Abstract
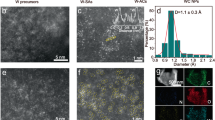
Single-atom catalysts have shown promising performance in various catalytic reactions. Catalytic metal sites supported on oxides or carbonaceous materials are usually strongly coordinated by oxygen or heteroatoms, which naturally affects their electronic environment and consequently their catalytic activity. Here, we reveal the stabilization of single-atom catalysts on tungsten carbides without the aid of heteroatom coordination for efficient catalysis of the oxygen evolution reaction (OER). Benefiting from the unique structure of tungsten carbides, the atomic FeNi catalytic sites are weakly bonded with the surface W and C atoms. The reported catalyst shows a low overpotential of 237 mV at 10 mA cm−2, which can even be lowered to 211 mV when the FeNi content is increased, a high turnover frequency value of 4.96 s−1 (η = 300 mV) and good stability (1,000 h). Density functional theory calculations show that either metallic Fe/Ni atoms or (hydro)oxide FeNi species are responsible for the high OER activity. We suggest that the application of inexpensive and durable WCx supports opens up a promising pathway to develop further single-atom catalysts for electrochemical catalytic reactions
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in the main text or the Supplementary Information. Source data are provided with this paper.
References
Seitz, L. C. et al. A highly active and stable IrOx/SrIrO3 catalyst for the oxygen evolution reaction. Science 353, 1011–1014 (2016).
Suntivich, J., May, K. J., Gasteiger, H. A., Goodenough, J. B. & Shao-Horn, Y. A perovskite oxide optimized for oxygen evolution catalysis from molecular orbital principles. Science 334, 1383–1385 (2011).
Seh, Z. W. et al. Combining theory and experiment in electrocatalysis: Insights into materials design. Science 355, eaad4998 (2017).
Smith, R. D. L. et al. Photochemical route for accessing amorphous metal oxide materials for water oxidation catalysis. Science 340, 60–63 (2013).
Yang, L. et al. Efficient oxygen evolution electrocatalysis in acid by a perovskite with face-sharing IrO6 octahedral dimers. Nat. Commun. 9, 5236 (2018).
Kanan, M. W. & Nocera, D. G. In situ formation of an oxygen-evolving catalyst in neutral water containing phosphate and Co2+. Science 321, 1072–1075 (2008).
Dincă, M., Surendranath, Y. & Nocera, D. G. Nickel-borate oxygen-evolving catalyst that functions under benign conditions. Proc. Natl Acad. Sci. USA 107, 10337–10341 (2010).
Zhao, S. et al. Ultrathin metal–organic framework nanosheets for electrocatalytic oxygen evolution. Nat. Energy 1, 16184 (2016).
Toma, F. M. et al. Efficient water oxidation at carbon nanotube–polyoxometalate electrocatalytic interfaces. Nat. Chem. 2, 826–831 (2010).
Yin, Q. et al. A fast soluble carbon-free molecular water oxidation catalyst based on abundant metals. Science 328, 342–345 (2010).
Zhao, Q., Yan, Z., Chen, C. & Chen, J. Spinels: controlled preparation, oxygen reduction/evolution reaction application, and beyond. Chem. Rev. 117, 10121–10211 (2017).
Yan, J. Q. et al. Single atom tungsten doped ultrathin α-Ni(OH)2 for enhanced electrocatalytic water oxidation. Nat. Commun. 10, 2149 (2019).
Subbaraman, R. et al. Trends in activity for the water electrolyser reactions on 3d M(Ni,Co,Fe,Mn) hydr(oxy)oxide catalysts. Nat. Mater. 11, 550–557 (2012).
Song, F. & Hu, X. Exfoliation of layered double hydroxides for enhanced oxygen evolution catalysis. Nat. Commun. 5, 4477 (2014).
Louie, M. W. & Bell, A. T. An investigation of thin-film Ni–Fe oxide catalysts for the electrochemical evolution of oxygen. J. Am. Chem. Soc. 135, 12329–12337 (2013).
Nong, H. N. et al. A unique oxygen ligand environment facilitates water oxidation in hole-doped IrNiOx core–shell electrocatalysts. Nat. Catal. 1, 841–851 (2018).
Bak, J., Bin Bae, H. & Chung, S.-Y. Atomic-scale perturbation of oxygen octahedra via surface ion exchange in perovskite nickelates boosts water oxidation. Nat. Commun. 10, 2713 (2019).
Zhang, B. et al. Homogeneously dispersed multimetal oxygen-evolving catalysts. Science 352, 333–337 (2016).
Li, P. et al. Boosting oxygen evolution of single-atomic ruthenium through electronic coupling with cobalt-iron layered double hydroxides. Nat. Commun. 10, 1711 (2019).
Zhang, B. et al. High-valence metals improve oxygen evolution reaction performance by modulating 3d metal oxidation cycle energetics. Nat. Catal. 3, 985–992 (2020).
Zhang, L., Zhou, M., Wang, A. & Zhang, T. Selective hydrogenation over supported metal catalysts: from nanoparticles to single atoms. Chem. Rev. 120, 683–733 (2020).
Li, Z. et al. Well-defined materials for heterogeneous catalysis: from nanoparticles to isolated single-atom sites. Chem. Rev. 120, 623–682 (2020).
Fei, H. et al. Single atom electrocatalysts supported on graphene or graphene-like carbons. Chem. Soc. Rev. 48, 5207–5241 (2019).
Xu, H., Cheng, D., Cao, D. & Zeng, X. C. A universal principle for a rational design of single-atom electrocatalysts. Nat. Catal. 1, 339–348 (2018).
Gu, J., Hsu, C.-S., Bai, L., Chen, H. M. & Hu, X. Atomically dispersed Fe3+ sites catalyze efficient CO2 electroreduction to CO. Science 364, 1091–1094 (2019).
Yang, X. F. et al. Single-atom catalysts: a new frontier in heterogeneous catalysis. Acc. Chem. Res. 46, 1740–1748 (2013).
Shi, Q., Zhu, C., Du, D. & Lin, Y. Robust noble metal-based electrocatalysts for oxygen evolution reaction. Chem. Soc. Rev. 48, 3181–3192 (2019).
Yan, Z. H. et al. Anion insertion enhanced electrodeposition of robust metal hydroxide/oxide electrodes for oxygen evolution. Nat. Commun. 9, 2373 (2018).
Kelly, T. G. & Chen, J. G. Metal overlayer on metal carbide substrate: unique bimetallic properties for catalysis and electrocatalysis. Chem. Soc. Rev. 41, 8021–8034 (2012).
Wan, W., Tackett, B. M. & Chen, J. G. Reactions of water and C1 molecules on carbide and metal-modified carbide surfaces. Chem. Soc. Rev. 46, 1807–1823 (2017).
Hou, Y. et al. Atomically dispersed nickel-nitrogen-sulfur species anchored on porous carbon nanosheets for efficient water oxidation. Nat. Commun. 10, 1392 (2019).
Fei, H. et al. General synthesis and definitive structural identification of MN4C4 single-atom catalysts with tunable electrocatalytic activities. Nat. Catal. 1, 63–72 (2018).
Kuai, C. et al. Phase segregation reversibility in mixed-metal hydroxide water oxidation catalysts. Nat. Catal. 3, 743–753 (2020).
Swierk, J. R., Klaus, S., Trotochaud, L., Bell, A. T. & Tilley, T. D. Electrochemical study of the energetics of the oxygen evolution reaction at nickel iron (oxy)hydroxide catalysts. J. Phys. Chem. C 119, 19022–19029 (2015).
Jiang, J. et al. Atomic-level insight into super-efficient electrocatalytic oxygen evolution on iron and vanadium co-doped nickel (oxy)hydroxide. Nat. Commun. 9, 2885 (2018).
Acknowledgements
We thank C. Eichenauer and M. Unterweger for assistance with TGA and X-ray diffraction analysis; we are also grateful to the high-performance computing centre and J. Zhou of Nanjing Tech University for support with computational resources. The authors also thank Ceshigo for technical support with DFT calculations and XAS data. We also thank ZELMI, TU-Berlin for supporting SEM measurements. This work is funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany’s Excellence Strategy-EXC 2008/1 (UniSysCat)-390540038 and Basismodul, Eigene Stelle (LI 3545/1-1)-449814841, postdoctoral funding from TU-Berlin (3-1323552-07-01, 3-2323552-07-01), National Key R&D Programme of China (2019YFA0110600, 2019YFA0110601), National Natural Science Foundation of China (11902144), Science and Technology Project of Sichuan Province (2020YFH0087) and Natural Science Foundation of the Jiangsu Higher Education Institutions (19KJB430022). This project has also received funding from the European Union’s Horizon 2020 Research and Innovation Programme under grant agreement no. 823717−ESTEEM3.
Author information
Authors and Affiliations
Contributions
S.L. and A.T. conceived and designed the experiments. S.L. synthesized the materials and performed catalytic tests. Y.W. and P.A.v.A. performed STEM imaging and analytical characterization, and analysed the STEM data. B.C., S.L. and C.C. conceived the module structure and carried out the DFT calculations. C.C. assisted with measurement of XAS and data analysis. M.-Y.Y. performed the SEM measurements. S.L., C.C. and A.T. wrote the whole paper together.
Corresponding authors
Ethics declarations
Competing interests
S.L. and A.T. have filed a provisional patent application regarding the synthesis of WCx-supported single-atom catalysts. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Materials thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Chemical reagents and materials. Characterizations and methods. Experimental sections. Electrochemical measurements. Calculation methods. Supplementary Figs. 1–44 and Tables 1–6. Captions for Videos 1–3.
Supplementary Video 1
The active atoms on WCx under STEM.
Supplementary Video 2
The active atoms at the edge of WCx under STEM.
Supplementary Video 3
The production of O2 on the electrode.
Source data
Source Data Fig. 1
Source data.
Source Data Fig. 2
Source data.
Source Data Fig. 3
Source data.
Source Data Fig. 4
Source data.
Source Data Fig. 5
Source data.
Rights and permissions
About this article
Cite this article
Li, S., Chen, B., Wang, Y. et al. Oxygen-evolving catalytic atoms on metal carbides. Nat. Mater. 20, 1240–1247 (2021). https://doi.org/10.1038/s41563-021-01006-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-021-01006-2
This article is cited by
-
Microenvironment reconstitution of highly active Ni single atoms on oxygen-incorporated Mo2C for water splitting
Nature Communications (2024)
-
Co-catalytic metal–support interactions in single-atom electrocatalysts
Nature Reviews Materials (2024)
-
Integrated energy storage and CO2 conversion using an aqueous battery with tamed asymmetric reactions
Nature Communications (2024)
-
Covalently Bonded Ni Sites in Black Phosphorene with Electron Redistribution for Efficient Metal-Lightweighted Water Electrolysis
Nano-Micro Letters (2024)
-
Atomically dispersed materials: Ideal catalysts in atomic era
Nano Research (2024)