Abstract
Diabetes mellitus is an important risk factor for a first cardiovascular event and for worse outcomes after a cardiovascular event has occurred. This situation might be caused, at least in part, by the prothrombotic status observed in patients with diabetes. Therefore, contemporary antithrombotic strategies, including more potent agents or drug combinations, might provide greater clinical benefit in patients with diabetes than in those without diabetes. In this Consensus Statement, our Working Group explores the mechanisms of platelet and coagulation activity, the current debate on antiplatelet therapy in primary cardiovascular disease prevention, and the benefit of various antithrombotic approaches in secondary prevention of cardiovascular disease in patients with diabetes. While acknowledging that current data are often derived from underpowered, observational studies or subgroup analyses of larger trials, we propose antithrombotic strategies for patients with diabetes in various cardiovascular settings (primary prevention, stable coronary artery disease, acute coronary syndromes, ischaemic stroke and transient ischaemic attack, peripheral artery disease, atrial fibrillation, and venous thromboembolism). Finally, we summarize the improvements in cardiovascular outcomes observed with the latest glucose-lowering drugs, and on the basis of the available evidence, we expand and integrate current guideline recommendations on antithrombotic strategies in patients with diabetes for both primary and secondary prevention of cardiovascular disease.
Similar content being viewed by others
Introduction
Diabetes mellitus is an important risk factor for a first cardiovascular event and for worse outcomes after a cardiovascular event has occurred. Cardiovascular disease in diabetes is a progressive process characterized by early endothelial dysfunction, oxidative stress, and vascular inflammation leading to monocyte recruitment and formation of foam cells and fatty streaks, which cause development of atherosclerotic plaques over years1. Compared with atherosclerotic plaques from individuals without diabetes, those from patients with diabetes are more vulnerable (rupture-prone), and therefore, these plaques are at increased risk of developing superimposed thrombosis because of increased amounts of soft extracellular lipids, inflammation, and prothrombotic milieu; this situation predisposes patients with diabetes to acute cardiovascular events1. Consequently, in principle, aggressive antithrombotic therapies might be associated with greater clinical benefit in patients with diabetes than in those without the condition. However, the ischaemic protection provided by antithrombotic drugs must be weighed against the drug-related bleeding risk.
This Consensus Statement from the Working Group on Thrombosis of the Italian Society of Cardiology provides up-to-date recommendations on primary and secondary prevention of atherothrombotic events in patients with diabetes. We explore the mechanisms of platelet and coagulation activity and analyse the current data on the risk–benefit balance of antiplatelet therapy for primary prevention of cardiovascular disease in patients with diabetes. We evaluate the differential protection provided by different antithrombotic therapies in secondary prevention of cardiovascular disease according to diabetic status in various settings (stable coronary artery disease (CAD), acute coronary syndrome (ACS), ischaemic cerebrovascular events, peripheral artery disease (PAD), and venous thromboembolism (VTE)). We also analyse the relationship between diabetes and thromboembolic risk in atrial fibrillation (AF) and the efficacy and safety of the non-vitamin K antagonist oral anticoagulants (NOACs) in patients with diabetes and AF. On the basis of the available evidence, we propose antithrombotic strategies that expand and integrate current guideline recommendations for patients with diabetes2,3,4 (Table 1). Finally, we discuss the cardiovascular outcomes data from randomized, controlled trials on the new classes of glucose-lowering drugs.
Platelet function and reactivity
Platelets of individuals with diabetes, compared with those of healthy controls, have dysregulation at both the receptor and the intracellular signal transduction levels, leading to hyperreactive adhesion, activation, degranulation, and aggregation5. Reduced insulin sensitivity causes increased signalling of P2Y purinoceptor 12 (P2Y12 receptor), the main platelet receptor for ADP6 (Box 1; Fig. 1). Hyperglycaemia and associated conditions, such as obesity, dyslipidaemia, and inflammation, modulate this phenotype5.
Increased platelet reactivity in diabetes involves higher levels of thrombin generation, increased production of thromboxane A2 (TXA2), hyperresponsiveness of proteinase-activated receptor 4 (PAR4) to thrombin and TXA2, and increased platelet membrane expression of P-selectin, adhesion molecules, and glycoprotein (GP) IIb/IIIa. Signalling of P2Y purinoceptor 12 (P2Y12 receptor) — the main platelet receptor for ADP — is also increased. Vascular synthesis of nitric oxide (NO) and prostaglandin I2 (PGI2; also known as prostacyclin) is decreased, and the production of reactive oxygen species (ROS) is increased. P2X, P2X purinoceptor; P2Y1, P2Y purinoceptor 1; vWF, von Willebrand factor.
In vitro studies from patients with diabetes have shown increased platelet membrane expression of P-selectin and glycoprotein IIb/IIIa (GPIIb/IIIa), as well as increased platelet response to adrenaline and thrombin5. Impairment of the arachidonic acid pathway accounts for platelet dysfunction in vivo, which can be improved through suppression of platelet cyclooxygenase 1 (COX1; also known as prostaglandin G/H synthase 1)7.
Platelets from patients with diabetes show increased turnover, resulting in a higher number of reticulated platelets, and increased adhesion to endothelial cells8. A reduction in platelet fluidity occurs through changes in membrane lipid structure or glycation of membrane proteins5. Furthermore, platelets from patients with diabetes have an increased expression of adhesion molecules, such as CD63 antigen, integrin α2 (CD49B), platelet endothelial cell adhesion molecule 1 (CD31), platelet glycoprotein 4 (CD36), and P-selectin (CD62P), as documented by flow cytometry9.
Another important determinant of platelet dysfunction in diabetes is the reduced vascular production of nitric oxide (NO) and prostaglandin I2 (PGI2; also known as prostacyclin), which physiologically inhibit platelet aggregation8. Other reactive oxygen species (ROS) might also contribute to in vivo platelet hyperactivation in diabetes8.
In patients with ACS or those undergoing coronary stent implantation, the recurrence of cardiovascular events is not infrequent despite using the current standard of care, which is dual antiplatelet therapy (DAPT) comprising acetylsalicylic acid (commonly referred to as aspirin) and a P2Y12-receptor blocker. Platelet-function studies have revealed that treatment with clopidogrel is associated with an overall modest level of P2Y12-receptor inhibition with interindividual variability, even when high loading doses are used10,11. ADP-induced high platelet reactivity (HPR) when taking clopidogrel has been identified as a marker of vascular risk in an international consensus document12.
Patients with diabetes have a more pronounced impairment in their response to clopidogrel than individuals without diabetes13,14,15,16,17. This finding was demonstrated in ex vivo testing of platelet reactivity and was also derived by subgroup analyses of clinical trials, in which patients with diabetes who were undergoing percutaneous coronary intervention (PCI) and receiving clopidogrel had a worse clinical course and a higher incidence of stent thrombosis than patients without diabetes13,14,15,16. The exact mechanism of this phenomenon remains unknown. Reduced insulin sensitivity in diabetes might cause increased signalling of P2Y12 receptors; in addition, insulin might physiologically reduce platelet aggregation by inhibiting the P2Y12 pathway through platelet insulin receptors, and this mechanism might be impaired in patients with diabetes18. Finally, diabetes is known to attenuate cytochrome P450 activity in animal models and in humans19,20,21; accordingly, in patients with diabetes who are receiving clopidogrel, HPR has been shown to be correlated with lower concentrations of the clopidogrel active metabolite22.
Another area of research is investigating the possible role of HPR in response to other platelet agonists, in particular arachidonic acid, which predominantly mirrors the effects of aspirin on platelet inhibition. Diabetes has been associated with HPR in response to arachidonic acid; aspirin-naive patients with diabetes seem to have elevated levels of platelet-derived thromboxane, and aspirin might be less effective in inhibiting thromboxane synthesis in patients with diabetes than in individuals without diabetes23. In the randomized ASPECT study24, in which multiple methods were used to assess responsiveness to aspirin (81 mg daily), patients with diabetes had greater platelet aggregability than those without diabetes, as measured by the VerifyNow assay (Accriva Diagnostics), urinary 11-dehydro-thromboxane B2 levels, and collagen-induced light transmittance aggregometry. Of note, increasing the dose of aspirin to >81 mg daily in patients with diabetes reduced platelet function and the prevalence of impaired response to the drug to values similar to those in patients without diabetes; this finding suggests that, in diabetes, low-dose aspirin does not provide adequate platelet inhibition and that higher aspirin dosing reduces the prevalence of low-responder patients24. In particular, a systematic review combining data from 31 studies showed that patients with diabetes were 36% more likely to have an impaired response to aspirin than those without diabetes and were 70% more likely to have a low drug response with aspirin 100 mg than with 101–325 mg daily25. A possible mechanism underlying HPR when receiving aspirin in patients with diabetes might be a faster recovery of platelet COX1 activity leading to incomplete thromboxane inhibition during the 24-h dosing interval. Indeed, aspirin 100 mg twice daily completely reversed the abnormal thromboxane B2 recovery in patients with diabetes26,27.
Coagulation activity
Multiple pathophysiological mechanisms might contribute to the prothrombotic environment associated with type 2 diabetes (Fig. 2; Table 2), a condition characterized by hyperglycaemia, hyperinsulinaemia, low-grade inflammation, and raised plasma triglyceride levels.
Fat tissue produces less adiponectin and is infiltrated by macrophages that release tumour necrosis factor (TNF), IL-1, and IL-6. This inflammatory state increases the synthesis of plasminogen activator inhibitor 1 (PAI1) and tissue factor (TF) by endothelial cells, as well as coagulation factors, carboxypeptidase B2 (also known as thrombin-activable fibrinolysis inhibitor; TAFI), PAI1, and acute phase proteins, such as complement C3, by the liver. TNF blocks the vasculoprotective insulin pathway involving insulin receptor substrate (IRS)–phosphoinositide 3-kinase (PI3K)–RACα serine/threonine-protein kinase (AKT) and activates inflammation through the signalling pathway involving c-Jun N-terminal kinase (JNK)–inhibitor of nuclear factor-κB kinase (IKK)–nuclear factor-κB (NF-κB). Impaired IRS–PI3K–AKT transduction alters nitric oxide (NO) and insulin-responsive glucose transporter type 4 (GLUT4; also known as SLC2A4) function, whereas the prothrombotic insulin pathway involving growth factor receptor-bound protein (GRB)–mitogen-activated protein kinase (MAPK) remains effective. Inflammation also blunts peroxisome proliferator-activated receptor-γ (PPARγ)-mediated synthesis of the anticoagulant tissue factor pathway inhibitor (TFPI). Hyperglycaemia increases production of reactive oxygen species (ROS) and contributes to endothelial dysfunction. Increased levels of glycated haemoglobin (HbA1c) alter the physiological transport and release of NO. Hyperglycaemia and triglyceridaemia favour the synthesis of coagulation factors and PAI1.
Hyperglycaemia has direct effects on gene transcription of coagulation factors, and hyperglycaemia-induced oxidative stress alters the natural vasculoprotective endothelial glycocalyx28,29. Insulin and proinsulin-like molecules in experimental systems promote the expression and secretion of plasminogen activator inhibitor 1 (PAI1) by hepatocytes and endothelial cells29,30.
Quantitative changes in coagulation factors in patients with diabetes involve higher plasma levels of fibrinogen (the soluble precursor of solid fibrin), coagulation factor VII, and coagulation factor XII; increased endothelial expression of tissue factor and tissue factor–coagulation factor VIIa complex activity; and a reduction in the anticoagulant protein tissue factor pathway inhibitor (TFPI)28,29. These changes culminate in increased thrombin generation and fibrin formation. Quantitative changes of fibrinolytic factors include increased plasma levels of PAI1 and carboxypeptidase B2 (also known as thrombin-activable fibrinolysis inhibitor)29,30. Levels of tissue-type plasminogen activator (t-PA) are also altered, with raised antigen concentrations and reduced t-PA activity29,30.
Qualitative changes of haemostatic factors include the glycation and oxidation of fibrinogen and plasminogen, as well as the incorporation of antiplasmin, PAI1, carboxypeptidase B2, and complement C3 into the fibrin mesh, resulting in a denser clot structure and in delayed spontaneous clot lysis28,29. Increased thrombin generation activates coagulation factor XIII, which cross links and further stabilizes the fibrin network29. Moreover, glycation of haemoglobin in patients with diabetes alters the physiological transport and release of NO by haemoglobin itself, with impaired peripheral vasodilatation, increased insulin resistance, and pro-inflammatory and prothrombotic effects31. An overall hypercoagulable state is demonstrated by the shortening of activated partial thromboplastin time in patients with diabetes compared with individuals without diabetes29.
In summary, our current understanding of the pathophysiological steps implicated in the increased thrombotic risk of patients with diabetes includes increased thrombin and fibrin generation, delayed clot lysis, and reduced NO bioavailability. Future therapies targeting the coagulation, fibrinolysis, and NO systems, rather than platelets alone, might open new frontiers in the prevention and treatment of cardiovascular diseases in these patients.
Primary prevention of cardiovascular disease
Data from Haffner and colleagues supported the concept that patients with diabetes but without previous CAD have a risk of future CAD events similar to that of patients without diabetes but with previous symptomatic CAD32; therefore, aspirin was considered to be of value for primary prevention in patients with diabetes. However, a meta-analysis of 13 studies, involving a total of 45,108 patients, has subsequently shown that the cardiovascular risk of patients with diabetes without previous cardiovascular events is significantly lower than that of patients without diabetes who have previous cardiovascular disease, refuting the notion that diabetes is an equivalent risk factor to CAD33. Nevertheless, the risk of cardiovascular events in patients with diabetes is twofold to fourfold greater than in individuals of the same age and sex without diabetes34,35; this risk should be balanced against the bleeding risk associated with aspirin. An individual patient-level meta-analysis by the Antithrombotic Trialists’ (ATT) Collaboration indicated a similar effect of aspirin on primary prevention of major cardiovascular events in individuals with or without diabetes, with a risk ratio (RR) of 0.88 (95% CI 0.67–1.15) and 0.87 (95% CI 0.79–0.96), respectively36.
Other studies have involved individuals with higher cardiovascular risk profiles than those included in the ATT meta-analysis. In the JPPP trial37, individuals aged 60–85 years with hypertension, dyslipidaemia, or diabetes were randomly assigned to aspirin 100 mg daily or placebo. The 5-year primary outcome event rate (cardiovascular death, nonfatal stroke, and nonfatal myocardial infarction (MI)) was not different in the two groups, but treatment with aspirin significantly increased the risk of extracranial bleeding. Conversely, in the CLIPS trial38, which was performed in patients with peripheral atherosclerosis but without a history of CAD, low-dose aspirin prevented serious vascular events compared with placebo, including in the subgroups with type 2 diabetes.
Six randomized clinical studies, in which patients with diabetes were considered as subgroups (1–22% of the overall trial cohort), have not provided definitive results on the benefit of aspirin in primary prevention of cardiovascular disease39,40,41,42,43,44. Additionally, three randomized trials on primary prevention of cardiovascular disease were conducted specifically in patients with diabetes45,46,47, one of them with a 10-year follow-up48. The oldest study used higher aspirin doses (650 mg daily) versus placebo45, whereas subsequent studies used lower aspirin doses (100 mg, or 81–100 mg daily in the JPAD study)46,47; none of the studies showed a significant reduction in cardiovascular events with aspirin use. A subsequent analysis of these nine studies found that aspirin treatment in patients with diabetes was associated with nonsignificant reductions in CAD (−9%) and cerebrovascular events (−11%) compared with control35. Other meta-analyses produced similar estimates49,50. Overall, these results suggest that aspirin, as currently prescribed, produces only a modest reduction in the risk of cardiovascular events when used for primary prevention in patients with diabetes51.
A word of caution concerns the heterogeneity and limitations of the primary prevention studies, including underpowering for sample size, differences in study designs, primary end points, risk profiles, and aspirin regimens, high rates of aspirin discontinuation during follow-up, and non-uniform reports of statin treatment prevalence. Despite these biases, the less-than-expected clinical benefit of aspirin in patients with diabetes might be explained by the presence of multiple thrombotic pathways involved in the pathogenesis of cardiovascular events and by the aforementioned platelet features (including residual hyperreactivity or increased turnover, especially in individuals with poor glycaemic control)52. Various aspirin formulations have been tested in attempts to improve its efficacy and/or limit adverse effects. An enteric-coated formulation was associated with increased rates of low responsiveness to the drug owing to reduced bioavailability compared with uncoated aspirin; this phenomenon might contribute to aspirin pseudo-resistance, with possibly lower clinical benefits in patients with diabetes receiving the coated formulation53. An extended-release, 162.5 mg formulation of aspirin was shown to provide a sustained antiplatelet effect over 24 h in patients with type 2 diabetes and multiple cardiovascular risk factors, with a favourable tolerability profile54; however, no extensive clinical data are available with this formulation. The previously mentioned increase in aspirin dose has been suggested to improve the effectiveness of the drug, but with inconsistent results and uncertain net benefit, the latter largely owing to the increased risk of adverse effects as a consequence of the reduced production of gastric prostaglandins, which protect against gastrointestinal bleeding, compared with the use of low doses of aspirin24,55. As discussed above, a twice-daily administration of low-dose aspirin has been tested in pharmacodynamic studies in patients with diabetes and concomitant CAD because this regimen might provide greater platelet inhibition than once-daily administration26,56; however, evidence of improved cardiovascular outcomes with twice-daily administration is still needed and is currently under investigation in the randomized ANDAMAN trial57.
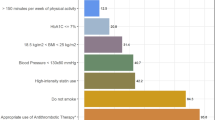
Regarding the bleeding risk of aspirin, a population-based study found a significant excess of both gastrointestinal and intracranial bleeding with aspirin compared with no aspirin use in individuals without diabetes but not in patients with diabetes58; this finding indirectly suggests a lower suppression of platelet function by standard aspirin doses in those with diabetes than in those without diabetes. Taken together, the overall available data on aspirin in patients with diabetes indicate a modest relative reduction in the risk of cardiovascular events in primary prevention offset against a 55% relative increase in the risk of extracranial bleeding, which is mainly gastrointestinal58,59. Therefore, we recommend that patients aged ≥50 years with a 10-year cardiovascular risk >10% according to the Systematic Coronary Risk Estimation (SCORE) risk charts59 and without high bleeding risk should initiate aspirin. Moreover, long-term follow-up of studies of aspirin in primary prevention of cardiovascular disease showed that low doses of this drug are associated with a reduction in colorectal cancer incidence and mortality from 5 years onwards60. Although data on cancer prevention with the use of aspirin specifically in patients with diabetes are lacking, this issue needs to be addressed, because patients with type 2 diabetes have an increased risk of some types of cancer, particularly of the pancreas and the colon–rectum61. Table 3 summarizes our recommendations on the use of aspirin in adults with diabetes and no pre-existing cardiovascular disease. Two primary prevention trials in diabetes (ASCEND62 and ACCEPT-D63) will help to assess the benefit–risk profile of low-dose aspirin in preventing multiple outcomes. The ASCEND trial62, in which adults with diabetes but no evident cardiovascular disease were randomly assigned to receive 100 mg of aspirin daily or placebo, was published in August 2018; in this study, the absolute benefits of aspirin in preventing serious vascular events were largely counterbalanced by the increased risk of bleeding.
Secondary prevention in CAD
Stable CAD
The presence of diabetes impairs clinical outcomes in patients with cardiovascular disease, in whom meta-analyses of randomized trials indicate an overall 40% increase in major adverse cardiovascular events (MACE; that is, MI, stroke, or cardiovascular death) compared with patients without diabetes36,64. In the setting of secondary prevention of cardiovascular disease in patients with diabetes and stable CAD, data from the ATT Collaboration on approximately 4,500 patients showed a 4.2% absolute reduction in cardiovascular events with aspirin compared with placebo at 60-month follow-up, which was similar to that observed in those without diabetes36. Clopidogrel has been extensively investigated in patients with stable CAD undergoing PCI or treated conservatively. Table 4 summarizes the main results of randomized trials evaluating antiplatelet therapy in patients with stable CAD for primary efficacy end points in the overall population and in the subgroup with diabetes. In the CAPRIE trial65, patients with established cardiovascular disease were randomly assigned to clopidogrel (75 mg daily) or aspirin (325 mg daily). The subgroup analysis including 3,866 patients with diabetes demonstrated that the use of clopidogrel instead of aspirin prevented 21 adverse events (vascular death, MI, stroke, and hospitalization for ischaemic or bleeding complications) per 1,000 patients per year (compared with 9 events prevented per 1,000 patients per year in individuals without diabetes)66. Of note, the cardiovascular protection provided by clopidogrel was even higher in the subgroup of patients with diabetes and receiving insulin therapy (38 events prevented per 1,000 patients per year)66.
The addition of clopidogrel to aspirin in stable patients was evaluated in the secondary prevention cohort (that is, patients with previous MI, stroke, or PAD) of the CHARISMA trial67. At 28-month follow-up in the subgroup of patients with diabetes, protection from cardiovascular events with the use of DAPT was no greater in patients with diabetes than in those without diabetes, but the bleeding risk was higher in those with diabetes68. Of note, the use of aspirin plus clopidogrel compared with aspirin alone led to a significant reduction in the combined end point including cardiovascular death, MI, or stroke only in patients without diabetic nephropathy (6.7% versus 7.7%; P = 0.048), whereas no significant benefit was observed in those with diabetic nephropathy (11.4% versus 12.0%; P = 0.84)68. Therefore, the current evidence indicates that the net clinical benefit of routinely adding clopidogrel to aspirin in patients with diabetes and stable CAD treated medically is questionable because the modest ischaemic protection is outweighed by the increased risk of bleeding.
The CREDO trial69 investigated the effects of preprocedural clopidogrel loading in addition to aspirin in patients undergoing elective PCI. Interestingly, in both the overall population and the subgroup with diabetes, pretreatment with 300 mg clopidogrel led to significant clinical benefit compared with downstream administration of clopidogrel, but only when the loading dose was given ≥6 h before the intervention. This finding essentially reflects the fact that several hours are needed to achieve maximal platelet inhibition after a 300 mg clopidogrel load70,71. Interestingly, a meta-analysis showed no reduction in MACE incidence with 12-month versus 6-month DAPT among patients with diabetes undergoing PCI with drug-eluting stent implantation for a variety of coronary syndromes72. On the basis of the available data, the antithrombotic strategies in patients with diabetes and different features of stable CAD are shown in Table 3.
The COMPASS trial73 investigators explored the safety and efficacy of inhibition of thrombin generation, as provided by rivaroxaban (2.5 mg twice daily) on a background of aspirin therapy or by rivaroxaban (5.0 mg twice daily) alone in patients with stable coronary or peripheral atherosclerotic disease (91% with CAD and 62% with previous MI). This study was prematurely stopped for superiority of the rivaroxaban plus aspirin versus aspirin alone group after a mean follow-up of 23 months in terms of MACE (cardiovascular death, MI, or stroke), all-cause death, and cardiovascular death. Consistent with the overall study results, in patients with diabetes (n = 6,922), the addition of rivaroxaban to aspirin resulted in a significantly lower incidence of MACE (HR 0.74, 95% CI 0.61–0.90) with higher rates of major bleeding (HR 1.70, 95% CI 1.25–2.31); the net clinical benefit was significantly in favour of combination therapy, without increased rates of intracranial bleeding. Future guidelines on clinical practice in patients with stable CAD are likely to include recommendations derived from the COMPASS study.
ACS
The clinical benefit provided by antiplatelet therapy in patients with diabetes and an ACS has been clearly demonstrated. Table 4 reports the main results of randomized trials evaluating antiplatelet therapy in patients with ACS for primary efficacy end points in the overall population and in the subgroup with diabetes. Meta-analysis data have shown that the reduction of MACE with aspirin versus placebo after MI is similar in patients with or without diabetes (from 22% to 18% in those with diabetes and from 16% to 13% in those without diabetes)36,64. These data also indicate that, despite aspirin therapy, the recurrence of ischaemic events in patients with diabetes and an ACS is approximately 20% over a mean of 2.5 years36. Although a persistently higher platelet reactivity when taking aspirin might partly explain this elevated recurrence rate74, the randomized CURRENT-OASIS 7 trial75 demonstrated no outcome improvement with higher-dose aspirin (300–325 mg daily versus 75–100 mg daily for 30 days) in a mixed population of patients with or without diabetes who were admitted to hospital for ACS, with no significant interaction between primary outcome and diabetic status.
Additional inhibition of the P2Y12 platelet receptor on a background of aspirin therapy, aimed at further reducing adverse events in patients with ACS, was first evaluated in the CURE trial76, in which patients with non-ST-segment elevation (NSTE)-ACS already receiving aspirin were randomly assigned to clopidogrel (300 mg loading dose followed by 75 mg daily maintenance dose) or placebo for a mean duration of 9 months. A lower rate of MACE was observed with the combination of clopidogrel and aspirin regardless of diabetic status; although no significant interaction was present, the relative reduction of adverse cardiovascular events at 1 year with clopidogrel in the 2,840 patients with diabetes was numerically lower than the reduction in those without diabetes (14% versus 20% relative risk reduction). This finding might be a result of the more pronounced interindividual variability of clopidogrel responsiveness in diabetes, resulting in a higher prevalence of HPR77,78,79.
As discussed above, the mechanisms behind poor clopidogrel-induced P2Y12 inhibition in patients with diabetes might include impaired drug metabolism leading to reduced active metabolite generation as well as dysregulation of the P2Y12 signalling pathway80,81. The OPTIMUS-3 study82 found that, in patients with diabetes, prasugrel was associated with faster and greater platelet inhibition, as well as lesser interindividual variability, than clopidogrel, even when the latter was given at high doses (600 mg loading plus 150 mg daily). Similar pharmacodynamic results were obtained with ticagrelor83. Given the pharmacodynamic drawbacks of clopidogrel in patients with diabetes, which are potentially overcome with the newer, more potent P2Y12 inhibitors, the subgroup analyses of diabetic populations from phase III studies comparing prasugrel and ticagrelor versus clopidogrel in the setting of ACS are of particular interest.
In the TRITON-TIMI 38 trial84, the use of prasugrel instead of standard-dose clopidogrel after PCI for ACS led to a higher relative risk reduction of MACE (cardiovascular death, stroke, and MI) at 15 months in patients with diabetes than in those without diabetes (30% versus 14%; P = 0.009 for interaction). Importantly, prasugrel did not increase the rates of major bleeding in the diabetes group. In this trial, the greatest reduction in adverse events with prasugrel was observed in patients receiving insulin therapy (37% relative risk reduction versus 26% in patients with diabetes who were not receiving insulin)84. The TRILOGY-ACS study85 included patients with NSTE-ACS managed without revascularization and randomly assigned to receive prasugrel or clopidogrel for up to 30 months in addition to aspirin. A subgroup analysis according to diabetes status showed no difference in the incidence of MACE (cardiovascular death, MI, or stroke) with either antiplatelet strategy in patients either with (n = 3,539) or without (n = 5,767) diabetes86. In the PLATO trial87, patients with ACS were randomly assigned to receive ticagrelor (180 mg loading dose and then 90 mg twice daily) or clopidogrel (300 or 600 mg loading dose and then 75 mg daily), irrespective of the subsequent therapeutic strategy. A lower rate of MACE at 1 year with ticagrelor was observed both in patients with diabetes and in those without diabetes (12% and 17% relative reductions, respectively). This ischaemic protection largely outweighed the elevation in non-CABG surgery-related major bleeding observed in the ticagrelor group. Of note, the use of ticagrelor had a strong effect in decreasing MACE, all-cause death, and stent thrombosis in patients with diabetes and haemoglobin A1c (HbA1c) ≥6%.
A pharmacodynamic comparison of prasugrel versus ticagrelor in patients with diabetes and CAD showed that ticagrelor exerts similar or greater inhibition of ADP-induced platelet reactivity than prasugrel in both the acute and the chronic phases of treatment88. A clinical comparison between prasugrel and ticagrelor is currently being investigated in the ongoing ISAR-REACT 5 trial89, in which diabetes is a predefined subgroup90. On the basis of the available evidence, we recommend consistent and highly effective platelet inhibition with the newer, more potent P2Y12 inhibitors for patients with ACS and concomitant diabetes, given their high baseline risk profile; therefore, DAPT with aspirin plus prasugrel or ticagrelor should be the first-line antiplatelet strategy up to 1 year in this setting, especially in patients without high bleeding risk (Table 3).
Given the poorer long-term cardiovascular outcomes after MI in patients with diabetes than in those without diabetes, DAPT prolongation is a particularly relevant issue in this setting. The DAPT study91 explored the efficacy and safety of prolonging DAPT beyond 1 year with aspirin plus a thienopyridine (prasugrel or clopidogrel) versus aspirin alone for 18 more months in patients with stable CAD or ACS who had undergone implantation of a drug-eluting stent. In the subgroup analysis according to diabetes status (n = 3,391 with and n = 8,257 without diabetes), a significant interaction existed between MI risk reduction and prolonged DAPT in favour of patients without diabetes (58% relative reduction versus 28% in patients with diabetes; P = 0.02 for interaction)92. Regardless of diabetes status, the rate of stent thrombosis was lower and the rate of bleeding events was higher in the DAPT group (stent thrombosis: 75% relative reduction in patients without diabetes versus 53% in patients with diabetes, P = 0.21 for interaction; moderate or severe bleeding: 71% relative increase in patients without diabetes versus 47% in patients with diabetes, P = 0.61 for interaction)92. Of note, withdrawal of thienopyridine in both groups resulted in a consistent numerical increase in early ischaemic events in patients with or without diabetes92.
The PEGASUS-TIMI 54 trial93 explored the efficacy and safety of DAPT prolongation (with ticagrelor 90 mg or 60 mg twice daily plus aspirin versus aspirin alone) in patients with a history of MI (in the past 1–3 years). The reduction in MACE with either ticagrelor dose was consistent in patients with or without diabetes. Given their higher risk of events, patients with diabetes had a greater absolute benefit from ticagrelor, with a 3-year number needed to treat for MACE (pooling results of both ticagrelor doses) of 67 compared with 91 in those without diabetes. Additionally, in patients with diabetes, ticagrelor reduced the rate of cardiovascular death by 22%. As in patients without diabetes, ticagrelor significantly increased the rates of nonfatal major bleeding in patients with diabetes (HR 2.56, P = 0.0004). Regardless of diabetic status, the net clinical outcome was better with the 60 mg than with the 90 mg ticagrelor dose — that is, similar ischaemic protection but lower bleeding risk.
The use of ticagrelor for secondary prevention of cardiovascular disease is being investigated specifically in patients with diabetes in the THEMIS trial94, where approximately 20,000 patients with type 2 diabetes and CAD, but without previous MI, are being randomly assigned to either ticagrelor (90 mg or 60 mg twice daily) or placebo for up to 4 years. As we wait for more evidence on the net clinical benefit of more aggressive antithrombotic therapies in patients with diabetes and ACS, especially in the long term, we summarize our therapeutic recommendations, which are based on currently available data, in Table 3.
Other antiplatelet strategies in CAD
Given the hyperreactivity of platelets in patients with diabetes, other antiplatelet strategies have been tested to improve cardiovascular outcomes in these individuals with CAD. Specifically, additional drugs have been investigated as adjunctive treatment to aspirin alone or to aspirin plus a P2Y12 inhibitor (triple therapy) to achieve a more complete, multi-pathway platelet inhibition.
Cilostazol, a selective and reversible inhibitor of phosphodiesterase 3 that has effects on various cell lines, including platelets, by increasing intracellular cAMP concentrations95, was evaluated in pharmacodynamic studies as part of a triple-therapy strategy in patients with diabetes and CAD. The OPTIMUS-2 study96 found that the addition of cilostazol to DAPT improved inhibition of platelet P2Y12 signalling. Moreover, cilostazol might decrease platelet aggregability in patients with diabetes and HPR when taking clopidogrel97. Clinical benefits of cilostazol in addition to DAPT were observed in a nonrandomized study of Asian patients undergoing PCI, mainly in terms of reduced rates of target-lesion revascularization and stent thrombosis98; in randomized studies, this improvement in outcomes was found to be greatest for patients with diabetes99,100,101. Other investigations have evaluated the effects of cilostazol in the setting of PCI for ACS. In the retrospective Korean Acute MI Registry, 4,203 patients with ST-segment elevation MI undergoing primary PCI received either DAPT (aspirin plus clopidogrel) or triple therapy (DAPT plus cilostazol)102; the latter strategy was associated with a significantly lower incidence of cardiac death, all-cause death, and MACE at 8-month follow-up. Interestingly, subgroup analysis showed that patients who were older (aged >65 years), female, or had diabetes derived greater benefit from triple therapy102. In another study, 1,212 Chinese patients with ACS undergoing PCI were randomly assigned to DAPT (aspirin plus clopidogrel) or DAPT plus cilostazol for 6 months103. Triple therapy resulted in a 35% relative reduction in MACE, without excess bleeding; the subgroup of 263 patients with diabetes derived particular benefit from the addition of cilostazol (HR 0.47, 95% CI 0.23–0.96)103.
Other attractive antiplatelet agents are thromboxane antagonists that potentially block the interaction of both aspirin-sensitive and aspirin-insensitive agonists, theoretically providing potent platelet inhibition, especially in patients with diabetes who show both platelet hyperreactivity and increased thromboxane-dependent platelet activation104. Among these drugs, terutroban and picotamide have been investigated in patients with cerebrovascular disease or PAD (see section below). Pharmacodynamic studies have evaluated EV-077, a thromboxane receptor antagonist and thromboxane synthase inhibitor, in patients with diabetes and stable CAD receiving monotherapy with aspirin or clopidogrel. In this setting, EV-077 provided further platelet inhibition through multiple signalling pathways; however, no specific in vivo data are available for EV-077105. Ridogrel, a thromboxane A2 (TXA2) synthase inhibitor and TXA2–prostaglandin endoperoxide receptor antagonist, was investigated in 907 patients with MI treated with thrombolysis and enrolled in the RAPT study106. Ridogrel was found to be not superior to aspirin in increasing the fibrinolytic efficacy of streptokinase, as assessed before discharge. In a post hoc analysis, fewer recurrent ischaemic cardiac events occurred in the ridogrel group, without an excess in serious bleeding complications. No specific data in the subgroup with diabetes are available from this investigation.
Targeting thrombin after ACS
Despite DAPT, patients with diabetes and established atherosclerosis have a high residual risk of recurrent events87. This situation is likely to be the direct consequence of endothelial dysfunction, oxidative stress, vascular inflammation, abnormal platelet reactivity, and decreased responsiveness to antiplatelet agents, but increased thrombin generation might also have a role107. Thrombin is a potent platelet agonist; therefore, thrombin inhibition might provide substantial suppression of platelet function in addition to reduced fibrin production. Inhibition of the effects of thrombin can be achieved by direct thrombin inhibitors, such as bivalirudin or dabigatran; by direct factor Xa inhibitors, such as apixaban, edoxaban, or rivaroxaban; or by platelet proteinase-activated receptor 1 (PAR1) antagonists, such as vorapaxar.
The effects of thrombin inhibition on cardiovascular outcomes in patients with ACS have been investigated in the ATLAS ACS–TIMI 46 trial108 with rivaroxaban (2.5 mg twice daily) and in the TRA 2°P-TIMI 50 trial109 with vorapaxar. In both studies, the newer agent under investigation was compared with placebo on a background of DAPT with aspirin plus clopidogrel. In the ATLAS ACS–TIMI 46 trial108, no significant interaction was observed between prevention of MACE, reduction in mortality, and increase in bleeding by rivaroxaban and diabetic status. In the TRA 2°P-TIMI 50 trial109, the net clinical benefit with vorapaxar in the population with previous MI was numerically more pronounced in patients with diabetes (HR 0.77, P = 0.006) than in those without diabetes (HR 0.88, P = 0.04). Various factors have precluded the widespread integration of the findings from these two studies into clinical practice. These factors include the delayed initiation of the additional antithrombotic agent after the coronary event, the lack of comparison data between rivaroxaban or vorapaxar in addition to DAPT with aspirin plus clopidogrel or aspirin plus either prasugrel or ticagrelor, and the bleeding concerns associated with triple antithrombotic therapy.
Secondary prevention after stroke or TIA
Diabetes is independently associated with an increased risk of ischaemic stroke, and the presence of diabetes after a stroke negatively affects recovery and predisposes to recurrent cerebral events110. Despite advances in pharmacotherapy, the optimal antiplatelet regimen for patients with diabetes and cerebrovascular disease (with or without concomitant CAD) remains debated because the use of more potent antithrombotic drugs after a stroke or transient ischaemic attack (TIA) might cause a considerable increase in the risk of intracranial bleeding111, casting uncertainty on the risk–benefit ratio in the setting of secondary prevention. No specific, large study on aspirin for secondary prevention of stroke in patients with diabetes exists, so the available data come from subgroup analyses of more general studies. Two large meta-analyses of cardiovascular secondary prevention trials, including patients with a previous stroke, indicate that the clinical benefit of aspirin compared with placebo is consistent in patients with or without diabetes36,64. As mentioned, in the CAPRIE trial65, the reduction of cardiovascular events with clopidogrel versus aspirin was more pronounced in patients with diabetes, and one-third of the patients in CAPRIE had a previous stroke or TIA.
The concept that the residual risk of recurrent events in patients with diabetes might be related to higher on-treatment platelet reactivity when receiving single antiplatelet therapy has led to investigations aimed at assessing whether DAPT or the newer P2Y12 inhibitors are associated with an increased clinical benefit in patients with diabetes and cerebral events. In randomized trials, the combination of low-dose aspirin and extended-release dipyridamole (ERDP) (400 mg daily) after a stroke showed a clinical benefit compared with aspirin alone in the ESPS-2 study112 and the ESPRIT trial113, in which patients with diabetes composed 15% and 18% of the trial cohorts, respectively. Conversely, outcomes after ischaemic stroke were similar with aspirin plus ERDP versus clopidogrel in the PRoFESS trial114 regardless of diabetic status (prevalence of diabetes 28%). The use of ERDP in clinical practice is currently discouraged and limited by the high incidence of adverse effects, such as headache and gastrointestinal symptoms.
In the CHARISMA trial67, >40% of patients had diabetes and approximately 36% had a previous stroke or TIA at randomization. In the subgroup analysis restricted to 4,320 patients with previous ischaemic stroke or TIA, the addition of clopidogrel was associated with a trend towards a reduction in recurrent stroke (4.9% versus 6.1%; HR 0.80, 95% CI 0.62–1.03)115. Among the overall population of the CHARISMA study67, the incidence of intracranial bleeding was similar (0.3%) in patients receiving aspirin plus clopidogrel or aspirin alone, and no significant difference in severe bleeding emerged in the subgroup analysis of patients with prior cerebrovascular disease (1.7% with placebo versus 1.9% with clopidogrel; HR 1.11, 95% CI 0.71–1.73)115. The efficacy and safety profile of the aspirin plus clopidogrel combination from these analyses formed the basis for subsequent specific randomized studies.
The MATCH trial116 investigators randomly allocated patients with stroke or TIA and at least one cardiovascular risk factor to clopidogrel versus aspirin plus clopidogrel. No significant reduction in stroke recurrence was observed in the DAPT group, whereas bleeding complications were increased. These findings were consistent in groups defined according to diabetes status. Conversely, in the CHANCE trial117, selectively performed in a Chinese population of 5,170 patients with minor stroke or TIA, DAPT with aspirin plus clopidogrel, given for 21 days after the index event, decreased the rate of recurrent stroke at 90 days compared with aspirin alone, without a significant increase in bleeding events, irrespective of diabetes status. The POINT trial118 investigated aspirin alone (50–325 mg daily) versus aspirin plus clopidogrel in patients with minor ischaemic stroke or high-risk TIA. The primary efficacy end point was a composite of ischaemic stroke, MI, or vascular death at 90 days. In the DAPT group, a significant 25% relative reduction in the main outcome measure and a 2.3-fold increased risk of major bleeding were observed. However, no outcome data according to diabetes status are so far available.
Terutroban, a selective thromboxane–prostaglandin receptor antagonist, was compared with aspirin in the PERFORM trial119 for the prevention of cerebral and cardiovascular events in patients with previous stroke or TIA. The incidence of the primary end point (a composite of ischaemic stroke, MI, or vascular death, not including haemorrhagic death) at 28 months was 11% in both groups (HR 1.02, 95% CI 0.94–1.12). Similar results were obtained in the subgroup of 5,299 patients with diabetes. However, a significant increase in any bleeding events was observed in patients receiving terutroban (HR 1.09, 95% CI 1.01–1.17); therefore, this agent is not an alternative to aspirin in patients with cerebral ischaemic events.
In the context of the newer, more potent antiplatelet agents, a history of any stroke or TIA is a contraindication for prasugrel therapy. Conversely, ticagrelor, which is contraindicated only after a haemorrhagic stroke, has been tested (180 mg loading dose followed by 90 mg twice daily for 90 days) versus aspirin in 13,199 patients within 24 h of a non-severe stroke or high-risk TIA in the SOCRATES trial120. Ticagrelor was not superior to aspirin in reducing the primary end point of stroke, MI, or death at 3 months in either the overall population or the subgroup (25% of patients) with diabetes (8.4% versus 9.5%; P = 0.99).
In summary, the available data do not provide clear evidence in support of DAPT (that is, aspirin plus clopidogrel versus aspirin or clopidogrel alone) or of any more potent antiplatelet agent in patients with diabetes and ischaemic cerebral events. Antiplatelet approaches in this setting are shown in Table 3.
Secondary prevention in PAD
Up to one-third of patients with PAD have diabetes121. The prevalence of PAD is higher among patients with diabetes, and in these individuals, PAD is more severe, progresses more rapidly, and entails a higher risk of recurrent ischaemic events and amputation than in those without diabetes122. In addition, PAD is a potent marker of systemic atherosclerosis and increased cardiovascular risk122. Data on antiplatelet strategies in this setting are limited and are often derived from subgroup analyses of investigations devoted primarily to the management of patients with CAD, in whom PAD was often asymptomatic or not clinically apparent.
In a meta-analysis of 18 trials, involving approximately 5,000 patients with PAD, those receiving aspirin had a modest, nonsignificant 12% lower risk of cardiovascular events than those receiving placebo123. This analysis was heavily driven by two studies performed on patients with diabetes, the POPADAD study47 (n = 1,276) and the VA-Cooperative trial124 (n = 231), which both showed no benefit of aspirin or of aspirin plus dipyridamole versus placebo. Various aspects might explain the apparent lower efficacy of aspirin in patients with PAD and diabetes. First, current evidence might be underpowered. Alternatively, or additionally, these patients might have phenotypic and biological differences from those with predominant CAD. For instance, the presence of PAD might render platelet activation more critically dependent on ADP than on TXA2 production104. Third, as stated above, the pharmacodynamics of aspirin might differ in patients with diabetes compared with those without diabetes24. Finally, the hypothesis that the inhibition of COX1-dependent TXA2 biosynthesis is insufficient in this setting is substantiated by data indicating that the use of picotamide, an inhibitor of both TXA2 receptors and TXA2 synthase, might increase survival versus aspirin in patients with diabetes and PAD (RR 0.55, 95% CI 0.31–0.98)125.
In an analysis of 6,452 patients with PAD included in the CAPRIE trial65, approximately one-third of whom had diabetes, clopidogrel recipients had a significant 24% and 22% relative risk reduction in cardiovascular mortality and cardiovascular events compared with aspirin, respectively, with a similar benefit in the subset of patients with diabetes. A post hoc analysis of the CHARISMA study126 on 3,096 patients with PAD demonstrated no benefit with aspirin plus clopidogrel versus aspirin alone in terms of reduction of the composite MACE end point; significant decreases in the rates of MI (HR 0.63, 95% CI 0.42–0.96, P = 0.029) and hospitalization for ischaemic events (HR 0.81, 95% CI 0.68–0.95, P = 0.011) were observed in the DAPT group at the expense of a twofold increased risk of minor bleeding. No specific outcome data are available in the subgroup of patients with PAD and concomitant diabetes in this trial.
The presence of PAD impairs cardiovascular prognosis in patients with a history of MI, leading to the rationale that multi-pathway platelet inhibition might improve clinical outcome in this setting127. In the PEGASUS-TIMI 54 trial128, 1,143 patients (5%) had known PAD at randomization, and 42% of these had diabetes. Given the high baseline risk profile of this PAD subgroup, the addition of ticagrelor to aspirin (versus aspirin alone) was associated with a greater absolute reduction in MACE in these patients than in patients without PAD (4.1% at 3 years and a number needed to treat of 25 versus 1.0% at 3 years and a number needed to treat of 100) without a significant increase in major bleeding128. Of note, the use of ticagrelor in patients with PAD significantly reduced major adverse limb events (a composite of acute limb ischaemia or peripheral revascularization: HR 0.65, 95% CI 0.44–0.95). No specific outcome data with ticagrelor plus aspirin versus aspirin alone have been published in the subgroup of patients with PAD and concomitant diabetes in this trial.
In patients with symptomatic PAD (more than one-third with diabetes) from the EUCLID trial129, ticagrelor was shown not to be superior to clopidogrel in reducing major cardiovascular and limb events, with similar rates of major bleeding. Among patients with symptomatic PAD (27% with diabetes) from the TRA 2°P-TIMI 50 trial130,131, the use of vorapaxar versus placebo did not significantly decrease MACE but did significantly reduced acute limb ischaemia and the rate of peripheral revascularization; the benefit in limb events was accompanied by an increased risk of bleeding, yielding an uncertain net benefit of vorapaxar in patients with PAD.
While acknowledging the paucity of data from specific studies, indications concerning antiplatelet therapy for cardiovascular prevention in patients with diabetes and PAD reflect the recommendations of the AHA/ACC and ESC guidelines on the general population of patients with PAD132,133 (Table 3). The cardiovascular protection afforded by rivaroxaban in addition to aspirin therapy versus aspirin alone in the COMPASS trial134 was consistent in the subgroups with PAD (27% of the overall population) or CAD and occurred regardless of diabetic status; in particular, patients with PAD and diabetes receiving rivaroxaban plus aspirin had a twofold higher absolute reduction of the composite end point, including cardiovascular death, MI, and stroke, than patients with PAD but without diabetes. Of note, in the PAD population of the COMPASS study, combination therapy was also associated with a significant 46% relative reduction in major adverse limb events, including major amputation. These findings are likely to influence future guideline recommendations. The ongoing VOYAGER PAD trial135, an international, randomized, double-blind, placebo-controlled study, is evaluating the efficacy and safety of the vascular dose of rivaroxaban (2.5 mg twice daily) in patients with symptomatic PAD undergoing peripheral surgical and/or endovascular revascularization136.
Prevention of thromboembolism in AF
Diabetes increases the risk of developing AF and worsens the prognosis of patients with AF137. In the ACCORD trial138, patients with new-onset AF and diabetes had an increased risk of all-cause death (HR 2.65), MI (HR 2.10), and heart failure (HF) (HR 3.80) compared with those without diabetes. The presence of diabetes raises the incidence of thromboembolic events (stroke or systemic embolism) in patients with AF, explaining the inclusion of diabetes in contemporary scores for stroke prediction139. In a meta-analysis of seven studies including >12,000 patients with AF, an overall 70% relative increase in the risk of thromboembolic complications was observed in patients with versus those without diabetes, with a yearly incidence ranging from 3.6% to 8.6%140. This variability reflects differences in study designs, definitions of outcome measures, patients’ baseline risk profile, concomitant therapies, and types of diabetic populations included. Analyses have identified a duration of diabetes >3 years as an independent predictor of ischaemic stroke, which is a stronger predictor than glycaemic control141. A subgroup analysis from the European PREFER in AF registry, involving >5,000 patients with AF, showed that patients with diabetes receiving insulin therapy had a more than twofold higher risk of stroke or systemic embolism at 1 year than patients without diabetes, whereas diabetes not treated with insulin did not entail a significantly increased risk142.
Oral anticoagulant therapy, historically with warfarin, is the cornerstone of treatment to reduce thromboembolic risk in patients with AF, including those with diabetes143. In the past decade, NOACs have revolutionized antithrombotic therapy to prevent AF-related thromboembolic events because of their favourable characteristics: predictable dose–response, rapid offset and onset, fixed doses, no interaction with food, limited interactions with other drugs, and no need for routine monitoring. A meta-analysis of four phase III trials comparing NOACs versus warfarin in patients with AF showed NOACs to be associated with a significant 19% relative reduction in the combined end point including any stroke or systemic embolism (P < 0.0001) and a 14% relative reduction in major bleeding (P < 0.06) compared with well-managed warfarin144. Among 71,683 patients receiving NOACs, the prevalence of diabetes ranged from 23.3% (in the RE-LY study) to 40.0% (in the ROCKET-AF study). Of note, in these trials, blood glucose levels and HbA1c values were not routinely available, and patients with a creatinine clearance <30 ml/min were excluded. Therefore, patients with severe diabetic nephropathy, who are at even higher risk of cardiovascular complications, were excluded145. In these phase III studies, no interaction was found between diabetes status and clinical efficacy of the NOACs versus warfarin, although the safety superiority of apixaban versus warfarin was lost among patients with AF and diabetes (P = 0.003 for interaction)145. Moreover, a study-level meta-analysis of these trials added more robust evidence on the topic146 (Fig. 3). Compared with warfarin, the use of NOACs decreased the rates of intracranial bleeding regardless of diabetic status (43% relative reduction versus warfarin in patients with diabetes and 38% in those without diabetes; P = 0.47 for interaction)146. Also, in patients with diabetes, use of NOACs versus warfarin significantly reduced the rate of cardiovascular death (RR 0.83, 95% CI 0.72–0.96, P = 0.01)146. To date, no data on a direct comparison between different NOACs in patients with diabetes are available; therefore, the choice of NOAC in patients with diabetes is not supported by specific evidence but should be guided by general principles and take into account the comorbidities associated with diabetes. On the basis of the available data, we recommend antithrombotic strategies for patients with AF and diabetes (Table 3).
Prevention and treatment of VTE
VTE encompasses a spectrum of diseases that extend from deep-vein thrombosis to pulmonary embolism. Patients with diabetes are characterized by a prothrombotic milieu and have an increased risk of incident and recurrent VTE147,148,149. However, the role of diabetes as an independent predictor of unprovoked or idiopathic VTE has been questioned150. Overall, diabetes is now considered only a weak predisposing factor for VTE, and from a practical point of view, its presence does not influence standard antithrombotic treatment151. Accordingly, no specific subgroup analyses have been performed on patients with diabetes in VTE trials evaluating the efficacy of vitamin K antagonists or aspirin versus placebo152 or, lately, in trials comparing NOACs and warfarin153.
Figure 4 indicates the different types of antithrombotic therapies at different time points after a VTE episode. Evaluation of diabetic status, as well as of other venous and arterial risk factors, is relevant in order to offer appropriate general lifestyle and prevention measures. Indeed, the presence of diabetes might guide the choice of oral anticoagulant therapy, call for closer patient follow-up, or act as a warning for thromboprophylaxis. Given the increased risk of major bleeding in patients with diabetes, NOACs might be the preferred therapeutic option, considering their better manageability and safety profile, especially in an outpatient setting153. Moreover, patients with diabetes require closer follow-up because they more frequently develop kidney disease or events related to concomitant PAD or CAD. These conditions might require the introduction of antiplatelet treatment even during the course of anticoagulation for a VTE episode. Patients with diabetes are often receiving polypharmacy; however, no interaction has been reported between the effects of NOACs and glucose-lowering drugs154. Finally, a contemporary issue is the real-world underutilization of thromboprophylaxis in vulnerable patient populations in the acute hospital care setting155. Owing to the prothrombotic status documented in patients with diabetes, thromboprophylaxis with anticoagulants should be strongly encouraged in these patients, when indicated.
New glucose-lowering drugs and thrombosis
The evidence reviewed in this Consensus Statement clearly identifies the alterations in mechanisms of thrombosis as important pathological factors for the development of macrovascular complications in patients with diabetes. Nevertheless, the effects of antidiabetic drugs on the pathophysiology of thrombosis are mostly unknown. Previous studies suggested that metformin decreases platelet activation156,157 through various mechanisms, including protection of mitochondrial function, reduction of membrane damage, and prevention of oxidative stress158,159. Similarly, thiazolidinediones have been shown to reduce prothrombotic mechanisms by modulating platelet function through the activation of 5ʹ-AMP-activated protein kinase and of peroxisome proliferator-activated receptor-γ160,161,162,163.
In the past decade, various new compounds with different mechanisms of action have been developed to counteract impairments in glucose metabolism164. Indeed, since 2008, regulatory agencies have requested pharmaceutical companies to perform randomized, controlled trials with cardiovascular events as outcome measures specifically to characterize the cardiovascular profile of new glucose-lowering drugs. Various studies have been completed (Table 5), and others are ongoing or being planned. Although their direct effects on coagulation and platelet activity are so far unknown, analysis of the available results from the cardiovascular outcome trials might help to identify the agents that are most likely to affect thrombosis in diabetes.
The EXAMINE165, SAVOR-TIMI 53 (ref.166), and TECOS167 studies were among the first cardiovascular outcome trials evaluating a new class of glucose-lowering drugs, the dipeptidyl peptidase 4 (DPP4) inhibitors. All these studies met the primary noninferiority end point for cardiovascular safety of these drugs versus placebo in patients with diabetes and established cardiovascular disease. However, the use of DPP4 inhibitors was not associated with significant benefits on the prespecified measures of cardiovascular outcome. Conversely, a signal of increased risk of HF with saxagliptin was highlighted in the SAVOR-TIMI 53 trial166, and a post hoc analysis of the EXAMINE trial168 indicated a nonsignificant 0.9% higher absolute incidence of hospitalization for HF in the alogliptin group.
The results of the EMPA-REG OUTCOME trial169 were a major breakthrough in the field, showing a significant 14% relative risk reduction in the rate of the composite primary end point, including cardiovascular death, MI, or stroke, with empagliflozin, an inhibitor of the sodium/glucose cotransporter 2 (SGLT2), causing glycosuria. Favourable cardiovascular effects have been subsequently obtained with another drug in the same class, canagliflozin170. Of note, the use of either empagliflozin or canagliflozin led to a significant decrease in hospitalizations for HF.
A lower incidence of cardiovascular events has been demonstrated with two different glucagon-like peptide 1 receptor (GLP1R) agonists, liraglutide and semaglutide171,172, but not with lixisenatide173. Finally, a borderline significant (P = 0.06) 0.9% absolute reduction in the primary cardiovascular outcome measure was found with exenatide in the EXCEL study174. Of note, unlike SGLT2 inhibitors, treatment with GLP1R agonists resulted in significant protection from individual components of the composite outcome measure (MI and stroke) but was neutral in terms of rates of HF.
Taken together, these data indicate that, despite a similar control on levels of HbA1c, the new glucose-lowering drugs have different effects on cardiovascular outcomes, with potential mechanisms of cardiovascular protection that are independent of glucose control (Fig. 5). The encouraging results of studies evaluating GLP1R agonists suggest that the protection from MI and stroke provided by these drugs derives from effects on the progression and destabilization of atherosclerotic plaques. These results also suggest that the incretin system is involved in the pathophysiology of arterial thrombosis. In this regard, platelets have been shown to express GLP1Rs constitutively, and in vitro stimulation of platelets with liraglutide or exenatide has inhibitory effects on platelet aggregation175,176. Conversely, SGLT2 inhibition resulted in a consistent reduction in HF-related events, suggesting that this class of agents acts through different pathways; however, mechanistic studies are needed to clarify the pathways of cardiovascular protection of these compounds. In particular, data on the effect of SGLT2 inhibitors on platelet function and thrombosis are lacking.
Several conditions contribute to a prothrombotic state in diabetes mellitus, including (but not limited to) hyperglycaemia, hypertension, obesity, insulin resistance, and impaired kidney function. The results of cardiovascular outcome trials and ad hoc studies suggest that both sodium/glucose cotransporter 2 (SGLT2) inhibitors and incretin hormones can counteract these conditions (to different extents and through different pathways), with possible benefits on the diabetic prothrombotic milieu. Some evidence also suggests a direct regulation of platelet activity by glucagon-like peptide 1 (dashed line). Thick lines indicate mainly direct actions; thin lines indicate mainly indirect effects. ROS, reactive oxygen species.
Conclusions
Patients with diabetes have a prothrombotic state and a higher risk of cardiovascular events than those without diabetes. Antithrombotic therapies in these patients might be associated with a higher absolute reduction of recurrent events than in patients without diabetes. More aggressive antithrombotic strategies might be specifically indicated in patients with diabetes and cardiovascular diseases, including the use of the newer, more potent P2Y12 inhibitors in patients with ACS, DAPT prolongation beyond 1 year in patients with MI, and short-term DAPT in selected patients with acute ischaemic stroke. However, the expected ischaemic protection from antithrombotic drugs must always be weighed against the risk of drug-related bleeding. Available evidence on the efficacy and safety of antithrombotic drugs in patients with diabetes at risk of or with cardiovascular diseases often derives from underpowered observational studies or subgroup analyses of trials not specifically performed in patients with diabetes; therefore, large investigations specifically focused on patients with diabetes are needed and should be encouraged.
References
Colwell, J. A., Lopes-Virella, M. & Halushka, P. V. Pathogenesis of atherosclerosis in diabetes mellitus. Diabetes Care 4, 121–133 (1981).
American Diabetes Association. 9. Cardiovascular disease and risk management: standards of medical care in diabetes-2018. Diabetes Care 41, S86–S104 (2018).
Fox, C. S. et al. Update on prevention of cardiovascular disease in adults with type 2 diabetes mellitus in light of recent evidence: a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care 38, 1777–1803 (2015).
Authors/Task Force Members et al. ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD: the Task Force on diabetes, pre-diabetes, and cardiovascular diseases of the European Society of Cardiology (ESC) and developed in collaboration with the European Association for the Study of Diabetes (EASD). Eur. Heart J. 34, 3035–3087 (2013).
Ferreiro, J. L. & Angiolillo, D. J. Diabetes and antiplatelet therapy in acute coronary syndrome. Circulation 123, 798–813 (2011).
Blache, D. et al. Glycated albumin with loss of fatty acid binding capacity contributes to enhanced arachidonate oxygenation and platelet hyperactivity: relevance in patients with type 2 diabetes. Diabetes 64, 960–972 (2015).
Sagel, J., Colwell, J. A., Crook, L. & Laimins, M. Increased platelet aggregation in early diabetus mellitus. Ann. Intern. Med. 82, 733–738 (1975).
Ferroni, P., Basili, S., Falco, A. & Davì, G. Platelet activation in type 2 diabetes mellitus. J. Thromb. Haemost. 2, 1282–1291 (2004).
Eibl, N. et al. Improved metabolic control decreases platelet activation markers in patients with type-2 diabetes. Eur. J. Clin. Invest. 34, 205–209 (2004).
Gurbel, P. A., Bliden, K. P., Hiatt, B. L. & O’Connor, C. M. Clopidogrel for coronary stenting: response variability, drug resistance, and the effect of pretreatment platelet reactivity. Circulation 107, 2908–2913 (2003).
Angiolillo, D. J. et al. High clopidogrel loading dose during coronary stenting: effects on drug response and interindividual variability. Eur. Heart J. 25, 1903–1910 (2004).
Bonello, L. et al. Consensus and future directions on the definition of high on-treatment platelet reactivity to adenosine diphosphate. J. Am. Coll. Cardiol. 56, 919–933 (2010).
Angiolillo, D. J. et al. Impact of platelet reactivity on cardiovascular outcomes in patients with type 2 diabetes mellitus and coronary artery disease. J. Am. Coll. Cardiol. 50, 1541–1547 (2007).
Angiolillo, D. J. et al. Impact of P2Y(12) inhibitory effects induced by clopidogrel on platelet procoagulant activity in type 2 diabetes mellitus patients. Thromb. Res. 124, 318–322 (2009).
Valenti, R. et al. Prognostic impact of high residual platelet reactivity after chronic total occlusion percutaneous coronary intervention in patients with diabetes mellitus. Int. J. Cardiol. 201, 561–567 (2015).
Marcucci, R. et al. Residual platelet reactivity is associated with clinical and laboratory characteristics in patients with ischemic heart disease undergoing PCI on dual antiplatelet therapy. Atherosclerosis 195, e217–e223 (2007).
Marcucci, R., Grifoni, E. & Giusti, B. On-treatment platelet reactivity: state of the art and perspectives. Vascul. Pharmacol. 77, 8–18 (2016).
Ferreira, I. A., Eybrechts, K. L., Mocking, A. I., Kroner, C. & Akkerman, J.-W. IRS-1 mediates inhibition of Ca2+ mobilization by insulin via the inhibitory G-protein Gi. J. Biol. Chem. 279, 3254–3264 (2004).
Goldstein, S., Simpson, A. & Saenger, P. Hepatic drug metabolism is increased in poorly controlled insulin-dependent diabetes mellitus. Acta Endocrinol. 123, 550–556 (1990).
Kudo, T. et al. Altered expression of CYP in TSOD mice: a model of type 2 diabetes and obesity. Xenobiotica 39, 889–902 (2009).
Patoine, D. et al. Modulation of CYP3a expression and activity in mice models of type 1 and type 2 diabetes. Pharmacol. Res. Perspect. 2, e00082 (2014).
Erlinge, D. et al. Patients with poor responsiveness to thienopyridine treatment or with diabetes have lower levels of circulating active metabolite, but their platelets respond normally to active metabolite added ex vivo. J. Am. Coll. Cardiol. 52, 1968–1977 (2008).
Watala, C. Blood platelet reactivity and its pharmacological modulation in (people with) diabetes mellitus. Curr. Pharm. Des. 11, 2331–2365 (2005).
DiChiara, J. et al. The effect of aspirin dosing on platelet function in diabetic and nondiabetic patients: an analysis from the aspirin-induced platelet effect (ASPECT) study. Diabetes 56, 3014–3019 (2007).
Simpson, S. H., Abdelmoneim, A. S., Omran, D. & Featherstone, T. R. Prevalence of high on-treatment platelet reactivity in diabetic patients treated with aspirin. Am. J. Med. 127, 95 (2014).
Dillinger, J.-G. et al. Biological efficacy of twice daily aspirin in type 2 diabetic patients with coronary artery disease. Am. Heart J. 164, 600–606 (2012).
Rocca, B. et al. The recovery of platelet cyclooxygenase activity explains interindividual variability in responsiveness to low-dose aspirin in patients with and without diabetes. J. Thromb. Haemost. 10, 1220–1230 (2012).
Vazzana, N., Ranalli, P., Cuccurullo, C. & Davì, G. Diabetes mellitus and thrombosis. Thromb. Res. 129, 371–377 (2012).
Yudkin, J. S. Abnormalities of coagulation and fibrinolysis in insulin resistance. Evidence for a common antecedent? Diabetes Care 22, C25–C30 (1999).
Alzahrani, S. H. & Ajjan, R. A. Coagulation and fibrinolysis in diabetes. Diab. Vasc. Dis. Res. 7, 260–273 (2010).
Andreotti, F. et al. Anemia contributes to cardiovascular disease through reductions in nitric oxide. J. Appl. Physiol. 122, 414–417 (2017).
Haffner, S. M., Lehto, S., Rönnemaa, T., Pyörälä, K. & Laakso, M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N. Engl. J. Med. 339, 229–234 (1998).
Bulugahapitiya, U., Siyambalapitiya, S., Sithole, J. & Idris, I. Is diabetes a coronary risk equivalent? Systematic review and meta-analysis. Diabet. Med. 26, 142–148 (2009).
Emerging Risk Factors Collaboration. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet 375, 2215–2222 (2010).
Pignone, M. et al. Aspirin for primary prevention of cardiovascular events in people with diabetes: a position statement of the American Diabetes Association, a scientific statement of the American Heart Association, and an expert consensus document of the American College. Circulation 121, 2694–2701 (2010).
Antithrombotic Trialists’ Collaboration. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet 373, 1849–1860 (2009).
Ikeda, Y. et al. Low-dose aspirin for primary prevention of cardiovascular events in Japanese patients 60 years or older with atherosclerotic risk factors: a randomized clinical trial. JAMA 312, 2510–2520 (2014).
Critical Leg Ischaemia Prevention Study Group. Prevention of serious vascular events by aspirin amongst patients with peripheral arterial disease: randomized, double-blind trial. J. Intern. Med. 261, 276–284 (2007).
Peto, R. et al. Randomised trial of prophylactic daily aspirin in British male doctors. Br. Med. J. (Clin. Res. Ed). 296, 313–316 (1988).
Steering Committee of the Physicians’ Health Study Research Group. Final report on the aspirin component of the ongoing Physicians’ Health Study. N. Engl. J. Med. 321, 129–135 (1989).
Fowkes, F. G. et al. Aspirin for prevention of cardiovascular events in a general population screened for a low ankle brachial index: a randomized controlled trial. JAMA 303, 841–848 (2010).
Hansson, L. et al. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. HOT study group. Lancet 351, 1755–1762 (1998).
Roncaglioni, M. C. & Collaborative Group of the Primary Prevention Project. Low-dose aspirin and vitamin E in people at cardiovascular risk: a randomised trial in general practice. Collaborative Group of the Primary Prevention project. Lancet 357, 89–95 (2001).
Ridker, P. M. et al. A randomized trial of low-dose aspirin in the primary prevention of cardiovascular disease in women. N. Engl. J. Med. 352, 1293–1304 (2005).
Kassoff, A. et al. Aspirin effects on mortality and morbidity in patients with diabetes mellitus: early treatment diabetic retinopathy study report 14. JAMA 268, 1292–1300 (1992).
Ogawa, H. et al. Low-dose aspirin for primary prevention of atherosclerotic events in patients with type 2 diabetes: a randomized controlled trial. JAMA 300, 2134–2141 (2008).
Belch, J. et al. The prevention of progression of arterial disease and diabetes (POPADAD) trial: factorial randomised placebo controlled trial of aspirin and antioxidants in patients with diabetes and asymptomatic peripheral arterial disease. BMJ 337, a1840 (2008).
Saito, Y. et al. Low-dose aspirin for primary prevention of cardiovascular events in patients with type 2 diabetes mellitus: 10-year follow-up of a randomized controlled trial. Circulation 135, 659–670 (2017).
De Berardis, G. et al. Aspirin for primary prevention of cardiovascular events in people with diabetes: meta-analysis of randomised controlled trials. BMJ 339, b4531 (2009).
Calvin, A. D. et al. Aspirin for the primary prevention of cardiovascular events: a systematic review and meta-analysis comparing patients with and without diabetes. Diabetes Care 32, 2300–2306 (2009).
Capodanno, D. & Angiolillo, D. J. Aspirin for primary cardiovascular risk prevention and beyond in diabetes mellitus. Circulation 134, 1579–1594 (2016).
Santilli, F., Pignatelli, P., Violi, F. & Davì, G. Aspirin for primary prevention in diabetes mellitus: from the calculation of cardiovascular risk and risk/benefit profile to personalised treatment. Thromb. Haemost. 114, 876–882 (2015).
Bhatt, D. L. et al. Enteric coating and aspirin nonresponsiveness in patients with type 2 diabetes mellitus. J. Am. Coll. Cardiol. 69, 603–612 (2017).
Gurbel, P. A. et al. Antiplatelet effect durability of a novel, 24-hour, extended-release prescription formulation of acetylsalicylic acid in patients with type 2 diabetes mellitus. Am. J. Cardiol. 118, 1941–1947 (2016).
Bethel, M. A. et al. Randomized controlled trial comparing impact on platelet reactivity of twice-daily with once-daily aspirin in people with type 2 diabetes. Diabet. Med. 33, 224–230 (2016).
Capodanno, D. et al. Pharmacodynamic effects of different aspirin dosing regimens in type 2 diabetes mellitus patients with coronary artery disease. Circ. Cardiovasc. Interv. 4, 180–187 (2011).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02520921 (2017).
De Berardis, G. et al. Association of aspirin use with major bleeding in patients with and without diabetes. JAMA 307, 2286–2294 (2012).
Piepoli, M. F. et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts) developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 37, 2315–2381 (2016).
Rothwell, P. M. et al. Short-term effects of daily aspirin on cancer incidence, mortality, and non-vascular death: analysis of the time course of risks and benefits in 51 randomised controlled trials. Lancet 379, 1602–1612 (2012).
Johnson, J. A. et al. Diabetes and cancer (1): evaluating the temporal relationship between type 2 diabetes and cancer incidence. Diabetologia 55, 1607–1618 (2012).
ASCEND Study Collaborative Group. Effects of aspirin for primary prevention in persons with diabetes mellitus. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa1804988 (2018).
De Berardis, G. et al. Aspirin and simvastatin combination for cardiovascular events prevention trial in diabetes (ACCEPT-D): design of a randomized study of the efficacy of low-dose aspirin in the prevention of cardiovascular events in subjects with diabetes mellitus treated with statins. Trials 8, 21 (2007).
Antiplatelet Trialists’ Collaboration. Collaborative overview of randomised trials of antiplatelet therapy — 1. Prevention of death, myocardial infarction, and stroke by prolonged antiplatelet therapy in various categories of patients. BMJ 308, 81–106 (1994).
CAPRIE Steering Committee. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 348, 1329–1339 (1996).
Bhatt, D. L. et al. Amplified benefit of clopidogrel versus aspirin in patients with diabetes mellitus. Am. J. Cardiol. 90, 625–628 (2002).
Bhatt, D. L. et al. Patients with prior myocardial infarction, stroke, or symptomatic peripheral arterial disease in the CHARISMA trial. J. Am. Coll. Cardiol. 49, 1982–1988 (2007).
Dasgupta, A. et al. Clinical outcomes of patients with diabetic nephropathy randomized to clopidogrel plus aspirin versus aspirin alone (a post hoc analysis of the clopidogrel for high atherothrombotic risk and ischemic stabilization, management, and avoidance [CHARISMA] trial). Am. J. Cardiol. 103, 1359–1363 (2009).
Steinhubl, S. R. et al. Early and sustained dual oral antiplatelet therapy following percutaneous coronary intervention: a randomized controlled trial. JAMA 288, 2411–2420 (2002).
Patti, G. et al. Randomized trial of high loading dose of clopidogrel for reduction of periprocedural myocardial infarction in patients undergoing coronary intervention: results from the ARMYDA-2 (Antiplatelet therapy for Reduction of MYocardial Damage during Angioplasty) study. Circulation 111, 2099–2106 (2005).
Taubert, D. et al. Pharmacokinetics of clopidogrel after administration of a high loading dose. Thromb. Haemost. 92, 311–316 (2004).
Gargiulo, G. et al. Short term versus long term dual antiplatelet therapy after implantation of drug eluting stent in patients with or without diabetes: systematic review and meta-analysis of individual participant data from randomised trials. BMJ 355, i5483 (2016).
Connolly, S. J. et al. Rivaroxaban with or without aspirin in patients with stable coronary artery disease: an international, randomised, double-blind, placebo-controlled trial. Lancet 391, 205–218 (2018).
Eikelboom, J. W. et al. Aspirin-resistant thromboxane biosynthesis and the risk of myocardial infarction, stroke, or cardiovascular death in patients at high risk for cardiovascular events. Circulation 105, 1650–1655 (2002).
CURRENT-OASIS 7 Investigators. Dose comparisons of clopidogrel and aspirin in acute coronary syndromes. N. Engl. J. Med. 363, 930–942 (2010).
Yusuf, S. et al. effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N. Engl. J. Med. 345, 494–502 (2001).
Angiolillo, D. J. et al. Platelet function profiles in patients with type 2 diabetes and coronary artery disease on combined aspirin and clopidogrel treatment. Diabetes 54, 2430–2435 (2005).
Mangiacapra, F. et al. Comparison of platelet reactivity and periprocedural outcomes in patients with versus without diabetes mellitus and treated with clopidogrel and percutaneous coronary intervention. Am. J. Cardiol. 106, 619–623 (2010).
Patti, G., Proscia, C. & Di Sciascio, G. Antiplatelet therapy in patients with diabetes mellitus and acute coronary syndrome. Circ. J. 78, 33–41 (2014).
Ueno, M. et al. Functional profile of the platelet P2Y12 receptor signalling pathway in patients with type 2 diabetes mellitus and coronary artery disease. Thromb. Haemost. 105, 730–732 (2011).
Angiolillo, D. J. et al. Impaired responsiveness to the platelet P2Y12 receptor antagonist clopidogrel in patients with type 2 diabetes and coronary artery disease. J. Am. Coll. Cardiol. 64, 1005–1014 (2014).
Angiolillo, D. J. et al. A pharmacodynamic comparison of prasugrel versus high-dose clopidogrel in patients with type 2 diabetes mellitus and coronary artery disease: results of the Optimizing anti-Platelet Therapy In diabetes MellitUS (OPTIMUS)-3 Trial. Eur. Heart J. 32, 838–846 (2011).
Laine, M. et al. Ticagrelor versus prasugrel in diabetic patients with an acute coronary syndrome. A pharmacodynamic randomised study. Thromb. Haemost. 111, 273–278 (2014).
Wiviott, S. D. et al. Greater clinical benefit of more intensive oral antiplatelet therapy with prasugrel in patients with diabetes mellitus in the trial to assess improvement in therapeutic outcomes by optimizing platelet inhibition with prasugrel–thrombolysis in myocardial infarction 38. Circulation 118, 1626–1636 (2008).
Roe, M. T. et al. Prasugrel versus clopidogrel for acute coronary syndromes without revascularization. N. Engl. J. Med. 367, 1297–1309 (2012).
Dalby, A. J. et al. Dual antiplatelet therapy in patients with diabetes and acute coronary syndromes managed without revascularization. Am. Heart J. 188, 156–166 (2017).
James, S. et al. Ticagrelor vs. clopidogrel in patients with acute coronary syndromes and diabetes: a substudy from the PLATelet inhibition and patient Outcomes (PLATO) trial. Eur. Heart J. 31, 3006–3016 (2010).
Franchi, F. et al. Pharmacodynamic comparison of prasugrel versus ticagrelor in patients with type 2 diabetes mellitus and coronary artery disease: the OPTIMUS (Optimizing Antiplatelet Therapy in Diabetes Mellitus)-4 study. Circulation 134, 780–792 (2016).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT01944800 (2017).
Schulz, S. et al. Randomized comparison of ticagrelor versus prasugrel in patients with acute coronary syndrome and planned invasive strategy — design and rationale of the iNtracoronary Stenting and Antithrombotic Regimen: Rapid Early Action for Coronary Treatment (ISAR-REACT) 5 trial. J. Cardiovasc. Transl Res. 7, 91–100 (2014).
Mauri, L. et al. Twelve or 30 months of dual antiplatelet therapy after drug-eluting stents. N. Engl. J. Med. 371, 2155–2166 (2014).
Meredith, I. T. et al. Diabetes mellitus and prevention of late myocardial infarction after coronary stenting in the randomized dual antiplatelet therapy study. Circulation 133, 1772–1782 (2016).
Bhatt, D. L. et al. Reduction in ischemic events with ticagrelor in diabetic patients with prior myocardial infarction in PEGASUS-TIMI 54. J. Am. Coll. Cardiol. 67, 2732–2740 (2016).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT01991795 (2018).
Goto, S. Cilostazol: potential mechanism of action for antithrombotic effects accompanied by a low rate of bleeding. Atheroscler. Suppl. 6, 3–11 (2005).
Angiolillo, D. J. et al. A randomized study assessing the impact of cilostazol on platelet function profiles in patients with diabetes mellitus and coronary artery disease on dual antiplatelet therapy: results of the OPTIMUS-2 study. Eur. Heart J. 29, 2202–2211 (2008).
Capranzano, P. et al. Pharmacodynamic effects of adjunctive cilostazol therapy in patients with coronary artery disease on dual antiplatelet therapy: impact of high on-treatment platelet reactivity and diabetes mellitus status. Catheter. Cardiovasc. Interv. 81, 42–49 (2013).
Lee, S.-W. et al. Triple versus dual antiplatelet therapy after coronary stenting: impact on stent thrombosis. J. Am. Coll. Cardiol. 46, 1833–1837 (2005).
Biondi-Zoccai, G. G. et al. Systematic review and meta-analysis of randomized clinical trials appraising the impact of cilostazol after percutaneous coronary intervention. Am. Heart J. 155, 1081–1089 (2008).
Lee, S.-W. et al. Drug-eluting stenting followed by cilostazol treatment reduces late restenosis in patients with diabetes mellitus the DECLARE-DIABETES Trial (a randomized comparison of triple antiplatelet therapy with dual antiplatelet therapy after drug-eluting stent implantation in diabetic patients). J. Am. Coll. Cardiol. 51, 1181–1187 (2008).
Ahn, Y. et al. Randomized comparison of cilostazol versus clopidogrel after drug-eluting stenting in diabetic patients — clilostazol for diabetic patients in drug-eluting stent (CIDES) trial. Circ. J. 72, 35–39 (2008).
Chen, K.-Y. et al. Triple versus dual antiplatelet therapy in patients with acute ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Circulation 119, 3207–3214 (2009).
Han, Y. et al. Cilostazol in addition to aspirin and clopidogrel improves long-term outcomes after percutaneous coronary intervention in patients with acute coronary syndromes: a randomized, controlled study. Am. Heart J. 157, 733–739 (2009).
Davì, G. et al. Thromboxane biosynthesis and platelet function in type II diabetes mellitus. N. Engl. J. Med. 322, 1769–1774 (1990).
Rollini, F. et al. Pharmacodynamic effects of EV-077 in patients with diabetes mellitus and coronary artery disease on aspirin or clopidogrel monotherapy: results of an in vitro pilot investigation. J. Thromb. Thrombolysis 37, 131–138 (2014).
The RAPT Investigators. Randomized trial of ridogrel, a combined thromboxane A2 synthase inhibitor and thromboxane A2/prostaglandin endoperoxide receptor antagonist, versus aspirin as adjunct to thrombolysis in patients with acute myocardial infarction. The Ridogrel Versus Aspirin Patency Trial (RAPT). Circulation 89, 588–595 (1994).
Undas, A., Wiek, I., Stêpien, E., Zmudka, K. & Tracz, W. Hyperglycemia is associated with enhanced thrombin formation, platelet activation, and fibrin clot resistance to lysis in patients with acute coronary syndrome. Diabetes Care 31, 1590–1595 (2008).
Mega, J. L. et al. Rivaroxaban in patients with a recent acute coronary syndrome. N. Engl. J. Med. 366, 9–19 (2012).
Cavender, M. A. et al. Vorapaxar in patients with diabetes mellitus and previous myocardial infarction: findings from the thrombin receptor antagonist in secondary prevention of atherothrombotic ischemic events-TIMI 50 trial. Circulation 131, 1047–1053 (2015).
Jørgensen, H., Nakayama, H., Raaschou, H. O. & Olsen, T. S. Stroke in patients with diabetes. The Copenhagen Stroke Study. Stroke 25, 1977–1984 (1994).
Capodanno, D., Alberts, M. & Angiolillo, D. J. Antithrombotic therapy for secondary prevention of atherothrombotic events in cerebrovascular disease. Nat. Rev. Cardiol. 13, 609–622 (2016).
Diener, H. C. et al. European Stroke Prevention Study. 2. Dipyridamole and acetylsalicylic acid in the secondary prevention of stroke. J. Neurol. Sci. 143, 1–13 (1996).
ESPRIT Study Group. Aspirin plus dipyridamole versus aspirin alone after cerebral ischaemia of arterial origin (ESPRIT): randomised controlled trial. Lancet 367, 1665–1673 (2006).
Sacco, R. L. et al. Aspirin and extended-release dipyridamole versus clopidogrel for recurrent stroke. N. Engl. J. Med. 359, 1238–1251 (2008).
Hankey, G. J. et al. Effect of clopidogrel plus ASA versus ASA early after TIA and ischaemic stroke: a substudy of the CHARISMA trial. Int. J. Stroke 6, 3–9 (2011).
Diener, H.-C. et al. Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high-risk patients (MATCH): randomised, double-blind, placebo-controlled trial. Lancet 364, 331–337 (2004).
Wang, Y. et al. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack. N. Engl. J. Med. 369, 11–19 (2013).
Johnston, S. C. et al. Clopidogrel and aspirin in acute ischemic stroke and high-risk TIA. N. Engl. J. Med. 379, 215–225 (2018).
Bousser, M.-G. et al. Terutroban versus aspirin in patients with cerebral ischaemic events (PERFORM): a randomised, double-blind, parallel-group trial. Lancet 377, 2013–2022 (2011).
Johnston, S. C. et al. Ticagrelor versus aspirin in acute stroke or transient ischemic attack. N. Engl. J. Med. 375, 35–43 (2016).
Marso, S. P. & Hiatt, W. R. Peripheral arterial disease in patients with diabetes. J. Am. Coll. Cardiol. 47, 921–929 (2006).
Jude, E. B., Oyibo, S. O., Chalmers, N. & Boulton, A. J. Peripheral arterial disease in diabetic and nondiabetic patients: a comparison of severity and outcome. Diabetes Care 24, 1433–1437 (2001).
Feldman, D. N. & Moussa, I. D. Efficacy of aspirin for secondary prevention in patients with peripheral artery disease. Expert Rev. Cardiovasc. Ther. 7, 1203–1207 (2009).
Colwell, J. A. et al. V. A. cooperative study of antiplatelet agents in diabetic patients after amputation for gangrene: unobserved, sudden, and unexpected deaths. J. Diabet. Compl. 3, 191–197 (1989).
Neri Serneri, G. G., Coccheri, S., Marubini, E. & Violi, F., Drug Evaluation in Atherosclerotic Vascular Disease in Diabetics (DAVID) Study Group. Picotamide, a combined inhibitor of thromboxane A2 synthase and receptor, reduces 2-year mortality in diabetics with peripheral arterial disease: the DAVID study. Eur. Heart J. 25, 1845–1852 (2004).
Cacoub, P. P. et al. Patients with peripheral arterial disease in the CHARISMA trial. Eur. Heart J. 30, 192–201 (2009).
Huynh, K. Antiplatelet therapy: ticagrelor reduces cardiac events in patients with PAD or diabetes. Nat. Rev. Cardiol. 13, 310–311 (2016).
Bonaca, M. P. et al. Ticagrelor for prevention of ischemic events after myocardial infarction in patients with peripheral artery disease. J. Am. Coll. Cardiol. 67, 2719–2728 (2016).
Hiatt, W. R. et al. Ticagrelor versus clopidogrel in symptomatic peripheral artery disease. N. Engl. J. Med. 376, 32–40 (2017).
Bonaca, M. P. et al. Vorapaxar in patients with peripheral artery disease: results from TRA2°P-TIMI 50. Circulation 127, 1522–1529 (2013).
Bonaca, M. P. et al. Acute limb ischemia and outcomes with vorapaxar in patients with peripheral artery disease: results from the trial to assess the effects of vorapaxar in preventing heart attack and stroke in patients with atherosclerosis–thrombolysis in myocardial infarction 50 (TRA2°P-TIMI 50). Circulation 133, 997–1005 (2016).
Gerhard-Herman, M. D. et al. 2016 AHA/ACC guideline on the management of patients with lower extremity peripheral artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 135, e726–e779 (2017).
European Stroke Organisation. ESC Guidelines on the diagnosis and treatment of peripheral artery diseases: document covering atherosclerotic disease of extracranial carotid and vertebral, mesenteric, renal, upper and lower extremity arteries: the Task Force on the Diagnosis and Treatment of Peripheral Artery Diseases of the European Society of Cardiology (ESC). Eur. Heart J. 32, 2851–2906 (2011).
Anand, S. S. et al. Rivaroxaban with or without aspirin in patients with stable peripheral or carotid artery disease: an international, randomised, double-blind, placebo-controlled trial. Lancet 391, 219–229 (2018).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02504216 (2018).
Capell, W. H. et al. Rationale and design for the Vascular Outcomes study of ASA along with rivaroxaban in endovascular or surgical limb revascularization for peripheral artery disease (VOYAGER PAD). Am. Heart J. 199, 83–91 (2018).
Huxley, R. R., Filion, K. B., Konety, S. & Alonso, A. Meta-analysis of cohort and case-control studies of type 2 diabetes mellitus and risk of atrial fibrillation. Am. J. Cardiol. 108, 56–62 (2011).
Fatemi, O. et al. Impact of intensive glycemic control on the incidence of atrial fibrillation and associated cardiovascular outcomes in patients with type 2 diabetes mellitus (from the Action to Control Cardiovascular Risk in Diabetes Study). Am. J. Cardiol. 114, 1217–1222 (2014).
Kirchhof, P. et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur. Heart J. 37, 2893–2962 (2016).
Stroke Risk in Atrial Fibrillation Working Group. Independent predictors of stroke in patients with atrial fibrillation: a systematic review. Neurology 69, 546–554 (2007).
Ashburner, J. M. et al. Effect of diabetes and glycemic control on ischemic stroke risk in AF patients: ATRIA study. J. Am. Coll. Cardiol. 67, 239–247 (2016).
Patti, G. et al. Insulin-requiring versus noninsulin-requiring diabetes and thromboembolic risk in patients with atrial fibrillation: PREFER in AF. J. Am. Coll. Cardiol. 69, 409–419 (2017).
Hart, R. G., Pearce, L. A. & Aguilar, M. I. Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann. Intern. Med. 146, 857–867 (2007).
Ruff, C. T. et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet 383, 955–962 (2014).
Itzhaki Ben Zadok, O. & Eisen, A. Use of non-vitamin K oral anticoagulants in people with atrial fibrillation and diabetes mellitus. Diabet. Med. 35, 548–556 (2018).
Patti, G., Di Gioia, G., Cavallari, I. & Nenna, A. Safety and efficacy of nonvitamin K antagonist oral anticoagulants versus warfarin in diabetic patients with atrial fibrillation: a study-level meta-analysis of phase III randomized trials. Diabetes. Metab. Res. Rev. 33, e2876 (2017).
Petrauskiene, V., Falk, M., Waernbaum, I., Norberg, M. & Eriksson, J. W. The risk of venous thromboembolism is markedly elevated in patients with diabetes. Diabetologia 48, 1017–1021 (2005).
Movahed, M.-R., Hashemzadeh, M. & Jamal, M. M. The prevalence of pulmonary embolism and pulmonary hypertension in patients with type II diabetes mellitus. Chest 128, 3568–3571 (2005).
Piazza, G. et al. Venous thromboembolism in patients with diabetes mellitus. Am. J. Med. 125, 709–716 (2012).
Heit, J. A. et al. Is diabetes mellitus an independent risk factor for venous thromboembolism? A population-based case-control study. Arterioscler. Thromb. Vasc. Biol. 29, 1399–1405 (2009).
Konstantinides, S. V. et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur. Heart J. 35, 3145–3151 (2014).
Castellucci, L. A. et al. Efficacy and safety outcomes of oral anticoagulants and antiplatelet drugs in the secondary prevention of venous thromboembolism: systematic review and network meta-analysis. BMJ 347, f5133 (2013).
Blann, A. D. & Lip, G. Y. H. Non-vitamin K antagonist oral anticoagulants (NOACs) for the management of venous thromboembolism. Heart 102, 975–983 (2016).
Di Minno, A. et al. Old and new oral anticoagulants: food, herbal medicines and drug interactions. Blood Rev. 31, 193–203 (2017).
Cohen, A. T. et al. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross-sectional study. Lancet 371, 387–394 (2008).
De Caterina, R. et al. The direct effects of metformin on platelet function in vitro. Eur. J. Clin. Pharmacol. 37, 211–213 (1989).
Formoso, G. et al. Decreased in vivo oxidative stress and decreased platelet activation following metformin treatment in newly diagnosed type 2 diabetic subjects. Diabetes Metab. Res. Rev. 24, 231–237 (2008).
Xin, G. et al. Metformin uniquely prevents thrombosis by inhibiting platelet activation and mtDNA release. Sci. Rep. 6, 36222 (2016).
Randriamboavonjy, V. et al. Metformin reduces hyper-reactivity of platelets from patients with polycystic ovary syndrome by improving mitochondrial integrity. Thromb. Haemost. 114, 569–578 (2015).
Khanolkar, M. P. et al. Rosiglitazone produces a greater reduction in circulating platelet activity compared with gliclazide in patients with type 2 diabetes mellitus — an effect probably mediated by direct platelet PPARgamma activation. Atherosclerosis 197, 718–724 (2008).
Schöndorf, T. et al. The fixed combination of pioglitazone and metformin improves biomarkers of platelet function and chronic inflammation in type 2 diabetes patients: results from the PIOfix study. J. Diabetes Sci. Technol. 5, 426–432 (2011).
Sidhu, J. S., Cowan, D., Tooze, J. A. & Kaski, J.-C. Peroxisome proliferator-activated receptor-gamma agonist rosiglitazone reduces circulating platelet activity in patients without diabetes mellitus who have coronary artery disease. Am. Heart J. 147, e25 (2004).
Li, D. et al. The effects of PPAR-gamma ligand pioglitazone on platelet aggregation and arterial thrombus formation. Cardiovasc. Res. 65, 907–912 (2005).
Maddaloni, E. & Pozzilli, P. SMART diabetes: the way to go (Safe and Multifactorial Approach to reduce the Risk for Therapy in diabetes). Endocrine 46, 3–5 (2014).
White, W. B. et al. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N. Engl. J. Med. 369, 1327–1335 (2013).
Scirica, B. M. et al. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N. Engl. J. Med. 369, 1317–1326 (2013).
Green, J. B. et al. Effect of sitagliptin on cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 373, 232–242 (2015).
Zannad, F. et al. Heart failure and mortality outcomes in patients with type 2 diabetes taking alogliptin versus placebo in EXAMINE: a multicentre, randomised, double-blind trial. Lancet 385, 2067–2076 (2015).
Zinman, B. et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 373, 2117–2128 (2015).
Neal, B. et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N. Engl. J. Med. 377, 644–657 (2017).
Marso, S. P. et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 375, 311–322 (2016).
Marso, S. P. et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 375, 1834–1844 (2016).
Pfeffer, M. A. et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N. Engl. J. Med. 373, 2247–2257 (2015).
Holman, R. R. et al. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 377, 1228–1239 (2017).
Cameron-Vendrig, A. et al. Glucagon-like peptide 1 receptor activation attenuates platelet aggregation and thrombosis. Diabetes 65, 1714–1723 (2016).
Barale, C. et al. Glucagon-like peptide 1-related peptides increase nitric oxide effects to reduce platelet activation. Thromb. Haemost. 117, 1115–1128 (2017).
Valgimigli, M. et al. 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS: the Task Force for dual antiplatelet therapy in coronary artery disease of the European Society of Cardiology (ESC) and of the European Association for Cardio-Thoracic Surgery (EACTS). Eur. Heart J. 4, 213–260 (2018).
Levine, G. N. et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 68, 1082–1115 (2016).
Acknowledgements
Reviewer information
Nature Reviews Cardiology thanks D. Angiolillo, R. Mehran, and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Consortia
Contributions
All authors researched data for the article and contributed to writing the manuscript. G.P. and I.C. discussed the content of the article, and G.P., I.C., and R.D.C. reviewed and edited it before submission.
Corresponding authors
Ethics declarations
Competing interests
G.P. is a speaker, consultant, and/or advisory board member for Amgen, AstraZeneca, Bayer, BMS-Pfizer, Boehringer Ingelheim, Daiichi Sankyo, Malesci, MSD, PIAM, Sanofi, and Sigma-Tau. F.A. is a speaker, consultant, and/or advisory board member for Actelion, Amgen, Bayer, BMS-Pfizer, Boehringer Ingelheim, Daiichi Sankyo, International Menarini Foundation, and Medscape. E.M. has received speaker fees from Merck-Serono and grant support from AstraZeneca and Lilly. D.P. is a speaker or has received consultant and/or advisory fees from Bayer, BMS-Pfizer, Boehringer Ingelheim, and Daiichi Sankyo. G.R. has received consultant and speaker fees from Bayer, Boehringer Ingelheim, and Daiichi Sankyo. R.D.C. has received fees from AstraZeneca, Bayer, BMS-Pfizer, Boehringer Ingelheim, Daiichi Sankyo, Lilly, Novartis, and Sanofi-Aventis and has received grant support from AstraZeneca, Bayer, BMS-Pfizer, Boehringer Ingelheim, and Daiichi Sankyo. The other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Patti, G., Cavallari, I., Andreotti, F. et al. Prevention of atherothrombotic events in patients with diabetes mellitus: from antithrombotic therapies to new-generation glucose-lowering drugs. Nat Rev Cardiol 16, 113–130 (2019). https://doi.org/10.1038/s41569-018-0080-2
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41569-018-0080-2
This article is cited by
-
Elevated plasma factor XI predicts cardiovascular events in patients with type 2 diabetes: a long-term observational study
Cardiovascular Diabetology (2023)
-
Heterogeneity of outcomes within diabetic patients with atrial fibrillation on edoxaban: a sub-analysis from the ETNA-AF Europe registry
Clinical Research in Cardiology (2023)
-
Managing thrombotic risk in patients with diabetes
Cardiovascular Diabetology (2022)
-
The effect of LDL-C status on the association between increased coronary artery calcium score and compositional plaque volume progression in statins-treated diabetic patients: evaluated using serial coronary CTAs
Cardiovascular Diabetology (2022)
-
Efficacy and safety of novel oral anticoagulants in patients with atrial nonvalvular atrial fibrillation and diabetes mellitus: a systematic review and meta-analysis
Journal of Translational Medicine (2022)