced clear cell renal cell carcinoma. Nat. Med. 26, 909–918 (2020).
Turajlic, S. et al. Insertion-and-deletion-derived tumour-specific neoantigens and the immunogenic phenotype: a pan-cancer analysis. Lancet Oncol. 18, 1009–1021 (2017).
Rooney, M. S., Shukla, S. A., Wu, C. J., Getz, G. & Hacohen, N. Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell. 160, 48–61 (2015).
SOGA, N. et al. Limited expression of cancer-testis antigens in renal cell carcinoma patients. Mol. Clin. Oncol. 1, 326–330 (2013).
Attermann, A. S., Bjerregaard, A. M., Saini, S. K., Grønbæk, K. & Hadrup, S. R. Human endogenous retroviruses and their implication for immunotherapeutics of cancer. Ann. Oncol. 29, 2183–2191 (2018).
Smith, C. C. et al. Endogenous retroviral signatures predict immunotherapy response in clear cell renal cell carcinoma. J. Clin. Invest. 128, 4804–4820 (2018).
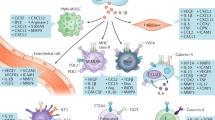
Giraldo, N. A. et al. Orchestration and prognostic significance of immune checkpoints in the microenvironment of primary and metastatic renal cell cancer. Clin. Cancer Res. 21, 3031–3040 (2015).
Jansen, C. S. et al. An intra-tumoral niche maintains and differentiates stem-like CD8 T cells. Nature 576, 465–470 (2019).
Ricketts, C. J. et al. The Cancer Genome Atlas comprehensive molecular characterization of renal cell carcinoma. Cell Rep. 23, 313–326.e5 (2018).
Thorsson, V. et al. The immune landscape of cancer. Immunity 48, 812–830.e14 (2018).
Creighton, C. J. et al. Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature 499, 43–49 (2013).
Iliopoulos, O., Levy, A. P., Jiang, C., Kaelin, W. G. & Goldberg, M. A. Negative regulation of hypoxia-inducible genes by the von Hippel-Lindau protein. Proc. Natl Acad. Sci. USA 93, 10595–10599 (1996).
Jubb, A. M. et al. Expression of vascular endothelial growth factor, hypoxia inducible factor 1α, and carbonic anhydrase IX in human tumours. J. Clin. Pathol. 57, 504–512 (2004).
Khan, K. A. & Kerbel, R. S. Improving immunotherapy outcomes with anti-angiogenic treatments and vice versa. Nat. Rev. Clin. Oncol. 15, 310–324 (2018).
Şenbabaoğlu, Y. et al. Tumor immune microenvironment characterization in clear cell renal cell carcinoma identifies prognostic and immunotherapeutically relevant messenger RNA signatures. Genome Biol. 17, 231 (2016).
Wang, T. et al. An empirical approach leveraging tumorgrafts to dissect the tumor microenvironment in renal cell carcinoma identifies missing link to prognostic inflammatory factors. Cancer Discov. 8, 1142–1155 (2018).
Clark, D. J. et al. Integrated proteogenomic characterization of clear cell renal cell carcinoma. Cell 179, 964–983.e31 (2019).
Yuen, K. C. et al. High systemic and tumor-associated IL8 correlates with reduced clinical benefit of PD-L1 blockade. Nat. Med. 26, 693–698 (2020).
Schalper, K. A. et al. Elevated serum interleukin 8 is associated with enhanced intratumor neutrophils and reduced clinical benefit with immune checkpoint inhibitors. Nat Med. 26, 688–692 (2020).
Bakouny, Z. & Choueiri, T. K. IL-8 and cancer prognosis on immunotherapy. Nat. Med. 26, 650–651 (2020).
Bakouny, Z. et al. Integrative molecular characterization of sarcomatoid and rhabdoid renal cell carcinoma reveals determinants of poor prognosis and response to immune checkpoint inhibitors. Nat. Commun. (in the press).
Rini, B. I. et al. Pembrolizumab (pembro) plus axitinib (axi) versus sunitinib as first-line therapy for metastatic renal cell carcinoma (mRCC): outcomes in the combined IMDC intermediate/poor risk and sarcomatoid subgroups of the phase 3 KEYNOTE-426 study [abstract]. J. Clin. Oncol. 37 (Suppl. 15), 4500 (2019).
McDermott, D. F. et al. CheckMate 214 post-hoc analyses of nivolumab plus ipilimumab or sunitinib in IMDC intermediate/poor-risk patients with previously untreated advanced renal cell carcinoma with sarcomatoid features [abstract]. J. Clin. Oncol. 37 (Suppl. 15), 4513 (2019).
Choueiri, T. K. et al. Efficacy and biomarker analysis of patients (pts) with advanced renal cell carcinoma (aRCC) with sarcomatoid histology (sRCC): subgroup analysis from the phase III JAVELIN renal 101 trial of first-line avelumab plus axitinib (A+Ax) vs sunitinib (S) [abstract 4823]. Ann. Oncol. 30 (Suppl. 5), v361 (2019).
Rini, B. I. et al. Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): a multicentre, open-label, phase 3, randomised controlled trial. Lancet 393, 2404–2415 (2019).
Rini, B. I. et al. Atezolizumab (atezo) + bevacizumab (bev) versus sunitinib (sun) in pts with untreated metastatic renal cell carcinoma (mRCC) and sarcomatoid (sarc) histology: IMmotion151 subgroup analysis [abstract]. J. Clin. Oncol. 37 (Suppl. 15), 4512 (2019).
Fridman, W. H., Zitvogel, L., Sautès-Fridman, C. & Kroemer, G. The immune contexture in cancer prognosis and treatment. Nat. Rev. Clin. Oncol. 14, 717–734 (2017).
Giraldo, N. A. et al. Tumor-infiltrating and peripheral blood T-cell immunophenotypes predict early relapse in localized clear cell renal cell carcinoma. Clin. Cancer Res. 23, 4416–4428 (2017).
Chevrier, S. et al. An immune atlas of clear cell renal cell carcinoma. Cell 169, 736–749.e18 (2017).
Pignon, J. C. et al. Irrecist for the evaluation of candidate biomarkers of response to nivolumab in metastatic clear cell renal cell carcinoma: analysis of a phase II prospective clinical trial. Clin. Cancer Res. 25, 2174–2184 (2019).
Siska, P. J. et al. Mitochondrial dysregulation and glycolytic insufficiency functionally impair CD8 T cells infiltrating human renal cell carcinoma. JCI Insight 2, e93411 (2017).
Beckermann, K. et al. Targeting metabolic dysregulation of T cells in kidney cancer [abstract]. J. Clin. Oncol. 38 (Suppl. 6), 722 (2020).
Miao, D. et al. Genomic correlates of response to immune checkpoint therapies in clear cell renal cell carcinoma. Science 359, 801–806 (2018).
Hegde, P. S., Karanikas, V. & Evers, S. The where, the when, and the how of immune monitoring for cancer immunotherapies in the era of checkpoint inhibition. Clin. Cancer Res. 22, 1865–1874 (2016).
Mariathasan, S. et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 554, 544–548 (2018).
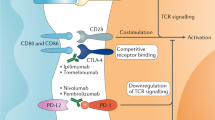
Marhelava, K., Pilch, Z., Bajor, M., Graczyk-Jarzynka, A. & Zagozdzon, R. Targeting negative and positive immune checkpoints with monoclonal antibodies in therapy of cancer. Cancers 11, 1756 (2019).
Granier, C. et al. Tim-3 expression on tumor-infiltrating PD-1+CD8+T cells correlates with poor clinical outcome in renal cell carcinoma. Cancer Res. 77, 1075–1082 (2017).
Cai, C. et al. Tim-3 expression represents dysfunctional tumor infiltrating T cells in renal cell carcinoma. World J. Urol. 34, 561–567 (2016).
Mach, N. et al. Phase (Ph) II study of MBG453 1 spartalizumab in patients (pts) with non-small cell lung cancer (NSCLC) and melanoma pretreated with anti-PD-1/L1 therapy [abstract 1202P]. Ann. Oncol. 30 (Suppl. 5), v491–v492 (2019).
Harding, J. J. et al. A phase Ia/Ib study of an anti-TIM-3 antibody (LY3321367) monotherapy or in combination with an anti-PD-L1 antibody (LY3300054): interim safety, efficacy, and pharmacokinetic findings in advanced cancers [abstract]. J. Clin. Oncol. 37 (Suppl. 8), 12 (2019).
Workman, C. J. & Vignali, D. A. A. The CD4-related molecule, LAG-3 (CD223), regulates the expansion of activated T cells. Eur. J. Immunol. 33, 970–979 (2003).
Andreae, S., Buisson, S. & Triebel, F. MHC class II signal transduction in human dendritic cells induced by a natural ligand, the LAG-3 protein (CD223). Blood 102, 2130–2137 (2003).
Zelba, H. et al. PD-1 and LAG-3 dominate checkpoint receptor-mediated T-cell inhibition in renal cell carcinoma. Cancer Immunol. Res. 7, 1891–1899 (2019).
Brignone, C., Escudier, B., Grygar, C., Marcu, M. & Triebel, F. A phase I pharmacokinetic and biological correlative study of IMP321, a novel MHC class II agonist, in patients with advanced renal cell carcinoma. Clin. Cancer Res. 15, 6225–6231 (2009).
Bendell J. C., et al. Phase Ia/Ib dose-escalation study of the anti-TIGIT antibody tiragolumab as a single agent and in combination with atezolizumab in patients with advanced solid tumors [abstract CT302]. Presented at the American Association for Cancer Research Virtual Annual Meeting, 2020. abstractsonline.com https://www.abstractsonline.com/pp8/#!/9045/presentation/11341 (2020).
Rodriguez-Abreu, D. et al. Primary analysis of a randomized, double-blind, phase II study of the anti-TIGIT antibody tiragolumab (tira) plus atezolizumab (atezo) versus placebo plus atezo as first-line (1L) treatment in patients with PD-L1-selected NSCLC (CITYSCAPE) [abstract]. J. Clin. Oncol. 38 (Suppl. 15), 9503 (2020).
Powderly, J. et al. CA-170, a first in class oral small molecule dual inhibitor of immune checkpoints PD-L1 and VISTA, demonstrates tumor growth inhibition in pre-clinical models and promotes T cell activation in phase 1 study [abstract 1141PD]. Ann. Oncol. 28 (Suppl. 5), v405–v406 (2017).
Chester, C., Sanmamed, M. F., Wang, J. & Melero, I. Immunotherapy targeting 4-1BB: mechanistic rationale, clinical results, and future strategies. Blood 131, 49–57 (2018).
Palazón, A. et al. The HIF-1α hypoxia response in tumor-infiltrating T lymphocytes induces functional CD137 (4-1BB) for immunotherapy. Cancer Discov. 2, 608–623 (2012).
Tolcher, A. W. et al. Phase Ib study of utomilumab (PF-05082566), a 4-1BB/CD137 agonist, in combination with pembrolizumab (MK-3475) in patients with advanced solid tumors. Clin. Cancer Res. 23, 5349–5357 (2017).
Alves Costa Silva, C., Facchinetti, F., Routy, B. & Derosa, L. New pathways in immune stimulation: targeting OX40. ESMO Open 5, e000573 (2020).
Glisson, B. S. et al. Phase 1 study of MEDI0562, a humanized OX40 agonist monoclonal antibody (mAb), in adult patients (pts) with advanced solid tumors [abstract 1052PD]. Ann. Oncol. 27 (Suppl. 6), vi361 (2016).
Curti, B. D. et al. OX40 is a potent immune-stimulating target in late-stage cancer patients. Cancer Res. 73, 7189–7198 (2013).
El-Khoueiry, A. B. et al. The relationship of pharmacodynamics (PD) and pharmacokinetics (PK) to clinical outcomes in a phase I study of OX40 agonistic monoclonal antibody (mAb) PF-04518600 (PF-8600) [abstract]. J. Clin. Oncol. 35 (Suppl. 15), 3027 (2017).
Spira, A. et al. Safety, tolerability, and pharmacokinetics of the OX40 agonist ABBV-368 in patients with advanced solid tumors [abstract 1149P]. Ann. Oncol. 29 (Suppl. 8), viii409 (2018).
Klapper, J. A. et al. High-dose interleukin-2 for the treatment of metastatic renal cell carcinoma: a retrospective analysis of response and survival in patients treated in the surgery branch at the National Cancer Institute between 1986 and 2006. Cancer 113, 293–301 (2008).
Charych, D. H. et al. NKTR-214, an engineered cytokine with biased IL2 receptor binding, increased tumor exposure, and marked efficacy in mouse tumor models. Clin. Cancer Res. 22, 680–690 (2016).
Diab, A. et al. NKTR-214 (CD122-biased agonist) plus nivolumab in patients with advanced solid tumors: preliminary phase 1/2 results of PIVOT [abstract]. J. Clin. Oncol. 36 (Suppl. 15), 3006 (2018).
Tugues, S. et al. New insights into IL-12-mediated tumor suppression. Cell Death Differ. 22, 237–246 (2015).
Fallon, J. et al. The immunocytokine NHS-IL12 as a potential cancer therapeutic. Oncotarget 5, 1869–1884 (2014).
Strauss, J. et al. First-in-human phase I trial of a tumor-targeted cytokine (NHS-IL12) in subjects with metastatic solid tumors. Clin. Cancer Res. 25, 99–109 (2019).
Strauss, J. et al. Phase Ib, open-label, dose-escalation study of M9241 (NHS-IL12) plus avelumab in patients (pts) with advanced solid tumours [abstract 1224P]. Ann. Oncol. 30 (Suppl. 5), v500–v501 (2019).
McDermott, D. F. et al. Safety and efficacy of the oral CXCR4 inhibitor X4P-001 + axitinib in advanced renal cell carcinoma patients: an analysis of subgroup responses by prior treatment [abstract 1186PD]. Ann. Oncol. 30 (Suppl. 5), v482–v483 (2019).
Badawy, A. A. B. Kynurenine pathway of tryptophan metabolism: regulatory and functional aspects. Int. J. Tryptophan. Res. 10, 1178646917691938 (2017).
Eleftheriadis, T., Pissas, G., Antoniadi, G., Liakopoulos, V. & Stefanidis, I. Indoleamine 2,3-dioxygenase depletes tryptophan, activates general control non-derepressible 2 kinase and down-regulates key enzymes involved in fatty acid synthesis in primary human CD4+ T cells. Immunology 146, 292–300 (2015).
Labadie, B. W., Bao, R. & Luke, J. J. Reimagining IDO pathway inhibition in cancer immunotherapy via downstream focus on the tryptophan-kynurenine-aryl hydrocarbon axis. Clin. Cancer Res. 25, 1462–1471 (2019).
Gandhi, R. et al. Activation of the aryl hydrocarbon receptor induces human type 1 regulatory T cell-like and Foxp3(+) regulatory T cells. Nat. Immunol. 11, 846–853 (2010).
Li, H. et al. Metabolomic adaptations and correlates of survival to immune checkpoint blockade. Nat. Commun. 10, 1–6 (2019).
Lara, P. et al. Epacadostat plus pembrolizumab in patients with advanced RCC: preliminary phase I/II results from ECHO-202/KEYNOTE-037 [abstract]. J. Clin. Oncol. 35 (Suppl. 15), 4515 (2017).
Long, G. V. et al. Epacadostat plus pembrolizumab versus placebo plus pembrolizumab in patients with unresectable or metastatic melanoma (ECHO-301/KEYNOTE-252): a phase 3, randomised, double-blind study. Lancet Oncol. 20, 1083–1097 (2019).
Kroemer, G., Galluzzi, L., Kepp, O. & Zitvogel, L. Immunogenic cell death in cancer therapy. Annu. Rev. Immunol. 31, 51–72 (2013).
Allard, B., Longhi, M. S., Robson, S. C. & Stagg, J. The ectonucleotidases CD39 and CD73: novel checkpoint inhibitor targets. Immunol. Rev. 276, 121–144 (2017).
Allard, D., Turcotte, M. & Stagg, J. Targeting A2 adenosine receptors in cancer. Immunol. Cell Biol. 95, 333–339 (2017).
Leone, R. D. & Emens, L. A. Targeting adenosine for cancer immunotherapy. J. Immunother. Cancer 6, 57 (2018).
Song, L. et al. Ecto-5′-nucleotidase (CD73) is a biomarker for clear cell renal carcinoma stem-like cells. Oncotarget 8, 31977–31992 (2017).
Giannakis, M. et al. Metabolomic correlates of response in nivolumab-treated renal cell carcinoma and melanoma patients [abstract]. J. Clin. Oncol. 35 (Suppl. 15), 3036 (2017).
Fong, L. et al. Adenosine 2A receptor blockade as an immunotherapy for treatment-refractory renal cell cancer. Cancer Discov. 10, 40–53 (2020).
Hirsch, L., Flippot, R., Escudier, B. & Albiges, L. Immunomodulatory roles of VEGF pathway inhibitors in renal cell carcinoma. Drugs 80, 1169–1181 (2020).
Plimack, E. R. et al. Pembrolizumab plus axitinib versus sunitinib as first-line therapy for advanced renal cell carcinoma (RCC): updated analysis of KEYNOTE-426 [abstract]. J. Clin. Oncol. 38 (Suppl. 15), 5001 (2020).
Palmer, A. C. & Sorger, P. K. Combination cancer therapy can confer benefit via patient-to-patient variability without drug additivity or synergy. Cell 171, 1678–1691.e13 (2017).
Kwak, E. L., Clark, J. W. & Chabner, B. Targeted agents: the rules of combination. Clin. Cancer Res. 13, 5232–5237 (2007).
Zappasodi, R., Merghoub, T. & Wolchok, J. D. Emerging concepts for immune checkpoint blockade-based combination therapies. Cancer Cell 33, 581–598 (2018).
Hammers, H. J. et al. Combination of dual immune checkpoint inhibition (ICI) with stereotactic radiation (SBRT) in metastatic renal cell carcinoma (mRCC) (RADVAX RCC) [abstract]. J. Clin. Oncol. 38 (Suppl. 6), 614 (2020).
Masini, C. et al. Nivolumab (NIVO) in combination with stereotactic body radiotherapy (SBRT) in pretreated patients (pts) with metastatic renal cell carcinoma (mRCC): first results of phase II NIVES study [abstract]. J. Clin. Oncol. 38 (Suppl. 6), 613 (2020).
Jonasch, E. et al. A first-in-human phase 1/2 trial of the oral HIF-2a inhibitor PT2977 in patients with advanced RCC [abstract 911PD]. Ann. Oncol. 30 (Suppl. 5), v361–v362 (2019).
Choueiri, T. K. & Kaelin, W. G. Targeting the HIF2–VEGF axis in renal cell carcinoma. Nat. Med. 26, 1519–1530 (2020).
Reynolds, K., Thomas, M. & Dougan, M. Diagnosis and management of hepatitis in patients on checkpoint blockade. Oncologist 23, 991–997 (2018).
Braun, D. A. & Wu, C. J. Antigen discovery and therapeutic targeting in hematologic malignancies. Cancer J. 23, 115–124 (2017).
Ott, P. A. & Wu, C. J. Cancer vaccines: steering T cells down the right path to eradicate tumors. Cancer Discov. 9, 476–481 (2019).
Maude, S. L., Teachey, D. T., Porter, D. L. & Grupp, S. A. CD19-targeted chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Blood 125, 4017–4023 (2015).
D’Agostino, M. & Raje, N. Anti-BCMA CAR T-cell therapy in multiple myeloma: can we do better? Leukemia 34, 21–34 (2020).
Slamon, D. J. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 344, 783–792 (2001).
Martinez, M. & Moon, E. K. CAR T cells for solid tumors: new strategies for finding, infiltrating, and surviving in the tumor microenvironment. Front Immunol. 10, 128 (2019).
Kaluz, S., Kaluzová, M., Liao, S. Y., Lerman, M. & Stanbridge, E. J. Transcriptional control of the tumor- and hypoxia-marker carbonic anhydrase 9: a one transcription factor (HIF-1) show? Biochim. Biophys. Acta 1795, 162–172 (2009).
Tostain, J., Li, G., Gentil-Perret, A. & Gigante, M. Carbonic anhydrase 9 in clear cell renal cell carcinoma: a marker for diagnosis, prognosis and treatment. Eur. J. Cancer 46, 3141–3148 (2010).
Lamers, C. H. J. et al. Treatment of metastatic renal cell carcinoma with autologous T-lymphocytes genetically retargeted against carbonic anhydrase IX: first clinical experience. J. Clin. Oncol. 24, e20–e22 (2006).
Lamers, C. H. J. et al. Treatment of metastatic renal cell carcinoma with CAIX CAR-engineered T cells: clinical evaluation and management of on-target toxicity. Mol. Ther. 21, 904–912 (2013).
Lamers, C. H. J., Klaver, Y., Gratama, J. W., Sleijfer, S. & Debets, R. Treatment of metastatic renal cell carcinoma (mRCC) with CAIX CAR-engineered T-cells – a completed study overview. Biochem. Soc. Trans. 44, 951–959 (2016).
Wang, Q. J. et al. Preclinical evaluation of chimeric antigen receptors targeting CD70-expressing cancers. Clin. Cancer Res. 23, 2267–2276 (2017).
Roybal, K. T. et al. Precision tumor recognition by T cells with combinatorial antigen-sensing circuits. Cell. 164, 770–779 (2016).
Diaconu, I. et al. Inducible caspase-9 selectively modulates the toxicities of CD19-specific chimeric antigen receptor-modified T cells. Mol. Ther. 25, 580–592 (2017).
Fraietta, J. A. et al. Disruption of TET2 promotes the therapeutic efficacy of CD19-targeted T cells. Nature 558, 307–312 (2018).
Weber, E. W., Maus, M. V. & Mackall, C. L. The emerging landscape of immune cell therapies. Cell 181, 46–62 (2020).
Li, H. et al. CAIX-specific CAR-T cells and sunitinib show synergistic effects against metastatic renal cancer models. J. Immunother. 43, 16–28 (2020).
Loustau, M. First CAR-T cell immunotherapy against HLA-G: targeting a unique ICP and TAA. Ann. Oncol. 30 (Suppl. 11), xi12 (2019).
Wang, Y. et al. Design and activity of 2nd generation, dual-targeted CAR T cell factories against ccRCC [abstract]. Cancer Res. 79 (Suppl. 13), 3179 (2019).
Suarez, E. R. et al. Chimeric antigen receptor T cells secreting anti-PD-L1 antibodies more effectively regress renal cell carcinoma in a humanized mouse model. Oncotarget. 7, 34341–34355 (2016).
Scott, A. M., Allison, J. P. & Wolchok, J. D. Monoclonal antibodies in cancer therapy. Cancer Immun. 12, 14 (2012).
Chamie, K. et al. Adjuvant weekly girentuximab following nephrectomy for high-risk renal cell carcinoma: the ARISER randomized clinical trial. JAMA Oncol. 3, 913–920 (2017).
Diamantis, N. & Banerji, U. Antibody-drug conjugates – an emerging class of cancer treatment. Br. J. Cancer 114, 362–367 (2016).
Muselaers, C. H. J. et al. Phase 2 study of lutetium 177-labeled anti-carbonic anhydrase IX monoclonal antibody girentuximab in patients with advanced renal cell carcinoma. Eur. Urol. 69, 767–770 (2016).
Coats, S. et al. Antibody-drug conjugates: future directions in clinical and translational strategies to improve the therapeutic index. Clin. Cancer Res. 25, 5441–5448 (2019).
Pal, S. K. et al. A phase 1 trial of SGN-CD70A in patients with CD70-positive, metastatic renal cell carcinoma. Cancer 125, 1124–1132 (2019).
McGregor, B. A. et al. Safety and efficacy of CDX-014, an antibody-drug conjugate directed against T cell immunoglobulin mucin-1 in advanced renal cell carcinoma. Invest. New Drugs 38, 1807–1814 (2020).
Thompson, J. A. et al. Phase I trials of anti-ENPP3 antibody–drug conjugates in advanced refractory renal cell carcinomas. Clin. Cancer Res. 24, 4399–4406 (2018).
Kollmannsberger, C. et al. A randomized phase II study of AGS-16C3F versus axitinib in previously treated patients with metastatic renal cell carcinoma. Oncologist https://doi.org/10.1002/onco.13628 (2021).
Rosenberg, S. A. et al. Observations on the systemic administration of autologous lymphokine-activated killer cells and recombinant interleukin-2 to patients with metastatic cancer. N. Engl. J. Med. 313, 1485–1492 (1985).
Dudley, M. E. et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 298, 850–854 (2002).
Rosenberg, S. A. et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin. Cancer Res. 17, 4550–4557 (2011).
Sarnaik, A. et al. Long-term follow up of lifileucel (LN-144) cryopreserved autologous tumor infiltrating lymphocyte therapy in patients with advanced melanoma progressed on multiple prior therapies [abstract]. J. Clin. Oncol. 38 (Suppl. 15), 10006 (2020).
Figlin, R. A. et al. Treatment of metastatic renal cell carcinoma with nephrectomy, interleukin-2 and cytokine-primed or CD8(+) selected tumor infiltrating lymphocytes from primary tumor. J. Urol. 158, 740–745 (1997).
Figlin, R. A. et al. Multicenter, randomized, phase III trial of CD8+ tumor-infiltrating lymphocytes in combination with recombinant interleukin-2 in metastatic renal cell carcinoma. J. Clin. Oncol. 17, 2521–2529 (1999).
Andersen, R. et al. T-cell responses in the microenvironment of primary renal cell carcinoma-implications for adoptive cell therapy. Cancer Immunol. Res. 6, 222–235 (2018).
Childs, R. et al. Regression of metastatic renal-cell carcinoma after nonmyeloablative allogeneic peripheral-blood stem-cell transplantation. N. Engl. J. Med. 343, 750–758 (2000).
Cherkasova, E. et al. Detection of an immunogenic HERV-E envelope with selective expression in clear cell kidney cancer. Cancer Res. 76, 2177–2185 (2016).
Morgan, R. A. et al. Cancer regression and neurological toxicity following anti-MAGE-A3 TCR gene therapy. J. Immunother. 36, 133–151 (2013).
Linette, G. P. et al. Cardiovascular toxicity and titin cross-reactivity of affinity-enhanced T cells in myeloma and melanoma. Blood 122, 863–871 (2013).
Kula, T. et al. T-scan: a genome-wide method for the systematic discovery of T cell epitopes. Cell 178, 1016–1028.e13 (2019).
Walter, S. et al. Multipeptide immune response to cancer vaccine IMA901 after single-dose cyclophosphamide associates with longer patient survival. Nat. Med. 18, 1254–1261 (2012).
Kirner, A., Mayer-Mokler, A. & Reinhardt, C. IMA901: a multi-peptide cancer vaccine for treatment of renal cell cancer. Hum. Vaccines Immunother. 10, 3179–3189 (2014).
Rini, B. I. et al. IMA901, a multipeptide cancer vaccine, plus sunitinib versus sunitinib alone, as first-line therapy for advanced or metastatic renal cell carcinoma (IMPRINT): a multicentre, open-label, randomised, controlled, phase 3 trial. Lancet Oncol. 17, 1599–1611 (2016).
Rausch, S. et al. Results of a phase 1/2 study in metastatic renal cell carcinoma patients treated with a patient-specific adjuvant multi-peptide vaccine after resection of metastases. Eur. Urol. Focus. 5, 604–607 (2019).
Figlin, R. A. et al. Results of the ADAPT phase 3 study of rocapuldencel-T in combination with sunitinib as first-line therapy in patients with metastatic renal cell carcinoma. Clin. Cancer Res. 26, 2327–2335 (2020).
Sarkizova, S. et al. A large peptidome dataset improves HLA class I epitope prediction across most of the human population. Nat. Biotechnol. 38, 199–209 (2020).
Ott, P. A. et al. An immunogenic personal neoantigen vaccine for patients with melanoma. Nature 547, 217–221 (2017).
Sahin, U. et al. Personalized RNA mutanome vaccines mobilize poly-specific therapeutic immunity against cancer. Nature 547, 222–226 (2017).
Keskin, D. B. et al. Neoantigen vaccine generates intratumoral T cell responses in phase Ib glioblastoma trial. Nature 565, 234–239 (2019).
Hilf, N. et al. Actively personalized vaccination trial for newly diagnosed glioblastoma. Nature 565, 240–245 (2019).
Lopez, J. S. et al. A phase Ib study to evaluate RO7198457, an individualized neoantigen specific immunotherapy (iNeST), in combination with atezolizumab in patients with locally advanced or metastatic solid tumors [abstract]. Cancer Res. 80 (Suppl. 16), CT301 (2020).
Verma, V. et al. PD-1 blockade in subprimed CD8 cells induces dysfunctional PD-1+ CD38hi cells and anti-PD-1 resistance. Nat. Immunol. 20, 1231–1243 (2019).
Pauken, K. E. et al. The PD-1 pathway regulates development and function of memory CD8+T cells following respiratory viral infection. Cell Rep. 31, 107827 (2020).
Hu, Z. et al. A cloning and expression system to probe T-cell receptor specificity and assess functional avidity to neoantigens. Blood 132, 1911–1921 (2018).
Grünwald, V. et al. Depth of remission is a prognostic factor for survival in patients with metastatic renal cell carcinoma. Eur. Urol. 67, 952–958 (2015).
Voss, M. H. et al. Depth of response (DepOR) analysis and correlation with clinical outcomes from JAVELIN Renal 101. J. Clin. Oncol. 38, 690–690 (2020).
Motzer, R. J. et al. Survival outcomes and independent response assessment with nivolumab plus ipilimumab versus sunitinib in patients with advanced renal cell carcinoma: 42-month follow-up of a randomized phase 3 clinical trial. J. Immunother. Cancer 8, 891 (2020).
Grünwald, V. et al. Association between depth of response and overall survival: exploratory analysis in patients with previously untreated advanced renal cell carcinoma (aRCC) in CheckMate 214 [abstract 950P]. Ann. Oncol. 30 (Suppl. 5), v382–v383 (2019).
Ravi, P. et al. Evaluation of the safety and efficacy of immunotherapy rechallenge in patients with renal cell carcinoma. JAMA Oncol. 6, 1606–1610 (2020).
Gul, A. et al. Salvage ipilimumab and nivolumab in patients with metastatic renal cell carcinoma after prior immune checkpoint inhibitors. J. Clin. Oncol. 38, 3088–3094 (2020).
Regan, M. M. et al. Treatment-free survival: a novel outcome measure of the effects of immune checkpoint inhibition — a pooled analysis of patients with advanced melanoma. J. Clin. Oncol. 37, 3350–3358 (2019).
Regan, M. M. et al. Treatment-free survival, with and without toxicity, as a novel outcome applied to immuno-oncology agents in advanced renal cell carcinoma [abstract 971P]. Ann. Oncol. 30 (Suppl. 5), v393–v394 (2019).
Acknowledgements
We acknowledge W. Kaelin for helpful discussion and comments. D.A.B. acknowledges support from the DF/HCC Kidney Cancer SPORE Career Enhancement Program (P50CA101942-15), the US Department of Defense (DOD) congressionally directed medical research programs (CDMRP) (KC170216 and KC190130) and the DOD Academy of Kidney Cancer Investigators (KC190128). L.H. is supported by the Fondation de France during her postdoctoral research fellowship at Dana-Farber Cancer Institute. E.M.V.A. acknowledges support from the US NIH (NCI-R01-CA227388 and U01-CA233100). C.J.W. is a Scholar of the Leukemia and Lymphoma Society, and is supported in part by the Parker Institute for Cancer Immunotherapy. C.J.W. acknowledges support from the US NIH (NCI-1RO1CA155010 and NIH/NCI U24 CA224331), and The G. Harold and Leila Y. Mathers Foundation. T.K.C. is supported in part by the Dana-Farber/Harvard Cancer Center Kidney SPORE (P50CA101942) and Cancer Center Support Grant (P30CA006516), the Kohlberg Chair at Harvard Medical School, the Trust Family, Michael Brigham, and Loker Pinard Funds for Kidney Cancer Research at DFCI, and various US National Cancer Institute (NCI), DOD, Research Foundations and industry grants.
Author information
Authors and Affiliations
Contributions
All authors made substantial contributions to all aspects of the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
D.A.B. has acted as a consultant of Adept Field Solutions, Blueprint Partnerships, Charles River Associates, Dedham Group, Defined Health, Insight Strategy, Octane Global, Slingshot Insights and Trinity Group, has received travel support from Bristol Myers Squibb and has received honoraria from LM Education/Exchange Services. Z.B. has received research support from Bristol Myers Squibb and Genentech. E.M.V.A. has served on the advisory boards of Enara Bio, Genome Medical, Manifold Bio, and Monte Rosa, has acted as a consultant of Invitae, Janssen and Tango Therapeutics, has received research funding from Bristol Myers Squibb-IION and Novartis, holds equity in Enara Bio, Genome Medical, Manifold Bio, Microsoft, Monte Rosa, Syapse and Tango Therapeutics, has received travel support from Roche/Genentech, and is listed on several institutional patents filed on chromatin mutations and immunotherapy response, and methods for clinical interpretation. C.J.W. holds equity in BioNtech. T.K.C. has acted as a consultant of AstraZeneca, Bristol Myers Squibb, Corvus, Eisai, Exelixis, Genentech, GlaxoSmithKline, Ipsen, Merck, Novartis, Peloton, Pfizer, Prometheus Labs, Roche, Surface Oncology, Tracon and Up-to-Date, and has received research funding from Astellas, AstraZeneca, Bristol Myers Squibb, Corvus, Exelixis, Genentech, GlaxoSmithKline, Merck, Novartis, Peloton, Pfizer and Tracon. The other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewer recognition statement
Nature Reviews Clinical Oncology thanks J. Bedke, R. Figlin, and C. Porta for their contribution to the peer review of this work.
Supplementary information
Rights and permissions
About this article
Cite this article
Braun, D.A., Bakouny, Z., Hirsch, L. et al. Beyond conventional immune-checkpoint inhibition — novel immunotherapies for renal cell carcinoma. Nat Rev Clin Oncol 18, 199–214 (2021). https://doi.org/10.1038/s41571-020-00455-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-020-00455-z
This article is cited by
-
Multi-omics and immunogenomics analysis revealed PFKFB3 as a targetable hallmark and mediates sunitinib resistance in papillary renal cell carcinoma: in silico study with laboratory verification
European Journal of Medical Research (2024)
-
Single-cell analysis reveals ADGRL4+ renal tubule cells as a highly aggressive cell type in clear cell renal cell carcinoma
Scientific Reports (2024)
-
STING agonist, SMA-2, inhibits clear cell renal cell carcinoma through improving tumor microenvironment
Molecular and Cellular Biochemistry (2024)
-
Exosomes in renal cell carcinoma: challenges and opportunities
Molecular Biology Reports (2024)
-
Circular RNA AGAP1 Stimulates Immune Escape and Distant Metastasis in Renal Cell Carcinoma
Molecular Biotechnology (2024)