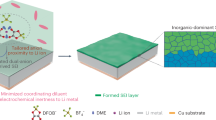
Abstract
Metal-free aqueous batteries can potentially address the projected shortages of strategic metals and safety issues found in lithium-ion batteries. More specifically, redox-active non-conjugated radical polymers are promising candidates for metal-free aqueous batteries because of the polymers’ high discharge voltage and fast redox kinetics. However, little is known regarding the energy storage mechanism of these polymers in an aqueous environment. The reaction itself is complex and difficult to resolve because of the simultaneous transfer of electrons, ions and water molecules. Here we demonstrate the nature of the redox reaction for poly(2,2,6,6-tetramethylpiperidinyloxy-4-yl acrylamide) by examining aqueous electrolytes of varying chao-/kosmotropic character using electrochemical quartz crystal microbalance with dissipation monitoring at a range of timescales. Surprisingly, the capacity can vary by as much as 1,000% depending on the electrolyte, in which certain ions enable better kinetics, higher capacity and higher cycling stability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated and analysed during this study are included in this Article and its Supplementary Information.
References
Lopez, J., Mackanic, D. G., Cui, Y. & Bao, Z. Designing polymers for advanced battery chemistries. Nat. Rev. Mater. 4, 312–330 (2019).
Nishide, H. & Oyaizu, K. Toward flexible batteries. Science 319, 737–738 (2008).
Poizot, P. et al. Opportunities and challenges for organic electrodes in electrochemical energy storage. Chem. Rev. 120, 6490–6557 (2020).
Nguyen, T. P. et al. Polypeptide organic radical batteries. Nature 593, 61–66 (2021).
Korley, L. T. J., Epps, T. H. III, Helms, B. A. & Ryan, A. J. Toward polymer upcycling—adding value and tackling circularity. Science 373, 66–69 (2021).
Rohland, P. et al. Redox-active polymers: the magic key towards energy storage—a polymer design guideline progress in polymer science. Prog. Polym. Sci. 125, 101474 (2022).
Liang, Y. & Yao, Y. Positioning organic electrode materials in the battery landscape. Joule 2, 1690–1706 (2018).
Lu, Y. & Chen, J. Prospects of organic electrode materials for practical lithium batteries. Nat. Rev. Chem. 4, 127–142 (2020).
Easley, A. D., Ma, T. & Lutkenhaus, J. L. Imagining circular beyond lithium-ion batteries. Joule 6, 1743–1749 (2022).
Sato, K. et al. Diffusion-cooperative model for charge transport by redox-active nonconjugated polymers. J. Am. Chem. Soc. 140, 1049–1056 (2018).
Tan, Y., Casetti, N. C., Boudouris, B. W. & Savoie, B. M. Molecular design features for charge transport in nonconjugated radical polymers. J. Am. Chem. Soc. 143, 11994–12002 (2021).
Oyaizu, K. & Nishide, H. Radical polymers for organic electronic devices: a radical departure from conjugated polymers? Adv. Mater. 21, 2339–2344 (2009).
Schon, T. B., McAllister, B. T., Li, P. F. & Seferos, D. S. The rise of organic electrode materials for energy storage. Chem. Soc. Rev. 45, 6345–6404 (2016).
Ma, T., Easley, A. D., Wang, S., Flouda, P. & Lutkenhaus, J. L. Mixed electron-ion-water transfer in macromolecular radicals for metal-free aqueous batteries. Cell Rep. Phys. Sci. 2, 100414 (2021).
Xie, Y., Zhang, K., Yamauchi, Y., Oyaizu, K. & Jia, Z. Nitroxide radical polymers for emerging plastic energy storage and organic electronics: fundamentals, materials, and applications. Mater. Horiz. 8, 803–829 (2021).
Janoschka, T. et al. An aqueous, polymer-based redox-flow battery using non-corrosive, safe, and low-cost materials. Nature 527, 78–81 (2015).
Wilcox, D. A., Agarkar, V., Mukherjee, S. & Boudouris, B. W. Stable radical materials for energy applications. Annu. Rev. Chem. Biomol. Eng. 9, 83–103 (2018).
Joo, Y., Agarkar, V., Sung, S. H., Savoie, B. M. & Boudouris, B. W. A nonconjugated radical polymer glass with high electrical conductivity. Science 359, 1391–1395 (2018).
Tan, Y. et al. Electronic and spintronic open-shell macromolecules, quo vadis? J. Am. Chem. Soc. 144, 626–647 (2022).
Goujon, N., Casado, N., Patil, N., Marcilla, R. & Mecerreyes, D. Organic batteries based on just redox polymers. Prog. Polym. Sci. 122, 101449 (2021).
Hatakeyama-Sato, K., Wakamatsu, H., Katagiri, R., Oyaizu, K. & Nishide, H. An ultrahigh output rechargeable electrode of a hydrophilic radical polymer/nanocarbon hybrid with an exceptionally large current density beyond 1 A cm–2. Adv. Mater. 30, e1800900 (2018).
Burgess, M., Hernandez-Burgos, K., Cheng, K. J., Moore, J. S. & Rodriguez-Lopez, J. Impact of electrolyte composition on the reactivity of a redox active polymer studied through surface interrogation and ion-sensitive scanning electrochemical microscopy. Analyst 141, 3842–3850 (2016).
Zhang, Y., Zhao, L., Liang, Y., Wang, X. & Yao, Y. Effect of electrolyte anions on the cycle life of a polymer electrode in aqueous batteries. eScience 2, 110–115 (2022).
Nimkar, A. et al. Influences of cations’ solvation on charge storage performance in polyimide anodes for aqueous multivalent ion batteries. ACS Energy Lett. 6, 2638–2644 (2021).
Wang, S., Li, F., Easley, A. D. & Lutkenhaus, J. L. Real-time insight into the doping mechanism of redox-active organic radical polymers. Nat. Mater. 18, 69–75 (2019).
Donald, H., Jenkins, B. & Marcus, Y. Viscosity B-coefficients of ions in solution. Chem. Rev. 95, 2695–2724 (1995).
Marcus, Y. Viscosity B-coefficients, structural entropies and heat capacities, and the effects of ions on the structure of water. J. Solut. Chem. 23, 831–848 (1994).
Hribar, B., Southall, N. T., Vlachy, V. & Dill, K. A. How ions affect the structure of water. J. Am. Chem. Soc. 124, 12302–12311 (2002).
Tietze, A. A. et al. On the nature of interactions between ionic liquids and small amino-acid-based biomolecules. ChemPhysChem 14, 4044–4064 (2013).
Bello, L. & Sing, C. E. Mechanisms of diffusive charge transport in redox-active polymer solutions. Macromolecules 53, 7658–7671 (2020).
Friebe, C. & Schubert, U. S. in Electrochemical Energy Storage: Next Generation Battery Concepts (ed Eichel R. A.) 65–99 (Springer International Publishing, 2019).
Andrieux, C. P. & Saveant, J. Electroneutrality coupling of electron hopping between localized sites with electroinactive counterion displacement. 1. Potential-step plateau currents. J. Phys. Chem. 92, 6761–6767 (1988).
Grey, C. P. & Tarascon, J. M. Sustainability and in situ monitoring in battery development. Nat. Mater. 16, 45–56 (2016).
Easley, A. D. et al. A practical guide to quartz crystal microbalance with dissipation monitoring of thin polymer films. J. Polym. Sci. 60, 1090–1107 (2021).
Wu, R., Matta, M., Paulsen, B. D. & Rivnay, J. Operando characterization of organic mixed ionic/electronic conducting materials. Chem. Rev. 122, 4493–4551 (2022).
Chen, H. & Ruckenstein, E. Hydrated ions: from individual ions to ion pairs to ion clusters. J. Phys. Chem. B 119, 12671–12676 (2015).
Flagg, L. Q., Giridharagopal, R., Guo, J. & Ginger, D. S. Anion-dependent doping and charge transport in organic electrochemical transistors. Chem. Mater. 30, 5380–5389 (2018).
Szumska, A. A. et al. Reversible electrochemical charging of n-type conjugated polymer electrodes in aqueous electrolytes. J. Am. Chem. Soc. 143, 14795–14805 (2021).
Lé, T. et al. Unveiling the ionic exchange mechanisms in vertically-oriented graphene nanosheet supercapacitor electrodes with electrochemical quartz crystal microbalance and a.c.-electrogravimetry. Electrochem. Commun. 93, 5–9 (2018).
Acknowledgements
The experimental work was supported by grant DE-SC0014006 funded by the US Department of Energy, Office of Science (T.M., R.M.T. and J.L.L.). The MD simulation work was supported by grant NSF-DMR-2119672 funded by the National Science Foundation, and the Texas A&M Institute for Data Science Career Initiation Fellowship (C.-H.L. and D.P.T.). The use of the Texas A&M University Soft Matter Facility (RRID:SCR_022482) and contribution of P. Wei are acknowledged.
Author information
Authors and Affiliations
Contributions
J.L.L. and T.M. conceived the study. T.M. developed the experimental procedures, carried out the experiments and analysed the data. T.M. and J.L.L. discussed the results and wrote the paper. R.M.T. performed the electron paramagnetic resonance and gel permeation chromatography tests. C.-H.L and D.P.T. conducted the MD simulation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Michel Armand and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–4, Figs. 1–45, discussions, references, PTAm synthesis, DFT computational details, MD computational details, kinetic investigations, Jones–Dole B coefficient, EQCM-D details, current collector|polymer film|electrolyte model, equations for anion and cation transfer in the PTAm reaction mechanism, and EIS calculations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, T., Li, CH., Thakur, R.M. et al. The role of the electrolyte in non-conjugated radical polymers for metal-free aqueous energy storage electrodes. Nat. Mater. 22, 495–502 (2023). https://doi.org/10.1038/s41563-023-01518-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-023-01518-z
This article is cited by
-
Reversible and high-density energy storage with polymers populated with bistable redox sites
Polymer Journal (2024)