Abstract
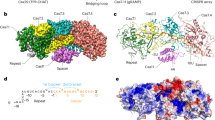
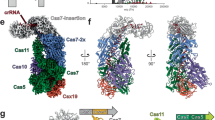
The recently discovered type III-E CRISPR-Cas effector Cas7-11 shows promise when used as an RNA manipulation tool, but its structure and the mechanisms underlying its function remain unclear. Here we present four cryo-EM structures of Desulfonema ishimotonii Cas7-11-crRNA complex in pre-target and target RNA-bound states, and the cryo-EM structure of DiCas7-11-crRNA bound to its accessory protein DiCsx29. These data reveal structural elements for pre-crRNA processing, target RNA cleavage and regulation. Moreover, a 3′ seed region of crRNA is involved in regulating RNA cleavage activity of DiCas7-11-crRNA-Csx29. Our analysis also shows that both the minimal mismatch of 4 nt to the 5′ handle of crRNA and the minimal matching of the first 12 nt of the spacer by the target RNA are essential for triggering the protease activity of DiCas7-11-crRNA-Csx29 towards DiCsx30. Taken together, we propose that target RNA recognition and cleavage regulate and fine-tune the protease activity of DiCas7-11-crRNA-Csx29, thus preventing auto-immune responses.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The 3D cryo-EM maps for the DiCas7-11-crRNA, DiCas7-11-crRNA-TR1, DiCas7-11-crRNA-TR2, DiCas7-11-crRNA-TR3 and DiCas7-11-crRNA-Csx29 complexes have been deposited in the Electron Microscopy Data Bank under accession numbers EMD-33955, EMD-33956, EMD-33957, EMD-33958 and EMD-33959. Atomic coordinates for the atomic model have been deposited in the Protein Data Bank (https://www.rcsb.org) under accession numbers 7YN9, 7YNA, 7YNB, 7YNC and 7YND. The structures used for structure comparison in this study were obtained from PDB 7WAH and 8D8N. Source data are provided with this paper.
References
Hampton, H. G., Watson, B. N. J. & Fineran, P. C. The arms race between bacteria and their phage foes. Nature 577, 327–336 (2020).
Makarova, K. S. et al. Evolution and classification of the CRISPR-Cas systems. Nat. Rev. Microbiol. 9, 467–477 (2011).
Makarova, K. S. et al. An updated evolutionary classification of CRISPR-Cas systems. Nat. Rev. Microbiol. 13, 722–736 (2015).
Sorek, R., Lawrence, C. M. & Wiedenheft, B. CRISPR-mediated adaptive immune systems in Bacteria and Archaea. Annu. Rev. Biochem. 82, 237–266 (2013).
Koonin, E. V., Makarova, K. S. & Zhang, F. Diversity, classification and evolution of CRISPR-Cas systems. Curr. Opin. Microbiol. 37, 67–78 (2017).
Koonin, E. V. & Makarova, K. S. Origins and evolution of CRISPR-Cas systems. Phil. Trans. R. Soc. Lond. B 374, 20180087 (2019).
van der Oost, J., Westra, E. R., Jackson, R. N. & Wiedenheft, B. Unravelling the structural and mechanistic basis of CRISPR-Cas systems. Nat. Rev. Microbiol. 12, 479–492 (2014).
Samai, P. et al. Co-transcriptional DNA and RNA cleavage during type III CRISPR-Cas immunity. Cell 161, 1164–1174 (2015).
Guo, M. H. et al. Coupling of ssRNA cleavage with DNase activity in type III-A CRISPR-Csm revealed by cryo-EM and biochemistry. Cell Res. 29, 305–312 (2019).
You, L. et al. Structure studies of the CRISPR-Csm complex reveal mechanism of co-transcriptional interference. Cell 176, 239–253.e16 (2019).
Jia, N. et al. Type III-A CRISPR-Cas Csm complexes: assembly, periodic RNA cleavage, DNase activity regulation, and autoimmunity. Mol. Cell 73, 264–277.e5 (2019).
Ozcan, A. et al. Programmable RNA targeting with the single-protein CRISPR effector Cas7-11. Nature 597, 720–725 (2021).
van Beljouw, S. P. B. et al. The gRAMP CRISPR-Cas effector is an RNA endonuclease complexed with a caspase-like peptidase. Science 373, 1349–1353 (2021).
Abudayyeh, O. O. et al. C2c2 is a single-component programmable RNA-guided RNA-targeting CRISPR effector. Science 353, aaf5573 (2016).
Liu, L. et al. The molecular architecture for RNA-guided RNA cleavage by Cas13a. Cell 170, 714–726.e10 (2017).
Zhang, C. et al. Structural basis for the RNA-guided ribonuclease activity of CRISPR-Cas13d. Cell 175, 212–223 (2018).
Knott, G. J. et al. Guide-bound structures of an RNA-targeting A-cleaving CRISPR-Cas13a enzyme. Nat. Struct. Mol. Biol. 24, 825–833 (2017).
Gootenberg, J. S. et al. Multiplexed and portable nucleic acid detection platform with Cas13, Cas12a, and Csm6. Science 360, 439–444 (2018).
Iwasaki, R. S. & Batey, R. T. SPRINT: a Cas13a-based platform for detection of small molecules. Nucleic Acids Res. 48, e101 (2020).
East-Seletsky, A. et al. Two distinct RNase activities of CRISPR-C2c2 enable guide-RNA processing and RNA detection. Nature 538, 270–273 (2016).
Tamulaitis, G. et al. Programmable RNA shredding by the type III-A CRISPR-Cas system of Streptococcus thermophilus. Mol. Cell 56, 506–517 (2014).
Staals, R. H. J. et al. Structure and activity of the RNA-targeting type III-B CRISPR-Cas complex of Thermus thermophilus. Mol. Cell 52, 135–145 (2013).
Sofos, N. et al. Structures of the Cmr-beta complex reveal the regulation of the immunity mechanism of type III-B CRISPR-Cas. Mol. Cell 79, 741–757 e7 (2020).
Niewoehner, O., Jinek, M. & Doudna, J. A. Evolution of CRISPR RNA recognition and processing by Cas6 endonucleases. Nucleic Acids Res. 42, 1341–1353 (2014).
Wang, R., Preamplume, G., Terns, M. P., Terns, R. M. & Li, H. Interaction of the Cas6 riboendonuclease with CRISPR RNAs: recognition and cleavage. Structure 19, 257–264 (2011).
Haurwitz, R. E., Jinek, M., Wiedenheft, B., Zhou, K. H. & Doudna, J. A. Sequence- and structure-specific RNA processing by a CRISPR endonuclease. Science 329, 1355–1358 (2010).
Hu, C. et al. Craspase is a CRISPR RNA-guided, RNA-activated protease. Science 377, 1278–1285 (2022).
Taylor, D. W. et al. Structures of the CRISPR-Cmr complex reveal mode of RNA target positioning. Science 348, 581–585 (2015).
Kato, K. et al. Structure and engineering of the type III-E CRISPR-Cas7-11 effector complex. Cell 185, 2324–2337.e16 (2022).
Huo, Y. et al. Cryo-EM structure of type III-A CRISPR effector complex. Cell Res. 28, 1195–1197 (2018).
Kazlauskiene, M., Kostiuk, G., Venclovas, C., Tamulaitis, G. & Siksnys, V. A cyclic oligonucleotide signaling pathway in type III CRISPR-Cas systems. Science 357, 605–609 (2017).
Niewoehner, O. et al. Type III CRISPR-Cas systems produce cyclic oligoadenylate second messengers. Nature 548, 543–548 (2017).
Gorski, S. A., Vogel, J. & Doudna, J. A. RNA-based recognition and targeting: sowing the seeds of specificity. Nat. Rev. Mol. Cell Biol. 18, 215–228 (2017).
Steens, J. A. et al. SCOPE enables type III CRISPR-Cas diagnostics using flexible targeting and stringent CARF ribonuclease activation. Nat. Commun. 12, 5033 (2021).
Wang, Y. L. et al. Structure of an argonaute silencing complex with a seed-containing guide DNA and target RNA duplex. Nature 456, 921–926 (2008).
Rouillon, C., Athukoralage, J. S., Graham, S., Gruschow, S. & White, M. F. Control of cyclic oligoadenylate synthesis in a type III CRISPR system. eLife 7, e36734 (2018).
Liu, X. et al. Target RNA activates the protease activity of Craspase to confer antiviral defense. Mol. Cell 82, 4503–4518 (2022).
Wang, S., Guo, M., Zhu, Y., Lin, Z. & Huang, Z. Cryo-EM structure of the type III-E CRISPR-Cas effector gRAMP in complex with TPR-CHAT. Cell Res. 32, 1128–1131 (2022).
Yu, G. et al. Structure and function of a bacterial type III-E CRISPR-Cas7-11 complex. Nat. Microbiol. 7, 2078–2088 (2022).
Strecker, J. et al. RNA-activated protein cleavage with a CRISPR-associated endopeptidase. Science 378, 874–881 (2022).
Kato, K. et al. RNA-triggered protein cleavage and cell growth arrest by the type III-E CRISPR nuclease-protease. Science 378, 882–889 (2022).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. eLife 7, e42166 (2018).
Kucukelbir, A., Sigworth, F. J. & Tagare, H. D. Quantifying the local resolution of cryo-EM density maps. Nat. Methods 11, 63–65 (2014).
Baek, M. et al. Accurate prediction of protein structures and interactions using a three-track neural network. Science 373, 871–876 (2021).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
Pettersen, E. F. et al. UCSF Chimera–a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Acknowledgements
We thank X. Huang, B. Zhu, X. Li and L. Chen for cryo-EM data collection at the Center for Biological Imaging (CBI, http://www.ibp.cas.cn/cbi/), Institute of Biophysics; D. Fan, L. Zhang, B. Huangfu and T. Wang at the CBI for cryo-EM grids screen; Z. Xie and L. Niu for mass spectrometry analysis at Lab of Proteomics of Institute of Biophysics, Chinese Academy of Sciences. This work was supported by grants from the Ministry of Science and Technology of China (2018YFE0203301) and Strategic Priority Research Program of the Chinese Academy of Sciences (XDB37010301) awarded to T.J.
Author information
Authors and Affiliations
Contributions
T.J. and Y.H. designed the project; Y.H. and H.Z. performed the full-length protein purification, cryo-EM sample preparation and data collection; Y.H. processed the cryo-EM data, built and refined the structure model; H.Z. constructed and purified the protein truncations and mutations. Y.H. and H.Z. performed biochemical assay. Q.D. assisted in model building and visualization; Y.H. and T.J. analysed the structures and wrote the manuscript; all authors discussed and commented on the results and the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Microbiology thanks Raymond Staals, Carl Salazar and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Purification of type III-E CRISPR-Cas effector complexes.
a, Purification of DiCas7-11-crRNA binary complex on Superdex 200. b, SDS-PADE of DiCas7-11-crRNA binary complex and best fractions were used for analysis. This gel is a representative of three replicate experiments. c, Analysis of the crRNA extracted from DiCas7-11-crRNA binary complex running on 16% urea-PAGE. This gel is a representative of three replicate experiments. d, Substrate cleavage assay of DiCas7-11-crRNA binary complex. This gel is a representative of three replicate experiments. e, Purification of DiCas7-11-crRNA-Csx29 ternary complex on Superdex 200. f, SDS-PADE of DiCas7-11-crRNA-Csx29 ternary binary complex and best fractions were used for analysis. This gel is a representative of three replicate experiments. g, Analysis of the crRNA extracted from DiCas7-11-crRNA-Csx29 ternary complex running on 16% urea-PAGE. This gel is a representative of three replicate experiments. h–j, Preparation of DiCas7-11-crRNA binary complex bound to tgRNA without 3’ anti-tag (TR1, h), tgRNA with matched 3’ anti-tag (TR2, i) and tgRNA with mismatched 3’ anti-tag (TR3, j).
Extended Data Fig. 2 Structural analysis of the DiCas7-11-crRNA binary complex.
a, Individual domain in DiCas7-11-crRNA binary complex. b, Structure comparison of Cas7L1, Cas7L2 and Cas7L3. c, ‘Lasso’ motif in Cas7L4. d, Surface potential map of insertion domain with crRNA showing the interactions between insertion domain and nucleotides 16–23 of crRNA. e, Surface potential map of DiCas7-11 with crRNA, showing the tgRNA binding channel.
Extended Data Fig. 3 Structural analysis of the interactions between the 5’ handle of the crRNA and Cas7L1-Cas7L2.
a, Cartoon depicting the 15 nt 5’ handle of the crRNA. b–f, Interactions between 5’ handle and Cas7L1-Cas7L2. g, Pre-crRNA processing assay of DiCas7-11 mutations. This gel is a representative of three replicate experiments. h, Structure comparison of DiCas7-11-crRNA-TR1 and Sb-gRAMP-crRNA-tgRNA showing their structure similarity. i-j, Sequence and structure comparison of the handles of Di-crRNA and Sb-crRNA showing their differences.
Extended Data Fig. 4 Structural study of the DiCas7-11-crRNA-TR1 complex.
a, Structural comparison of the DiCas7-11-crRNA binary complex and the DiCas7-11-crRNA-tgRNA (TR1) ternary complex based on crRNA alignment. b, Vector length correlation with the domain movement scale corresponding to (a) shows the conformational change that occurs in DiCas7-11-crRNA after binding to the tgRNA. Cas11L and the insertion domain are emphasized by dotted line boxes. c, Close-up views of the movements of the insertion domain and the Cas11L after tgRNA binding. d, Surface potential map of the insertion domain with partial crRNA-TR1 duplex in DiCas7-11-crRNA-TR1 complex. e, Structure comparison of DiCas7-11-crRNA-TR1 with Cas7-11-crRNA-tgRNA (PDB: 7WAH) showing their similarity. f, Schematic representation of the tgRNA with a truncated 5’ end used in substrate cleavage assay. All of the substrates were Cy3-labelled at the 5’ end. g, Substrate cleavage assay of DiCas7-11 (Δ Insertion)-crRNA and DiCas7-11 (Δ Cas11L)-crRNA comparing to DiCas7-11-crRNA. This gel is a representative of three replicate experiments. h, Comparison of the substrate cleavage activity between DiCas7-11-crRNA and DiCas7-11(Δ Insertion)-crRNA shows that deletion of the insertion domain accelerates the substrate cleavage activity. This gel is a representative of three replicate experiments. i, Substrate cleavage assay assessing the cleavage activity of DiCas7-11-crRNA towards substrates shown in (f). This gel is a representative of three replicate experiments. j, DiCas7-11-crRNA containing the Cas7L4-Insertion deletion has lost its substrate cleavage activity. This gel is a representative of three replicate experiments.
Extended Data Fig. 5 Pull-down assay of DiCsx29, DiCsx30, and DiCsx31 targeting DiCas7-11.
Lane1-4, 8×his-DiCsx29 co-expressed with DiCas7-11 in the present of crRNA template. Lane 1, cell lysate precipitate; Lane 2, cell lysate supernatant; Lane 3-4, beads elution with buffer containing 50 mM and 250 mM imidazole. Lane 5-8, 6×his-DiCsx30 co-expressed with DiCas7-11 in the present of crRNA template. Lane 5, cell lysate precipitate; Lane 6, cell lysate supernatant; Lane 7-8, beads elution with buffer containing 50 mM and 250 mM imidazole. Lane 9-12, 6×his-DiCsx31 co-expressed with DiCas7-11 in the present of crRNA template. Lane 9, cell lysate precipitate; Lane 10, cell lysate supernatant; Lane 11-12, beads elution with buffer containing 50 mM and 250 mM imidazole. This gel is a representative of three replicate experiments.
Extended Data Fig. 6 Cryo-EM study of the DiCas7-11-crRNA-Csx29 ternary complex.
a, b, Domains arrangement of DiCas7-11 and DiCsx29. c, Cryo-EM map of the Cas7-11-crRNA-Csx29 ternary complex at resolution of 3.97 Å, and color of each domain of DiCas7-11 and DiCsx29 is identical to that shown in (a) and (b).
Extended Data Fig. 7 Interaction analysis of the DiCas7-11-crRNA-Csx29 ternary complex.
a, Structure of the DiCas7-11-crRNA-Csx29 ternary complex. DiCas7-11 is shown in pink, DiCsx29 is shown in sky-blue, and the helices in the TPR domain of DiCsx29 are labelled with numbers accordingly. b, List of interactions across the DiCas7-11and DiCsx29 interface. Residues in DiCas7-11 and DiCsx29 are shown in pink and sky-blue, respectively.
Extended Data Fig. 8 Structural analysis of the DiCas7-11-crRNA-Csx29 ternary complex.
a, Close-up view of the FR1, FR2 and crRNA in DiCas7-11-crRNA-Csx29 ternary complex. FR1 and FR2 are shown as surface in magenta and deep-salmon respectively. b, c, Structure of the DiCas7-11-crRNA-Csx29 ternary complex in different orientation. DiCas7-11 is shown in pink, DiCsx29 is shown in sky-blue, FR1 is shown as surface in magenta, FR2 is shown as surface in deep-salmon, and the substrate cleavage sites are shown as sphere colored in green. d, Surface presentation of DiCas7-11-Csx29 with crRNA showing nucleotides 18-23 of crRNA are exposed to solvent as seed region. e, f, Structure of the DiCas7-11-crRNA-Csx29 ternary complex shown as surface in different orientation with a hypothetical tgRNA. DiCas7-11 is shown in pink, DiCsx29 is shown in sky-blue, FR1 is shown in magenta, FR2 is shown in deep-salmon, the 5’ handle of the crRNA is shown as cartoon colored in grey, the spacer of the crRNA is shown as cartoon colored in orange, and the supposed tgRNA (TR1) from DiCas7-11-crRNA-TR1 complex is shown as cartoon colored in red.
Extended Data Fig. 9 Structure comparison of DiCas7-11-crRNA-Csx29 ternary complex with Sb-gRAMP-crRNA-tgRNA ternary complex (PDB: 8D8N).
a, b, Structure comparison of DiCas7-11-crRNA-Csx29 ternary complex with Sb-gRAMP-crRNA-tgRNA ternary complex showing the similarity between DiCas7-11 and Sb-gRAMP. c, Close-view of FR1 and FR2 of DiCas7-11 and their corresponding parts of Sb-gRAMP. d, e, Sequence alignment of FR1 and FR2 of DiCas7-11 with their corresponding regions in Sb-gRAMP showing their differences.
Supplementary information
Supplementary Information
Supplementary Figs. 1–5, and Tables 1 and 2.
Source data
Source Data Fig. 1
Unprocessed gel of Fig. 1g.
Source Data Fig. 3
Unprocessed gel of Fig. 3e.
Source Data Fig. 4
Unprocessed gels of Fig. 4i.
Source Data Fig. 5
Unprocessed gels of Fig. 5c,e,g.
Source Data Extended Data Fig. 1
Unprocessed gels of Extended Data Fig. 1b–d,f,g.
Source Data Extended Data Fig. 3
Unprocessed gel of Extended Data Fig. 3g
Source Data Extended Data Fig. 4
Unprocessed gels of Extended Data Fig. 4g–j.
Source Data Extended Data Fig. 5
Unprocessed gel of Extended Data Fig. 5
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huo, Y., Zhao, H., Dong, Q. et al. Cryo-EM structure and protease activity of the type III-E CRISPR-Cas effector. Nat Microbiol 8, 522–532 (2023). https://doi.org/10.1038/s41564-022-01316-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-022-01316-4
This article is cited by
-
RNA targeting and cleavage by the type III-Dv CRISPR effector complex
Nature Communications (2024)