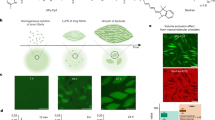
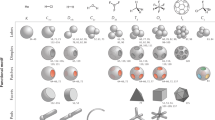
Abstract
Colloid science has recently grown substantially owing to the innovative use of silane coupling agents (SCAs), especially 3-trimethoxysilylpropyl methacrylate (TPM). SCAs were previously used mainly as modifying agents, but their ability to form droplets and condense onto pre-existing structures has enabled their use as a versatile and powerful tool to create novel anisotropic colloids with increasing complexity. In this Review, we highlight the advances in complex colloid synthesis facilitated by the use of TPM and show how this has driven remarkable new applications. The focus is on TPM as the current state-of-the-art in colloid science, but we also discuss other silanes and their potential to make an impact. We outline the remarkable properties of TPM colloids and their synthesis strategies, and discuss areas of soft matter science that have benefited from TPM and other SCAs.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Hueckel, T., Hocky, G. M. & Sacanna, S. Total synthesis of colloidal matter. Nat. Rev. Mater. 6, 1053–1069 (2021).
Kim, Y.-J., Moon, J.-B., Hwang, H., Kim, Y. S. & Yi, G.-R. Advances in colloidal building blocks: toward patchy colloidal clusters. Adv. Mater. 35, 2203045 (2023).
Jin, X., Yuan, X., Chen, K., Xie, H. & Chen, C. Role of 3-methacryloxypropyltrimethoxysilane in dentin bonding. ACS Omega 7, 15892–15900 (2022).
Marsden, J. G. in Handbook of Adhesives (ed. Skeist, I.) 536–548 (Springer, 1990).
Badley, R. D., Ford, W. T., McEnroe, F. J. & Assink, R. A. Surface modification of colloidal silica. Langmuir 6, 792–801 (1990).
van Blaaderen, A. & Vrij, A. Synthesis and characterization of monodisperse colloidal organo-silica spheres. J. Colloid Interface Sci. 156, 1–18 (1993).
Gellermann, C., Storch, W. & Wolter, H. Synthesis and characterization of the organic surface modifications of monodisperse colloidal silica. J. Solgel Sci. Technol. 8, 173–176 (1997).
Philipse, A. P. & Vrij, A. Preparation and properties of nonaqueous model dispersions of chemically modified, charged silica spheres. J. Colloid Interface Sci. 128, 121–136 (1989).
Liu, B. et al. Switching plastic crystals of colloidal rods with electric fields. Nat. Commun. 5, 3092 (2014).
Van Helden, A. K., Jansen, J. W. & Vrij, A. Preparation and characterization of spherical monodisperse silica dispersions in nonaqueous solvents. J. Colloid Interface Sci. 81, 354–368 (1981).
van Blaaderen, A., Peetermans, J., Maret, G. & Dhont, J. K. G. Long‐time self‐diffusion of spherical colloidal particles measured with fluorescence recovery after photobleaching. J. Chem. Phys. 96, 4591–4603 (1992).
Bagwe, R. P., Hilliard, L. R. & Tan, W. Surface modification of silica nanoparticles to reduce aggregation and non-specific binding. Langmuir 22, 4357–4362 (2006).
Stöber, W., Fink, A. & Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci. 26, 62–69 (1968).
Giesche, H. Synthesis of monodispersed silica powders I. Particle properties and reaction kinetics. J. Eur. Ceram. Soc. 14, 189–204 (1994).
Giesche, H. Synthesis of monodispersed silica powders II. Controlled growth reaction and continuous production process. J. Eur. Ceram. Soc. 14, 205–214 (1994).
Bogush, G. H., Tracy, M. A. & Zukoski, C. F. Preparation of monodisperse silica particles: control of size and mass fraction. J. Non-Cryst. Solids 104, 95–106 (1988).
Ghimire, P. P. & Jaroniec, M. Renaissance of Stöber method for synthesis of colloidal particles: new developments and opportunities. J. Colloid Interface Sci. 584, 838–865 (2021).
Jungmann, N., Schmidt, M. & Maskos, M. Characterization of polyorganosiloxane nanoparticles in aqueous dispersion by asymmetrical flow field-flow fractionation. Macromolecules 34, 8347–8353 (2001).
Ma, C. & Kimura, Y. Preparation of nano-particles of poly(phenylsilsesquioxane)s by emulsion polycondensation of phenylsilanetriol formed in aqueous solution. Polym. J. 34, 709–713 (2002).
Ma, C., Taniguchi, I., Miyamoto, M. & Kimura, Y. Formation of stable nanoparticles of poly(phenyl/methylsilsesquioxane) in aqueous solution. Polym. J. 35, 270–275 (2003).
Bronstein, L. M. et al. Controlled synthesis of novel metalated poly(aminohexyl)-(aminopropyl)silsesquioxane colloids. Langmuir 19, 7071–7083 (2003).
Nakamura, M. & Ishimura, K. One-pot synthesis and characterization of three kinds of thiol–organosilica nanoparticles. Langmuir 24, 5099–5108 (2008).
Hah, H. J., Kim, J. S., Jeon, B. J., Koo, S. M. & Lee, Y. E. Simple preparation of monodisperse hollow silica particles without using templates. Chem. Commun. 34, 1712–1713 (2003).
Wang, Q., Liu, Y. & Yan, H. Mechanism of a self-templating synthesis of monodispersed hollow silica nanospheres with tunable size and shell thickness. Chem. Commun. 21, 2339–2341 (2007).
Segers, M., Arfsten, N., Buskens, P. & Möller, M. A facile route for the synthesis of sub-micron sized hollow and multiporous organosilica spheres. RSC Adv. 4, 20673–20676 (2014).
Segers, M., Sliepen, M., Kraft, D. J., Möller, M. & Buskens, P. Synthesis of sub-micron sized hollow, and nanoporous phenylsiloxane spheres through use of phenyltrimethoxysilane as surfmer: insights into the surfactant and factors influencing the particle architecture. Colloids Surf. A Physicochem. Eng. Asp. 497, 378–384 (2016).
Mori, H. Design and synthesis of functional silsesquioxane-based hybrids by hydrolytic condensation of bulky triethoxysilanes. Int. J. Polym. Sci. 2012, 173624 (2012).
Miller, C. R. et al. Functionalized organosilica microspheres via a novel emulsion-based route. Langmuir 21, 9733–9740 (2005).
Lee, Y.-G., Park, J.-H., Oh, C., Oh, S.-G. & Kim, Y. C. Preparation of highly monodispersed hybrid silica spheres using a one-step sol–gel reaction in aqueous solution. Langmuir 23, 10875–10878 (2007).
Meng, Z. et al. Preparation of highly monodisperse hybrid silica nanospheres using a one-step emulsion reaction in aqueous solution. Langmuir 25, 7879–7883 (2009).
Deng, T.-S. et al. One-step synthesis of highly monodisperse hybrid silica spheres in aqueous solution. J. Colloid Interface Sci. 329, 292–299 (2009).
Nair, B. P. & Pavithran, C. Bifunctionalized hybrid silica spheres by hydrolytic cocondensation of 3-aminopropyltriethoxysilane and vinyltriethoxysilane. Langmuir 26, 730–735 (2010).
Dirè, S., Tagliazucca, V., Callone, E. & Quaranta, A. Effect of functional groups on condensation and properties of sol–gel silica nanoparticles prepared by direct synthesis from organoalkoxysilanes. Mater. Chem. Phys. 126, 909–917 (2011).
Lu, Z., Sun, L., Nguyen, K., Gao, C. & Yin, Y. Formation mechanism and size control in one-pot synthesis of mercapto-silica colloidal spheres. Langmuir 27, 3372–3380 (2011).
Chiu, S.-J., Wang, S.-Y., Chou, H.-C., Liu, Y.-L. & Hu, T.-M. Versatile synthesis of thiol- and amine-bifunctionalized silica nanoparticles based on the Ouzo effect. Langmuir 30, 7676–7686 (2014).
Chou, H.-C., Chiu, S.-J., Liu, Y.-L. & Hu, T.-M. Direct formation of S-nitroso silica nanoparticles from a single silica source. Langmuir 30, 812–822 (2014).
Hong, F. C.-N. & Yan, C.-J. Preparation and application of monodisperse spherical polymethylsilsesquioxane (PMSQ) beads by sol-gel method. Adv. Powder Technol. 29, 1632–1639 (2018).
Zhang, H., Zhang, J., Wu, C., Zhang, B. & Zhang, Q. Preparation of functionalized SiO2 microspheres by one step method. Silicon 11, 2819–2827 (2019).
Chen, G.-T. & Hu, T.-M. Stable encapsulation of methylene blue in polysulfide organosilica colloids for fluorescent tracking of nanoparticle uptake in cells. ACS Omega 6, 32109–32119 (2021).
Giasuddin, A. B. M., Cartwright, A. & Britt, D. W. Silica nanoparticles synthesized from 3,3,3-propyl(trifluoro)trimethoxysilane or n-propyltrimethoxysilane for creating superhydrophobic surfaces. ACS Appl. Nano Mater. 4, 4092–4102 (2021).
Huo, Z. & Chen, L. Base-deactivated and alkaline-resistant chromatographic stationary phase based on functionalized polymethylsilsesquioxane microspheres. J. Sep. Sci. 43, 389–397 (2020).
Suzuki, T. M. et al. Direct synthesis of amino-functionalized monodispersed mesoporous silica spheres and their catalytic activity for nitroaldol condensation. J. Mol. Catal. A Chem. 280, 224–232 (2008).
Wani, A. et al. Surface functionalization of mesoporous silica nanoparticles controls loading and release behavior of mitoxantrone. Pharm. Res. 29, 2407–2418 (2012).
Fujii, Y., Zhou, S., Shimada, M. & Kubo, M. Synthesis of monodispersed hollow mesoporous organosilica and silica nanoparticles with controllable shell thickness using soft and hard templates. Langmuir 39, 4571–4582 (2023).
El Moujarrad, I. et al. Size-tuning of hollow periodic mesoporous organosilica nanoparticles (HPMO-NPs) using a dual templating strategy. J. Solgel Sci. Technol. 107, 302–311 (2023).
Zou, H. & Ren, Y. Synthetic strategies for nonporous organosilica nanoparticles from organosilane. Nanoscale 15, 10484–10497 (2023).
Obey, T. M. & Vincent, B. Novel monodisperse ‘silicone oil’/water emulsions. J. Colloid Interface Sci. 163, 454–463 (1994).
Anderson, K. R., Obey, T. M. & Vincent, B. Surfactant-stabilized silicone oil in water emulsions. Langmuir 10, 2493–2494 (1994).
Zoldesi, C. I. & Imhof, A. Synthesis of monodisperse colloidal spheres, capsules, and microballoons by emulsion templating. Adv. Mater. 17, 924–928 (2005).
Zoldesi, C. I., Ivanovska, I. L., Quilliet, C., Wuite, G. J. L. & Imhof, A. Elastic properties of hollow colloidal particles. Phys. Rev. E 78, 051401 (2008).
Quilliet, C., Zoldesi, C., Riera, C., van Blaaderen, A. & Imhof, A. Anisotropic colloids through non-trivial buckling. Eur. Phys. J. E 27, 13–20 (2008).
Marechal, M., Kortschot, R. J., Demirörs, A. F., Imhof, A. & Dijkstra, M. Phase behavior and structure of a new colloidal model system of bowl-shaped particles. Nano Lett. 10, 1907–1911 (2010).
Jose, J., Kamp, M., van Blaaderen, A. & Imhof, A. Unloading and reloading colloidal microcapsules with apolar solutions by controlled and reversible buckling. Langmuir 30, 2385–2393 (2014).
Elbers, N. A., Jose, J., Imhof, A. & van Blaaderen, A. Bulk scale synthesis of monodisperse PDMS droplets above 3 μm and their encapsulation by elastic shells. Chem. Mater. 27, 1709–1719 (2015).
Kamp, M. et al. Electric-field-induced lock-and-key interactions between colloidal spheres and bowls. Chem. Mater. 28, 1040–1048 (2016).
Sacanna, S., Kegel, W. K. & Philipse, A. P. Thermodynamically stable Pickering emulsions. Phys. Rev. Lett. 98, 158301 (2007).
Sacanna, S., Irvine, W. T. M., Chaikin, P. M. & Pine, D. J. Lock and key colloids. Nature 464, 575–578 (2010).
van der Wel, C. et al. Preparation of colloidal organosilica spheres through spontaneous emulsification. Langmuir 33, 8174–8180 (2017).
Sacanna, S., Rossi, L. & Pine, D. J. Magnetic click colloidal assembly. J. Am. Chem. Soc. 134, 6112–6115 (2012).
Sacanna, S. et al. Shaping colloids for self-assembly. Nat. Commun. 4, 1688 (2013).
Kamp, M., de Nijs, B., Baumberg, J. J. & Scherman, O. A. Contact angle as a powerful tool in anisotropic colloid synthesis. J. Colloid Interface Sci. 581, 417–426 (2021).
Middleton, C., Hannel, M. D., Hollingsworth, A. D., Pine, D. J. & Grier, D. G. Optimizing the synthesis of monodisperse colloidal spheres using holographic particle characterization. Langmuir 35, 6602–6609 (2019).
Abdelaziz, M. A. et al. Ultrasonic chaining of emulsion droplets. Phys. Rev. Res. 3, 043157 (2021).
Crothers, R. A., Orr, N. H. P., van der Meer, B., Dullens, R. P. A. & Yanagishima, T. Characterization and optimization of fluorescent organosilica colloids for 3D confocal microscopy prepared under “zero-flow” conditions. Langmuir 39, 5306–5314 (2023).
Chang, F. et al. Controllable synthesis of patchy particles with tunable geometry and orthogonal chemistry. J. Colloid Interface Sci. 582, 333–341 (2021).
Diaz A, J. A., Oh, J. S., Yi, G.-R. & Pine, D. J. Photo-printing of faceted DNA patchy particles. Proc. Natl Acad. Sci. USA 117, 10645–10653 (2020).
Issa, A. A. & Luyt, A. S. Kinetics of alkoxysilanes and organoalkoxysilanes polymerization: a review. Polymers 11, E537 (2019).
Pantoja, M., Velasco, F., Broekema, D., Abenojar, J. & del Real, J. C. The influence of pH on the hydrolysis process of γ-methacryloxypropyltrimethoxysilane, analyzed by FT-IR, and the silanization of electrogalvanized steel. J. Adhes. Sci. Technol. 24, 1131–1143 (2010).
Hu, T.-M., Chou, H.-C. & Lin, C.-Y. Facile green synthesis of organosilica nanoparticles by a generic “salt route”. J. Colloid Interface Sci. 539, 634–645 (2019).
Brochier Salon, M.-C., Bayle, P.-A., Abdelmouleh, M., Boufi, S. & Belgacem, M. N. Kinetics of hydrolysis and self condensation reactions of silanes by NMR spectroscopy. Colloids Surf. A Physicochem. Eng. Asp. 312, 83–91 (2008).
Oh, C., Shim, S.-B., Lee, Y.-G. & Oh, S.-G. Effects of the concentrations of precursor and catalyst on the formation of monodisperse silica particles in sol–gel reaction. Mater. Res. Bull. 46, 2064–2069 (2011).
van Bommel, M. J., Bernards, T. N. M. & Boonstra, A. H. The influence of the addition of alkyl-substituted ethoxysilane on the hydrolysis–condensation process of TEOS. J. Non-Cryst. Solids 128, 231–242 (1991).
van Bommel, M. J., ten Wolde, P. M. C. & Bernards, T. N. M. The influence of methacryloxypropyltrimethoxysilane on the sol-gel process of TEOS. J. Solgel Sci. Technol. 2, 167–170 (1994).
Wu, C., Wu, Y., Xu, T. & Yang, W. Study of sol–gel reaction of organically modified alkoxysilanes. Part I: investigation of hydrolysis and polycondensation of phenylaminomethyl triethoxysilane and tetraethoxysilane. J. Non-Cryst. Solids 352, 5642–5651 (2006).
Yang, H., Lu, X. & Xin, Z. One-step synthesis of nonspherical organosilica particles with tunable morphology. Langmuir 34, 11723–11728 (2018).
Liang, R., Fang, X., Qiu, B. & Zou, H. One-step synthesis of golf ball-like thiol-functionalized silica particles. Soft Matter 16, 9113–9120 (2020).
Eisenberg, P. et al. Cagelike precursors of high-molar-mass silsesquioxanes formed by the hydrolytic condensation of trialkoxysilanes. Macromolecules 33, 1940–1947 (2000).
Borovin, E., Callone, E., Ribot, F. & Diré, S. Mechanism and kinetics of oligosilsesquioxane growth in the in situ water production sol–gel route: dependence on water availability. Eur. J. Inorg. Chem. 2016, 2166–2174 (2016).
Tunstall-Garcia, H., Charles, B. L. & Evans, R. C. The role of polyhedral oligomeric silsesquioxanes in optical applications. Adv. Photonics Res. 2, 2000196 (2021).
Pescarmona, P. P., Aprile, C. & Swaminathan, S. in New and Future Developments in Catalysis (ed. Suib, S. L.) 385–422 (Elsevier, 2013).
Urata, C. et al. Aqueous colloidal mesoporous nanoparticles with ethenylene-bridged silsesquioxane frameworks. J. Am. Chem. Soc. 133, 8102–8105 (2011).
Neibloom, D., Bevan, M. A. & Frechette, J. Surfactant-stabilized spontaneous 3-(trimethoxysilyl) propyl methacrylate nanoemulsions. Langmuir 36, 284–292 (2020).
Neibloom, D., Bevan, M. A. & Frechette, J. Droplet formation and growth mechanisms in reaction-induced spontaneous emulsification of 3-(trimethoxysilyl) propyl methacrylate. Langmuir 37, 11625–11636 (2021).
Zheng, Y., Davis, C. R., Howarter, J. A., Erk, K. A. & Martinez, C. J. Spontaneous emulsions: adjusting spontaneity and phase behavior by hydrophilic–lipophilic difference-guided surfactant, salt, and oil selection. Langmuir 38, 4276–4286 (2022).
Sacanna, S., Irvine, W. T. M., Rossi, L. & Pine, D. J. Lock and key colloids through polymerization-induced buckling of monodisperse silicon oil droplets. Soft Matter 7, 1631–1634 (2011).
Ohta, T., Nagao, D., Ishii, H. & Konno, M. Preparation of oil-containing, polymeric particles having a single depression with various shapes. Soft Matter 8, 4652–4658 (2012).
Wu, H., Du, X., Meng, X., Qiu, D. & Qiao, Y. A three-tiered colloidosomal microreactor for continuous flow catalysis. Nat. Commun. 12, 6113 (2021).
van der Wel, C., van de Stolpe, G. L., Verweij, R. W. & Kraft, D. J. Micrometer-sized TPM emulsion droplets with surface-mobile binding groups. J. Phys. Condens. Matter 30, 094005 (2018).
Shah, Z. H. et al. Synthesis of two-patch particles with controlled patch size via nonequilibrium solidification of droplets on rods. Polymer 177, 91–96 (2019).
Shah, Z. H. et al. Highly efficient chemically-driven micromotors with controlled snowman-like morphology. Chem. Commun. 56, 15301–15304 (2020).
Doura, T., Tamanoi, F. & Nakamura, M. Miniaturization of thiol-organosilica nanoparticles induced by an anionic surfactant. J. Colloid Interface Sci. 526, 51–62 (2018).
Vogel, R. et al. Fluorescent organosilica micro- and nanoparticles with controllable size. J. Colloid Interface Sci. 310, 144–150 (2007).
Surawski, P. P. T., Battersby, B. J., Vogel, R., Lawrie, G. & Trau, M. Modification and optimization of organosilica microspheres for peptide synthesis and microsphere-based immunoassays. Mol. BioSyst. 5, 826–831 (2009).
Kamp, M. et al. Cascaded nano-optics to probe microsecond atomic scale phenomena. Proc. Natl Acad. Sci. 117, 14819–14826 (2020).
Wang, Y. et al. Three-dimensional lock and key colloids. J. Am. Chem. Soc. 136, 6866–6869 (2014).
Chen, Y., Chen, H.-R. & Shi, J.-L. Construction of homogenous/heterogeneous hollow mesoporous silica nanostructures by silica-etching chemistry: principles, synthesis, and applications. Acc. Chem. Res. 47, 125–137 (2014).
Hunks, W. J. & Ozin, G. A. Challenges and advances in the chemistry of periodic mesoporous organosilicas (PMOs). J. Mater. Chem. 15, 3716–3724 (2005).
Ren, X. & Tsuru, T. Organosilica-based membranes in gas and liquid-phase separation. Membranes 9, 107 (2019).
Kadowaki, K., Ishii, H., Nagao, D. & Konno, M. Imprinting dimples on narrowly dispersed polymeric spheres by heterocoagulation between hard polymer particles and soft oil droplets. Langmuir 32, 11600–11605 (2016).
Graf, C., Vossen, D. L. J., Imhof, A. & van Blaaderen, A. A general method to coat colloidal particles with silica. Langmuir 19, 6693–6700 (2003).
Ma, J., Liu, Y., Bao, Y., Liu, J. & Zhang, J. Research advances in polymer emulsion based on “core–shell” structure particle design. Adv. Colloid Interface Sci. 197–198, 118–131 (2013).
Deng, T.-S. & Marlow, F. Synthesis of monodisperse polystyrene@vinyl-SiO2 core–shell particles and hollow SiO2 spheres. Chem. Mater. 24, 536–542 (2012).
Liu, Y. et al. Core–shell particles for simultaneous 3D imaging and optical tweezing in dense colloidal materials. Adv. Mater. 28, 8001–8006 (2016).
Weijgertze, H. M. H., Kegel, W. K. & Zanini, M. Patchy rough colloids as Pickering stabilizers. Soft Matter 16, 8002–8012 (2020).
Lan, Y. et al. Unexpected stability of aqueous dispersions of raspberry-like colloids. Nat. Commun. 9, 3614 (2018).
Kamp, M. et al. Selective depletion interactions in mixtures of rough and smooth silica spheres. Langmuir 32, 1233–1240 (2016).
Van Blaaderen, A., Imhof, A., Hage, W. & Vrij, A. Three-dimensional imaging of submicrometer colloidal particles in concentrated suspensions using confocal scanning laser microscopy. Langmuir 8, 1514–1517 (1992).
Liu, Y. et al. Colloidal organosilica spheres for three-dimensional confocal microscopy. Langmuir 35, 7962–7969 (2019).
Kamp, M. et al. Regiospecific nucleation and growth of silane coupling agent droplets onto colloidal particles. J. Phys. Chem. C 121, 19989–19998 (2017).
Liu, M., Dong, F., Jackson, N. S., Ward, M. D. & Weck, M. Customized chiral colloids. J. Am. Chem. Soc. 142, 16528–16532 (2020).
Kamp, M. et al. Multivalent patchy colloids for quantitative 3D self-assembly studies. Langmuir 36, 2403–2418 (2020).
Wang, Z. et al. Active patchy colloids with shape-tunable dynamics. J. Am. Chem. Soc. 141, 14853–14863 (2019).
Aubret, A., Martinet, Q. & Palacci, J. Metamachines of pluripotent colloids. Nat. Commun. 12, 36398 (2021).
Gong, Z., Hueckel, T., Yi, G.-R. & Sacanna, S. Patchy particles made by colloidal fusion. Nature 550, 234 (2017).
He, M. et al. Colloidal diamond. Nature 585, 524–529 (2020).
He, M., Gales, J. P., Shen, X., Kim, M. J. & Pine, D. J. Colloidal particles with triangular patches. Langmuir 37, 7246–7253 (2021).
Oh, J. S., Yi, G.-R. & Pine, D. J. Reconfigurable transitions between one- and two-dimensional structures with bifunctional DNA-coated Janus colloids. ACS Nano 14, 15786–15792 (2020).
Segers, M., Vermeer, I., Möller, M., Verheijen, M. & Buskens, P. Synthesis and characterization of hybrid particles obtained in a one-pot process through simultaneous sol-gel reaction of (3-mercaptopropyl)trimethoxysilane and emulsion polymerization of styrene. Colloids Interfaces 1, 7 (2017).
Kim, D.-Y. et al. Microfluidic preparation of monodisperse polymeric microspheres coated with silica nanoparticles. Sci. Rep. 8, 8525 (2018).
Opdam, J., Tuinier, R., Hueckel, T., J. Snoeren, T. & Sacanna, S. Selective colloidal bonds via polymer-mediated interactions. Soft Matter 16, 7438–7446 (2020).
García-Santamaría, F., Míguez, H., Ibisate, M., Meseguer, F. & López, C. Refractive index properties of calcined silica submicrometer spheres. Langmuir 18, 1942–1944 (2002).
Seet, K. Y. T. et al. Refractometry of organosilica microspheres. Appl. Opt. 46, 1554–1561 (2007).
Dong, F., Liu, M., Grebe, V., Ward, M. D. & Weck, M. Assembly of shape-tunable colloidal dimers in a dielectrophoretic field. Chem. Mater. 32, 6898–6905 (2020).
Zhu, J., Wang, H. & Zhang, Z. Shape-tunable Janus micromotors via surfactant-induced dewetting. Langmuir 37, 4964–4970 (2021).
Iwamatsu, M. Size-dependent contact angle and the wetting and drying transition of a droplet adsorbed onto a spherical substrate: line-tension effect. Phys. Rev. E 94, 042803 (2016).
Liu, B. & Böker, A. Measuring rotational diffusion of colloidal spheres with confocal microscopy. Soft Matter 12, 6033–6037 (2016).
Yanagishima, T., Liu, Y., Tanaka, H. & Dullens, R. Particle-level visualization of hydrodynamic and frictional couplings in dense suspensions of spherical colloids. Phys. Rev. X 11, 021056 (2021).
Reculusa, S. et al. Hybrid dissymmetrical colloidal particles. Chem. Mater. 17, 3338–3344 (2005).
Lv, X. et al. Synthesis of snowman-shaped photocatalytic microrotors and mechanical micropumps. ChemNanoMat 7, 902–905 (2021).
Wang, L., Shi, S., Luo, Z., Qu, N. & Liu, B. Hierarchical, highly open microtubes and columnar liquid crystals self-assembled from symmetrical and asymmetrical colloidal rings. Angew. Chem. Int. Ed. 61, e202112507 (2022).
Luo, Z., Li, S., Wang, L. & Liu, B. Asymmetrical ring-shaped colloidal particles for self-assembly and superhydrophobic coatings. Chem. Commun. 58, 5757–5760 (2022).
Liu, M., Zheng, X., Grebe, V., Pine, D. J. & Weck, M. Tunable assembly of hybrid colloids induced by regioselective depletion. Nat. Mater. 19, 1354–1361 (2020).
van Ravensteijn, B. G. P., Kamp, M., van Blaaderen, A. & Kegel, W. K. General route toward chemically anisotropic colloids. Chem. Mater. 25, 4348–4353 (2013).
Reculusa, S., Mingotaud, C., Bourgeat-Lami, E., Duguet, E. & Ravaine, S. Synthesis of daisy-shaped and multipod-like silica/polystyrene nanocomposites. Nano Lett. 4, 1677–1682 (2004).
Nagao, D., Hashimoto, M., Hayasaka, K. & Konno, M. Synthesis of anisotropic polymer particles with soap-free emulsion polymerization in the presence of a reactive silane coupling agent. Macromol. Rapid Commun. 29, 1484–1488 (2008).
Wang, Y. et al. Colloids with valence and specific directional bonding. Nature 491, 51–55 (2012).
Forestier, T., Ferrié, M. & Ravaine, S. Synthesis of hematite/silica/polymer composite colloids with a tunable morphology. Colloid Polym. Sci. 291, 187–192 (2013).
Shibata, N., Nagao, D., Ishii, H. & Konno, M. Preparation of various Janus composite particles with two components differently combined. Colloid Polym. Sci. 291, 137–142 (2013).
Zhang, W. et al. Effective structure control of colloidal molecules and the morphology evolution mechanism investigation. Langmuir 37, 12429–12437 (2021).
Sacanna, S. & Pine, D. J. Shape-anisotropic colloids: building blocks for complex assemblies. Curr. Opin. Colloid Interface Sci. 16, 96–105 (2011).
Tian, L. et al. Particle-click-particle: colloidal clusters from click seeded emulsion polymerization. Polym. Chem. 13, 1084–1089 (2022).
Yi, C. et al. Self-limiting directional nanoparticle bonding governed by reaction stoichiometry. Science 369, 1369–1374 (2020).
Kim, J.-W., Larsen, R. J. & Weitz, D. A. Synthesis of nonspherical colloidal particles with anisotropic properties. J. Am. Chem. Soc. 128, 14374–14377 (2006).
Park, J.-G., Forster, J. D. & Dufresne, E. R. High-yield synthesis of monodisperse dumbbell-shaped polymer nanoparticles. J. Am. Chem. Soc. 132, 5960–5961 (2010).
Kraft, D. J. et al. Surface roughness directed self-assembly of patchy particles into colloidal micelles. Proc. Natl Acad. Sci. USA 109, 10787 (2012).
Peng, B., Vutukuri, H. R., van Blaaderen, A. & Imhof, A. Synthesis of fluorescent monodisperse non-spherical dumbbell-like model colloids. J. Mater. Chem. 22, 21893–21900 (2012).
Gui, H. et al. Preparation of asymmetric particles by controlling the phase separation of seeded emulsion polymerization with ethanol/water mixture. J. Colloid Interface Sci. 618, 496–506 (2022).
Ma, F., Wang, S., Wu, D. T. & Wu, N. Electric-field-induced assembly and propulsion of chiral colloidal clusters. Proc. Natl Acad. Sci. USA 112, 6307–6312 (2015).
Meester, V. & Kraft, D. J. Spherical, dimpled, and crumpled hybrid colloids with tunable surface morphology. Langmuir 32, 10668–10677 (2016).
Kim, J.-W., Larsen, R. J. & Weitz, D. A. Uniform nonspherical colloidal particles with tunable shapes. Adv. Mater. 19, 2005–2009 (2007).
Peng, B. et al. Site-specific growth of polymers on silica rods. Soft Matter 10, 9644–9650 (2014).
Mann, D. et al. Protecting patches in colloidal synthesis of Au semishells. Chem. Commun. 53, 3898–3901 (2017).
Youssef, M., Hueckel, T., Yi, G.-R. & Sacanna, S. Shape-shifting colloids via stimulated dewetting. Nat. Commun. 7, 12216 (2016).
Wang, Y., McGinley, J. T. & Crocker, J. C. Dimpled polyhedral colloids formed by colloidal crystal templating. Langmuir 33, 3080–3087 (2017).
Shillingford, C., Kim, B. M. & Weck, M. Capillary assembly of liquid particles. Small 16, 1907523 (2020).
Oh, J. S., Lee, S., Glotzer, S. C., Yi, G.-R. & Pine, D. J. Colloidal fibers and rings by cooperative assembly. Nat. Commun. 10, 3936 (2019).
Edmond, K. V. et al. Large-scale synthesis of colloidal bowl-shaped particles. Soft Matter 17, 6176–6181 (2021).
Schade, N. B. et al. Tetrahedral colloidal clusters from random parking of bidisperse spheres. Phys. Rev. Lett. 110, 148303 (2013).
Shelke, Y., Marín-Aguilar, S., Camerin, F., Dijkstra, M. & Kraft, D. J. Exploiting anisotropic particle shape to electrostatically assemble colloidal molecules with high yield and purity. J. Colloid Interface Sci. 629, 322–333 (2023).
Liu, Y., Wang, J., Imaz, I. & Maspoch, D. Assembly of colloidal clusters driven by the polyhedral shape of metal–organic framework particles. J. Am. Chem. Soc. 143, 12943–12947 (2021).
Mani, E. et al. Sheet-like assemblies of spherical particles with point-symmetrical patches. J. Chem. Phys. 136, 144706 (2012).
Noya, E. G., Zubieta, I., Pine, D. J. & Sciortino, F. Assembly of clathrates from tetrahedral patchy colloids with narrow patches. J. Chem. Phys. 151, 094502 (2019).
Xu, Z., Hueckel, T., Irvine, W. T. M. & Sacanna, S. Transmembrane transport in inorganic colloidal cell-mimics. Nature 597, 220–224 (2021).
Zheng, X., Liu, M., He, M., Pine, D. J. & Weck, M. Shape-shifting patchy particles. Angew. Chem. Int. Ed. 129, 5599–5603 (2017).
Stuij, S. et al. Revealing polymerization kinetics with colloidal dipatch particles. Phys. Rev. Lett. 127, 108001 (2021).
Swinkels, P. J. M. et al. Revealing pseudorotation and ring-opening reactions in colloidal organic molecules. Nat. Commun. 12, 2810 (2021).
Nguyen, V. D., Dang, M. T., Nguyen, T. A. & Schall, P. Critical Casimir forces for colloidal assembly. J. Phys. Condens. Matter 28, 043001 (2016).
Swinkels, M., Gong, P. J., Sacanna, S., Noya, E. & Schall, P. Phases of surface-confined trivalent colloidal particles. Soft Matter 19, 3414–3422 (2023).
Elacqua, E., Zheng, X., Shillingford, C., Liu, M. & Weck, M. Molecular recognition in the colloidal world. Acc. Chem. Res. 50, 2756–2766 (2017).
Barrow, S. J., Kasera, S., Rowland, M. J., del Barrio, J. & Scherman, O. A. Cucurbituril-based molecular recognition. Chem. Rev. 115, 12320–12406 (2015).
Elacqua, E., Zheng, X. & Weck, M. Light-mediated reversible assembly of polymeric colloids. ACS Macro Lett. 6, 1060–1065 (2017).
Zheng, X., Wang, Y., Wang, Y., Pine, D. J. & Weck, M. Thermal regulation of colloidal materials architecture through orthogonal functionalizable patchy particles. Chem. Mater. 28, 3984–3989 (2016).
Zhang, T., Lyu, D., Xu, W., Mu, Y. & Wang, Y. Programming self-assembled materials with DNA-coated colloids. Front. Phys. 9, 330 (2021).
Wang, Y. et al. Crystallization of DNA-coated colloids. Nat. Commun. 6, 7253 (2015).
Wang, Y. et al. Synthetic strategies toward DNA-coated colloids that crystallize. J. Am. Chem. Soc. 137, 10760–10766 (2015).
Khalaf, R., Viamonte, A., Ducrot, E., Mérindol, R. & Ravaine, S. Transfer of multi-DNA patches by colloidal stamping. Nanoscale 15, 573–577 (2022).
Lee, S., Zheng, C. Y., Bujold, K. E. & Mirkin, C. A. A cross-linking approach to stabilizing stimuli-responsive colloidal crystals engineered with DNA. J. Am. Chem. Soc. 141, 11827–11831 (2019).
Petukhov, A. V., Tuinier, R. & Vroege, G. J. Entropic patchiness: effects of colloid shape and depletion. Curr. Opin. Colloid Interface Sci. 30, 54–61 (2017).
Badaire, S., Cottin-Bizonne, C. & Stroock, A. D. Experimental investigation of selective colloidal interactions controlled by shape, surface roughness, and steric layers. Langmuir 24, 11451–11463 (2008).
Colón-Meléndez, L. et al. Binding kinetics of lock and key colloids. J. Chem. Phys. 142, 174909 (2015).
Liu, M. et al. Two-dimensional (2D) or quasi-2D superstructures from DNA-coated colloidal particles. Angew. Chem. Int. Ed.133, 5808–5812 (2021).
Liljeström, V., Chen, C., Dommersnes, P., Fossum, J. O. & Gröschel, A. H. Active structuring of colloids through field-driven self-assembly. Curr. Opin. Colloid Interface Sci. 40, 25–41 (2019).
Driscoll, M. & Delmotte, B. Leveraging collective effects in externally driven colloidal suspensions: experiments and simulations. Curr. Opin. Colloid Interface Sci. 40, 42–57 (2019).
Rossi, L. Magnetic colloids as building blocks for complex structures: Preparation and assembly. In Frontiers of Nanoscience Vol. 13 (eds Chakrabarti, D. & Sacanna, S.) Ch. 1 (Elsevier, 2019).
Meijer, J.-M. & Rossi, L. Preparation, properties, and applications of magnetic hematite microparticles. Soft Matter 17, 2354–2368 (2021).
Van Blaaderen, A. et al. Manipulating the self assembly of colloids in electric fields. Eur. Phys. J. Spec. Top. 222, 2895–2909 (2013).
Jia, Z., Sacanna, S. & Lee, S. S. Dielectrophoretic assembly of dimpled colloids into open packing structures. Soft Matter 13, 5724–5730 (2017).
Jia, Z., Youssef, M., Samper, A., Sacanna, S. & Lee, S. S. Reversible solid-state phase transitions in confined two-layer colloidal crystals. Colloid Polym. Sci. 298, 1611–1617 (2020).
Collins, K. A. et al. Electric-field-induced reversible phase transitions in two-dimensional colloidal crystals. Langmuir 31, 10411–10417 (2015).
Boon, N. et al. Screening of heterogeneous surfaces: charge renormalization of Janus particles. J. Phys. Condens. Matter 22, 104104 (2010).
Song, P. et al. Patchy particle packing under electric fields. J. Am. Chem. Soc. 137, 3069–3075 (2015).
Wang, Z., Wang, Z., Li, J. & Wang, Y. Directional and reconfigurable assembly of metallodielectric patchy particles. ACS Nano 15, 5439–5448 (2021).
Yan, J. et al. Reconfiguring active particles by electrostatic imbalance. Nat. Mater. 15, 1095–1099 (2016).
Abrikosov, A. I., Sacanna, S., Philipse, A. P. & Linse, P. Self-assembly of spherical colloidal particles with off-centered magnetic dipoles. Soft Matter 9, 8904–8913 (2013).
Sprinkle, B., Wee, E. B., van der, Luo, Y., Driscoll, M. M. & Donev, A. Driven dynamics in dense suspensions of microrollers. Soft Matter 16, 7982–8001 (2020).
van der Wee, E. B. et al. A simple catch: fluctuations enable hydrodynamic trapping of microrollers by obstacles. Sci. Adv. 9, eade0320 (2023).
Kang, N., Zhu, J., Zhang, X., Wang, H. & Zhang, Z. Reconfiguring self-assembly of photoresponsive hybrid colloids. J. Am. Chem. Soc. 144, 4754–4758 (2022).
Palacci, J., Sacanna, S., Steinberg, A. P., Pine, D. J. & Chaikin, P. M. Living crystals of light-activated colloidal surfers. Science 339, 936–940 (2013).
Aubret, A., Youssef, M., Sacanna, S. & Palacci, J. Targeted assembly and synchronization of self-spinning microgears. Nat. Phys. 14, 1114–1118 (2018).
Gonzalez Ortiz, D., Pochat-Bohatier, C., Cambedouzou, J., Bechelany, M. & Miele, P. Current trends in Pickering emulsions: particle morphology and applications. Engineering 6, 468–482 (2020).
Dekker, R. I. et al. Is there a difference between surfactant-stabilised and Pickering emulsions? Soft Matter 19, 1941–1951 (2023).
Kegel, W. K. & Groenewold, J. Scenario for equilibrium solid-stabilized emulsions. Phys. Rev. E 80, 030401 (2009).
Hasnain, J. et al. Spontaneous emulsification induced by nanoparticle surfactants. J. Chem. Phys. 153, 224705 (2020).
Ketzetzi, S., de Graaf, J. & Kraft, D. J. Diffusion-based height analysis reveals robust microswimmer-wall separation. Phys. Rev. Lett. 125, 238001 (2020).
Ebbens, S. J. Active colloids: progress and challenges towards realising autonomous applications. Curr. Opin. Colloid Interface Sci. 21, 14–23 (2016).
Notingher, I. et al. In situ characterisation of living cells by Raman spectroscopy. Spectroscopy 16, 43–51 (2002).
Hess, H., Howard, J. & Vogel, V. A piconewton forcemeter assembled from microtubules and kinesins. Nano Lett. 2, 1113–1115 (2002).
Hess, H., Bachand, G. D. & Vogel, V. Powering nanodevices with biomolecular motors. Chem. Eur. J. 10, 2110–2116 (2004).
Kabir, A. Md. R., Kageyama, Y. & Kakugo, A. Molecular actuators and their applications in molecular robotics. In Encyclopedia of Robotics (eds Ang, M. H. et al.) (Springer, 2020).
Ikram, M. et al. Light-activated fuel-free Janus metal organic framework colloidal motors for the removal of heavy metal ions. ACS Appl. Mater. Interfaces 13, 51799–51806 (2021).
Xu, Z., Hueckel, T., Irvine, W. T. M. & Sacanna, S. Caged colloids. Chem. Mater. 35, 6357–6363 (2023).
Peng, B., Liu, Y., Aarts, D. G. A. L. & Dullens, R. P. A. Stabilisation of hollow colloidal TiO2 particles by partial coating with evenly distributed lobes. Soft Matter 17, 1480–1486 (2021).
Liang, R. & Zou, H. Removal of aqueous Hg(II) by thiol-functionalized nonporous silica microspheres prepared by one-step sol–gel method. RSC Adv. 10, 18534–18542 (2020).
Ren, G., Wang, W., Shang, M., Zou, H. & Cheng, S. Using a macroporous silver shell to coat sulfonic acid group-functionalized silica spheres and their applications in catalysis and surface-enhanced Raman scattering. Langmuir 31, 10517–10523 (2015).
Hu, H. et al. Reversible and precise self-assembly of Janus metal-organosilica nanoparticles through a linker-free approach. ACS Nano 10, 7323–7330 (2016).
Huang, J. et al. Tracking interfacial single-molecule pH and binding dynamics via vibrational spectroscopy. Sci. Adv. 7, eabg1790 (2021).
Morphew, D., Shaw, J., Avins, C. & Chakrabarti, D. Programming hierarchical self-assembly of patchy particles into colloidal crystals via colloidal molecules. ACS Nano 12, 2355–2364 (2018).
Neophytou, A., Manoharan, V. N. & Chakrabarti, D. Self-assembly of patchy colloidal rods into photonic crystals robust to stacking faults. ACS Nano 15, 2668–2678 (2021).
Yang, S. & Li, Y. Fluorescent hybrid silica nanoparticles and their biomedical applications. WIREs Nanomed. Nanobiotechnol. 12, e1603 (2020).
Nakamura, M., Mochizuki, C., Kuroda, C., Shiohama, Y. & Nakamura, J. Size effect of fluorescent thiol-organosilica particles on their distribution in the mouse spleen. Colloids Surf. B Biointerfaces 228, 113397 (2023).
Encinas, N. et al. Mixed-charge pseudo-zwitterionic mesoporous silica nanoparticles with low-fouling and reduced cell uptake properties. Acta Biomater. 84, 317–327 (2019).
Nakamura, M. Biomedical applications of organosilica nanoparticles toward theranostics. Nanotechnol. Rev. 1, 469–491 (2012).
Shah, Z. H., Sockolich, M., Rivas, D. & Das, S. Fabrication and open-loop control of three-lobed nonspherical Janus microrobots. MRS Adv. 8, 1028–1032 (2023).
Picchetti, P. et al. Breaking with light: stimuli-responsive mesoporous organosilica particles. Chem. Mater. 32, 392–399 (2020).
Riccio, D. A., Nugent, J. L. & Schoenfisch, M. H. Stöber synthesis of nitric oxide-releasing S-nitrosothiol-modified silica particles. Chem. Mater. 23, 1727–1735 (2011).
Grober, D. et al. Unconventional colloidal aggregation in chiral bacterial baths. Nat. Phys. 19, 1680–1688 (2023).
Acknowledgements
S.S. acknowledges support from the US Army Research Office under award number W911NF-21-1-0011. R.P.A.D. acknowledges financial support from the European Research Council (ERC Consolidator Grant number 724834 OMCIDC).
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to discussion of the content. M.K. substantially wrote the first draft of the article. All authors reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Chemistry thanks Hua Zou, Bing Liu, Huaguang Wang and Serge Ravaine for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Critical point
-
Temperature above which the components of a medium are miscible.
- Debye length
-
Characteristic colloidal length scale that expresses how far the effect of a colloid’s electrostatic charge persists in the medium at hand owing to screening by the charge carriers (ionic species) in the medium.
- Depletion force
-
Effective attractive force between colloidal particles arising when suspended in a solution of solutes (for example, polymer or smaller particles). The solutes cannot reach the colloidal particles closer than their own radius (excluded volume), which causes the solutes to be excluded from the spaces between the particles (overlap volume). This results in a net attractive force between the colloidal particles.
- Dielectrophoresis
-
(DEP). The motion of colloidal particles in a non-uniform electric field, driven by induced polarization acquired in the field provided the dielectric constant of particles differs from that of the medium.
- Diffusiophoresis
-
The motion of particles (colloids, (macro)molecules, and so on) caused by a bulk concentration gradient of dissolved solutes in the medium.
- Induced-charge electrophoresis
-
(ICEP). The motion of colloidal particles in an alternating electric field, driven by asymmetric fluid flow on account of the asymmetric surface properties of a particle. The asymmetric fluid flow itself is caused by induced-charge electro-osmosis or the effect of the alternating electric field on the ion clouds around colloidal particles.
- Nematic phase
-
Type of liquid crystal in which anisotropic colloidal particles are oriented along a common direction but exhibit no positional order.
- Pair potential
-
The potential energy between two colloidal particles as a function of the distance between them.
- Protein corona
-
Coating of (non-bound) biomolecules, usually proteins, around the surface of a nanoparticle. This protein corona often assembles when colloids are suspended in biological media.
- (Refractive) Index matching
-
In a dispersion, the refractive index of the medium can be matched to that of the colloids by careful choice of media. This is called index matching. It is usually done to reduce scattering of light from the particles, improving the quality of images taken by (confocal) fluorescence microscopy.
- Sedimentation–diffusion equilibrium
-
In a colloidal dispersion, particles will settle owing to the gravitational force, whereas diffusion causes an upward particle flux. The balance of these two effects results in an equilibrium height distribution of the particles, called the sedimentation–diffusion equilibrium.
- Spinodal decomposition
-
Spontaneous separation of a medium into two (or more) co-existing phases.
- Yukawa interaction (screened Coulombic potential)
-
The pair potential between two charged colloidal particles in a medium, wherein the ions in the medium screen the colloidal electrostatic charges.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kamp, M., Sacanna, S. & Dullens, R.P.A. Spearheading a new era in complex colloid synthesis with TPM and other silanes. Nat Rev Chem (2024). https://doi.org/10.1038/s41570-024-00603-4
Accepted:
Published:
DOI: https://doi.org/10.1038/s41570-024-00603-4