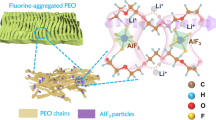
Abstract
Metal fluoride conversion cathodes offer a pathway towards developing lower-cost Li-ion batteries. Unfortunately, such cathodes suffer from extremely poor performance at elevated temperatures, which may prevent their use in large-scale energy storage applications. Here we report that replacing commonly used organic electrolytes with solid polymer electrolytes may overcome this hurdle. We demonstrate long-cycle stability for over 300 cycles at 50 °C attained in high-capacity (>450 mAh g−1) FeF2 cathodes. The absence of liquid solvents reduced electrolyte decomposition, while mechanical properties of the solid polymer electrolyte enhanced cathode structural stability. Our findings suggest that the formation of an elastic, thin and homogeneous cathode electrolyte interphase layer on active particles is a key for stable performance. The successful operation of metal fluorides at elevated temperatures opens a new avenue for their practical applications and future successful commercialization.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data used in this study are available from the authors upon reasonable request.
References
Tarascon, J. M. & Armand, M. Issues and challenges facing rechargeable lithium batteries. Nat. Rev. 414, 359–367 (2001).
Nitta, N., Wu, F., Lee, J. T. & Yushin, G. Li-ion battery materials: present and future. Mater. Today 18, 252–264 (2015).
Wang, Q. et al. Thermal runaway caused fire and explosion of lithium ion battery. J. Power Sources 208, 210–224 (2012).
Turcheniuk, K., Bondarev, D., Singhal, V. & Yushin, G. Ten years left to redesign lithium-ion batteries. Nature 559, 467–470 (2018).
Wu, F. & Yushin, G. Conversion cathodes for rechargeable lithium and lithium-ion batteries. Energy Environ. Sci. 10, 435–459 (2017).
Badway, F. et al. Structure and electrochemistry of copper fluoride nanocomposites utilizing mixed conducting matrices. Chem. Mater. 19, 4129–4141 (2007).
Wang, X. et al. Carbon nanotube–CoF2 multifunctional cathode for lithium ion batteries: effect of electrolyte on cycle stability. Small 11, 5164–5173 (2015).
Gordon, D. et al. Mixed metal difluorides as high capacity conversion-type cathodes: impact of composition on stability and performance. Adv. Energy Mater 8, 1800213 (2018).
Huang, Q. et al. Fading mechanisms and voltage hysteresis in FeF2–NiF2 solid solution cathodes for lithium and lithium-ion batteries. Small 15, 1804670 (2019).
Fu, W. et al. Iron fluoride-carbon nanocomposite nanofibers as free-standing cathodes for high-energy lithium batteries. Adv. Funct. Mater. 28, 1801711 (2018).
Munao, D., Van Erven, J., Valvo, M., Garcia-Tamayo, E. & Kelder, E. Role of the binder on the failure mechanism of Si nano-composite electrodes for Li-ion batteries. J. Power Sources 196, 6695–6702 (2011).
Kim, S. W., Seo, D. H., Gwon, H., Kim, J. & Kang, K. Fabrication of FeF3 nanoflowers on CNT branches and their application to high power lithium rechargeable batteries. Adv. Mater. 22, 5260–5264 (2010).
Zhao, E. B. et al. Lithium-Iron (III) fluoride battery with double surface protection. Adv. Energy Mater. 8, 1800721 (2018).
Mansour, A., Badway, F., Yoon, W.-S., Chung, K. & Amatucci, G. In situ X-ray absorption spectroscopic investigation of the electrochemical conversion reactions of CuF2–MoO3 nanocomposite. J. Solid State Chem. 183, 3029–3038 (2010).
Badway, F. et al. Enabling aspects of metal halide nanocomposites for reversible energy storage. Mater. Res. Soc. Symp. Proc. 972, 0972-AA07-01-BB08-01 (2006).
Kim, S. et al. Improved performance in FeF2 conversion cathodes through use of a conductive 3D scaffold and Al2O3 ALD coating. Adv. Funct. Mater. 27, 1702783 (2017).
Fan, X. et al. High energy-density and reversibility of iron fluoride cathode enabled via an intercalation-extrusion reaction. Nat. Commun. 9, 2324 (2018).
Gu, W. et al. Lithium–iron fluoride battery with in situ surface protection. Adv. Funct. Mater. 26, 1507–1516 (2016).
Ebner, M., Marone, F., Stampanoni, M. & Wood, V. Visualization and quantification of electrochemical and mechanical degradation in Li ion batteries. Science 342, 716–720 (2013).
Gorecki, W., Jeannin, M., Belorizky, E., Roux, C. & Armand, M. Physical properties of solid polymer electrolyte PEO (LiTFSI) complexes. J. Phys. Condens. Matter 7, 6823 (1995).
Liu, W. et al. Enhancing ionic conductivity in composite polymer electrolytes with well-aligned ceramic nanowires. Nat. Energy 2, 17035 (2017).
Liu, W., Lin, D., Sun, J., Zhou, G. & Cui, Y. Improved lithium ionic conductivity in composite polymer electrolytes with oxide-ion conducting nanowires. ACS Nano 10, 11407–11413 (2016).
Tao, T., Lu, S. & Chen, Y. A review of advanced flexible lithium-ion batteries. Adv. Mater. Technol. 3, 1700375 (2018).
Pereira, N. & Amatucci, G. Iron oxyfluoride electrodes for electrochemical energy storage. US patent 8,623,549 B2 (2014).
Li, L. et al. Origins of large voltage hysteresis in high-energy-density metal fluoride lithium-ion battery conversion electrodes. J. Am. Chem. Soc. 138, 2838–2848 (2016).
Manthiram, A., Yu, X. & Wang, S. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater. 2, 16103 (2017).
Mao, X., Bai, Y., Yu, J. & Ding, B. Flexible and highly temperature resistant polynanocrystalline zirconia nanofibrous membranes designed for air filtration. J. Am. Ceram. Soc. 99, 2760–2768 (2016).
Noh, H.-J. et al. Cathode material with nanorod structure—an application for advanced high-energy and safe lithium batteries. Chem. Mater. 25, 2109–2115 (2013).
Zhou, J. S., Zhang, D., Zhang, X. T., Song, H. H. & Chen, X. H. Carbon-nanotube-encapsulated FeF2 nanorods for high-performance lithium-ion cathode materials. ACS Appl. Mater. Interfaces 6, 21223–21229 (2014).
Gu, W., Magasinski, A., Zdyrko, B. & Yushin, G. Metal fluorides nanoconfined in carbon nanopores as reversible high capacity cathodes for Li and Li-ion rechargeable batteries: FeF2 as an example. Adv. Energy Mater 5, 1401148 (2015).
Tang, M. et al. High-temperature electrochemical performance of FeF3/C nanocomposite as a cathode material for lithium-ion batteries. J. Mater. Eng. Perform. 27, 624–629 (2018).
Badway, F., Pereira, N., Cosandey, F. & Amatucci, G. G. Carbon-metal fluoride nanocomposites: structure and electrochemistry of FeF3: C. J. Electrochem. Soc. 150, A1209–A1218 (2003).
Sina, M. et al. Investigation of SEI layer formation in conversion iron fluoride cathodes by combined STEM/EELS and XPS. J. Phys. Chem. C 119, 9762–9773 (2015).
Reddy, M. A. et al. CFx derived Carbon–FeF2 nanocomposites for reversible lithium storage. Adv. Energy Mater. 3, 308–313 (2013).
Cabana, J., Monconduit, L., Larcher, D. & Palacin, M. R. Beyond intercalation-based Li-ion batteries: the state of the art and challenges of electrode materials reacting through conversion reactions. Adv. Mater. 22, E170–E192 (2010).
Dingreville, R., Qu, J. & Cherkaoui, M. Surface free energy and its effect on the elastic behavior of nano-sized particles, wires and films. J Mech. Phys. Solids 53, 1827–1854 (2005).
Zhang, D., Yan, H., Zhu, Z., Zhang, H. & Wang, J. Electrochemical stability of lithium bis (oxatlato) borate containing solid polymer electrolyte for lithium ion batteries. J. Power Sources 196, 10120–10125 (2011).
Pereira, N., Badway, F., Wartelsky, M., Gunn, S. & Amatucci, G. Iron oxyfluorides as high capacity cathode materials for lithium batteries. J. Electrochem. Soc. 156, A407–A416 (2009).
Ko, J. K. et al. Transport, phase reactions, and hysteresis of iron fluoride and oxyfluoride conversion electrode materials for lithium batteries. ACS Appl. Mater. Interfaces 6, 10858–10869 (2014).
Choukourov, A. et al. Vacuum thermal degradation of poly (ethylene oxide). J. Phys. Chem. B 113, 2984–2989 (2009).
Wang, J., Heuer, L. & Joseph, D. Aging properties of semidilute aqueous solutions of polyethylene oxide seeded with silica nanoparticles. J. Rheol. 49, 1303–1316 (2005).
Zheng, J. et al. 3D visualization of inhomogeneous multi-layered structure and Young’s modulus of the solid electrolyte interphase (SEI) on silicon anodes for lithium ion batteries. Phys. Chem. Chem. Phys. 16, 13229–13238 (2014).
Peled, E., Golodnitsky, D. & Ardel, G. Advanced model for solid electrolyte interphase electrodes in liquid and polymer electrolytes. J. Electrochem. Soc. 144, L208–L210 (1997).
Gmitter, A. J., Halajko, A., Sideris, P. J., Greenbaum, S. G. & Amatucci, G. G. Subsurface diffusion of oxide electrolyte decomposition products in metal fluoride nanocomposite electrodes. Electrochim. Acta 88, 735–744 (2013).
Amine, K. et al. Mechanism of capacity fade of MCMB/Li1.1[Ni1/3Mn1/3Co1/3]0.9O2 cell at elevated temperature and additives to improve its cycle life. J. Mater. Chem. 21, 17754–17759 (2011).
Song, B., Liu, Z., Lai, M. O. & Lu, L. Structural evolution and the capacity fade mechanism upon long-term cycling in Li-rich cathode material. Phys. Chem. Chem. Phys. 14, 12875–12883 (2012).
Wang, F. et al. Ternary metal fluorides as high-energy cathodes with low cycling hysteresis. Nat. Commun. 6, 6668 (2015).
Vijayalakshmi, S., Chakraborty, J. & Madras, G. Thermal and microwave-assisted oxidative degradation of poly (ethylene oxide). J. Appl. Polym. Sci. 96, 2090–2096 (2005).
Acknowledgements
This project was largely supported by the Army Research Office (grant No. W911NF−17-1-0053). The authors also wish to acknowledge fellowship support of Q. Huang and X. Ren by the China Scholarship Council.
Author information
Authors and Affiliations
Contributions
Q.H. contributed to the synthesis and electrochemical testing. Q.H., X.R., A.M., A.-Y.S., Y.X. and D.K. contributed to sample characterizations. K.T., Q.H. and G.Y. discussed and interpreted the experimental results. Q.H., K.T. and G.Y. wrote the paper. G.Y. conceived the idea.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–9.
Rights and permissions
About this article
Cite this article
Huang, Q., Turcheniuk, K., Ren, X. et al. Cycle stability of conversion-type iron fluoride lithium battery cathode at elevated temperatures in polymer electrolyte composites. Nat. Mater. 18, 1343–1349 (2019). https://doi.org/10.1038/s41563-019-0472-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-019-0472-7
This article is cited by
-
Converting intercalation-type cathode in spent lithium-ion batteries into conversion-type cathode
Nano Research (2024)
-
Stress and Manufacturability in Solid-State Lithium-Ion Batteries
International Journal of Precision Engineering and Manufacturing-Green Technology (2023)
-
Tactics to optimize conversion-type metal fluoride/sulfide/oxide cathodes toward advanced lithium metal batteries
Nano Research (2023)
-
Weaving tangled webs
Nature Materials (2022)
-
Amorphous iron fluorosulfate as a high-capacity cathode utilizing combined intercalation and conversion reactions with unexpectedly high reversibility
Nature Energy (2022)