Abstract
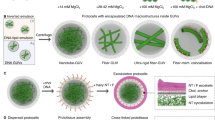
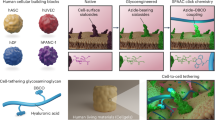
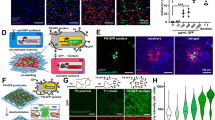
Although several new types of synthetic cell-like entities are now available, their structural integration into spatially interlinked prototissues that communicate and display coordinated functions remains a considerable challenge. Here we describe the programmed assembly of synthetic prototissue constructs based on the bio-orthogonal adhesion of a spatially confined binary community of protein–polymer protocells, termed proteinosomes. The thermoresponsive properties of the interlinked proteinosomes are used collectively to generate prototissue spheroids capable of reversible contractions that can be enzymatically modulated and exploited for mechanochemical transduction. Overall, our methodology opens up a route to the fabrication of artificial tissue-like materials capable of collective behaviours, and addresses important emerging challenges in bottom-up synthetic biology and bioinspired engineering.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The authors declare that all relevant data supporting the findings of this study are available within the paper and its Supplementary Information files. Additional data are available from the corresponding author upon reasonable request.
References
Mantri, S. & Sapra, K. T. Evolving protocells to prototissues: rational design of a missing link. Biochem. Soc. Trans. 41, 1159–1165 (2013).
Lentini, R. et al. Two-way chemical communication between artificial and natural cells. ACS Cent. Sci. 3, 117–123 (2017).
Qiao, H. et al. Encapsulation of nucleic acids into giant unilamellar vesicles by freeze–thaw: a way protocells may form. Orig. Life Evol. Biosph. 47, 499–510 (2016).
Altamura, E. et al. Highly oriented photosynthetic reaction centers generate a proton gradient in synthetic protocells. Proc. Natl Acad. Sci. USA 114, 3837–3842 (2017).
Adamala, K. P., Engelhart, A. E. & Szostak, J. W. Collaboration between primitive cell membranes and soluble catalysts. Nat. Commun. 7, 11041 (2016).
Jang, W. S. et al. Enzymatically triggered rupture of polymersomes. Soft Matter 12, 1014–1020 (2016).
Peyret, A. et al. Polymersome popping by light-induced osmotic shock under temporal, spatial, and spectral control. Angew. Chem. Int. Ed. 56, 1566–1570 (2017).
Schmitt, C., Lippert, A. H., Bonakdar, N., Sandoghdar, V. & Voll, L. M. Compartmentalization and transport in synthetic vesicles. Front. Bioeng. Biotechnol. 4, 19 (2016).
Che, H. & van Hest, J. C. M. Stimuli-responsive polymersomes and nanoreactors. J. Mater. Chem. B 4, 4632–4647 (2016).
Bellomo, E. G., Wyrsta, M. D., Pakstis, L., Pochan, D. J. & Deming, T. J. Stimuli-responsive polypeptide vesicles by conformation-specific assembly. Nat. Mater. 3, 244–248 (2004).
Holowka, E. P., Sun, V. Z., Kamei, D. T. & Deming, T. J. Polyarginine segments in block copolypeptides drive both vesicular assembly and intracellular delivery. Nat. Mater. 6, 52–57 (2007).
Xiao, Q. et al. Bioactive cell-like hybrids coassembled from (glyco)dendrimersomes with bacterial membranes. Proc. Natl Acad. Sci. USA 113, E1134–E1141 (2016).
Percec, V. et al. Self-assembly of Janus dendrimers into uniform dendrimersomes and other complex architectures. Science 328, 1009–1014 (2010).
Akkarachaneeyakorn, K., Li, M., Davis, S. A. & Mann, S. Secretion and reversible assembly of extracellular-like matrix by enzyme-active colloidosome-based protocells. Langmuir 32, 2912–2919 (2016).
Sun, S. et al. Chemical signaling and functional activation in colloidosome-based protocells. Small 12, 1920–1927 (2016).
Li, M., Huang, X. & Mann, S. Spontaneous growth and division in self-reproducing inorganic colloidosomes. Small 10, 3291–3298 (2014).
Li, M., Harbron, R. L., Weaver, J. V., Binks, B. P. & Mann, S. Electrostatically gated membrane permeability in inorganic protocells. Nat. Chem. 5, 529–536 (2013).
Yin, Y. et al. Non-equilibrium behaviour in coacervate-based protocells under electric-field-induced excitation. Nat. Commun. 7, 10658 (2016).
Williams, D. S., Patil, A. J. & Mann, S. Spontaneous structuration in coacervate-based protocells by polyoxometalate-mediated membrane assembly. Small 10, 1830–1840 (2014).
Tang, D. T.-Y. et al. Fatty acid membrane assembly on coacervate microdroplets as a step towards a hybrid protocell model. Nat. Chem. 6, 527–533 (2014).
Tang, D. T.-Y., van Swaay, D., deMello, A., Ross Anderson, J. L. & Mann, S. In vitro gene expression within membrane-free coacervate protocells. Chem. Commun. 51, 11429–11432 (2015).
Qiao, Y., Li, M., Booth, R. & Mann, S. Predatory behaviour in synthetic protocell communities. Nat. Chem. 9, 110–119 (2017).
Huang, X. et al. Interfacial assembly of protein–polymer nano-conjugates into stimulus-responsive biomimetic protocells. Nat. Commun. 4, 2239 (2013).
Huang, X., Li, M. & Mann, S. Membrane-mediated cascade reactions by enzyme-polymer proteinosomes. Chem. Commun. 50, 6278–6280 (2014).
Huang, X., Patil, A. J., Li, M. & Mann, S. Design and construction of higher-order structure and function in proteinosome-based protocells. J. Am. Chem. Soc. 136, 9225–9234 (2014).
Liu, X. et al. Hierarchical proteinosomes for programmed release of multiple components. Angew. Chem. Int. Ed. 55, 7095–7100 (2016).
Wen, P. et al. Coordinated membrane fusion of proteinosomes by contact-induced hydrogel self-healing. Small 13, 1700467 (2017).
Jin, H. et al. Reversible and large-scale cytomimetic vesicle aggregation: light-responsive host–guest interactions. Angew. Chem. Int. Ed. 50, 10352–10356 (2011).
Jin, H. et al. Cytomimetic large-scale vesicle aggregation and fusion based on host–guest interaction. Langmuir 28, 2066–2072 (2012).
Jin, H., Huang, W., Zheng, Y., Zhou, Y. & Yan, D. Construction of macroscopic cytomimetic vesicle aggregates based on click chemistry: controllable vesicle fusion and phase separation. Chemistry 18, 8641–8646 (2012).
Jin, H. et al. Three-component vesicle aggregation driven by adhesion interactions between Au nanoparticles and polydopamine-coated nanotubes. Chem. Commun. 50, 6157–6160 (2014).
Bai, Y. et al. A double droplet trap system for studying mass transport across a droplet–droplet interface. Lab. Chip 10, 1281–1285 (2010).
Zagnoni, M. & Cooper, J. M. A microdroplet-based shift register. Lab. Chip 10, 3069–3073 (2010).
Stanley, C. E. et al. A microfluidic approach for high-throughput droplet interface bilayer (DIB) formation. Chem. Commun. 46, 1620–1622 (2010).
Elani, Y., Law, R. V. & Ces, O. Vesicle-based artificial cells as chemical microreactors with spatially segregated reaction pathways. Nat. Commun. 5, 5305 (2014).
Dixit, S. S., Kim, H., Vasilyev, A., Eid, A. & Faris, G. W. Light-driven formation and rupture of droplet bilayers. Langmuir 26, 6193–6200 (2010).
Holden, M. A., Needham, D. & Bayley, H. Functional bionetworks from nanoliter water droplets. J. Am. Chem. Soc. 129, 8650–8655 (2007).
Leptihn, S. et al. Constructing droplet interface bilayers from the contact of aqueous droplets in oil. Nat. Protoc. 8, 1048–1057 (2013).
Hwang, W. L., Chen, M., Cronin, B., Holden, M. A. & Bayley, H. Asymmetric droplet interface bilayers. J. Am. Chem. Soc. 130, 5878–5879 (2008).
Milianta, P. J., Muzzio, M., Denver, J., Cawley, G. & Lee, S. Water permeability across symmetric and asymmetric droplet interface bilayers: interaction of cholesterol sulfate with DPhPC. Langmuir 31, 12187–12196 (2015).
Villar, G., Heron, A. J. & Bayley, H. Formation of droplet networks that function in aqueous environments. Nat. Nanotech. 6, 803–808 (2011).
Booth, M. J., Schild, V. R., Graham, A. D., Olof, S. N. & Bayley, H. Light-activated communication in synthetic tissues. Sci. Adv. 2, e1600056 (2016).
Villar, G., Graham, A. D. & Bayley, H. A tissue-like printed material. Science 340, 48–52 (2013).
Gobbo, P., Novoa, S., Biesinger, M. C. & Workentin, M. S. Interfacial strain-promoted alkyne-azide cycloaddition (I-SPAAC) for the synthesis of nanomaterial hybrids. Chem. Commun. 49, 3982–3984 (2013).
Gobbo, P. et al. Versatile strained alkyne modified water-soluble AuNPs for interfacial strain promoted azide-alkyne cycloaddition (I-SPAAC). J. Mater. Chem. B 2, 1764–1769 (2014).
Luo, W. et al. ‘Shine & click’ photo-induced interfacial unmasking of strained alkynes on small water-soluble gold nanoparticles. Chemistry 23, 1052–1059 (2017).
Gibson, L. J. & Ashby, M. F. Cellular Solids—Structure and Properties (Cambridge Univ. Press, Cambridge, 2014).
Liu, J., Sun, N., Bruce, M. A., Wu, J. C. & Butte, M. J. Atomic force mechanobiology of pluripotent stem cell-derived cardiomyocytes. PLoS ONE 7, e37559 (2012).
du Roure, O. et al. Force mapping in epithelial cell migration. Proc. Natl Acad. Sci. USA 102, 2390–2395 (2005).
Sabass, B., Koch, M. D., Liu, G., Stone, H. A. & Shaevitz, J. W. Force generation by groups of migrating bacteria. Proc. Natl Acad. Sci. USA 114, 7266–7271 (2017).
Acknowledgements
The authors thank NSERC Canada (PDF-487171–2016) and EU Horizon 2020 (Marie Skłodowska-Curie grant no. 701876) for funding, and the NMR/Mass Spectrometry Facility, the Wolfson Bioimaging Facility (A. Leard; BBSRC grant no. BB/L014181/1) and Chemical Imaging Facility (EPSRC grant ‘Atoms to Applications’, EP/K035746/1) for help with physical characterization. The authors also thank I. Manners, J. Finnegan and S. Briggs for assistance with GPC/DSC measurements, D. Woolfson for use of dichroism and plate reader spectrometers (BBSRC/EPSRC Bristol Synthetic Biology Research Centre, grant no. BB/L01386X/1), D. Gubala for help with AFM measurements, N. Martin and R. Booth for fruitful discussions, T. Ferrugia for assistance with the development of a customized microscope heating stage, and T. Liverpool for mathematical discussions.
Author information
Authors and Affiliations
Contributions
P.G., A.J.P., M.L. and S.M. conceived the experiments. P.G. performed the experiments. P.G. and R.H. performed the force measurements. W.H.B. and P.G. developed the theoretical model for the prototissue expansion force. All authors undertook data analysis, discussed the results, and contributed to drafts of the manuscript. P.G. and S.M. wrote the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Video Legends 1–5, Supplementary Notes 1–3, Supplementary Figures 1–34, Supplementary Table 1, Supplementary References 1–2
Supplementary Video 1
Caged prototissue spheroid
Supplementary Video 2
Uncaged prototissue spheroid
Supplementary Video 3
Prototissue spheroid undergoing reversible contractions
Supplementary Video 4
Prototissue undergoing reversible contractions with buckling protocells
Supplementary Video 5
Communication between compartments in an uncaged prototissue spheroid
Rights and permissions
About this article
Cite this article
Gobbo, P., Patil, A.J., Li, M. et al. Programmed assembly of synthetic protocells into thermoresponsive prototissues. Nature Mater 17, 1145–1153 (2018). https://doi.org/10.1038/s41563-018-0183-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-018-0183-5