Abstract
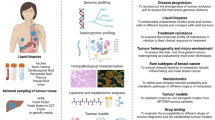
A research autopsy is a post-mortem medical procedure performed on a deceased individual with the primary goal of collecting tissue to support basic and translational research. This approach has increasingly been used to investigate the pathophysiological mechanisms of cancer evolution, metastasis and treatment resistance. In this Review, we discuss the rationale for the use of research autopsies in cancer research and provide an evidence-based discussion of the quality of post-mortem tissues compared with other types of biospecimens. We also discuss the advantages of using post-mortem tissues over other types of biospecimens, including the large amounts of tissue that can be obtained and the extent of multiregion sampling that is achievable, which is not otherwise possible in living patients. We highlight how the research autopsy has supported the identification of the clonal origins and modes of spread among metastases, the extent that selective pressures imposed by treatments cause bottlenecks leading to parallel and convergent tumour evolution, and the creation of rare tissue banks and patient-derived model systems. Finally, we comment on the future of the research autopsy as an integral component of precision medicine strategies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Buja, L. M., Barth, R. F., Krueger, G. R., Brodsky, S. V. & Hunter, R. L. The importance of the autopsy in medicine: perspectives of pathology colleagues. Acad. Pathol. 6, 2374289519834041 (2019).
Blokker, B. M. et al. Conventional autopsy versus minimally invasive autopsy with postmortem MRI, CT, and CT-guided biopsy: comparison of diagnostic performance. Radiology 289, 658–667 (2018).
Kretzschmar, H. Brain banking: opportunities, challenges and meaning for the future. Nat. Rev. Neurosci. 10, 70–78 (2009).
Hajdu, S. I. A note from history: the first printed case reports of cancer. Cancer 116, 2493–2498 (2010).
Mariette, C. et al. Consensus on the pathological definition and classification of poorly cohesive gastric carcinoma. Gastric Cancer 22, 1–9 (2019).
Ghosh, S. K. Giovanni Battista Morgagni (1682–1771): father of pathologic anatomy and pioneer of modern medicine. Anat. Sci. Int. 92, 305–312 (2017).
Paget, S. The distribution of secondary growths in cancer of the breast. Lancet 133, 571–573 (1889). A seminal large post-mortem study of patients with breast cancer that led to the seed and soil hypothesis of metastasis.
Fidler, I. J. Metastasis: quantitative analysis of distribution and fate of tumor emboli labeled with 125I-5-iodo-2′-deoxyuridine. J. Natl. Cancer Inst. 45, 773–782 (1970). The first study to provide experimental support for the seed and soil hypothesis.
Fidler, I. J. & Kripke, M. L. Metastasis results from preexisting variant cells within a malignant tumor. Science 197, 893–895 (1977).
Minn, A. J. et al. Distinct organ-specific metastatic potential of individual breast cancer cells and primary tumors. J. Clin. Invest. 115, 44–55 (2005).
Minn, A. J. et al. Lung metastasis genes couple breast tumor size and metastatic spread. Proc. Natl Acad. Sci. USA 104, 6740–6745 (2007).
Minn, A. J. et al. Genes that mediate breast cancer metastasis to lung. Nature 436, 518–524 (2005).
Bos, P. D. et al. Genes that mediate breast cancer metastasis to the brain. Nature 459, 1005–1009 (2009). This study and Minn et al. (J. Clin. Invest., 2005), Minn et al. (Proc. Natl Acad. Sci. USA, 2007) and Minn et al. (Nature, 2005) are the first studies to illustrate the molecular mechanisms of organotropism of breast cancer metastasis.
Rubin, M. A. et al. Rapid (‘warm’) autopsy study for procurement of metastatic prostate cancer. Clin. Cancer Res. 6, 1038–1045 (2000). The first published report describing the creation of a PDX from post-mortem tissue obtained from a patient with prostate cancer.
Morrissey, C. et al. Effects of androgen deprivation therapy and bisphosphonate treatment on bone in patients with metastatic castration-resistant prostate cancer: results from the University of Washington rapid autopsy series. J. Bone Miner. Res. 28, 333–340 (2013).
Iacobuzio-Donahue, C. A. et al. DPC4 gene status of the primary carcinoma correlates with patterns of failure in patients with pancreatic cancer. J. Clin. Oncol. 27, 1806–1813 (2009).
Liu, W. et al. Copy number analysis indicates monoclonal origin of lethal metastatic prostate cancer. Nat. Med. 15, 559–565 (2009).
Abbosh, C. et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature 545, 446–451 (2017).
Gundem, G. et al. The evolutionary history of lethal metastatic prostate cancer. Nature 520, 353–357 (2015).
Savas, P. et al. The subclonal architecture of metastatic breast cancer: results from a prospective community-based rapid autopsy program “CASCADE”. PLOS Med. 13, e1002204 (2016).
Razavi, P. et al. The genomic landscape of endocrine-resistant advanced breast cancers. Cancer Cell 34, 427–438.e6 (2018).
Yegnasubramanian, S. et al. DNA hypomethylation arises later in prostate cancer progression than CpG island hypermethylation and contributes to metastatic tumor heterogeneity. Cancer Res. 68, 8954–8967 (2008).
Yegnasubramanian, S. et al. Hypermethylation of CpG islands in primary and metastatic human prostate cancer. Cancer Res. 64, 1975–1986 (2004).
Pisapia, D. J. et al. Next-generation rapid autopsies enable tumor evolution tracking and generation of preclinical models. JCO Precis. Oncol. 2017, 1–13 (2017).
Wu, J. M. et al. Heterogeneity of breast cancer metastases: comparison of therapeutic target expression and promoter methylation between primary tumors and their multifocal metastases. Clin. Cancer Res. 14, 1938–1946 (2008).
Turajlic, S. et al. Tracking cancer evolution reveals constrained routes to metastases: TRACERx Renal. Cell 173, 581–594 (2018).
Vaught, J. Biobanking comes of age: the transition to biospecimen science. Annu. Rev. Pharmacol. Toxicol. 56, 211–228 (2016).
Carithers, L. J. et al. A novel approach to high-quality postmortem tissue procurement: The GTEx Project. Biopreserv. Biobank. 13, 311–319 (2015). Description of the GTEx project, which is designed to support understanding of the relationship between genomic variation and gene expression based on a large series of post-mortem-collected tissues.
Farrell, M. M. & Levin, D. L. Brain death in the pediatric patient: historical, sociological, medical, religious, cultural, legal, and ethical considerations. Crit. Care Med. 21, 1951–1965 (1993).
De Georgia, M. A. History of brain death as death: 1968 to the present. J. Crit. Care 29, 673–678 (2014).
Jakušovaitė, I. et al. Determination of death: metaphysical and biomedical discourse. Med. 52, 205–210 (2016).
Pozhitkov, A. E. & Noble, P. A. Gene expression in the twilight of death. BioEssays 39, 1700066 (2017).
Grizzle, W. E., Otali, D., Sexton, K. C. & Atherton, D. S. Effects of cold ischemia on gene expression: a review and commentary. Biopreserv. Biobank. 14, 548–558 (2016).
Green, D. R. & Llambi, F. Cell death signaling. Cold Spring Harb. Perspect. Biol. 7, a006080 (2015).
Shemie, S. D. & Gardiner, D. Circulatory arrest, brain arrest and death determination. Front. Cardiovasc. Med. 13, 15 (2018).
Bate-Smith, E. C. & Bendall, J. R. Factors determining the time course of rigor mortis. J. Physiol. 110, 47–65 (1949).
Goldberg, D. et al. Changing metrics of organ procurement organization performance in order to increase organ donation rates in the United States. Am. J. Transplant. 17, 3183–3192 (2017).
Jimeno, A. et al. A direct pancreatic cancer xenograft model as a platform for cancer stem cell therapeutic development. Mol. Cancer Ther. 8, 310–314 (2009).
Tiriac, H. et al. Organoid profiling identifies common responders to chemotherapy in pancreatic cancer. Cancer Discov. 8, 1112–1129 (2018).
Pauli, C. et al. Personalized in vitro and in vivo cancer models to guide precision medicine. Cancer Discov. 7, 462–477 (2017).
Aguirre, A. J. et al. Real-time genomic characterization of advanced pancreatic cancer to enable precision medicine. Cancer Discov. 8, 1096–1111 (2018).
Li, J. Z. et al. Sample matching by inferred agonal stress in gene expression analyses of the brain. BMC Genomics 8, 336 (2007).
Lee, D. C. et al. A lactate-induced response to hypoxia. Cell 161, 595–609 (2015).
Xu, Y. et al. Glycolysis determines dichotomous regulation of T cell subsets in hypoxia. J. Clin. Invest. 126, 2678–2688 (2016).
Lawless, S. J. et al. Glucose represses dendritic cell-induced T cell responses. Nat. Commun. 8, 15620 (2017).
Bär, W., Kratzer, A., Mächler, M. & Schmid, W. Postmortem stability of DNA. Forensic Sci. Int. 39, 59–70 (1988).
Sijen, T. Molecular approaches for forensic cell type identification: on mRNA, miRNA, DNA methylation and microbial markers. Forensic Sci. Int. Genet. 18 September, 21–32 (2015).
Vass, A. A. The elusive universal post-mortem interval formula. Forensic Sci. Int. 204, 34–40 (2011).
Dumache, R., Ciocan, V., Muresan, C., Rogobete, A. F. & Enache, A. Circulating microRNAs as promising biomarkers in forensic body fluids identification. Clin. Lab. 61, 1129–1135 (2015).
Freire-Aradas, A., Phillips, C. & Lareu, M. V. Forensic individual age estimation with DNA: From initial approaches to methylation tests. Forensic Sci. Rev. 29, 121–144 (2017).
Woerner, A. E. et al. Forensic human identification with targeted microbiome markers using nearest neighbor classification. Forensic Sci. Int. Genet. 38 Jan, 130–139 (2019).
Van den Berge, M., Wiskerke, D., Gerretsen, R. R. R., Tabak, J. & Sijen, T. DNA and RNA profiling of excavated human remains with varying postmortem intervals. Int. J. Leg. Med. 130, 1471–1480 (2016).
Bauer, M. RNA in forensic science. Forensic Sci. Int. Genet. 1, 69–74 (2007).
Budowle, B., Schmedes, S. E. & Wendt, F. R. Increasing the reach of forensic genetics with massively parallel sequencing. Forensic Sci. Med. Pathol. 13, 342–349 (2017).
Hunt, R. W., D’Onise, K., Nguyen, A. M. T. & Venugopal, K. Where patients with cancer die: a population-based study, 1990 to 2012. J. Palliat. Care Nov 28, 825859718814813 (2018).
Gao, W. et al. A population-based conceptual framework for evaluating the role of healthcare services in place of death. Healthcare 6, 107 (2018).
Bryant, V. A. et al. Childhood neoplasms presenting at autopsy: a 20-year experience. Pediatr. Blood Cancer 64, e26474 (2017).
Suzuki, H., Tanifuji, T., Abe, N. & Fukunaga, T. Causes of death in forensic autopsy cases of malnourished persons. Leg. Med. 15, 7–11 (2013).
Carithers, L. J. & Moore, H. M. The genotype-tissue expression (GTEx) project. Biopreserv. Biobank. 13, 307–308 (2015).
Wainberg, M. et al. Opportunities and challenges for transcriptome-wide association studies. Nat. Genet. 51, 592–599 (2019).
Rivas, M. A. et al. Effect of predicted protein-truncating genetic variants on the human transcriptome. Science 348, 666–669 (2015).
Mohammadi, P., Castel, S. E., Brown, A. A. & Lappalainen, T. Quantifying the regulatory effect size of cis-acting genetic variation using allelic fold change. Genome Res. 27, 1859–1871 (2017).
Melé, M. et al. The human transcriptome across tissues and individuals. Science 348, 660–665 (2015).
Tukiainen, T. et al. Landscape of X chromosome inactivation across human tissues. Nat. 550, 244–248 (2017).
Saha, A. et al. Co-expression networks reveal the tissue-specific regulation of transcription and splicing. Genome Res. 27, 1843–1858 (2017).
Zhu, Y., Wang, L., Yin, Y. & Yang, E. Systematic analysis of gene expression patterns associated with postmortem interval in human tissues. Sci. Rep. 7, 5435 (2017).
Fan, J. & Iacobuzio-Donahue, C. A. The science of rapid research autopsy. in Autopsy in the 21st Century: Best Practices and Future Diretions (eds. Hooper, J. E. & Williamson, A.) 151–166 (Springer International Publishing, 2018).
Embuscado, E. E. E. et al. Immortalizing the complexity of cancer metastasis: genetic features of lethal metastatic pancreatic cancer obtained from rapid autopsy. Cancer Biol. Ther. 4, 548–554 (2005).
Hooper, J. E. et al. A patient-derived xenograft model of parameningeal embryonal rhabdomyosarcoma for preclinical studies. Sarcoma 1, 826124 (2015).
Misuraca, K. L., Cordero, F. J. & Becher, O. J. Pre-clinical models of diffuse intrinsic pontine glioma. Front. Oncol. 5, 172 (2015).
Nguyen, H. M. et al. LuCaP prostate cancer patient-derived xenografts reflect the molecular heterogeneity of advanced disease and serve as models for evaluating cancer therapeutics. Prostate 77, 654–671 (2017).
Cocariu, E. A. et al. Correlations between the autolytic changes and postmortem interval in refrigerated cadavers. Rom. J. Intern. Med. 54, 105–112 (2016).
Fan, J. et al. Quantification of nucleic acid quality in postmortem tissues from a cancer research autopsy program. Oncotarget 7, 66906–66921 (2016).
Tomita, H. et al. Effect of agonal and postmortem factors on gene expression profile: Quality control in microarray analyses of postmortem human brain. Biol. Psychiatry 55, 346–352 (2004).
Harrison, P. J. et al. The relative importance of premortem acidosis and postmortem interval for human brain gene expression studies: selective mRNA vulnerability and comparison with their encoded proteins. Neurosci. Lett. 200, 151–154 (1995).
Gilkes, D. M., Semenza, G. L. & Wirtz, D. Hypoxia and the extracellular matrix: drivers of tumour metastasis. Nat. Rev. Cancer 14, 430–439 (2014).
Nobre, A. R., Entenberg, D., Wang, Y., Condeelis, J. & Aguirre-Ghiso, J. A. The different routes to metastasis via hypoxia-regulated programs. Trends Cell Biol. 28, 941–956 (2018).
Maley, C. C. et al. Classifying the evolutionary and ecological features of neoplasms. Nat. Rev. Cancer 17, 605–619 (2017). A commentary article proposing an objective set of metrics to quantify cell-intrinsic and cell-extrinsic factors that influence the clonal evolution of cancer.
Turajlic, S., Sottoriva, A., Graham, T. & Swanton, C. Resolving genetic heterogeneity in cancer. Nat. Rev. Genet. 20, 404–416 (2019).
Greaves, M. Darwinian medicine: a case for cancer. Nat. Rev. Cancer 7, 213–221 (2007).
Wilson, B. A., Garud, N. R., Feder, A. F., Assaf, Z. J. & Pennings, P. S. The population genetics of drug resistance evolution in natural populations of viral, bacterial and eukaryotic pathogens. Mol. Ecol. 25, 42–66 (2016).
Kreiner, J. M., Stinchcombe, J. R. & Wright, S. I. Population genomics of herbicide resistance: adaptation via evolutionary rescue. Annu. Rev. Plant Biol. Apr 29, 611–635 (2017).
Gillies, R. J., Verduzco, D. & Gatenby, R. A. Evolutionary dynamics of carcinogenesis and why targeted therapy does not work. Nat. Rev. Cancer 12, 487–493 (2012).
Ibrahim-Hashim, A. et al. Defining cancer subpopulations by adaptive strategies rather than molecular properties provides novel insights into intratumoral evolution. Cancer Res. 77, 2242–2254 (2017).
Spunt, S. L. et al. The clinical, research, and social value of autopsy after any cancer death. Cancer 118, 3002–3009 (2012).
Van Der Linden., A. et al. Post-mortem tissue biopsies obtained at minimally invasive autopsy: an RNA-quality analysis. PLOS ONE 9, e115675 (2014).
Gerlinger, M. et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 366, 883–892 (2012).
de Bruin, E. C., McGranahan, N. & Swanton, C. Analysis of intratumor heterogeneity unravels lung cancer evolution. Mol. Cell. Oncol. 2, e985549 (2015).
Kim, S. K. et al. Comprehensive analysis of genetic aberrations linked to tumorigenesis in regenerative nodules of liver cirrhosis. J. Gastroenterol. 54, 628–640 (2019).
Zhang, A. W. et al. Interfaces of malignant and immunologic clonal dynamics in ovarian. Cancer. Cell 173, 1755–1769 (2018).
Naxerova, K. et al. Origins of lymphatic and distant metastases in human colorectal cancer. Science 357, 55–60 (2017).
McPherson, A. et al. Divergent modes of clonal spread and intraperitoneal mixing in high-grade serous ovarian cancer. Nat. Genet. 48, 758–767 (2016).
Faltas, B. M. et al. Clonal evolution of chemotherapy-resistant urothelial carcinoma. Nat. Genet. 48, 1490–1499 (2016).
Brown, D. et al. Phylogenetic analysis of metastatic progression in breast cancer using somatic mutations and copy number aberrations. Nat. Commun. 8, 14944 (2017).
Yachida, S. et al. Distant metastasis occurs late during the genetic evolution of pancreatic cancer. Nat. 467, 1114–1117 (2010).
Ascierto, M. L. M. L. et al. Transcriptional mechanisms of resistance to anti-PD-1 therapy. Clin. Cancer Res. 23, 3168–3180 (2017).
Sottoriva, A. et al. A Big Bang model of human colorectal tumor growth. Nat. Genet. 47, 209–216 (2015).
Sun, R. et al. Between-region genetic divergence reflects the mode and tempo of tumor evolution. Nat. Genet. 49, 1015–1024 (2017).
Aryee, M. J. et al. DNA methylation alterations exhibit intraindividual stability and interindividual heterogeneity in prostate cancer metastases. Sci. Transl. Med. 5, 169ra10 (2013).
Turajlic, S. et al. Deterministic evolutionary trajectories influence primary tumor growth: TRACERx Renal. Cell 173, 595–610 (2018).
Reiter, J. G. J. G. et al. Minimal functional driver gene heterogeneity among untreated metastases. Science 361, 1033–1037 (2018).
Berquist, S. W. et al. Systemic therapy in the management of localized and locally advanced renal cell carcinoma: current state and future perspectives. Int. J. Urol. 26, 532–542 (2019).
Makohon-Moore, A. P. et al. Limited heterogeneity of known driver gene mutations among the metastases of individual patients with pancreatic cancer. Nat. Genet. 49, 358–366 (2017).
Borazanci, E. et al. Pancreatic cancer: ‘a riddle wrapped in a mystery inside an enigma’. Clin. Cancer Res. 23, 1629–1637 (2017).
Rosenthal, R. et al. Neoantigen-directed immune escape in lung cancer evolution. Nature 567, 479–485 (2019).
Mitchell, T. J. et al. Timing the landmark events in the evolution of clear cell renal cell cancer: TRACERx Renal. Cell 173, 611–623 (2018).
Jones, S. et al. Comparative lesion sequencing provides insights into tumor evolution. Proc. Natl Acad. Sci. USA 105, 4283–4288 (2008). The first study to use whole-exome sequencing data to derive estimates of the evolutionary life history of a neoplasm.
Körber, V. et al. Evolutionary trajectories of IDHWT glioblastomas reveal a common path of early tumorigenesis instigated years ahead of initial diagnosis. Cancer Cell 35, 692–704.e12 (2019).
Wu, R. et al. Genomic landscape and evolutionary trajectories of ovarian cancer precursor lesions. J. Pathol. 248, 41–50 (2019).
Matsuda, Y. et al. The prevalence and clinicopathological characteristics of high-grade pancreatic intraepithelial neoplasia autopsy study evaluating the entire pancreatic parenchyma. Pancreas 46, 658–664 (2017).
Groot, V. P. et al. Systematic review on the treatment of isolated local recurrence of pancreatic cancer after surgery; re-resection, chemoradiotherapy and SBRT. HPB 19, 83–92 (2017).
Makohon-Moore, A. P. et al. Precancerous neoplastic cells can move through the pancreatic ductal system. Nature 561, 201–205 (2018).
Martincorena, I. et al. Somatic mutant clones colonize the human esophagus with age. Science 362, 911–917 (2018).
Yizhak, K. et al. RNA sequence analysis reveals macroscopic somatic clonal expansion across normal tissues. Science 364, eaaw0726 (2019).
Biswas, R. et al. Genomic profiling of multiple sequentially acquired tumor metastatic sites from an “exceptional responder” lung adenocarcinoma patient reveals extensive genomic heterogeneity and novel somatic variants driving treatment response. Mol. Case Stud. 2, a001263 (2016).
Sanchez-Vega, F. et al. EGFR and MET amplifications determine response to HER2 inhibition in ERBB2-amplified esophagogastric cancer. Cancer Discov. 9, 199–209 (2019).
Juric, D. et al. Convergent loss of PTEN leads to clinical resistance to a PI(3)Kα inhibitor. Nature 518, 240–244 (2015).
Topalian, S. L., Drake, C. G. & Pardoll, D. M. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell 27, 450–461 (2015).
Sohal, D. P. S. et al. Metastatic pancreatic cancer: American Society of Clinical Oncology clinical practice guideline. J. Clin. Oncol. 34, 2784–2796 (2016).
Jaber, Y., Reichard, C. A. & Chapin, B. F. Emerging role of cytoreductive prostatectomy in patients with metastatic disease. Transl. Androl. Urol. 7, S505–S513 (2018).
Winter, J. M. et al. Survival after resection of pancreatic adenocarcinoma: results from a single institution over three decades. Ann. Surg. Oncol. 19, 169–175 (2012).
Yachida, S. et al. Establishment and characterization of a new cell line, A99, from a primary small cell carcinoma of the pancreas. Pancreas 40, 905–910 (2011).
Skapek, S. X. et al. Rhabdomyosarcoma. Nat. Rev. Dis. Prim. 5, 1 (2019).
Alabran, J. L. et al. Overcoming autopsy barriers in pediatric cancer research. Pediatr. Blood Cancer 60, 204–209 (2013).
Hawkins, D. S., Spunt, S. L. & Skapek, S. X. Children’s Oncology Group’s 2013 blueprint for research: soft tissue sarcomas. Pediatr. Blood Cancer 60, 1001–1008 (2013).
Monje, M. et al. Hedgehog-responsive candidate cell of origin for diffuse intrinsic pontine glioma. Proc. Natl Acad. Sci. USA 108, 4453–4458 (2011).
Caretti, V. et al. Human pontine glioma cells can induce murine tumors. Acta Neuropathol. 127, 897–909 (2014).
Sanchez, H. & Chamberlin, G. Utilizing the autopsy for quality improvement. in Autopsy in the 21st Century: Best Practices and Future Directions (eds. Hooper, J. E. & Williamson, A.) 19–38 (Springer International, 2018).
Cabot, R. C. Diagnostic pitfalls identified during a study of three thousand autopsies. J. Am. Med. Assoc. LIX, 2295–2298 (1912). A study illustrating the rates of incidental and unappreciated clinically relevant findings found at autopsy.
Shojania, K. G., Burton, E. C., McDonald, K. M. & Goldman, L. Changes in rates of autopsy-detected diagnostic errors over time: a systematic review. JAMA 289, 2849–2856 (2003).
Chow, S.-C. Adaptive clinical trial design. Annu. Rev. Med. 65, 405–415 (2014).
Gallo, P. et al. Adaptive designs in clinical drug development—an executive summary of the PhRMA working group. J. Biopharm. Stat. 16, 275–283 (2006).
Freidlin, B. & Korn, E. L. Biomarker-adaptive clinical trial designs. Pharmacogenomics 11, 1679–1682 (2010).
Regev, A. et al. The human cell atlas. Elife 6, e27041 (2017).
Manjili, M. H. Tumor dormancy and relapse: from a natural byproduct of evolution to a disease state. Cancer Res. 77, 2564–2569 (2017).
Chukwueke, U. N. & Brastianos, P. K. Sequencing brain metastases and opportunities for targeted therapies. Pharmacogenomics 18, 585–594 (2017).
Palmieri, D., Chambers, A. F., Felding-Habermann, B., Huang, S. & Steeg, P. S. The biology of metastasis to a sanctuary site. Clin. Cancer Res. 13, 1656–1662 (2007).
Beerenwinkel, N., Greenman, C. D. & Lagergren, J. Computational cancer biology: an evolutionary perspective. PLOS Comput. Biol. 12, e1004717 (2016).
Levitin, H. M., Yuan, J. & Sims, P. A. Single-cell transcriptomic analysis of tumor heterogeneity. Trends Cancer 4, 264–268 (2018).
Reiter, J. G. et al. Reconstructing metastatic seeding patterns of human cancers. Nat. Commun. 8, 14114 (2017). This article presents a phylogenetic algorithm developed specifically for the analysis of multiregion-sequenced tumour samples.
Zaccaria, S., El-Kebir, M., Klau, G. W. & Raphael, B. J. Phylogenetic copy-number factorization of multiple tumor samples. J. Comput. Biol. 25, 689–708 (2018).
Niknafs, N., Beleva-Guthrie, V., Naiman, D. Q. & Karchin, R. Subclonal hierarchy inference from somatic mutations: automatic reconstruction of cancer evolutionary trees from multi-region next generation sequencing. PLOS Comput. Biol. 11, e1004416 (2015).
Malikic, S., McPherson, A. W., Donmez, N. & Sahinalp, C. S. Clonality inference in multiple tumor samples using phylogeny. Bioinformatics 31, 1349–1356 (2015).
El-Kebir, M., Satas, G. & Raphael, B. J. Inferring parsimonious migration histories for metastatic cancers. Nat. Genet. 50, 718–726 (2018).
Tang, Z., Kang, B., Li, C., Chen, T. & Zhang, Z. GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res. 47, W556–W560 (2019).
Ma, Y. & Wei, P. FunSPU: a versatile and adaptive multiple functional annotation-based association test of whole-genome sequencing data. PLOS Genet. 15, e1008081 (2019).
Mounir, M. et al. New functionalities in the TCGAbiolinks package for the study and integration of cancer data from GDC and GTEx. PLOS Comput. Biol. 15, e1006701 (2019).
Achkar, T., Wilson, J., Simon, J., Rosenzweig, M. & Puhalla, S. Metastatic breast cancer patients: attitudes toward tissue donation for rapid autopsy. Breast Cancer Res. Treat. 155, 159–164 (2016).
Tsitsikas, D. A., Brothwell, M., Chin Aleong, J.-A. & Lister, A. T. The attitudes of relatives to autopsy: a misconception. J. Clin. Pathol. 64, 412–414 (2011).
Alsop, K. et al. A community-based model of rapid autopsy in end-stage cancer patients. Nat. Biotechnol. 34, 1010–1014 (2016). A comprehensive description of the logistics of running a large-scale research autopsy programme for patients with cancer.
Siminoff, L. A. et al. Impact of cognitive load on family decision makers’ recall and understanding of donation requests for the genotype-tissue expression (GTEx) project. J. Clin. Ethics 29, 20–30 (2018).
Hoadley, K. A. et al. Multiplatform analysis of 12 cancer types reveals molecular classification within and across tissues of origin. Cell 158, 929–944 (2014).
Acknowledgements
The authors acknowledge grant support from the US National Institutes of Health (grants R01 CA179991 and R35 CA220508 to C.I.A,-D) and salary support from the Parker Institute for Cancer Immunotherapy to T.J.H.
Author information
Authors and Affiliations
Contributions
All authors researched data for the manuscript and made substantial contributions to the discussion of the content. C.A.I.-D., J.E.H. and T.J.H. wrote the manuscript. C.A.I.-D., P.B., R.K., J.E.H. and T.J.H. reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
C.I.A.-D. and T.J.H. have received research support from Bristol-Myers Squibb. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Cancer thanks M. Rubin, S. Turajlic and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Rapid autopsy
-
An autopsy that is performed within 2 h of cardiopulmonary arrest.
- Warm autopsy
-
An autopsy that is performed so rapidly that the deceased person’s body has not yet cooled to room temperature.
- Somatic mosaicism
-
The presence of two or more genetically distinct populations of cells within an individual.
- Tumour dormancy
-
A state in which viable cancer cells remain quiescent for a prolonged period.
- Sanctuary sites
-
Tissues within the body in which cancer cells are protected from pharmacological agents or other therapies.
Rights and permissions
About this article
Cite this article
Iacobuzio-Donahue, C.A., Michael, C., Baez, P. et al. Cancer biology as revealed by the research autopsy. Nat Rev Cancer 19, 686–697 (2019). https://doi.org/10.1038/s41568-019-0199-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41568-019-0199-4
This article is cited by
-
Tumor classification of gastrointestinal liver metastases using CT-based radiomics and deep learning
Cancer Imaging (2023)
-
Locally sourced: site-specific immune barriers to metastasis
Nature Reviews Immunology (2023)
-
A medley of resistance in ovarian cancers
Nature Genetics (2023)
-
Application of high-throughput single-nucleus DNA sequencing in pancreatic cancer
Nature Communications (2023)
-
Proteome alterations in human autopsy tissues in relation to time after death
Cellular and Molecular Life Sciences (2023)