Abstract
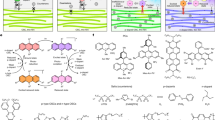
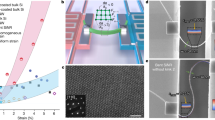
Rapid increase in the power conversion efficiency of organic solar cells (OSCs) has been achieved with the development of non-fullerene small-molecule acceptors (NF-SMAs). Although the morphological stability of these NF-SMA devices critically affects their intrinsic lifetime, their fundamental intermolecular interactions and how they govern property–function relations and morphological stability of OSCs remain elusive. Here, we discover that the diffusion of an NF-SMA into the donor polymer exhibits Arrhenius behaviour and that the activation energy Ea scales linearly with the enthalpic interaction parameters χH between the polymer and the NF-SMA. Consequently, the thermodynamically most unstable, hypo-miscible systems (high χ) are the most kinetically stabilized. We relate the differences in Ea to measured and selectively simulated molecular self-interaction properties of the constituent materials and develop quantitative property–function relations that link thermal and mechanical characteristics of the NF-SMA and polymer to predict relative diffusion properties and thus morphological stability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data represented in Fig. 3a,b are provided with the paper as source data. Other datasets generated and/or analysed during the current study are available from the corresponding authors upon request.
Code availability
The code used for the simulation of the Flory–Huggins phase diagram is available from the corresponding author upon request.
References
Zhang, G. et al. Nonfullerene acceptor molecules for bulk heterojunction organic solar cells. Chem. Rev. 118, 3447–3507 (2018).
Baran, D. et al. Reducing the efficiency–stability–cost gap of organic photovoltaics with highly efficient and stable small molecule acceptor ternary solar cells. Nat. Mater. 16, 363–369 (2016).
Du, X. et al. Efficient polymer solar cells based on non-fullerene acceptors with potential device lifetime approaching 10 years. Joule 3, 215–226 (2019).
Cha, H. et al. An efficient, ‘burn in’ free organic solar cell employing a nonfullerene electron acceptor. Adv. Mater. 29, 1701156 (2017).
Du, X. et al. Unraveling the microstructure-related device stability for polymer solar cells based on nonfullerene small–molecular acceptors. Adv. Mater. 32, 1908305 (2020).
Li, N. et al. Abnormal strong burn-in degradation of highly efficient polymer solar cells caused by spinodal donor–acceptor demixing. Nat. Commun. 8, 14541 (2017).
Ghasemi, M. et al. Delineation of thermodynamic and kinetic factors that control stability in non-fullerene organic solar cells. Joule 3, 1328–1348 (2019).
Wong, H. C. et al. Morphological stability and performance of polymer–fullerene solar cells under thermal stress: the impact of photoinduced PC60BM oligomerization. ACS Nano 8, 1297–1308 (2014).
Zhu, Y. et al. Rational strategy to stabilize an unstable high-efficiency binary nonfullerene organic solar cells with a third component. Adv. Energy Mater. 9, 1900376 (2019).
Ye, L. et al. Quantitative relations between interaction parameter, miscibility and function in organic solar cells. Nat. Mater. 17, 253–260 (2018).
Hu, H. et al. Effect of ring-fusion on miscibility and domain purity: key factors determining the performance of PDI-based nonfullerene organic solar cells. Adv. Energy Mater. 8, 1800234 (2018).
Ma, W. et al. Domain purity, miscibility, and molecular orientation at donor/acceptor interfaces in high performance organic solar cells: paths to further improvement. Adv. Energy Mater. 3, 864–872 (2013).
Treat, N. D. et al. Polymer–fullerene miscibility: a metric for screening new materials for high-performance organic solar cells. J. Am. Chem. Soc. 134, 15869–15879 (2012).
Zhang, C. et al. Understanding the correlation and balance between the miscibility and optoelectronic properties of polymer–fullerene solar cells. J. Mater. Chem. A 5, 17570–17579 (2017).
Ghasemi, M. et al. Panchromatic sequentially cast ternary polymer solar cells. Adv. Mater. 29, 1604603 (2017).
Li, S. et al. An unfused-core-based nonfullerene acceptor enables high-efficiency organic solar cells with excellent morphological stability at high temperatures. Adv. Mater. 30, 1705208 (2018).
Yuan, J. et al. Single-junction organic solar cell with over 15% efficiency using fused-ring acceptor with electron-deficient core. Joule 3, 1140–1151 (2019).
Yang, W. et al. Simultaneous enhanced efficiency and thermal stability in organic solar cells from a polymer acceptor additive. Nat. Commun. 11, 1218 (2020).
Ye, L. et al. Miscibility–function relations in organic solar cells: significance of optimal miscibility in relation to percolation. Adv. Energy Mater. 8, 1703058 (2018).
Treat, N. D., Mates, T. E., Hawker, C. J., Kramer, E. J. & Chabinyc, M. L. Temperature dependence of the diffusion coefficient of PCBM in poly(3-hexylthiophene). Macromolecules 46, 1002–1007 (2013).
Zhao, W. et al. Fullerene-free polymer solar cells with over 11% efficiency and excellent thermal stability. Adv. Mater. 28, 4734–4739 (2016).
Treat, N. D. et al. Interdiffusion of PCBM and P3HT reveals miscibility in a photovoltaically active blend. Adv. Energy Mater. 1, 82–89 (2011).
Ye, L. et al. Quenching to the percolation threshold in organic solar cells. Joule 3, 443–458 (2019).
Yazmaciyan, A. et al. Recombination losses above and below the transport percolation threshold in bulk heterojunction organic solar cells. Adv. Energy Mater. 8, 1703339 (2018).
Bartelt, J. A. et al. The importance of fullerene percolation in the mixed regions of polymer–fullerene bulk heterojunction solar cells. Adv. Energy Mater. 3, 364–374 (2013).
Li, S. et al. Energy-level modulation of small-molecule electron acceptors to achieve over 12% efficiency in polymer solar cells. Adv. Mater. 28, 9423–9429 (2016).
Flory, P. J. Principles of Polymer Chemistry (Cornell Univ. Press, 1953).
Rubinstein, M. & Colby, R. H. Polymer Physics (Oxford Univ. Press, 2003).
Batzer, H. & Kreibich, U. Connections between glass transition-temperature, thermodynamic and mechanic values for predicting material properties from the chemical-structure. Die Angew. Makromol. Chem. 105, 113–130 (1982).
Kreibich, U. & Batzer, H. Influence of the segment structure and crosslinking on the glass-transition Tg-possibilities of predicting Tg using the values of cohesive energy ecoh. Die Angew. Makromol. Chem. 83, 57–112 (1979).
Lee, C. J. Correlations of elastic modulus, cohesive energy density and heat capacity jump of glassy polymers. Polyrn. Eng. Sci. 27, 1015–1017 (1987).
Roberts, R. J., Rowe, R. C. & York, P. The relationship between Young’s modulus of elasticity of organic solids and their molecular structure. Powder Technol. 65, 139–146 (1991).
Root, S. E., Alkhadra, M. A., Rodriquez, D., Printz, A. D. & Lipomi, D. J. Measuring the glass transition temperature of conjugated polymer films with ultraviolet–visible spectroscopy. Chem. Mater. 29, 2646–2654 (2017).
Sharma, A., Pan, X., Campbell, J. A., Andersson, M. R. & Lewis, D. A. Unravelling the thermomechanical properties of bulk heterojunction blends in polymer solar cells. Macromolecules 50, 3347–3354 (2017).
Balar, N. et al. The importance of entanglements in optimizing the mechanical and electrical performance of all-polymer solar cells. Chem. Mater. 31, 5124–5132 (2019).
Sperling, L. H. Introduction to Physical Polymer Science (John Wiley & Sons, 2005).
Lodge, T. P. Reconciliation of the molecular weight dependence of diffusion and viscosity in entangled polymers. Phys. Rev. Lett. 83, 3218–3221 (1999).
Thomas, N. L. & Windle, A. H. A theory of case II diffusion. Polymer 23, 529–542 (1982).
Matsuoka, S. (ed.) Relaxation Phenomena in Polymers (Hanser, 1992).
Balar, N. & O’Connor, B. T. Correlating crack onset strain and cohesive fracture energy in polymer semiconductor films. Macromolecules 50, 8611–8618 (2017).
Stafford, C. M. et al. A buckling-based metrology for measuring the elastic moduli of polymeric thin films. Nat. Mater. 3, 545–550 (2004).
Virkar, A. A., Mannsfeld, S., Bao, Z. & Stingelin, N. Organic semiconductor growth and morphology considerations for organic thin-film transistors. Adv. Mater. 22, 3857–3875 (2010).
Kim, J.-S. et al. Tuning mechanical and optoelectrical properties of poly(3-hexylthiophene) through systematic regioregularity control. Macromolecules 48, 4339–4346 (2015).
Collins, B. A. et al. Molecular miscibility of polymer−fullerene blends. J. Phys. Chem. Lett. 1, 3160–3166 (2010).
Standard Test Methods for Photovoltaic Modules in Cyclic Temperature and Humidity Environments ASTM E1171-15 (ASTM International, 2015).
White, J. R. On internal stress and activation volume in polymers. J. Mater. Sci. 16, 3249–3262 (1981).
Mehrer, H. Diffusion in Solids: Fundamentals, Methods, Materials, Diffusion-Controlled Processes (Springer Science & Business Media, 2007).
Deppe, D. D., Miller, R. D. & Torkelson, J. M. Small molecule diffusion in a rubbery polymer near Tg: effects of probe size, shape, and flexibility. J. Polym. Sci., B: Polym. Phys. 34, 2987–2997 (1996).
Carpenter, J. H. et al. Competition between exceptionally long-range alkyl sidechain ordering and backbone ordering in semiconducting polymers and its impact on electronic and optoelectronic properties. Adv. Funct. Mater. 29, 1806977 (2019).
Alkhadra, M. A. et al. Quantifying the fracture behavior of brittle and ductile thin films of semiconducting polymers. Chem. Mater. 29, 10139–10149 (2017).
Kozub, D. R. et al. Polymer crystallization of partially miscible polythiophene/fullerene mixtures controls morphology. Macromolecules 44, 5722–5726 (2011).
Abraham, M. J. et al. GROMACS: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1-2, 19–25 (2015).
Jorgensen, W. L., Maxwell, D. S. & Tirado-Rives, J. Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225–11236 (1996).
Frisch, M. J. et al. Gaussian 16 Rev. C.01 (Wallingford, 2016).
Ryno, S. M. & Risko, C. Deconstructing the behavior of donor–acceptor copolymers in solution & the melt: the case of PTB7. Phys. Chem. Chem. Phys. 21, 7802–7813 (2019).
Acknowledgements
Y.Q., Z.P., H.H., H.A. and initial work by M.G. was supported by Office of Naval Research (ONR) grant no. N000141712204 and KAUST’s Center Partnership Fund (no. 3321). N.B. and B.T.O. acknowledge support by a National Science Foundation (NSF) grant (no. CMMI-1554322). T.K., A.A. and recent work by M.G. was supported by NCSU start-up funds to A.A., J.R. and W.Y. acknowledge support by an NSF grant (no. CBET-1639429). C.R. and W.M. acknowledge the support of the ONR (N00014-18-1-2448) and the NSF under Cooperative Agreement no. 1849213; supercomputing resources were provided by the Department of Defense (DoD) through the DoD High-Performance Computing Modernization Program (project no. ONRDC40433481) and by the University of Kentucky Information Technology Department and Center for Computational Sciences. SIMS measurements were performed at the Analytical Instrumentation Facility at NCSU, which is partially supported by the State of North Carolina and the National Science Foundation. C. Zhou is acknowledged for providing support for SIMS measurements. The DSC instrument was purchased with UNC-GA ROI funds. C. Zhu, A. Hexemer and C. Wang of the ALS provided instrument maintenance. E. Gomez and J. Litofsky are acknowledged for providing the initial Flory–Huggins program code. L. Ye and M. Balik (NCSU) are acknowledged for fruitful discussion and input. A. Dinku is acknowledged for maintaining shared ORaCEL facilities and sharing some PBDB-T2F:Y6 stability data for reference. F. He and T. Zhao are acknowledged for help with attaining molecular weight data via high temperature gel permeation chromatography. H. Yan is acknowledged for providing ITIC-4Cl NF-SMA. I. Angunawela is acknowledged for performing complementary shelflife measurements of P3HT:EH-IDTBR devices.
Author information
Authors and Affiliations
Contributions
H.A. and B.T.O. conceived the scientific framework with the help of M.G. M.G. designed experimental protocols, coordinated the experimental work, performed the SIMS, DSC measurements and analysed the SIMS and DSC data with the help of Z.P. Z.P. performed the complementary SIMS measurements of P3HT:NF-SMA. M.G. fabricated solar cell devices and performed subsequent stability tests with the help of H.H., T.K. and Y.Q., and with supervision by A.A. B.T.O. and N.B. designed the mechanical test experiments. N.B. prepared the films needed for mechanical test measurements and performed DMA and elastic modulus measurements. H.H. fabricated the complementary FTAZ:IT-M devices. J.J.R. synthesized the FTAZ polymers, supervised by W.Y. M.B. synthesized P3HT, supervised by I.M. W.M. performed molecular dynamics simulations, supervised by C.R. M.G., H.A. and B.T.O. drafted the paper. All authors contributed to the editing and interpretation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Materials thanks Mats Andersson, Andrew T. Kleinschmidt, Darren Lipomi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–13, equations (1)–(15), Tables 1–9 and Discussion.
Source data
Source Data Fig. 3
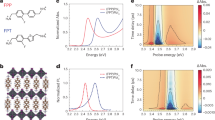
DSC thermograms of neat polymers and NF-SMAs and time-temperature superposition of neat polymers.
Rights and permissions
About this article
Cite this article
Ghasemi, M., Balar, N., Peng, Z. et al. A molecular interaction–diffusion framework for predicting organic solar cell stability. Nat. Mater. 20, 525–532 (2021). https://doi.org/10.1038/s41563-020-00872-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-020-00872-6
This article is cited by
-
Nanoscale doping of polymeric semiconductors with confined electrochemical ion implantation
Nature Nanotechnology (2024)
-
Perovskite–organic tandem solar cells
Nature Reviews Materials (2024)
-
The role of interfacial donor–acceptor percolation in efficient and stable all-polymer solar cells
Nature Communications (2024)
-
A case study of comparing two dimerized acceptor molecules built by different branch-connected and terminal-connected approaches
Science China Chemistry (2024)
-
SrTiO3–TiO2 Litchi-Like Hollow Nanospheres for Superior Photocatalytic Hydrogen Production
Catalysis Letters (2024)