Abstract
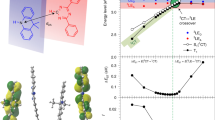
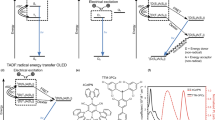
A spin-flip from a triplet to a singlet excited state, that is, reverse intersystem crossing (RISC), is an attractive route for improving light emission in organic light-emitting diodes, as shown by devices using thermally activated delayed fluorescence (TADF). However, device stability and efficiency roll-off remain challenging issues that originate from a slow RISC rate (kRISC). Here, we report a TADF molecule with multiple donor units that form charge-resonance-type hybrid triplet states leading to a small singlet–triplet energy splitting, large spin–orbit couplings, and a dense manifold of triplet states energetically close to the singlets. The kRISC in our TADF molecule is as fast as 1.5 × 107 s−1, a value some two orders of magnitude higher than typical TADF emitters. Organic light-emitting diodes based on this molecule exhibit good stability (estimated T90 about 600 h for 1,000 cd m−2), high maximum external quantum efficiency (>29.3%) and low efficiency roll-off (<2.3% at 1,000 cd m−2).
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available in the University of Cambridge Repository (https://doi.org/10.17863/CAM.52923). Related research results are available from the corresponding authors upon reasonable request.
References
Baldo, M. A. et al. Highly efficient phosphorescent emission from organic electroluminescent devices. Nature 395, 151–154 (1998).
Einzinger, M. et al. Sensitization of silicon by singlet exciton fission in tetracene. Nature 571, 90–94 (2019).
Samuel, I. D. W. & Turnbull, G. A. Organic semiconductor lasers. Chem. Rev. 107, 1272–1295 (2007).
Kohler, A. & Bassler, H. Triplet states in organic semiconductors. Mater. Sci. Eng. R 66, 71–109 (2009).
Adachi, C., Baldo, M. A., Thompson, M. E. & Forrest, S. R. Nearly 100% internal phosphorescence efficiency in an organic light emitting device. J. Appl. Phys. 90, 5048–5051 (2001).
Ma, Y., Zhang, H., Shen, J. & Che, C. Electroluminescence from triplet metal-ligand charge-transfer excited state of transition metal complexes. Synth. Met. 94, 245–248 (1998).
Köhler, A. & Beljonne, D. The singlet–triplet exchange energy in conjugated polymers. Adv. Funct. Mater. 14, 11–18 (2004).
Kondakov, D. Y. Characterization of triplet–triplet annihilation in organic light-emitting diodes based on anthracene derivatives. J. Appl. Phys. 102, 114504 (2007).
Ly, K. T. et al. Near-infrared organic light-emitting diodes with very high external quantum efficiency and radiance. Nat. Photon. 11, 63–68 (2017).
Li, L. K. et al. Strategies towards rational design of gold(iii) complexes for high-performance organic light-emitting devices. Nat. Photon. 13, 185–191 (2019).
Hamze, R. et al. Eliminating nonradiative decay in Cu(i) emitters: >99% quantum efficiency and microsecond lifetime. Science 363, 601–606 (2019).
Ai, X. et al. Efficient radical-based light-emitting diodes with doublet emission. Nature 563, 536–540 (2018).
Uoyama, H., Goushi, K., Shizu, K., Nomura, H. & Adachi, C. Highly efficient organic light-emitting diodes from delayed fluorescence. Nature 492, 234–238 (2012).
Zhang, Q. et al. Efficient blue organic light-emitting diodes employing thermally activated delayed fluorescence. Nat. Photon. 8, 326–332 (2014).
Liu, Y., Li, C., Ren, Z., Yan, S. & Bryce, M. R. All-organic thermally activated delayed fluorescence materials for organic light-emitting diodes. Nat. Rev. Mater. 3, 18020 (2018).
Cui, L.-S. et al. Controlling synergistic oxidation processes for efficient and stable blue thermally activated delayed fluorescence devices. Adv. Mater. 28, 7620–7625 (2016).
Noda, H., Nakanotani, H. & Adachi, C. Excited state engineering for efficient reverse intersystem crossing. Sci. Adv. 4, eaao6910 (2018).
Cui, L.-S. et al. Long-lived efficient delayed fluorescence organic light-emitting diodes using n-type hosts. Nat. Commun. 8, 2250 (2017).
Chan, C.-Y. et al. Efficient and stable sky-blue delayed fluorescence organic light-emitting diodes with CIEy below 0.4. Nat. Commun. 9, 5036 (2018).
Turro, N. J., Ramamurthy, V. & Scaiano, J. C. in Principles of Molecular Photochemistry: An Introduction Ch. 3, 113–118 (University Science Books, 2009).
Kondo, Y. et al. Narrowband deep-blue organic light-emitting diode featuring an organoboron-based emitter. Nat. Photon. 13, 678–682 (2019).
Kotadiya, N., Blom, P. W. M. & Wetzelaer, G.-J. A. H. Efficient and stable single-layer organic light-emitting diodes based on thermally activated delayed fluorescence. Nat. Photon. 13, 765–769 (2019).
Ahn, D. H. et al. Highly efficient blue thermally activated delayed fluorescence emitters based on symmetrical and rigid oxygen-bridged boron acceptors. Nat. Photon. 13, 540–546 (2019).
Wu, T.-L. et al. Diboron compound-based organic light-emitting diodes with high efficiency and reduced efficiency roll-off. Nat. Photon. 12, 235–240 (2018).
Lee, D. R. et al. Design strategy for 25% external quantum efficiency in green and blue thermally activated delayed fluorescent devices. Adv. Mater. 27, 5861–5867 (2015).
Wu, K. et al. De novo design of excited-state intramolecular proton transfer emitters via a thermally activated delayed fluorescence channel. J. Am. Chem. Soc. 140, 8877–8886 (2018).
Lin, T.-A. et al. Sky-blue organic light emitting diode with 37% external quantum efficiency using thermally activated delayed fluorescence from spiroacridine–triazine hybrid. Adv. Mater. 28, 6976–6983 (2016).
Zhang, Y. et al. Multi‐resonance induced thermally activated delayed fluorophores for narrowband green OLEDs. Angew. Chem. Int. Ed. 131, 1–7 (2019).
El-Sayed, M. A. The radiationless processes involving change of multiplicity in the diazenes. J. Chem. Phys. 36, 573–574 (1962).
Olivier, Y. et al. Nature of the singlet and triplet excitations mediating thermally activated delayed fluorescence. Phys. Rev. Mater. 1, 75602 (2017).
Marian, C. M. Spin–orbit coupling and intersystem crossing in molecules. WIREs Comput. Mol. Sci 2, 187–203 (2012).
Etherington, M. K., Gibson, J., Higginbotham, H. F., Penfold, T. J. & Monkman, A. P. Revealing the spin–vibronic coupling mechanism of thermally activated delayed fluorescence. Nat. Commun. 7, 13680 (2016).
Penfold, T. J., Gindensperger, E., Daniel, C. & Marian, C. M. Spin–vibronic mechanism for intersystem crossing. Chem. Rev. 118, 6975–7025 (2018).
Murawski, C., Leo, K. & Gather, M. C. Efficiency roll-off in organic light-emitting diodes. Adv. Mater. 25, 6801–6827 (2013).
Einzinger, M. et al. Shorter exciton lifetimes via an external heavy-atom effect: alleviating the effects of bimolecular processes in organic light-emitting diodes. Adv. Mater. 29, 1701987 (2017).
Reineke, S., Schwartz, G., Walzer, K. & Leo, K. Reduced efficiency roll-off in phosphorescent organic light emitting diodes by suppression of triplet–triplet annihilation. Appl. Phys. Lett. 91, 123508 (2007).
Lee, J. et al. Hot excited state management for long-lived blue phosphorescent organic light-emitting diodes. Nat. Commun. 8, 15566 (2017).
Munkhbat, B., Wersäll, M., Baranov, D. G., Antosiewicz, T. J. & Shegai, T. Suppression of photo-oxidation of organic chromophores by strong coupling to plasmonic nanoantennas. Sci. Adv. 4, eaas9552 (2018).
Chen, X.-K. et al. A new design strategy for efficient thermally activated delayed fluorescence organic emitters: from twisted to planar structures. Adv. Mater. 29, 1702767 (2017).
Chen, X. K., Zhang, S. F., Fan, J. X. & Ren, A. M. Nature of highly efficient thermally activated delayed fluorescence in organic light-emitting diode emitters: nonadiabatic effect between excited states. J. Phys. Chem. C 119, 9728–9733 (2015).
Noda, H. et al. Critical role of intermediate electronic states for spin-flip processes in charge-transfer-type organic molecules with multiple donors and acceptors. Nat. Mater. 18, 1084–1090 (2019).
Evans, E. W. et al. Vibrationally assisted intersystem crossing in benchmark thermally activated delayed fluorescence molecules. J. Phys. Chem. Lett. 9, 4053–4058 (2018).
Kuang, Z. R. et al. Conformational relaxation and thermally activated delayed fluorescence in anthraquinone-based intramolecular charge-transfer compound. J. Phys. Chem. C 122, 3727–3737 (2018).
Zhu, Z.-Q., Klimes, K., Holloway, S. & Li, J. Efficient cyclometalated platinum(ii) complex with superior operational stability. Adv. Mater. 29, 1605002 (2017).
Yokoyama, D. Molecular orientation in small-molecule organic light-emitting diodes. J. Mater. Chem. 21, 19187–19202 (2011).
Chen, X.-K., Kim, D. & Bredas, J.-L. Thermally activated delayed fluorescence (TADF) path toward efficient electroluminescence in purely organic materials: molecular level insight. Acc. Chem. Res. 51, 2215–2224 (2018).
Chen, X.-K. et al. Intramolecular noncovalent interactions facilitate thermally activated delayed fluorescence (TADF). J. Phys. Chem. Lett. 10, 3260–3268 (2019).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyser. J. Comput. Chem. 33, 580–592 (2012).
Frisch, M. J. et al. Gaussian 16, revision C.01 (Gaussian, Inc., 2016).
Gao, X. et al. Evaluation of spin–orbit couplings with linear-response time-dependent density functional methods. J. Chem. Theory Comput. 13, 515–524 (2017).
Stoll, S. & Schweiger, A. EasySpin, a comprehensive software package for spectral simulation and analysis in EPR. J. Magn. Reson. 178, 42–55 (2006).
Furno, M., Meerheim, R., Hofmann, S., Lüssem, B. & Leo, K. Efficiency and rate of spontaneous emission in organic electroluminescent devices. Phys. Rev. B 85, 115205 (2012).
Acknowledgements
L.-S.C., A.J.G., E.W.E. and R.H.F. acknowledge the Engineering and Physical Sciences Research Council (EPSRC) for funding (EP/M01083X/1, EP/M005143/1). The Centre for Advanced ESR (CAESR) is supported by UK EPSRC (EP/L011972/1). We thank Diamond Light Source (UK) for synchrotron beamtime on I19 (CY21497). X.-K.C. and J.-L.B. acknowledge support from the Georgia Institute of Technology, Georgia Research Alliance, Vasser-Woolley Foundation and Kyulux. H.Y., Z.-S.L., H.N. and C.A acknowledge the Japan Science and Technology Agency, ERATO, Adachi Molecular Exciton Engineering Project for funding (JPMJER1305). Y.L. acknowledges a stipend from the Chinese Scholarship Council. S.-F.Z. acknowledges financial support by Guangxi Department of Science and Technology (no. AD19110030), Department of Education (no. 2019KY0394) and the start-up funds provided by Guangxi University of Science and Technology. E.W.E also thanks the Leverhulme Trust for funding (ECF-2019-054). We thank J. Y. Lee for providing the control TADF materials and C. Zhong for discussions.
Author information
Authors and Affiliations
Contributions
L.-S.C. designed the molecules and carried out device fabrication and measurements. A.J.G. conducted the transient absorption experiments. S.-F.Z. performed the theoretical calculations under the supervision of X.-K.C. Y.L. performed the molecular orientation and optical simulations under the supervision of S.R. H.Y. and Z.-S.L carried out steady-state and time-resolved photophysical properties under the supervision of C.A. E.W.E. and W.K.M conducted the transient electron spin resonance measurements and analysed the results. T.K.R. performed single-crystal X-ray diffraction and analysed the results. H.N. participated in the discussion of the photophysical mechanism. L.-S.C., A.J.G., X.-K.C., J.-L.B. and R.H.F. analysed the data and wrote the manuscript. All authors discussed the progress of research and reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–26, Scheme 1 and Tables 1–7.
Crystallographic Data 1
Crystallographic Data of 5Cz-TRZ.
Rights and permissions
About this article
Cite this article
Cui, LS., Gillett, A.J., Zhang, SF. et al. Fast spin-flip enables efficient and stable organic electroluminescence from charge-transfer states. Nat. Photonics 14, 636–642 (2020). https://doi.org/10.1038/s41566-020-0668-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41566-020-0668-z
This article is cited by
-
A figure of merit for efficiency roll-off in TADF-based organic LEDs
Nature (2024)
-
Tunable afterglow for mechanical self-monitoring 3D printing structures
Nature Communications (2024)
-
Efficient, narrow-band, and stable electroluminescence from organoboron-nitrogen-carbonyl emitter
Nature Communications (2024)
-
Understanding of complex spin up-conversion processes in charge-transfer-type organic molecules
Nature Communications (2024)
-
Suppression of Dexter transfer by covalent encapsulation for efficient matrix-free narrowband deep blue hyperfluorescent OLEDs
Nature Materials (2024)