Abstract
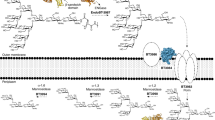
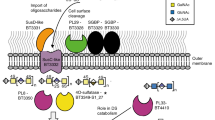
Glycans are major nutrients for the human gut microbiota (HGM). Arabinogalactan proteins (AGPs) comprise a heterogenous group of plant glycans in which a β1,3-galactan backbone and β1,6-galactan side chains are conserved. Diversity is provided by the variable nature of the sugars that decorate the galactans. The mechanisms by which nutritionally relevant AGPs are degraded in the HGM are poorly understood. Here we explore how the HGM organism Bacteroides thetaiotaomicron metabolizes AGPs. We propose a sequential degradative model in which exo-acting glycoside hydrolase (GH) family 43 β1,3-galactanases release the side chains. These oligosaccharide side chains are depolymerized by the synergistic action of exo-acting enzymes in which catalytic interactions are dependent on whether degradation is initiated by a lyase or GH. We identified two GHs that establish two previously undiscovered GH families. The crystal structures of the exo-β1,3-galactanases identified a key specificity determinant and departure from the canonical catalytic apparatus of GH43 enzymes. Growth studies of Bacteroidetes spp. on complex AGP revealed 3 keystone organisms that facilitated utilization of the glycan by 17 recipient bacteria, which included B. thetaiotaomicron. A surface endo-β1,3-galactanase, when engineered into B. thetaiotaomicron, enabled the bacterium to utilize complex AGPs and act as a keystone organism.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and the Supplementary Information. The crystal structure data sets generated (coordinate files and structure factors) have been deposited in the Protein Data Bank (PDB) and are listed in Supplementary Table 6 together with the PDB accession codes.
Change history
20 September 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Clemente, J. C., Ursell, L. K., Parfrey, L. W. & Knight, R. The impact of the gut microbiota on human health: an integrative view. Cell 148, 1258–1270 (2012).
El Kaoutari, A., Armougom, F., Gordon, J. I., Raoult, D. & Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 11, 497–504 (2013).
Koropatkin, N. M., Cameron, E. A. & Martens, E. C. How glycan metabolism shapes the human gut microbiota. Nat. Rev. Microbiol. 10, 323–335 (2012).
Porter, N. T. & Martens, E. C. The critical roles of polysaccharides in gut microbial ecology and physiology. Annu. Rev. Microbiol. 71, 349–369 (2017).
Gilbert, H. J., Stalbrand, H. & Brumer, H. How the walls come crumbling down: recent structural biochemistry of plant polysaccharide degradation. Curr. Opin. Plant Biol. 11, 338–348 (2008).
Lombard, V., Golaconda Ramulu, H., Drula, E., Coutinho, P. M. & Henrissat, B. The carbohydrate-active enzymes database (CAZy) in 2013. Nucleic Acids Res. 42, D490–D495 (2014).
Davies, G. & Henrissat, B. Structures and mechanisms of glycosyl hydrolases. Structure 3, 853–859 (1995).
Ndeh, D. & Gilbert, H. J. Biochemistry of complex glycan depolymerisation by the human gut microbiota. FEMS Microbiol. Rev. 42, 146–164 (2018).
Martens, E. C., Koropatkin, N. M., Smith, T. J. & Gordon, J. I. Complex glycan catabolism by the human gut microbiota: the Bacteroidetes Sus-like paradigm. J. Biol. Chem. 284, 24673–24677 (2009).
Larsbrink, J. et al. A discrete genetic locus confers xyloglucan metabolism in select human gut Bacteroidetes. Nature 506, 498–502 (2014).
Luis, A. S. et al. Dietary pectic glycans are degraded by coordinated enzyme pathways in human colonic Bacteroides. Nat. Microbiol. 3, 210–219 (2018).
Fincher, G. B., Stone, B. A. & Clarke, A. E. Arabinogalactan-proteins: structure, biosynthesis, and function. Annu. Rev. Plant Phys. 34, 47–70 (1983).
Vidal, S., Williams, P., Doco, T., Moutounet, M. & Pellerin, P. The polysaccharides of red wine: total fractionation and characterization. Carbohydr. Polym. 54, 439–447 (2003).
Capek, P., Matulova, M., Navarini, L. & Suggi-Liverani, F. Structural features of an arabinogalactan-protein isolated from instant coffee powder of Coffea arabica beans. Carbohydr. Polym. 80, 180–185 (2010).
Dauqan, E. & Abdullah, A. Utilization of gum arabic for industries and human health. Am. J. Appl. Sci. 10, 1270–1279 (2013).
McNamara, M. K. & Stone, B. A. Isolation, characterization and chemical synthesis of a galactosyl-hydroxyproline linkage compound from wheat endosperm arabinogalactan-peptide. Lebensm. Wiss. Technol. 14, 182–187 (1981).
Ichinose, H. et al. Characterization of an exo-β-1,3-galactanase from Clostridium thermocellum. Appl. Environ. Microbiol. 72, 3515–3523 (2006).
Munoz-Munoz, J. et al. An evolutionarily distinct family of polysaccharide lyases removes rhamnose capping of complex arabinogalactan proteins. J. Biol. Chem. 292, 13271–13283 (2017).
Munoz-Munoz, J., Cartmell, A., Terrapon, N., Henrissat, B. & Gilbert, H. J. Unusual active site location and catalytic apparatus in a glycoside hydrolase family. Proc. Natl Acad. Sci. USA 114, 4936–4941 (2017).
Calame, W., Weseler, A. R., Viebke, C., Flynn, C. & Siemensma, A. D. Gum arabic establishes prebiotic functionality in healthy human volunteers in a dose-dependent manner. Br. J. Nutr. 100, 1269–1275 (2008).
Martens, E. C. et al. Recognition and degradation of plant cell wall polysaccharides by two human gut symbionts. PLoS Biol. 9, e1001221 (2011).
Mewis, K., Lenfant, N., Lombard, V. & Henrissat, B. Dividing the large glycoside hydrolase family 43 into subfamilies: a motivation for detailed enzyme characterization. Appl. Environ. Microbiol. 82, 1686–1692 (2016).
Kotake, T. et al. Endo-β-1,3-galactanase from winter mushroom Flammulina velutipes. J. Biol. Chem. 286, 27848–27854 (2011).
Cartmell, A. et al. The structure and function of an arabinan-specific α-1,2-arabinofuranosidase identified from screening the activities of bacterial GH43 glycoside hydrolases. J. Biol. Chem. 286, 15483–15495 (2011).
Kitazawa, K. et al. β-galactosyl Yariv reagent binds to the β-1,3-galactan of arabinogalactan proteins. Plant Physiol. 161, 1117–1126 (2013).
Nakamura, A. et al. “Newton’s cradle” proton relay with amide-imidic acid tautomerization in inverting cellulase visualized by neutron crystallography. Sci. Adv. 1, e1500263 (2015).
Gloster, T. M., Turkenburg, J. P., Potts, J. R., Henrissat, B. & Davies, G. J. Divergence of catalytic mechanism within a glycosidase family provides insight into evolution of carbohydrate metabolism by human gut flora. Chem. Biol. 15, 1058–1067 (2008).
Rakoff-Nahoum, S., Coyne, M. J. & Comstock, L. E. An ecological network of polysaccharide utilization among human intestinal symbionts. Curr. Biol. 24, 40–49 (2014).
Rakoff-Nahoum, S., Foster, K. R. & Comstock, L. E. The evolution of cooperation within the gut microbiota. Nature 533, 255–259 (2016).
Sharma, S. K., Corrales, G. & Penadés, S. Single step stereoselective synthesis of unprotected 2,4-dinitrophenyl glycosides. Tetrahedron Lett. 36, 5627–5630 (1995).
Vonhoff, S., Heightman, T. D. & Vasella, A. Inhibition of glycosidases by lactam oximes: Influence of the aglycon in disaccharide analogues. Helv. Chim. Acta 81, 1710–1725 (1998).
Cavanagh, J., Fairbrother, W. J., Palmer, A. G. & Skelton, N. J. Protein NMR Spectroscopy: Principles and Practice (Academic Press, San Diego, 1996).
Vranken, W. F. et al. The CCPN data model for NMR spectroscopy: development of a software pipeline. Proteins 59, 687–696 (2005).
Bock, K. & Pedersen, C. Study of CH-13 coupling-constants in pentapyranoses and some of their derivatives. Acta Chem. Scand. B 29, 258–264 (1975).
Koropatkin, N. M., Martens, E. C., Gordon, J. I. & Smith, T. J. Starch catabolism by a prominent human gut symbiont is directed by the recognition of amylose helices. Structure 16, 1105–1115 (2008).
Cuskin, F. et al. Human gut Bacteroidetes can utilize yeast mannan through a selfish mechanism. Nature 517, 165–169 (2015).
Rodriguez-Ortega, M. J. et al. Characterization and identification of vaccine candidate proteins through analysis of the group A Streptococcus surface proteome. Nat. Biotechnol. 24, 191–197 (2006).
Davis, S. et al. Expanding proteome coverage with charge ordered parallel ion analysis (CHOPIN) combined with broad specificity proteolysis. J. Proteome. Res. 16, 1288–1299 (2017).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Cox, J. et al. Andromeda: a peptide search engine integrated into the MaxQuant environment. J. Proteome. Res. 10, 1794–1805 (2011).
MacLean, B. et al. Skyline: an open source document editor for creating and analyzing targeted proteomics experiments. Bioinformatics 26, 966–968 (2010).
Mi, H. et al. PANTHER version 11: expanded annotation data from Gene Ontology and Reactome pathways, and data analysis tool enhancements. Nucleic Acids Res. 45, D183–D189 (2017).
Zhou, M., Boekhorst, J., Francke, C. & Siezen, R. J. LocateP: genome-scale subcellular-location predictor for bacterial proteins. BMC Bioinformatics 9, 173 (2008).
Evans, P. R. An introduction to data reduction: space-group determination, scaling and intensity statistics. Acta Crystallogr. D 67, 282–292 (2011).
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010).
Evans, P. Scaling and assessment of data quality. Acta Crystallogr. D 62, 72–82 (2006).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Cowtan, K. The Buccaneer software for automated model building. 1. Tracing protein chains. Acta Crystallogr. D 62, 1002–1011 (2006).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Winn, M. D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. D 67, 235–242 (2011).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010).
Terrapon, N., Lombard, V., Gilbert, H. J. & Henrissat, B. Automatic prediction of polysaccharide utilization loci in Bacteroidetes species. Bioinformatics 31, 647–655 (2015).
Terrapon, N., Weiner, J., Grath, S., Moore, A. D. & Bornberg-Bauer, E. Rapid similarity search of proteins using alignments of domain arrangements. Bioinformatics 30, 274–281 (2014).
Cartmell, A. et al. How members of the human gut microbiota overcome the sulfation problem posed by glycosaminoglycans. Proc. Natl Acad. Sci. USA 114, 7037–7042 (2017).
Acknowledgements
This work was supported in part by an Advanced Grant from the European Research Council (grant no. 322820) awarded to H.J.G. and B.H. supporting D.A.N., A.C., J.M.-M., J.B. and N.T., and a Wellcome Trust Senior Investigator Award to H.J.G. (grant no. WT097907MA) that supported E.C.L. The Biotechnology and Biological Research Council project ‘Ricefuel’ (grant number BB/K020358/1) awarded to H.J.G. supported A.L. We thank Diamond Light Source for access to beamline I02, I04-1 and I24 (mx1960, mx7854 and mx9948) that contributed to the results presented here.
Author information
Authors and Affiliations
Contributions
Enzyme characterization and oligosaccharide purification were performed by A.C., D.A.N. and J.M.-M. Gene deletion strains were constructed by D.N. and A.L. Co-culturing experiments were carried out by A.C, J.B. and D.A.N. Western blots were carried out by D.A.N. Phylogenetic reconstruction and metagenomic analysis were performed by N.T. and B.H. Bacterial growth and transcriptomic experiments: E.C.L. and D.A.N. X-ray protein crystallography was carried out by A.C., A.B. J.M.-M. NMR experiments were performed by A.C. and K.S. Mass spectrometry was carried out by J.G., L.Y. and P.D. Chemical synthesis was performed by P.Z.F., S.S. and S.J.W. T.H., M.T. and E.C.L. performed the whole-cell proteomics. Experiments were designed by H.J.G. A.C. J.M.-M. and D.A.N. The manuscript was written by H.J.G. with substantial contributions from N.T., B.H. and S.J.W. Figures were prepared by J.M.-M. and E.C.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Tables 1–7, Supplementary Figures 1–15.
Rights and permissions
About this article
Cite this article
Cartmell, A., Muñoz-Muñoz, J., Briggs, J.A. et al. A surface endogalactanase in Bacteroides thetaiotaomicron confers keystone status for arabinogalactan degradation. Nat Microbiol 3, 1314–1326 (2018). https://doi.org/10.1038/s41564-018-0258-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-018-0258-8
This article is cited by
-
Bifidobacterial GH146 β-l-arabinofuranosidase for the removal of β1,3-l-arabinofuranosides on plant glycans
Applied Microbiology and Biotechnology (2024)
-
Chemoproteomic identification of a DPP4 homolog in Bacteroides thetaiotaomicron
Nature Chemical Biology (2023)
-
Nutritional and host environments determine community ecology and keystone species in a synthetic gut bacterial community
Nature Communications (2023)
-
Fungal β-glucan-facilitated cross-feeding activities between Bacteroides and Bifidobacterium species
Communications Biology (2023)
-
Carbohydrate complexity limits microbial growth and reduces the sensitivity of human gut communities to perturbations
Nature Ecology & Evolution (2023)