Abstract
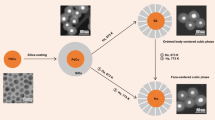
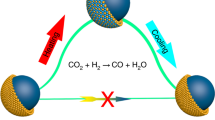
Extending the toolbox from mono- to bimetallic catalysts is key in realizing efficient chemical processes1. Traditionally, the performance of bimetallic catalysts featuring one active and one selective metal is optimized by varying the metal composition1,2,3, often resulting in a compromise between the catalytic properties of the two metals4,5,6. Here we show that by designing the atomic distribution of bimetallic Au–Pd nanocatalysts, we obtain a synergistic catalytic performance in the industrially relevant selective hydrogenation of butadiene. Our single-crystalline Au-core Pd-shell nanorods were up to 50 times more active than their alloyed and monometallic counterparts, while retaining high selectivity. We find a shell-thickness-dependent catalytic activity, indicating that not only the nature of the surface but also several subsurface layers play a crucial role in the catalytic performance, and rationalize this finding using density functional theory calculations. Our results open up an alternative avenue for the structural design of bimetallic catalysts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All raw data are available upon request by contacting the corresponding authors. Source data are provided with this paper.
References
Zhang, L., Zhou, M., Wang, A. & Zhang, T. Selective hydrogenation over supported metal catalysts: from nanoparticles to single atoms. Chem. Rev. 120, 683–733 (2020).
Wu, C. H. et al. Bimetallic synergy in cobalt–palladium nanocatalysts for CO oxidation. Nat. Catal. 2, 78–85 (2019).
Studt, F. et al. Identification of non-precious metal alloy catalysts for selective hydrogenation of acetylene. Science 320, 1320–1322 (2008).
Enache, D. I. et al. Solvent-free oxidation of primary alcohols to aldehydes using Au-Pd/TiO2 catalysts. Science 311, 362–365 (2006).
Hugon, A., Delannoy, L., Krafft, J. M. & Louis, C. Selective hydrogenation of 1,3-butadiene in the presence of an excess of alkenes over supported bimetallic gold palladium catalysts. J. Phys. Chem. C 114, 10823–10835 (2010).
Masoud, N., Delannoy, L., Calers, C. & Gallet, J. J. Silica-supported Au–Ag catalysts for the selective hydrogenation of butadiene. Chem. Cat. Chem. 9, 2418–2425 (2017).
Lucci, F. R. et al. Selective hydrogenation of 1,3-butadiene on platinum–copper alloys at the single-atom limit. Nat. Commun. 6, 8550 (2015).
Wang, G. H. et al. Platinum–cobalt bimetallic nanoparticles in hollow carbon nanospheres for hydrogenolysis of 5-hydroxymethylfurfural. Nat. Mater. 13, 293–300 (2014).
Bu, L. et al. Biaxially strained PtPb/Pt core/shell nanoplate boosts oxygen reduction catalysis. Science 354, 1410–1414 (2016).
Wang, X. et al. Palladium–platinum core–shell icosahedra with substantially enhanced activity and durability towards oxygen reduction. Nat. Commun. 6, 7594 (2015).
Strasser, P. et al. Lattice-strain control of the activity in dealloyed core–shell fuel cell catalysts. Nat. Chem. 2, 454–460 (2010).
Zugic, B. et al. Dynamic restructuring drives catalytic activity on nanoporous gold–silver alloy catalysts. Nat. Mater. 16, 558–565 (2016).
Ahmadi, M., Behafarid, F., Cui, C., Strasser, P. & Cuenya, B. R. Long-range segregation phenomena in shape-selected bimetallic nanoparticles: chemical state effects. ACS Nano 7, 9195–9204 (2013).
Wrasman, C. J. et al. Synthesis of colloidal Pd/Au dilute alloy nanocrystals and their potential for selective catalytic oxidations. J. Am. Chem. Soc. 140, 12930–12939 (2018).
Luneau, M. et al. Dilute Pd/Au alloy nanoparticles embedded in colloid-templated porous SiO2: stable Au-based oxidation catalysts. Chem. Mater. 31, 5759–5768 (2019).
Lee, J. D. et al. Tuning the electrocatalytic oxygen reduction reaction activity of Pt–Co nanocrystals by cobalt concentration with atomic-scale understanding. ACS Appl. Mater. Interfaces 11, 26789–26797 (2019).
Gilroy, K. D., Ruditskiy, A., Peng, H. C., Qin, D. & Xia, Y. Bimetallic nanocrystals: syntheses, properties, and applications. Chem. Rev. 116, 10414–10472 (2016).
Deng, T. S. et al. Oxidative etching and metal overgrowth of gold nanorods within mesoporous silica shells. Chem. Mater. 27, 7196–7230 (2015).
Zhang, Y. J. et al. Probing the electronic structure of heterogeneous metal interfaces by transition metal shelled gold nanoparticle-enhanced Raman spectroscopy. J. Phys. Chem. C 120, 20684–20691 (2016).
van der Hoeven, J. E. S. et al. In situ observation of atomic redistribution in alloying gold–silver nanorods. ACS Nano 12, 8467–8476 (2018).
Yu, W. Y., Mullen, G. M. & Mullins, C. B. Hydrogen adsorption and absorption with Pd–Au bimetallic surfaces. J. Phys. Chem. C 117, 19535–19543 (2013).
Molnár, A., Sárkány, A. & Varga, M. Hydrogenation of carbon–carbon multiple bonds: chemo-, regio- and stereo-selectivity. J. Mol. Catal. A Chem. 173, 185–221 (2001).
Silvestre-Albero, J., Rupprechter, G. & Freund, H. J. From Pd nanoparticles to single crystals: 1,3-butadiene hydrogenation on well-defined model catalysts. Chem. Commun. 80–82 (2006).
Bachiller-Baeza, B. et al. Detecting the genesis of a high-performance carbon-supported Pd sulfide nanophase and its evolution in the hydrogenation of butadiene. ACS Catal. 5, 5235–5241 (2015).
McEwan, L., Juliusa, M., Robertsa, S. & Fletchera, J. C. Q. A review of the use of gold catalysts in selective hydrogenation reactions. Gold. Bull. 43, 298–306 (2010).
Hugon, A., Delannoy, L. & Louis, C. Supported gold catalysts for selective hydrogenation of 1,3-butadiene in the presence of an excess of alkenes. Gold. Bull. 41, 127–138 (2008).
Masoud, N. et al. Superior stability of Au/SiO2 compared to Au/TiO2 catalysts for the selective hydrogenation of butadiene. ACS Catal. 7, 5594–5603 (2017).
Kolli, N. E., Delannoy, L. & Louis, C. Bimetallic Au–Pd catalysts for selective hydrogenation of butadiene: influence of the preparation method on catalytic properties. J. Catal. 297, 79–92 (2013).
Goris, B. et al. Atomic-scale determination of surface facets in gold nanorods. Nat. Mater. 11, 930–935 (2012).
Pinheiro, A., Zei, M., Luo, M. & Ertl, G. The epitaxial growth of Pd electrodeposition on Au(100) studied by LEED and RHEED. Surf. Sci. 600, 641–650 (2006).
Dolle, P. et al. Strained Pd films, by epitaxial growth on Au(110), to control catalytic properties. Surf. Sci. 518, 1–13 (2002).
Bertolini, J. C. et al. Electronic properties of supported Pd aggregates in relation with their reactivity for 1,3-butadiene hydrogenation. Catal. Lett. 6, 215–223 (1990).
Piccolo, L., Piednoir, A. & Bertolini, J. C. Pd-Au single-crystal surfaces: segregation properties and catalytic activity in the selective hydrogenation of 1,3-butadiene. Surf. Sci. 592, 169–181 (2005).
Acknowledgements
We thank R. Beerthuis and J.W. de Rijk for useful discussions and technical support. We thank N. Masoud for providing the gold catalyst reference data. We thank S. Dussi for critically reading the manuscript. We thank S. Zanoni for useful discussions regarding the carbon monoxide infrared spectroscopy measurements. This project received funding from the European Research Council under the European Union’s Horizon 2020 research and innovation programme (ERC-2014-CoG no. 648991) and the European Union’s Seventh Framework Programme (FP-2007-2013; European Research Council Advanced Grant Agreement #291667 HierarSACol). J.E.S.v.d.H. also acknowledges the graduate programme of the Debye Institute for Nanomaterials Science (Utrecht University), which is facilitated by grant 022.004.016 of the Netherlands Organisation for Scientific Research. J.J. and F.S. gratefully acknowledge support by the state of Baden-Württemberg through bwHPC (bwunicluster and JUSTUS, RV bw17D011) as well as financial support from the Helmholtz Association.
Author information
Authors and Affiliations
Contributions
J.E.S.v.d.H. performed the experiments supervised by A.v.B. and P.E.d.J.; J.J. performed the calculations under the supervision of F.S.; L.A.O. synthesized the NRs supervised by J.E.S.v.d.H.; and G.T. assisted in the catalysis experiments. R.J.A.v.D.-M. performed the inductively coupled plasma atomic emission spectroscopy measurements. J.-M.K. and C.L. conducted and interpreted the CO-DRIFTS analysis. J.E.S.v.d.H. and P.E.d.J. wrote the paper with the contributions of all the authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Materials thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Figs. 1–16 and Tables 1–4.
Computational Data 1
Cartesian coordinates used to compute the binding energies of butadiene (C4H6, Fig. 4a,b), propene (C3H6, Fig. 4b) and butene (C4H8, Fig. 4b) in the sigma and pi binding configuration, and for the hydrogen binding energy (2H) on the Au, 1Pd, 2Pd, 3Pd, 4Pd and 8Pd structures. The hydrogen and butadiene binding energies on an 8Pd structure with Au lattice spacing (Fig. 4c). The coordinates are given for a 111, 100 or 110 fcc surface structure.
Source data
Source Data Fig. 1
Source data for Fig. 1b.
Source Data Fig. 2
Source data for Fig. 2b–d.
Source Data Fig. 3
Source data for Fig. 3a–c.
Source Data Fig. 4
Source data for Fig. 4a–c.
Rights and permissions
About this article
Cite this article
van der Hoeven, J.E.S., Jelic, J., Olthof, L.A. et al. Unlocking synergy in bimetallic catalysts by core–shell design. Nat. Mater. 20, 1216–1220 (2021). https://doi.org/10.1038/s41563-021-00996-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-021-00996-3
This article is cited by
-
Strategies to improve hydrogen activation on gold catalysts
Nature Reviews Chemistry (2024)
-
Probing the atomically diffuse interfaces in Pd@Pt core-shell nanoparticles in three dimensions
Nature Communications (2023)
-
Conjugated dual size effect of core-shell particles synergizes bimetallic catalysis
Nature Communications (2023)
-
Synthesis of core/shell nanocrystals with ordered intermetallic single-atom alloy layers for nitrate electroreduction to ammonia
Nature Synthesis (2023)
-
A study of size-controlled Au@Cu2O nanocomposite for highly improved methyl orange catalytic performances
Journal of Materials Science (2023)