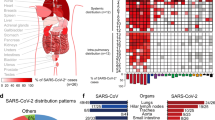
Abstract
The emergence of SARS-CoV-2 has resulted in millions of deaths as a result of COVID-19. Suitable models were missing at the beginning of the pandemic, and studies investigating disease pathogenesis relied on patients who had succumbed to COVID-19. Since then, autopsies of patients have substantially contributed to our understanding of the pathogenesis of COVID-19 and associated major organ complications. Here we summarize how autopsies have complemented experimental studies, mainly in animal models, and how they have facilitated critical knowledge of COVID-19 to improve daily clinical practice and develop therapeutic interventions. Employing advanced histopathologic and molecular genetic methods in post-mortem tissues, the COVID-19 pandemic has highlighted the importance of autopsies for virology research and clinical practice in current and emerging infectious diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Zhu, N. et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 382, 727–733 (2020).
Andersen, K. G., Rambaut, A., Lipkin, W. I., Holmes, E. C. & Garry, R. F. The proximal origin of SARS-CoV-2. Nat. Med. 26, 450–452 (2020).
Lytras, S., Xia, W., Hughes, J., Jiang, X. & Robertson, D. L. The animal origin of SARS-CoV-2. Science 373, 968–970 (2021).
Infection Prevention and Control for the Safe Management of a Dead Body in the Context of COVID-19: Interim Guidance, 24 March 2020 (World Health Organization, 2020).
Msemburi, W. et al. The WHO estimates of excess mortality associated with the COVID-19 pandemic. Nature 613, 130–137 (2023).
Muñoz-Fontela, C. et al. Animal models for COVID-19. Nature 586, 509–515 (2020).
Munster, V. J. et al. Respiratory disease in rhesus macaques inoculated with SARS-CoV-2. Nature 585, 268–272 (2020).
Beer, J. et al. Impaired immune response drives age-dependent severity of COVID-19. J. Exp. Med. 219, e20220621 (2022).
Winkler, E. S. et al. SARS-CoV-2 infection of human ACE2-transgenic mice causes severe lung inflammation and impaired function. Nat. Immunol. 21, 1327–1335 (2020).
Liu, Q. et al. Gross examination report of a COVID-19 death autopsy. Fa Yi Xue Za Zhi 36, 21–23 (2020).
Wichmann, D. et al. Autopsy findings and venous thromboembolism in patients with COVID-19. Ann. Intern. Med. 173, 268–277 (2020).
The REMAP-CAP, ACTIV-4a and ATTACC Investigators. Therapeutic anticoagulation with heparin in critically ill patients with COVID-19. N. Engl. J. Med. 385, 777–789 (2021).
Puelles, V. G. et al. Multiorgan and renal tropism of SARS-CoV-2. N. Engl. J. Med. 383, 590–592 (2020).
De Cock, K. M., Zielinski-Gutiérrez, E. & Lucas, S. B. Learning from the dead. N. Engl. J. Med. 381, 1889–1891 (2019).
Trypsteen, W., Van Cleemput, J., Snippenberg, W. V., Gerlo, S. & Vandekerckhove, L. On the whereabouts of SARS-CoV-2 in the human body: a systematic review. PLoS Pathog. 16, e1009037 (2020).
Wu, Z. & McGoogan, J. M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA 323, 1239–1242 (2020).
von Stillfried, S. et al. First report from the German COVID-19 autopsy registry. Lancet Reg. Health Eur. 15, 100330 (2022).
Lamers, M. M. & Haagmans, B. L. SARS-CoV-2 pathogenesis. Nat. Rev. Microbiol. 20, 270–284 (2022).
Damiani, S. et al. Pathological post-mortem findings in lungs infected with SARS-CoV-2. J. Pathol. 253, 31–40 (2021).
Menter, T. et al. Postmortem examination of COVID-19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings in lungs and other organs suggesting vascular dysfunction. Histopathology 77, 198–209 (2020).
Matthay, M. A. et al. Acute respiratory distress syndrome. Nat. Rev. Dis. Primers 5, 18 (2019).
Acosta, M. A. T. & Singer, B. D. Pathogenesis of COVID-19-induced ARDS: implications for an ageing population. Eur. Respir. J. 56, 2002049 (2020).
Nienhold, R. et al. Two distinct immunopathological profiles in autopsy lungs of COVID-19. Nat. Commun. 11, 5086 (2020).
Delorey, T. M. et al. COVID-19 tissue atlases reveal SARS-CoV-2 pathology and cellular targets. Nature 595, 107–113 (2021).
Melms, J. C. et al. A molecular single-cell lung atlas of lethal COVID-19. Nature 595, 114–119 (2021).
Kory, P. & Kanne, J. P. SARS-CoV-2 organising pneumonia: ‘Has there been a widespread failure to identify and treat this prevalent condition in COVID-19?’. BMJ Open Respir. Res. 7, e000724 (2020).
Dorward, D. A. et al. Tissue-specific immunopathology in fatal COVID-19. Am. J. Respir. Crit. Care Med. 203, 192–201 (2021).
Rendeiro, A. F. et al. The spatial landscape of lung pathology during COVID-19 progression. Nature 593, 564–569 (2021).
Nie, X. et al. Multi-organ proteomic landscape of COVID-19 autopsies. Cell 184, 775–791.e14 (2021).
Junqueira, C. et al. FcγR-mediated SARS-CoV-2 infection of monocytes activates inflammation. Nature 606, 576–584 (2022).
Wu, H. et al. Postmortem high-dimensional immune profiling of severe COVID-19 patients reveals distinct patterns of immunosuppression and immunoactivation. Nat. Commun. 13, 269 (2022).
Ackermann, M. et al. The fatal trajectory of pulmonary COVID-19 is driven by lobular ischemia and fibrotic remodelling. eBioMedicine https://doi.org/10.1016/j.ebiom.2022.104296 (2022).
Varga, Z. et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet 395, 1417–1418 (2020).
Ackermann, M. et al. Pulmonary vascular endothelialitis, thrombosis and angiogenesis in COVID-19. N. Engl. J. Med. 383, 120–128 (2020).
Perico, L. et al. Immunity, endothelial injury and complement-induced coagulopathy in COVID-19. Nat. Rev. Nephrol. 17, 46–64 (2021).
Bryce, C. et al. Pathophysiology of SARS-CoV-2: the Mount Sinai COVID-19 autopsy experience. Mod. Pathol. 34, 1456–1467 (2021).
Krasemann, S. et al. Assessing and improving the validity of COVID-19 autopsy studies—a multicentre approach to establish essential standards for immunohistochemical and ultrastructural analyses. eBioMedicine https://doi.org/10.1016/j.ebiom.2022.104193 (2022).
Fajnzylber, J. et al. SARS-CoV-2 viral load is associated with increased disease severity and mortality. Nat. Commun. 11, 5493 (2020).
Corey, L. et al. SARS-CoV-2 variants in patients with immunosuppression. N. Engl. J. Med. 385, 562–566 (2021).
Van Cleemput, J. et al. Organ-specific genome diversity of replication-competent SARS-CoV-2. Nat. Commun. 12, 6612 (2021).
Legrand, M. et al. Pathophysiology of COVID-19-associated acute kidney injury. Nat. Rev. Nephrol. 17, 751–764 (2021).
Braun, F. et al. SARS-CoV-2 renal tropism associates with acute kidney injury. Lancet 396, 597–598 (2020).
Hanson, P. J. et al. Characterization of COVID-19-associated cardiac injury: evidence for a multifactorial disease in an autopsy cohort. Lab. Invest. 102, 814–825 (2022).
Zheng, Y.-Y., Ma, Y.-T., Zhang, J.-Y. & Xie, X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 17, 259–260 (2020).
Bräuninger, H. et al. Cardiac SARS-CoV-2 infection is associated with pro-inflammatory transcriptomic alterations within the heart. Cardiovasc. Res. 118, 542–555 (2022).
Varatharaj, A. et al. Neurological and neuropsychiatric complications of COVID-19 in 153 patients: a UK-wide surveillance study. Lancet Psychiatry 7, 875–882 (2020).
Schwabenland, M. et al. Deep spatial profiling of human COVID-19 brains reveals neuroinflammation with distinct microanatomical microglia-T-cell interactions. Immunity 54, 1594–1610 (2021).
Matschke, J. et al. Neuropathology of patients with COVID-19 in Germany: a post-mortem case series. Lancet Neurol. 19, 919–929 (2020).
Lee, M.-H. et al. Microvascular injury in the brains of patients with COVID-19. N. Engl. J. Med. 384, 481–483 (2020).
Schurink, B. et al. Viral presence and immunopathology in patients with lethal COVID-19: a prospective autopsy cohort study. Lancet Microbe 1, e290–e299 (2020).
Stein, S. R. et al. SARS-CoV-2 infection and persistence in the human body and brain at autopsy. Nature 612, 758–763 (2022).
Yang, A. C. et al. Dysregulation of brain and choroid plexus cell types in severe COVID-19. Nature 595, 565–571 (2021).
Fullard, J. F. et al. Single-nucleus transcriptome analysis of human brain immune response in patients with severe COVID-19. Genome Med. 13, 118 (2021).
Mavrikaki, M., Lee, J. D., Solomon, I. H. & Slack, F. J. Severe COVID-19 is associated with molecular signatures of aging in the human brain. Nat. Aging 2, 1130–1137 (2022).
Jackson, C. B., Farzan, M., Chen, B. & Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 23, 3–20 (2022).
Muus, C. et al. Single-cell meta-analysis of SARS-CoV-2 entry genes across tissues and demographics. Nat. Med. 27, 546–559 (2021).
Alexandersen, S., Chamings, A. & Bhatta, T. R. SARS-CoV-2 genomic and subgenomic RNAs in diagnostic samples are not an indicator of active replication. Nat. Commun. 11, 6059 (2020).
Burton, J. L. & Underwood, J. Clinical, educational and epidemiological value of autopsy. Lancet 369, 1471–1480 (2007).
Fusco, F. M. et al. A 2009 cross-sectional survey of procedures for post-mortem management of highly infectious disease patients in 48 isolation facilities in 16 countries: data from EuroNHID. Infection 44, 57–64 (2016).
Heinrich, F. et al. Postmortem antigen-detecting rapid diagnostic tests to predict infectivity of SARS-CoV-2-associated deaths. Emerg. Infect. Dis. 28, 244–247 (2022).
Marsoof, A. The disposal of COVID-19 dead bodies: impact of Sri Lanka’s response on fundamental rights. J. Hum. Rights Pract. 13, 669–689 (2021).
Walker, D. H. et al. Pathologic and virologic study of fatal Lassa fever in man. Am. J. Pathol. 107, 349–356 (1982).
Okokhere, P. et al. Clinical and laboratory predictors of Lassa fever outcome in a dedicated treatment facility in Nigeria: a retrospective, observational cohort study. Lancet Infect. Dis. 18, 684–695 (2018).
Keita, A. K. et al. Resurgence of Ebola virus in 2021 in Guinea suggests a new paradigm for outbreaks. Nature 597, 539–543 (2021).
Diallo, B. et al. Resurgence of Ebola virus disease in Guinea linked to a survivor with virus persistence in seminal fluid for more than 500 days. Clin. Infect. Dis. 63, 1353–1356 (2016).
Keita, M. et al. Subsequent mortality in survivors of Ebola virus disease in Guinea: a nationwide retrospective cohort study. Lancet Infect. Dis. 19, 1202–1208 (2019).
Khan, M. et al. Visualizing in deceased COVID-19 patients how SARS-CoV-2 attacks the respiratory and olfactory mucosae but spares the olfactory bulb. Cell 184, 5932–5949 (2021).
Krasemann, S. et al. The blood–brain barrier is dysregulated in COVID-19 and serves as a CNS entry route for SARS-CoV-2. Stem Cell Rep. 17, 307–320 (2022).
Carithers, L. J. et al. A novel approach to high-quality postmortem tissue procurement: the GTEx Project. Biopreserv. Biobanking 13, 311–319 (2015).
Cable, J. et al. Lessons from the pandemic: responding to emerging zoonotic viral diseases—a Keystone Symposia report. Ann. NY Acad. Sci. 1518, 209–225 (2022).
Ferreira, P. G. et al. The effects of death and post-mortem cold ischemia on human tissue transcriptomes. Nat. Commun. 9, 490 (2018).
Pechal, J. L., Schmidt, C. J., Jordan, H. R. & Benbow, M. E. A large-scale survey of the postmortem human microbiome, and its potential to provide insight into the living health condition. Sci. Rep. 8, 5724 (2018).
Layne, S. P., Walters, K.-A., Kash, J. C. & Taubenberger, J. K. More autopsy studies are needed to understand the pathogenesis of severe COVID-19. Nat. Med. 28, 427–428 (2022).
Jonigk, D. et al. Organ manifestations of COVID-19: what have we learned so far (not only) from autopsies? Virchows Arch. 481, 139–159 (2022).
Acknowledgements
We thank T. L. E. Wong for proofreading the final version of the manuscript. Original figures were created using Adobe Illustrator (Adobe) and BioRender.com.
Author information
Authors and Affiliations
Contributions
F.H. and S.K. conceptualized the manuscript and drafted the figures. K.D.M. contributed and critically reviewed pathology-related content and histological images. M.G. contributed and critically reviewed neuropathology-related content. M.B. contributed and critically reviewed microbiology- and animal-model-related content. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The Institute of Legal Medicine (F.H.) and Institute of Neuropathology (S.K. and M.G.) were funded by the research consortium NATON (Nationales Obduktionsnetzwerk; National Autopsy Network). The NATON project (grant no. 01KX2121) is part of the National Network University Medicine, funded by the Federal Ministry of Education and Research, Germany. The National Network University Medicine is coordinated at the Charité–Universitätsmedizin Berlin and supervised by the German Aerospace Center (DLR Project Management Agency). The funders had no role in study design, data collection and analysis, the decision to publish or manuscript preparation.
Peer review
Peer review information
Nature Microbiology thanks Edwin Veldhuis Kroeze, Jan von der Thüsen and Linos Vandekerckhove for their contributions to the peer review of this Work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Heinrich, F., Mertz, K.D., Glatzel, M. et al. Using autopsies to dissect COVID-19 pathogenesis. Nat Microbiol 8, 1986–1994 (2023). https://doi.org/10.1038/s41564-023-01488-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-023-01488-7
This article is cited by
-
The neurobiology of SARS-CoV-2 infection
Nature Reviews Neuroscience (2024)