Abstract
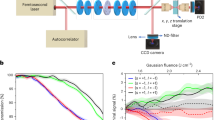
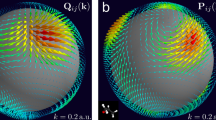
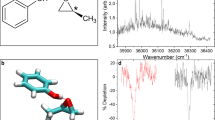
Chiral effects appear in a wide variety of natural phenomena and are of fundamental importance in science, from particle physics to metamaterials. The standard technique of chiral discrimination—photoabsorption circular dichroism—relies on the magnetic properties of a chiral medium and yields an extremely weak chiral response. Here, we propose and demonstrate an orders of magnitude more sensitive type of circular dichroism in neutral molecules: photoexcitation circular dichroism. This technique does not rely on weak magnetic effects, but takes advantage of the coherent helical motion of bound electrons excited by ultrashort circularly polarized light. It results in an ultrafast chiral response and the efficient excitation of a macroscopic chiral density in an initially isotropic ensemble of randomly oriented chiral molecules. We probe this excitation using linearly polarized laser pulses, without the aid of further chiral interactions. Our time-resolved study of vibronic chiral dynamics opens a way to the efficient initiation, control and monitoring of chiral chemical change in neutral molecules at the level of electrons.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Cotton, A. Recherches sur l’absorption et la dispersion de la lumière par les milieux doués du pouvoir rotatoire. J. Phys. Theor. Appl. 5, 237–244 (1896).
Barron, L. D. Molecular Light Scattering and Optical Activity. 2nd edn (Cambridge University Press: Cambridge, 2004).
Berova, N, Nakanishi, K. & Woody, R. Circular Dichroism: Principles and Applications. 2nd edn, (Wiley: New York, NY, 2000).
Ritchie, B. Theory of the angular distribution of photoelectrons ejected from optically active molecules and molecular negative ions. Phys. Rev. A 13, 1411–1415 (1976).
Powis, I. Photoelectron circular dichroism of the randomly oriented chiral molecules glyceraldehyde and lactic acid. J. Chem. Phys. 112, 301–310 (2000).
Böwering, N. et al. Asymmetry in photoelectron emission from chiral molecules induced by circularly polarized light. Phys. Rev. Lett. 86, 1187–1190 (2001).
Lux, C. et al. Circular dichroism in the photoelectron angular distributions of camphor and fenchone from multiphoton ionization with femtosecond laser pulses. Angew. Chem. Int. Ed. 51, 5001–5005 (2012).
Lehmann, C. S., Ram, N. B., Powis, I. & Janssen, M. H. M. Imaging photoelectron circular dichroism of chiral molecules by femtosecond multiphoton coincidence detection. J. Chem. Phys. 139, 234–307 (2013).
Pitzer, M. et al. Direct determination of absolute molecular stereochemistry in gas phase by coulomb explosion imaging. Science 341, 1096–1100 (2013).
Herwig, P. et al. Imaging the absolute configuration of a chiral epoxide in the gas phase. Science 342, 1084–1086 (2013).
Patterson, D., Schnell, M. & Doyle, J. M. Enantiomer-specific detection of chiral molecules via microwave spectroscopy. Nature 497, 475–477 (2013).
Yachmenev, A. & Yurchenko, S. N. Detecting chirality in molecules by linearly polarized laser fields. Phys. Rev. Lett. 117, 033001 (2016).
Comby, A. et al. Relaxation dynamics in photoexcited chiral molecules studied by time-resolved photoelectron circular dichroism: toward chiral femtochemistry. J. Phys. Chem. Lett. 7, 4514–4519 (2016).
Nahon, L., Garcia, G. A. & Powis, I. Valence shell one-photon photoelectron circular dichroism in chiral systems. J. Electron Spectrosc. Relat. Phenom. 204, 322–334 (2015).
Nahon, L. et al. Determination of accurate electron chiral asymmetries in fenchone and camphor in the VUV range: sensitivity to isomerism and enantiomeric purity. Phys. Chem. Chem. Phys. 18, 12696–12706 (2016).
Barron, L. D. True and false chirality and absolute asymmetric synthesis. J. Am. Chem. Soc. 108, 5539–5542 (1986).
Tang, Y. & Cohen, A. E. Optical chirality and its interaction with matter. Phys. Rev. Lett. 104, 163901 (2010).
Cherepkov, N. A. & Raşeev, G. Linear dichroism in the angular distribution of photoelectrons. J. Chem. Phys. 103, 8238–8246 (1995).
Garcia, G. A., Nahon, L., Daly, S. & Powis, I. Vibrationally induced inversion of photoelectron forward–backward asymmetry in chiral molecule photoionization by circularly polarized light. Nat. Commun. 4, 2132 (2013).
Stener, M., Di Tommaso, D., Fronzoni, G., Decleva, P. & Powis, I. Theoretical study on the circular dichroism in core and valence photoelectron angular distributions of camphor enantiomers. J. Chem. Phys. 124, 024326 (2006).
Ulrich, V. et al. Giant chiral asymmetry in the C1s core level photoemission from randomly oriented fenchone enantiomers. J. Phys. Chem. A 112, 3544–3549 (2008).
Wang, T. et al. Femtosecond single-shot imaging of nanoscale ferromagnetic order in Co/Pd multilayers using resonant x-ray holography. Phys. Rev. Lett. 108, 267403 (2012).
Spezzani, C. et al. Coherent light with tunable polarization from single-pass free-electron lasers. Phys. Rev. Lett. 107, 084801 (2011).
Allaria, E. et al. Highly coherent and stable pulses from the FERMI seeded free-electron laser in the extreme ultraviolet. Nat. Photon. 6, 699–704 (2012).
Fleischer, A., Kfir, O., Diskin, T., Sidorenko, P. & Cohen, O. Spin angular momentum and tunable polarization in high-harmonic generation. Nat. Photon. 8, 543–549 (2014).
Ferré, A. et al. A table-top ultrashort light source in the extreme ultraviolet for circular dichroism experiments. Nat. Photon. 9, 93–98 (2015).
Lux, C., Wollenhaupt, M., Sarpe, C. & Baumert, T. Photoelectron circular dichroism of bicyclic ketones from multiphoton ionization with femtosecond laser pulses. ChemPhysChem 16, 115–137 (2015).
Eibenberger, S., Doyle, J. & Patterson, D. Enantiomer-specific state transfer of chiral molecules. Phys. Rev. Lett. 118, 123002 (2017).
Bredtmann, T., Ivanov, M. & Dixit, G. X-ray imaging of chemically active valence electrons during a pericyclic reaction. Nat. Commun. 5, 5589 (2014).
Lunnemann, S., Kuleff, A. I. & Cederbaum, L. S. Ultrafast charge migration in 2-phenylethyl-N,N-dimethylamine. Chem. Phys. Lett. 450, 232–235 (2008).
Breidbach, J. & Cederbaum, L. S. Universal attosecond response to the removal of an electron. Phys. Rev. Lett. 94, 033901 (2005).
Remacle, F. & Levine, R. D. An electronic time scale in chemistry. Proc. Natl Acad. Sci. USA 103, 6793–6798 (2006).
Kuleff, A. I. & Cederbaum, L. S. Charge migration in different conformers of glycine: The role of nuclear geometry. Chem. Phys. 338, 320–328 (2007).
Lepine, F. Ivanov, M. Y. & Vrakking, M. J. J. Attosecond molecular dynamics: Fact or fiction? Nat. Photon. 8, 195–204 (2014).
Leone, S. R. et al. What will it take to observe processes in ‘real time’? Nat. Photon. 8, 162–166 (2014).
Kuleff, A. I. & Cederbaum, L. S. Ultrafast correlation-driven electron dynamics. J. Phys. B 47, 124002 (2014).
Calegari, F. et al. Ultrafast electron dynamics in phenylalanine initiated by attosecond pulses. Science 346, 336–339 (2014).
Weinkauf, R., Schlag, E. W., Martinez, T. J. & Levine, R. D. Nonstationary electronic states and site-selective reactivity. J. Phys. Chem. A 101, 7702–7710 (1997).
Pulm, F., Schramm, J., Hormes, J., Grimme, S. & Peyerimhoff, S. D. Theoretical and experimental investigations of the electronic circular dichroism and absorption spectra of bicyclic ketones. Chem. Phys. 224, 143–155 (1997).
Acknowledgements
We thank R. Bouillaud and L. Merzeau for technical assistance. We thank M. Ivanov, F. Morales, A. Stolow and T. Elsaesser for stimulating discussions. We acknowledge financial support from the Agence Nationale pour la Recherche (ANR-14-CE32-0014 MISFITS), the University of Bordeaux and the Conseil Regional de Nouvelle-Aquitaine (2.1.3-09010502 COLA2 project). Z.M. and O.S. gratefully acknowledge the support from Deutsche Forschungsgemeinschaft, project Sm 292-5/1, A.G.H. gratefully acknowledges the support from Deutsche Forschungsgemeinschaft, projects IV 152/7-1 and HA 8552/2-1. A.F.O. and O.S. gratefully acknowledge the MEDEA project, which has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska Curie grant agreement no. 641789 (H2020-MSCA-ITN-2014-641789-MEDEA (Marie Skłodowska Curie Innovative Training Networks)). S.B. acknowledges the support of a NSERC Vanier Canada Graduate Scholarship. R.G. acknowledges financial support from the Agence Nationale pour la Recherche through the XSTASE project (ANR-14-CE32-0010). The authors gratefully acknowledge the support of their collaboration through European Cooperation in Science and Technology (COST), programme CM1204 XLIC. This project has received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme no. 682978 - EXCITERS.
Author information
Authors and Affiliations
Contributions
S.B., A.C., R.G., Y.M. and V.B., performed the experiment. D.D. and S.P. operated the laser system. S.B., A.C., B.F., G.A.G., L.N., B.P., Y.M. and V.B. analysed the data. B.F. and B.P. performed the molecular geometry and dynamical calculations. A.G.H., A.F.O., Z.M. and O.S. developed the analytical theory, A.F.O. and O.S. derived triple-product chirality measures for PXCD and PXECD and analysed their connection and properties. S.B. wrote the first version of the manuscript; all authors contributed to writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures 1–12, Supplementary Note, Supplementary References
Rights and permissions
About this article
Cite this article
Beaulieu, S., Comby, A., Descamps, D. et al. Photoexcitation circular dichroism in chiral molecules. Nature Phys 14, 484–489 (2018). https://doi.org/10.1038/s41567-017-0038-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41567-017-0038-z
This article is cited by
-
Propagation properties of higher-order cosine-hyperbolic-Gaussian beams in a chiral medium
Optical and Quantum Electronics (2024)
-
Geometric magnetism and anomalous enantio-sensitive observables in photoionization of chiral molecules
Communications Physics (2023)
-
Condensation and asymmetric amplification of chirality in achiral molecules adsorbed on an achiral surface
Nature Communications (2023)
-
Paraxial propagation of Hermite cosine-hyperbolic-Gaussian beams in a chiral medium
Optical and Quantum Electronics (2023)
-
Chiral molecular imprinting-based SERS detection strategy for absolute enantiomeric discrimination
Nature Communications (2022)