Abstract
In all organisms, the DNA sequence and the structural organization of chromosomes affect gene expression. The extremely thermophilic crenarchaeon Sulfolobus has one circular chromosome with three origins of replication. We previously revealed that this chromosome has defined A and B compartments that have high and low gene expression, respectively. As well as higher levels of gene expression, the A compartment contains the origins of replication. To evaluate the impact of three-dimensional organization on genome evolution, we characterized the effect of replication origins and compartmentalization on primary sequence evolution in eleven Sulfolobus species. Using single-nucleotide polymorphism analyses, we found that distance from an origin of replication was associated with increased mutation rates in the B but not in the A compartment. The enhanced polymorphisms distal to replication origins suggest that replication termination may have a causal role in their generation. Further mutational analyses revealed that the sequences in the A compartment are less likely to be mutated, and that there is stronger purifying selection than in the B compartment. Finally, we applied the Assay for Transposase-Accessible Chromatin using sequencing (ATAC-seq) to show that the B compartment is less accessible than the A compartment. Taken together, our data suggest that compartmentalization of chromosomal DNA can influence chromosome evolution in Sulfolobus. We propose that the A compartment serves as a haven for stable maintenance of gene sequences, while sequences in the B compartment can be diversified.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All sequencing data used in this study have been deposited to the NCBI Sequence Read Archive (SRA) under project number PRJNA814106. Source data are provided with this paper.
Code availability
No custom code was generated for this work.
References
Bryant, J. A., Sellars, L. E., Busby, S. J. W. & Lee, D. J. Chromosome position effects on gene expression in Escherichia coli K-12. Nucleic Acids Res. 42, 11383–11392 (2014).
Mitelman, F., Johansson, B. & Mertens, F. The impact of translocations and gene fusions on cancer causation. Nat. Rev. Cancer 7, 233–245 (2007).
Kempfer, R. & Pombo, A. Methods for mapping 3D chromosome architecture. Nat. Rev. Genet. https://doi.org/10.1038/s41576-019-0195-2 (2019).
Wit, Ede & Laat, Wde A decade of 3C technologies: insights into nuclear organization. Genes Dev. 26, 11–24 (2012).
Crane, E. et al. Condensin-driven remodelling of X chromosome topology during dosage compensation. Nature 523, 240–244 (2015).
Guo, Y. et al. CRISPR inversion of CTCF sites alters genome topology and enhancer/promoter function. Cell 162, 900–910 (2015).
Lioy, V. S., Junier, I., Lagage, V., Vallet, I. & Boccard, F. Distinct activities of bacterial condensins for chromosome management in Pseudomonas aeruginosa. Cell Rep. 33, 108344 (2020).
Collombet, S. et al. Parental-to-embryo switch of chromosome organization in early embryogenesis. Nature https://doi.org/10.1038/s41586-020-2125-z (2020).
Le, T. B. K., Imakaev, M. V., Mirny, L. A. & Laub, M. T. High-resolution mapping of the spatial organization of a bacterial chromosome. Science 342, 731–734 (2013).
Lieberman-Aiden, E. et al. Comprehensive mapping of long-range interactions reveals folding principles of the human genome. Science 326, 289–293 (2009).
Takemata, N. & Bell, S. D. Multi-scale architecture of archaeal chromosomes. Mol. Cell 81, 473–487.e6 (2021).
van Bemmel, J. G. et al. The bipartite TAD organization of the X-inactivation center ensures opposing developmental regulation of Tsix and Xist. Nat. Genet. 51, 1024–1034 (2019).
Kraft, K. et al. Serial genomic inversions induce tissue-specific architectural stripes, gene misexpression and congenital malformations. Nat. Cell Biol. 21, 305–310 (2019).
Lupiáñez, D. G. et al. Disruptions of topological chromatin domains cause pathogenic rewiring of gene-enhancer interactions. Cell 161, 1012–1025 (2015).
Paliou, C. et al. Preformed chromatin topology assists transcriptional robustness of Shh during limb development. Proc. Natl Acad. Sci. USA 116, 12390–12399 (2019).
Berthelot, C., Muffato, M., Abecassis, J. & Roest Crollius, H. The 3D organization of chromatin explains evolutionary fragile genomic regions. Cell Rep. 10, 1913–1924 (2015).
Tao, J.-F. et al. Influence of chromatin 3D organization on structural variations of the Arabidopsis thaliana genome. Mol. Plant 10, 340–344 (2017).
Zhang, Y. et al. Spatial organization of the mouse genome and its role in recurrent chromosomal translocations. Cell 148, 908–921 (2012).
Cockram, C., Thierry, A., Gorlas, A., Lestini, R. & Koszul, R. Euryarchaeal genomes are folded into SMC-dependent loops and domains, but lack transcription-mediated compartmentalization. Mol. Cell 81, 459–472.e10 (2021).
Takemata, N., Samson, R. Y. & Bell, S. D. Physical and functional compartmentalization of archaeal chromosomes. Cell 179, 165–179.e18 (2019).
Greci, M. D. & Bell, S. D. Archaeal DNA replication. Annu. Rev. Microbiol. 74, 65–80 (2020).
Takemata, N. & Bell, S. D. Emerging views of genome organization in Archaea. J. Cell Sci. 133, jcs243782 (2020).
Guo, L. et al. Genome analyses of icelandic strains of Sulfolobus islandicus, model organisms for genetic and virus–host interaction studies. J. Bacteriol. 193, 1672–1680 (2011).
Krause, D. J., Didelot, X., Cadillo-Quiroz, H. & Whitaker, R. J. Recombination shapes genome architecture in an organism from the archaeal domain. Genome Biol. Evol. 6, 170–178 (2014).
Mao, D. & Grogan, D. W. How a genetically stable extremophile evolves: modes of genome diversification in the archaeon Sulfolobus acidocaldarius. J. Bacteriol. 199, e00177–17 (2017).
Andersson, A. F. et al. Replication-biased genome organisation in the crenarchaeon Sulfolobus. BMC Genomics 11, 454 (2010).
Flynn, K. M., Vohr, S. H., Hatcher, P. J. & Cooper, V. S. Evolutionary rates and gene dispensability associate with replication timing in the archaeon Sulfolobus islandicus. Genome Biol. Evol. 2, 859–869 (2010).
Paul, S., Million-Weaver, S., Chattopadhyay, S., Sokurenko, E. & Merrikh, H. Accelerated gene evolution through replication–transcription conflicts. Nature 495, 512–515 (2013).
Srivatsan, A., Tehranchi, A., MacAlpine, D. M. & Wang, J. D. Co-orientation of replication and transcription preserves genome integrity. PLOS Genet. 6, e1000810 (2010).
Du, X. et al. Potential non-B DNA regions in the human genome are associated with higher rates of nucleotide mutation and expression variation. Nucleic Acids Res. 42, 12367–12379 (2014).
Guiblet, W. M. et al. Long-read sequencing technology indicates genome-wide effects of non-B DNA on polymerization speed and error rate. Genome Res. 28, 1767–1778 (2018).
Guiblet, W. M. et al. Non-B DNA: a major contributor to small- and large-scale variation in nucleotide substitution frequencies across the genome. Nucleic Acids Res. 49, 1497–1516 (2021).
Wang, G. & Vasquez, K. M. Impact of alternative DNA structures on DNA damage, DNA repair, and genetic instability. DNA Repair 19, 143–151 (2014).
Brázda, V. et al. G4Hunter web application: a web server for G-quadruplex prediction. Bioinformatics 35, 3493–3495 (2019).
Kejnovsky, E., Tokan, V. & Lexa, M. Transposable elements and G-quadruplexes. Chromosome Res. 23, 615–623 (2015).
Yadav, V., Hemansi, Kim, N., Tuteja, N. & Yadav, P. G quadruplex in plants: a ubiquitous regulatory element and its biological relevance. Front. Plant Sci. 8, 1163 (2017).
Ramsden, D. A., Weed, B. D. & Reddy, Y. V. R. V(D)J recombination: born to be wild. Semin. Cancer Biol. 20, 254–260 (2010).
Cree, S. L., Chua, E. W., Crowther, J., Dobson, R. C. J. & Kennedy, M. A. G-quadruplex structures bind to EZ-Tn5 transposase. Biochimie 177, 190–197 (2020).
Buenrostro, J. D., Giresi, P. G., Zaba, L. C., Chang, H. Y. & Greenleaf, W. J. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat. Methods 10, 1213–1218 (2013).
Buenrostro, J. D., Wu, B., Chang, H. Y. & Greenleaf, W. J. ATAC-seq: a method for assaying chromatin accessibility genome-wide. Curr. Protoc. Mol. Biol. 109, 21.29.1–21.29.9 (2015).
Couturier, E. & Rocha, E. P. C. Replication-associated gene dosage effects shape the genomes of fast-growing bacteria but only for transcription and translation genes. Mol. Microbiol. 59, 1506–1518 (2006).
Yaffe, E. & Tanay, A. Probabilistic modeling of Hi-C contact maps eliminates systematic biases to characterize global chromosomal architecture. Nat. Genet. 43, 1059–1065 (2011).
Imakaev, M. et al. Iterative correction of Hi-C data reveals hallmarks of chromosome organization. Nat. Methods 9, 999–1003 (2012).
Chandler, M. G. & Pritchard, R. H. The effect of gene concentration and relative gene dosage on gene output in Escherichia coli. Mol. Gen. Genet. 138, 127–141 (1975).
Bernander, R. & Poplawski, A. Cell cycle characteristics of thermophilic archaea. J. Bacteriol. 179, 4963–4969 (1997).
Rocha, E. P. C. The organization of the bacterial genome. Annu. Rev. Genet. 42, 211–233 (2008).
Mei, Q. et al. Two mechanisms of chromosome fragility at replication-termination sites in bacteria. Sci. Adv. 7, eabe2846 (2021).
Duggin, I. G., McCallum, S. A. & Bell, S. D. Chromosome replication dynamics in the archaeon Sulfolobus acidocaldarius. Proc. Natl Acad. Sci. USA 105, 16737–16742 (2008).
Duggin, I. G., Dubarry, N. & Bell, S. D. Replication termination and chromosome dimer resolution in the archaeon Sulfolobus solfataricus. EMBO J. 30, 145–153 (2011).
Samson, R. Y. et al. Specificity and function of archaeal DNA replication initiator proteins. Cell Rep. 3, 485–496 (2013).
Dorazi, R., Götz, D., Munro, S., Bernander, R. & White, M. F. Equal rates of repair of DNA photoproducts in transcribed and non-transcribed strands in Sulfolobus solfataricus. Mol. Microbiol. 63, 521–529 (2007).
Robinson, N. P. & Bell, S. D. Extrachromosomal element capture and the evolution of multiple replication origins in archaeal chromosomes. Proc. Natl Acad. Sci. USA 104, 5806–5811 (2007).
Zhang, C., Phillips, A. P. R., Wipfler, R. L., Olsen, G. J. & Whitaker, R. J. The essential genome of the crenarchaeal model Sulfolobus islandicus. Nat. Commun. 9, 4908 (2018).
Contreras-Moreira, B. & Vinuesa, P. GET_HOMOLOGUES, a versatile software package for scalable and robust microbial pangenome analysis. Appl. Environ. Microbiol. 79, 7696–7701 (2013).
Darling, A. C. E., Mau, B., Blattner, F. R. & Perna, N. T. Mauve: multiple alignment of conserved genomic sequence with rearrangements. Genome Res. 14, 1394–1403 (2004).
Fagan, R. P. & Fairweather, N. F. Biogenesis and functions of bacterial S-layers. Nat. Rev. Microbiol. 12, 211–222 (2014).
Horvath, P. & Barrangou, R. CRISPR/Cas, the immune system of bacteria and archaea. Science 327, 167–170 (2010).
Korber-Irrgang, B. HIV signature and sequence variation analysis. Comput. Anal. HIV Mol. Seq. 4, 55–72 (2000).
Deng, L., Zhu, H., Chen, Z., Liang, Y. X. & She, Q. Unmarked gene deletion and host-vector system for the hyperthermophilic crenarchaeon Sulfolobus islandicus. Extremophiles 13, 735 (2009).
Suzuki, T. et al. Sulfolobus tokodaii sp. nov. (f. Sulfolobus sp. strain 7), a new member of the genus Sulfolobus isolated from Beppu Hot Springs, Japan. Extremophiles 6, 39–44 (2002).
Brock, T. D., Brock, K. M., Belly, R. T. & Weiss, R. L. Sulfolobus: a new genus of sulfur-oxidizing bacteria living at low pH and high temperature. Arch. Mikrobiol. 84, 54–68 (1972).
Itoh, T. et al. Sulfuracidifex tepidarius gen. nov., sp. nov. and transfer of Sulfolobus metallicus Huber and Stetter 1992 to the genus Sulfuracidifex as Sulfuracidifex metallicus comb. nov. Int. J. Syst. Evol. Microbiol. https://doi.org/10.1099/ijsem.0.003981 (2020).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Takemata, N. & Bell, S. D. High-resolution analysis of chromosome conformation in hyperthermophilic archaea. STAR Protoc. 2, 100562 (2021).
Cockram, C., Thierry, A. & Koszul, R. Generation of gene-level resolution chromosome contact maps in bacteria and archaea. STAR Protoc. 2, 100512 (2021).
Samson, R. Y., Obita, T., Freund, S. M., Williams, R. L. & Bell, S. D. A role for the ESCRT system in cell division in Archaea. Science 322, 1710–1713 (2008).
Madeira, F. et al. The EMBL-EBI search and sequence analysis tools APIs in 2019. Nucleic Acids Res. 47, W636–W641 (2019).
Guindon, S. et al. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst. Biol. 59, 307–321 (2010).
Lemoine, F. et al. NGPhylogeny.fr: new generation phylogenetic services for non-specialists. Nucleic Acids Res. 47, W260–W265 (2019).
Letunic, I. & Bork, P. Interactive tree of life (iTOL) v3: an online tool for the display and annotation of phylogenetic and other trees. Nucleic Acids Res. 44, W242–W245 (2016).
Acknowledgements
Work in S.D.B.’s lab was supported by NIH R01GM135178 and the College of Arts and Sciences, Indiana University. R.Y.S. was supported by the College of Arts and Sciences, Indiana University. We thank N. Takemata for advice and assistance with software for the 3C studies. Sequencing was performed by the Center for Genomics and Bioinformatics, Indiana University.
Author information
Authors and Affiliations
Contributions
C.B. designed experimental approaches, conducted experimental work, analysed the data and wrote the initial draft of the manuscript. R.Y.S. designed experimental approaches, conducted experimental work, analysed the data and edited the manuscript. S.D.B. designed experimental approaches, analysed the data and co-wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Microbiology thanks Remus Dame, Frédéric Boccard and Thorsten Allers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
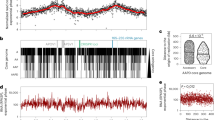
Extended Data Fig. 1 The effects of inactivating origins of replication in Sulfolobus islandicus REY15A.
a. Diagram of the Sulfolobus islandicus REY15A chromosome. The three origins of replication are shown as open circles on the chromosome. b. Marker frequency analysis of wild-type cells (left) and cells lacking Orc1-1 and Orc1-3 (right). Marker ratios of sequence tag abundance across the chromosome of exponentially growing cells normalized to non-replicating stationary phase cells are plotted relative to genome position. Genome coordinates are shown on the x-axes. The locations of active origins of replication are indicated above the plots. c. ChIP-seq analysis of ClsN enrichment on wild-type (top) and Δorc1-1, orc1-3 (bottom) chromosomes. All ChIP data are normalized to input DNA. Genome coordinates are shown on the x-axis. d. Scatterplot showing ClsN enrichment in the Δorc1-1, orc1-3 strain versus the wild-type strain for each data bin plotted in panel C. e. Transcript abundance profiles of wild-type (top) and Δorc1-1, orc1-3 strains (bottom) calculated for each gene. Genome coordinates are shown on the x-axis. F. Scatterplot showing RNA abundance in the Δorc1-1, orc1-3 strain versus the wild-type strain for each protein-coding gene.
Extended Data Fig. 2 SNP density and gene orientation for Sulfolobus islandicus REY15A.
Violin plot of the SNP density in genes oriented head-on or codirectionally with respect to replication, for A and B compartments. The p-value of the Wilcoxon test (two-sided) is indicated and the horizontal line represents the median.
Extended Data Fig. 3 Raw results of ATACseq analysis in Sulfolobus islandicus E233S.
a. Raw accessibility score for fixed cells, non-fixed cells and purified genomic DNA, plotted along the chromosome. b. DNA abundance in the genomic DNA from replicate 3, used to determine the transposition bias. c. Distribution density of paired-end sequenced insert size, mapped to the A or B compartment. d. Correlation between the raw accessibility score of replicates.
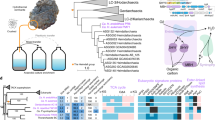
Extended Data Fig. 4 Phylogeny of the Sulfolobales.
16S rRNA phylogeny of Sulfolobales species with at least one complete genome sequenced, computed by ML. Species analyzed in this article are indicated in bold. Chi2-based likelihood is indicated when lower than 90%.
Extended Data Fig. 5 3C-seq contact heat-maps and marker frequency analysis (MFA) for Sulfurisphaera tokodaii strain 7.
a. Heat-map representing the contact score of pairs of 5-kb bins for iced-normalized 3C-seq data. b. Pearson-correlation analysis of the matrix presented in A. C. Marker Frequency Analysis. Read count ratios of exponentially growing cells normalized to non-replicating stationary phase cells are plotted relative to genome position.
Extended Data Fig. 6 3C-seq contact heat-maps and marker frequency analysis (MFA) for Sulfuracidifex tepidarius JCM16833.
a. Heat-map representing the contact score of pairs of 5-kb bins for iced-normalized 3C-seq data. b. Pearson-correlation analysis of the matrix presented in A. c. Marker Frequency Analysis. Read count ratios of exponentially growing cells normalized to non-replicating stationary phase cells are plotted relative to genome position.
Extended Data Fig. 7 Comparison of normalization methods for the quantification of 3C contact scores for Sulfuracidifex tepidarius JCM16833.
The distribution of the enzyme AluI restriction sites, DNA abundance in the analyzed cell population and various contact scores are plotted along the chromosome. Dot color indicates the compartment.
Extended Data Fig. 8 Primary chromosome organization in Sulfolobus acidocaldarius DSM639.
a. Chromosome organization, including the localization of the compartments, the origins of replication and the putative zones where replication forks collapse b to d. SNP density, ClsN enrichment and transcription level of protein-coding genes plotted in function of their distance to the nearest origin of replication for the A (red circle) and B (blue square) compartments. Continuous lines represent linear regressions for the A compartment in red, and the B compartment in blue. Pearson correlation p-values and coefficients are indicated for the A and B compartments. e. Violin plot of the distance to the nearest origin of replication for the dispensable and essential protein-coding genes of the A and B compartments. f. Violin plot of the G4 count per 10 kb window for A and B compartments. g. Violin plot of the SNP density in essential or dispensable protein-coding genes for the A and B compartments. h. Violin plot of the SNP density in genes oriented head-on or codirectionally with respect to replication, for A and B compartments. i. dN/dS of protein-coding genes plotted along their position on the chromosome. The A and B compartment localizations are indicated in red and blue respectively. Black vertical lines represent protein-coding genes that do not have orthologues in all the strains of the Sulfolobus acidocaldarius dataset and for which no dN/dS value was calculated. j. Violin plots of dN/dS value of protein-coding genes and of essential or dispensable protein-coding genes in the A and B compartments. dN/dS presented a bimodal distribution. The p-value of the Kolmogorov-Smirnov test (two-sided) is indicated at the top in bold. Two-sided student tests were performed for values higher or lower than the anti-mode and their p-values are indicated in in italic. For violin plots, except in J, the p-value of the Wilcoxon test (two-sided) is indicated and the horizontal line represents the median.
Extended Data Fig. 9 Dotplot of orthologous genes between pairs of Sulfolobales strains.
Vertical and Horizontal red backgrounds indicate the A compartment in the corresponding strains. The dot color indicates the gene compartment conservation between the two strains.
Supplementary information
Source data
Source Data Fig. 1
Data table.
Source Data Fig. 2
Data table.
Source Data Fig. 3
Data table.
Source Data Fig. 4
Data table.
Source Data Fig. 5
Data table.
Source Data Fig. 6
Data table.
Source Data Extended Data Fig. 1
Data table.
Source Data Extended Data Fig. 2
Data table.
Source Data Extended Data Fig. 3
Data table.
Source Data Extended Data Fig. 4
Data table.
Source Data Extended Data Fig. 5
Data table.
Source Data Extended Data Fig. 6
Data table.
Source Data Extended Data Fig. 7
Data table.
Source Data Extended Data Fig. 8
Data table.
Source Data Extended Data Fig. 9
Data table.
Rights and permissions
About this article
Cite this article
Badel, C., Samson, R.Y. & Bell, S.D. Chromosome organization affects genome evolution in Sulfolobus archaea. Nat Microbiol 7, 820–830 (2022). https://doi.org/10.1038/s41564-022-01127-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-022-01127-7
This article is cited by
-
The chromatin landscape of the euryarchaeon Haloferax volcanii
Genome Biology (2023)
-
Chromosome architecture in an archaeal species naturally lacking structural maintenance of chromosomes proteins
Nature Microbiology (2023)
-
The cell biology of archaea
Nature Microbiology (2022)