Abstract
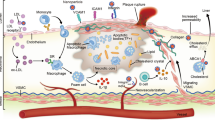
Atherosclerosis is a chronic disease of the large arteries and the underlying cause of myocardial infarction and stroke. Atherosclerosis is driven by cholesterol accumulation and subsequent inflammation in the vessel wall. Despite the clinical successes of lipid-lowering treatments, atherosclerosis remains one of the major threats to human health worldwide. Over the past 20 years, insights into cardiovascular immunopathology have provided a plethora of new potential therapeutic targets to reduce the risk of atherosclerosis and have shifted the therapeutic focus from lipids to inflammation. In 2017, the CANTOS trial demonstrated for the first time the beneficial effects of targeting inflammation to treat cardiovascular disease by showing that IL-1β inhibition can reduce the recurrence rate of cardiovascular events in a large cohort of patients. At the same time, preclinical studies have highlighted nanotechnology approaches that facilitate the specific targeting of innate immune cells, which could potentially generate more effective immunomodulatory treatments to induce disease regression and prevent the recurrence of cardiovascular events. The clinical translation of such nanoimmunotherapies and their application to treat patients with ischaemic heart disease are challenges that lie ahead.
Key points
-
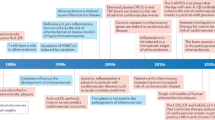
The therapeutic focus in atherosclerosis has shifted from lipid lowering to treating inflammation.
-
In the past decade, novel therapeutic targets for atherosclerosis have been identified as our understanding of the complex immune processes involved in this pathology has increased.
-
Advances in bioengineering have yielded innovative techniques to produce libraries of nanomaterials that engage immune cells.
-
The combined advances in nanoengineering and immunobiology have fuelled the development of novel nanoimmunotherapies, mainly aimed at modulating innate immune responses in cardiovascular diseases.
-
Large studies in animal models focusing on efficacy as well as safety are required to pave the way for clinical translation of cardiovascular nanoimmunotherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Quillard, T., Franck, G., Mawson, T., Folco, E. & Libby, P. Mechanisms of erosion of atherosclerotic plaques. Curr. Opin. Lipidol. 28, 434–441 (2017).
Melanie, C. M., Roland, L., Harald, S., Ruediger, N. & Wick, S. G. Atherosclerosis research from past to present — on the track of two pathologists with opposing views, Carl von Rokitansky and Rudolf Virchow. Virchows Arch. 449, 96–103 (2006).
Moore, K. J. & Tabas, I. Macrophages in the pathogenesis of atherosclerosis. Cell 145, 341–355 (2011).
Caro, C. G., Fitz-Gerald, J. M. & S. R. Arterial wall shear and distribution of early atheroma in man. Nature 223, 1159–1161 (1969).
Malek, A. M. & Alper, S. L. Hemodynamic shear stress and its role in atherosclerosis. JAMA 282, 2035–2042 (2014).
Chatzizisis, Y. S. et al. Role of endothelial shear stress in the natural history of coronary atherosclerosis and vascular remodeling. molecular, cellular, and vascular behavior. J. Am. Coll. Cardiol. 49, 2379–2393 (2007).
van Hinsbergh, V. W. M., Eringa, E. C. & Daemen, M. J. A. P. Neovascularization of the atherosclerotic plaque: interplay between atherosclerotic lesion, adventitia-derived microvessels and perivascular fat. Curr. Opin. Lipidol. 26, 405–411 (2015).
Swirski, F. K. et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 235, 612–616 (2009).
Swirski, F. K. et al. Ly-6 Chi monocytes dominate hypercholesterolemia-associated monocytosis and give rise to macrophages in atheromata. J. Clin. Invest. 117, 195–205 (2007).
Yvan-Charvet, L. et al. ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation. Science 328, 1689–1693 (2010).
Dutta, P. et al. Myocardial infarction accelerates atherosclerosis. Nature 487, 325–329 (2012).
Rosengren, A. et al. Association of psychosocial risk factors with risk of acute myocardial infarction in 11119 cases and 13648 controls from 52 countries (the INTERHEART study): case-control study. Lancet 364, 953–962 (2004).
Heidt, T. et al. Chronic variable stress activates hematopoietic stem cells. Nat. Med. 20, 754–758 (2014).
Fuster, J. J. et al. Clonal hematopoiesis associated with TET2 deficiency accelerates atherosclerosis development in mice. Science 355, 842–847 (2017).
Jaiswal, S. et al. Clonal hematopoiesis and risk of atherosclerotic cardiovascular disease. N. Engl. J. Med. 377, 111–121 (2017).
Gomez Perdiguero, E. et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors Most haematopoietic cells renew from adult haematopoietic stem cells (HSCs). Nature 518, 547–551 (2015).
Robbins, C. S. et al. Local proliferation dominates lesional macrophage accumulation in atherosclerosis. Nat. Med. 19, 1166–1172 (2013).
Brandl, R. et al. Topographic analysis of proliferative activity in carotid endarterectomy specimens by immunocytochemical detection of the cell cycle-related antigen Ki-67. Circulation 96, 3360–3368 (1997).
Iwasaki, A. & Medzhitov, R. Control of adaptive immunity by the innate immune system. Nat. Immunol. 16, 343–353 (2015).
Duewell, P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010).
Stewart, C. R. et al. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nat. Immunol. 11, 155–161 (2010).
Sheedy, F. J. et al. CD36 coordinates NLRP3 inflammasome activation by facilitating intracellular nucleation of soluble ligands into particulate ligands in sterile inflammation. Nat. Immunol. 14, 812–820 (2013).
Brinkmann, V. et al. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535 (2004).
Warnatsch, A., Ioannou, M., Wang, Q. & Papayannopoulos, V. Neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science 349, 316–320 (2015).
Small, D. M. & Shipley, G. G. Physical-chemical basis of lipid deposition in atherosclerosis. Science 185, 222–229 (1974).
Childs, B. G. et al. Senescent intimal foam cells are deleterious at all stages of atherosclerosis. Science 354, 472–477 (2016).
Koltsova, E. K. et al. Dynamic T cell-APC interactions sustain chronic inflammation in atherosclerosis. J. Clin. Invest. 122, 3114–3126 (2012).
Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017).
Pérez-Medina, C. et al. Nanoreporter PET predicts the efficacy of anti-cancer nanotherapy. Nat. Commun. 7, 11838 (2016).
Zhao, Y. et al. Augmenting drug-carrier compatibility improves tumour nanotherapy efficacy. Nat. Commun. 7, 11221 (2016).
Pérez-Medina, C. et al. In vivo PET imaging of HDL in multiple atherosclerosis models. JACC Cardiovasc. Imag. 9, 950–961 (2016).
Weissleder, R., Nahrendorf, M. & Pittet, M. J. Imaging macrophages with nanoparticles. Nat. Mater. 13, 125–138 (2014).
Barenholz, Y. Doxil® — The first FDA-approved nano-drug: lessons learned. J. Control. Release 160, 117–134 (2012).
Wagner, V., Dullaart, A., Bock, A. K. & Zweck, A. The emerging nanomedicine landscape. Nat. Biotechnol. 24, 1211–1217 (2006).
Zhang, L. et al. Nanoparticles in medicine: therapeutic applications and developments. Clin. Pharmacol. Ther. 83, 761–769 (2008).
Fitzgerald, K. et al. Effect of an RNA interference drug on the synthesis of proprotein convertase subtilisin/kexin type 9 (PCSK9) and the concentration of serum LDL cholesterol in healthy volunteers: A randomised, single-blind, placebo-controlled, phase 1 trial. Lancet 383, 60–68 (2014).
van der Valk, F. M. et al. Prednisolone-containing liposomes accumulate in human atherosclerotic macrophages upon intravenous administration. Nanomedicine 11, 1039–1046 (2015).
van der Valk, F. M. et al. Liposomal prednisolone promotes macrophage lipotoxicity in experimental atherosclerosis. Nanomedicine 12, 1463–1470 (2016).
Milla, P., Dosio, F. & Cattel, L. PEGylation of proteins and liposomes: a powerful and flexible strategy to improve the drug delivery. Curr. Drug Metab. 13, 105–119 (2012).
Parodi, A. et al. Synthetic nanoparticles functionalized with biomimetic leukocyte membranes possess cell-like functions. Nat. Nanotechnol. 8, 61–68 (2012).
Rodriguez, P. L. et al. Minimal ‘Self’ peptides that inhibit phagocytic clearance and enhance delivery of nanoparticles. Science 339, 971–975 (2013).
Lobatto, M. E. et al. Atherosclerosis targeting mechanism of long-circulating nanoparticles established by multimodal imaging. ACS Nano 9, 1837–1847 (2015).
Winter, P. M. et al. Molecular imaging of angiogenesis in early-stage atherosclerosis with αvβ3-integrin-targeted nanoparticles. Circulation 108, 2270–2274 (2003).
Winter, P. M. et al. Endothelial αvβ3 integrin-targeted fumagillin nanoparticles inhibit angiogenesis in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 26, 2103–2109 (2006).
Kamaly, N. et al. Targeted interleukin-10 nanotherapeutics developed with a microfluidic chip enhance resolution of inflammation in advanced atherosclerosis. ACS Nano 10, 5280–5292 (2016).
Fredman, G. et al. Targeted nanoparticles containing the proresolving peptide Ac2-26 protect against advanced atherosclerosis in hypercholesterolemic mice. Sci. Transl. Med. 7, 275ra20 (2015).
Mulder, W. J. M. et al. Molecular imaging of macrophages in atherosclerotic plaques using bimodal PEG-micelles. Magn. Reson. Med. 58, 1164–1170 (2007).
Sanchez-Gaytan, B. L. et al. Real-time monitoring of nanoparticle formation by FRET imaging. Angew. Chemie Int. Ed. 56, (2923–2926 (2017).
Tang, J. et al. Immune cell screening of a nanoparticle library improves atherosclerosis therapy. Proc. Natl Acad. Sci. USA 113, E6731–E6740 (2016).
Geissmann, F. et al. Development of monocytes, macrophages, and dendritic cells. Science 327, 656–661 (2010).
Serbina, N. V. & Pamer, E. G. Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nat. Immunol. 7, 311–317 (2006).
Hamers, A. A. J. et al. Bone marrow–specific deficiency of nuclear receptor Nur77 enhances atherosclerosis. Circ. Res. 110, 428–438 (2012).
Hanna, R. N. et al. NR4A1 (Nur77) deletion polarizes macrophages toward an inflammatory phenotype and increases atherosclerosis. Circ. Res. 110, 416–427 (2012).
Moore, K. J., Sheedy, F. J. & Fisher, E. A. Macrophages in atherosclerosis: a dynamic balance. Nat. Rev. Immunol. 13, 709–721 (2013).
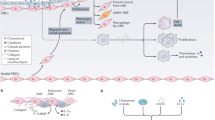
Hilgendorf, I., Swirski, F. K. & Robbins, C. S. Monocyte fate in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 35, 272–279 (2015).
Leuschner, F. et al. Therapeutic siRNA silencing in inflammatory monocytes in mice. Nat. Biotechnol. 29, 1005–1010 (2011).
Lee, H. et al. Molecularly self-assembled nucleic acid nanoparticles for targeted in vivo siRNA delivery. Nat. Nanotechnol. 7, 389–393 (2012).
Nahrendorf, M., Pittet, M. J. & Swirski, F. K. Monocytes: protagonists of infarct inflammation and repair after myocardial infarction. Circulation 121, 2437–2445 (2010).
Getts, D. R. et al. Therapeutic inflammatory monocyte modulation using immune-modifying microparticles. Sci. Transl. Med. 6, 219ra7 (2014).
Lameijer, M. et al. Efficacy and safety assessment of a TRAF6-targeted nanoimmunotherapy in atherosclerotic mice and non-human primates. Nat. Biomed. Eng. 2, 279–292 (2018).
Seijkens, T. T. P. et al. Targeting CD40-induced TRAF6 signaling in macrophages reduces atherosclerosis. J. Am. Coll. Cardiol. 71, 527–542 (2018).
Dahlman, J. E. et al. In vivo endothelial siRNA delivery using polymeric nanoparticles with low molecular weight. Nat. Nanotechnol. 9, 648–655 (2014).
Sager, H. B. et al. RNAi targeting multiple cell adhesion molecules reduces immune cell recruitment and vascular inflammation after myocardial infarction. Sci. Transl. Med. 8, 342ra80 (2016).
Honold, L. & Nahrendorf, M. Resident and monocyte-derived macrophages in cardiovascular disease. Circ. Res. 122, 113–127 (2018).
Lewis, D. R. et al. Sugar-based amphiphilic nanoparticles arrest atherosclerosis in vivo. Proc. Natl Acad. Sci. USA 112, 2693–2698 (2015).
Beldman, T. J. et al. Hyaluronan nanoparticles selectively target plaque-associated macrophages and improve plaque stability in atherosclerosis. ACS Nano 11, 5785–5799 (2017).
Nakashiro, S. et al. Pioglitazone-incorporated nanoparticles prevent plaque destabilization and rupture by regulating monocyte/macrophage differentiation in ApoE−/− mice. Arterioscler. Thromb. Vasc. Biol. 36, 491–500 (2016).
Brown, J. D. & Plutzky, J. Peroxisome proliferator-activated receptors as transcriptional nodal points and therapeutic targets. Circulation 115, 518–533 (2007).
Courties, G. et al. In vivo silencing of the transcription factor IRF5 reprograms the macrophage phenotype and improves infarct healing. J. Am. Coll. Cardiol. 63, 1556–1566 (2014).
Krausgruber, T. et al. IRF5 promotes inflammatory macrophage polarization and TH1-TH17 responses. Nat. Immunol. 12, 231–238 (2011).
Duivenvoorden, R. et al. A statin-loaded reconstituted high-density lipoprotein nanoparticle inhibits atherosclerotic plaque inflammation. Nat. Commun. 5, 3065 (2014).
Tobert, J. A. Lovastatin and beyond: the history of the HMG-CoA reductase inhibitors. Nat. Rev. Drug Discov. 2, 517–526 (2003).
Katsuki, S. et al. Nanoparticle-mediated delivery of pitavastatin inhibits atherosclerotic plaque destabilization/rupture in mice by regulating the recruitment of inflammatory monocytes. Circulation 129, 896–906 (2014).
Tang, J. et al. Inhibiting macrophage proliferation suppresses atherosclerotic plaque inflammation. Sci. Adv. 1, e1400223 (2015).
Dutta, P. et al. Macrophages retain hematopoietic stem cells in the spleen via VCAM-1. J. Exp. Med. 212, 497–512 (2015).
Tak, T., Tesselaar, K., Pillay, J., Borghans, J. A. M. & Koenderman, L. What’s your age again? Determination of human neutrophil half-lives revisited. J. Leukoc. Biol. 94, 595–601 (2013).
Soehnlein, O., Steffens, S., Hidalgo, A. & Weber, C. Neutrophils as protagonists and targets in chronic inflammation. Nat. Rev. Immunol. 17, 248–261 (2017).
Drechsler, M., Megens, R. T. A., van Zandvoort, M., Weber, C. & Soehnlein, O. Hyperlipidemia-triggered neutrophilia promotes early atherosclerosis. Circulation 122, 1837–1845 (2010).
Ionita, M. G. et al. High neutrophil numbers in human carotid atherosclerotic plaques are associated with characteristics of rupture-prone lesions. Arterioscler. Thromb. Vasc. Biol. 30, 1842–1848 (2010).
Hellings, W. E. et al. Composition of carotid atherosclerotic plaque is associated with cardiovascular outcome: a prognostic study. Circulation 121, 1941–1950 (2010).
Guasti, L. et al. Neutrophils and clinical outcomes in patients with acute coronary syndromes and/or cardiac revascularization: a systematic review on more than 34,000 subjects. Thromb. Haemost. 106, 591–599 (2011).
Wang, Z., Li, J., Cho, J. & Malik, A. B. Prevention of vascular inflammation by nanoparticle targeting of adherent neutrophils. Nat. Nanotechnol. 9, 204–210 (2014).
Chu, D., Gao, J. & Wang, Z. Neutrophil-mediated delivery of therapeutic nanoparticles across blood vessel barrier for treatment of inflammation and infection. ACS Nano 9, 11800–11811 (2015).
Singha, S. et al. Peptide-MHC-based nanomedicines for autoimmunity function as T cell receptor microclustering devices. Nat. Nanotechnol. 12, 701–710 (2017).
Clemente-casares, X. et al. Expanding antigen-specific regulatory networks to treat autoimmunity. Nature 530, 434–440 (2016).
Fayad, Z. A. et al. Safety and efficacy of dalcetrapib on atherosclerotic disease using novel non-invasive multimodality imaging (dal-PLAQUE): a randomised clinical trial. Lancet 378, 1547–1559 (2011).
Acknowledgements
The authors are supported by grants from the Netherlands Organization for Scientific Research: ZonMW Veni 016156059 (R.D.), ZonMW Vidi 91713324 (W.J.M.M.), and ZonMW Vici 91818622 (W.J.M.M.); AHA grant 17PRE33660729 and the Foundation “De Drie Lichten” in the Netherlands (M.L.S.); AHA grant 16SDG31390007 (C.P.M.); NIH grants R01 HL118440, R01 HL125703, and P01 HL131478 (W.J.M.M.), R01 EB009638 (Z.A.F.), and R01 HL144072 (W.J.M.M. and Z.A.F.); NIH Program of Excellence in Nanotechnology (PEN) Award HHSN368201000045C (Z.A.F.); and the Massachusetts General Hospital Research Scholar Award (M.N.).
Author information
Authors and Affiliations
Contributions
R.D. and W.J.M.M. wrote the manuscript. M.L.S. designed the figures for initial submission. All the authors reviewed and edited the article before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
7th Framework Programme NanoAthero: http://www.nanoathero.eu.
National Heart, Lung, and Blood Institute’s Program of Excellence in Nanotechnology: http://nhlbi-pen.net.
Rights and permissions
About this article
Cite this article
Duivenvoorden, R., Senders, M.L., van Leent, M.M.T. et al. Nanoimmunotherapy to treat ischaemic heart disease. Nat Rev Cardiol 16, 21–32 (2019). https://doi.org/10.1038/s41569-018-0073-1
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41569-018-0073-1
This article is cited by
-
Therapeutic angiogenesis and tissue revascularization in ischemic vascular disease
Journal of Biological Engineering (2023)
-
Regulating trained immunity with nanomedicine
Nature Reviews Materials (2022)
-
Synthetic recovery of impulse propagation in myocardial infarction via silicon carbide semiconductive nanowires
Nature Communications (2022)
-
Nano-enabled immunomodulation
Nature Nanotechnology (2021)
-
Exploring the most stable aptamer/target molecule complex by the stochastic tunnelling-basin hopping-discrete molecular dynamics method
Scientific Reports (2021)