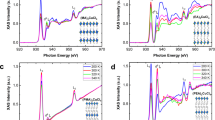
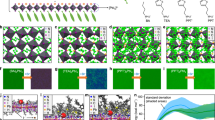
Abstract
Reduced-dimensional metal halide perovskites (RDPs) have attracted significant attention in recent years due to their promising light harvesting and emissive properties. We sought to increase the systematic understanding of how RDPs are formed. Here we report that layered intermediate complexes formed with the solvent provide a scaffold that facilitates the nucleation and growth of RDPs during annealing, as observed via in situ X-ray scattering. Transient absorption spectroscopy of RDP single crystals and films enables the identification of the distribution of quantum well thicknesses. These insights allow us to develop a kinetic model of RDP formation that accounts for the experimentally observed size distribution of wells. RDPs exhibit a thickness distribution (with sizes that extend above n = 5) determined largely by the stoichiometric proportion between the intercalating cation and solvent complexes. The results indicate a means to control the distribution, composition and orientation of RDPs via the selection of the intercalating cation, the solvent and the deposition technique.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper are available from the corresponding author upon request.
References
Shi, D. et al. Low trap-state density and long carrier diffusion in organolead trihalide perovskite single crystals. Science 347, 519–522 (2015).
Wehrenfennig, C., Eperon, G. E., Johnston, M. B., Snaith, H. J. & Herz, L. M. High charge carrier mobilities and lifetimes in organolead trihalide perovskites. Adv. Mater. 26, 1584–1589 (2014).
Xing, G. et al. Long-range balanced electron- and hole-transport lengths in organic–inorganic CH3NH3PbI3. Science 342, 344–347 (2013).
Yang, W. S. et al. High-performance photovoltaic perovskite layers fabricated through intramolecular exchange. Science 348, 1234–1237 (2015).
Bi, D. et al. Polymer-templated nucleation and crystal growth of perovskite films for solar cells with efficiency greater than 21%. Nat. Energy 1, 16142 (2016).
Tan, H. et al. Efficient and stable solution-processed planar perovskite solar cells via contact passivation. Science 726, 722–726 (2017).
Pan, J. et al. Highly efficient perovskite-quantum-dot light-emitting diodes by surface engineering. Adv. Mater. 28, 8718–8725 (2016).
Yakunin, S. et al. Low-threshold amplified spontaneous emission and lasing from colloidal nanocrystals of caesium lead halide perovskites. Nat. Commun. 6, 8056 (2015).
Sutherland, B. R. & Sargent, E. H. Perovskite photonic sources. Nat. Photon. 10, 295–302 (2016).
Bryant, D. et al. Light and oxygen induced degradation limits the operational stability of methylammonium lead triiodide perovskite solar cells. Energy Environ. Sci. 9, 1655–1660 (2016).
Yuan, Y. & Huang, J. Ion migration in organometal trihalide perovskite and its impact on photovoltaic efficiency and stability. Acc. Chem. Res. 49, 286–293 (2016).
Berhe, T. A. et al. Organometal halide perovskite solar cells: degradation and stability.Energy Environ. Sci. 9, 323–356 (2016).
Leo, K. Perovskite photovoltaics: Signs of stability. Nat. Nanotech. 10, 574–575 (2015).
Mitzi, D. B. in Progress in Inorganic Chemistry Vol. 48 (ed. Karlin, K.D) 1–121 (Wiley, Hoboken, NJ, 1999).
Mitzi, D. B., Feild, C. A., Harrison, W. T. A. & Guloy, A. M. Conducting tin halides with a layered organic-based perovskite structure. Nature 369, 467–469 (1994).
Yuan, M. et al. Perovskite energy funnels for efficient light-emitting diodes.Nat. Nanotech. 11, 872–877 (2016).
Quan, L. N. et al. Ligand-stabilized reduced-dimensionality perovskites. J. Am. Chem. Soc. 138, 2649–2655 (2016).
Byun, J. et al. Efficient visible quasi-2D perovskite light-emitting diodes. Adv. Mater. 28, 7515–7520 (2016).
Tsai, H. et al. High-efficiency two-dimensional Ruddlesden–Popper perovskite solar cells. Nature 536, 312–316 (2016).
Grancini, G. et al. One-year stable perovskite solar cells by 2D/3D interface engineering. Nat. Commun. 8, 1–8 (2017).
Wang, N. et al. Perovskite light-emitting diodes based on solution-processed self-organized multiple quantum wells. Nat. Photon. 10, 699–704 (2016).
Liu, J., Leng, J., Wu, K., Zhang, J. & Jin, S. Observation of internal photoinduced electron and hole separation in hybrid 2-dimensional perovskite films. J. Am. Chem. Soc. 139, 1432–1435 (2017).
Milot, R. L. et al. Charge-carrier dynamics in 2D hybrid metal-halide perovskites. Nano Lett. 16, 7001–7007 (2016).
Schlipf, J. & Müller-Buschbaum, P. Structure of Organometal Halide Perovskite Films as Determined with grazing-incidence X-ray scattering methods. Adv. Energy Mater. 7, 1700131 (2017).
Kong, W. et al. Characterization of an abnormal photoluminescence behavior upon crystal-phase transition of perovskite CH3NH3PbI3. Phys. Chem. Chem. Phys. 17, 16405–16411 (2015).
Yamada, Y. et al. Dynamic optical properties of CH3NH3PbI3 single crystals as revealed by one- and two-photon excited photoluminescence measurements. J. Am. Chem. Soc. 137, 10456–10459 (2015).
Ma, J. & Wang, L. Nanoscale charge localization induced by random orientations of organic molecules in hybrid perovskite CH3NH3PbI3. Nano Lett. 15, 248–253 (2014).
Stoumpos, C. C. et al. Ruddlesden–Popper hybrid lead iodide perovskite 2D homologous semiconductors. Chem. Mater. 28, 2852–2867 (2016).
Moreels, I. et al. Size-dependent optical properties of colloidal PbS quantum dots. ACS Nano 3, 3023–3030 (2009).
Wu, X., Trinh, M. T. & Zhu, X. Y. Excitonic many-body interactions in two-dimensional lead iodide perovskite quantum wells. J. Phys. Chem. C 119, 14714–14721 (2015).
Leguy, A. M. A. et al. Experimental and theoretical optical properties of methylammonium lead halide perovskites. Nanoscale 8, 6317–6327 (2016).
Wang, Y., Suna, A., Mahler, W. & Kasowski, R. PbS in polymers: from molecules to bulk solids. J. Chem. Phys. 87, 7315–7322 (1987).
Yang, Y. et al. Observation of a hot-phonon bottleneck in lead-iodide perovskites. Nat. Photon. 10, 53–59 (2015).
Jeon, N. J. et al. Solvent engineering for high-performance inorganic–organic hybrid perovskite solar cells. Nat. Mater. 13, 897–903 (2014).
Munir, R. et al. Hybrid perovskite thin-film photovoltaics: in situ diagnostics and importance of the precursor solvate phases. Adv. Mater. 29, 1604113 (2017).
Rong, Y. et al. Solvent engineering towards controlled grain growth in perovskite planar heterojunction solar cells. Nanoscale 7, 10595–10599 (2015).
Barrit, D. et al. Hybrid perovskite solar cells: in situ investigation of solution-processed PbI2 reveals metastable precursors and a pathway to producing porous thin films. J. Mater. Res. 32, 1899–1907 (2017).
Wang, F. et al. Two-dimensional semiconductor nanocrystals: properties, templated formation, and magic-size nanocluster intermediates. Acc. Chem. Res. 48, 13–21 (2014).
Liu, Y., Wang, F., Wang, Y., Gibbons, P. C. & Buhro, W. E. Lamellar assembly of cadmium selenide nanoclusters into quantum belts. J. Am. Chem. Soc. 133, 17005–17013 (2011).
Morrison, P. J., Loomis, R. A. & Buhro, W. E. Synthesis and growth mechanism of lead sulfide quantum platelets in lamellar mesophase templates. Chem. Mater. 26, 5012–5019 (2014).
Reidenger, A. et al. An intrinsic growth instability in isotropic materials leads to quasi-two-dimensional nanoplatelets. Nat. Mater. 16, 743–748 (2017).
Yu, L. et al. Programmable and coherent crystallization of semiconductors. Sci. Adv. 3, 1–10 (2017).
Park, K. H., Jang, K. & Son, S. U. Synthesis, optical properties, and self-assembly of ultrathin hexagonal In2S3 nanoplates. Angew. Chem. Int. Ed. 45, 4608–4612 (2006).
Zhang, Y., Sun, X., Si, R., You, L. & Yan, C. Single-crystalline and monodisperse LaF3 triangular nanoplates from a single-source precursor. J. Am. Chem. Soc. 127, 3260–3261 (2005).
Chen, A. Z. et al. Origin of vertical orientation in two-dimensional metal halide perovskites and its effect on photovoltaic performance. Nat. Commun. 9, 1–7 (2018).
Lee, E. M. Y. & Tisdale, W. A. Determination of exciton diffusion length by transient photoluminescence quenching and its application to quantum dot films. J. Phys. Chem. C 119, 9005–9015 (2015).
Cao, D. H., Stoumpos, C. C., Farha, O. K., Hupp, J. T. & Kanatzidis, M. G. 2D homologous perovskites as light-absorbing materials for solar cell applications. J. Am. Chem. Soc. 137, 7843–7850 (2015).
Ilavsky, J. Nika: software for two-dimensional data reduction. J. Appl. Crystallogr. 45, 324–328 (2012).
Jiang, Z. GIXSGUI: a MATLAB toolbox for grazing-incidence X-ray scattering data visualization and reduction, and indexing of buried three-dimensional periodic nanostructured films. J. Appl. Crystallogr. 48, 917–926 (2015).
Macrae, C. F. et al. Mercury CSD 2.0—new features for the visualization and investigation of crystal structures. J. Appl. Crystallogr. 41, 466–470 (2008).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Acknowledgements
This article is based in part on work supported by the Ontario Research Fund Research Excellence Program, by the Natural Sciences and Engineering Research Council (NSERC) of Canada, by the US Department of the Navy, Office of Naval Research (grant no. N00014-17-1-2524) and by the King Abdullah University of Science and Technology (KAUST) award no. KUS-11-009-21. Use of the Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, is supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences, under contract no. DE-AC02-76SF00515. A.G. is supported by NSF GRFP (DGE-1147470). CHESS is supported by the NSF and NIH/NIGMS via NSF award DMR-1332208. We acknowledge D.-M. Smilgies for assistance with GISAXS measurements and E. Dauzon for assistance with spinning in situ GIWAXS measurements at the D-line at CHESS. Some of the synchrotron measurements were performed at the HXMA beamline in the CLS, which is funded by the Canada Foundation for Innovation, the NSERC, the Canadian Institutes of Health Research, the Government of Saskatchewan, Western Economic Diversification Canada and the University of Saskatchewan. Measurements were also conducted at the NSRRC in Hsinchu, Taiwan. The authors acknowledge the technical assistance and scientific guidance of C. Y. Kim at the CLS, and U.-S. Jeong at the NSRRC. The authors thank G. Walters, O. Ouellette, L. N. Quan and H. Tan for fruitful discussions.
Author information
Authors and Affiliations
Contributions
R.Q.-B. and E.H.S. designed and directed this study. R.Q.-B. prepared the samples. R.Q.-B., A.G.-P., R.M. and Z.Y. carried out the synchrotron X-ray scattering experiments. R.Q.-B. and A.G.-P. performed the analysis of the X-ray scattering data, supported and advised by M.F.T. R.Q.-B. carried out the TA spectroscopy measurements and both R.Q.-B. and A.H.P. performed the analysis of this data. R.Q.-B. and A.H.P. fabricated and tested the solar cells. S.O.K., A.A., M.F.T. and E.H.S. supervised the work. R.Q.-B., A.G.-P., M.F.T. and E.H.S. wrote the manuscript with critical input from all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Supplementary Figures 1–32, Supplementary Tables 1–2, Supplementary References 1–18
Supplementary Video 1
Growth of 〈n〉 = 5 in DMF. Video depicting the evolution of the GIWAXS patterns over the course of the annealing process (analysed in Fig. 3) for the following sample: growth of 〈n〉 = 5 in DMF
Supplementary Video 2
Growth of 〈n〉 = 5 in NMP. Video depicting the evolution of the GIWAXS patterns over the course of the annealing process (analysed in Fig. 3) for the following sample: growth of 〈n〉 = 5 in NMP
Supplementary Video 3
Growth of 〈n〉 = 5 in DMSO. Video depicting the evolution of the GIWAXS patterns over the course of the annealing process (analysed in Fig. 3) for the following sample: growth of 〈n〉 = 5 in DMSO
Supplementary Video 4
Growth of 〈n〉 = 10 in DMSO. Video depicting the evolution of the GIWAXS patterns over the course of the annealing process (analysed in Fig. 3) for the following sample: growth of 〈n〉 = 10 in DMSO
Rights and permissions
About this article
Cite this article
Quintero-Bermudez, R., Gold-Parker, A., Proppe, A.H. et al. Compositional and orientational control in metal halide perovskites of reduced dimensionality. Nature Mater 17, 900–907 (2018). https://doi.org/10.1038/s41563-018-0154-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-018-0154-x
This article is cited by
-
Double-side 2D/3D heterojunctions for inverted perovskite solar cells
Nature (2024)
-
Heterojunction formed via 3D-to-2D perovskite conversion for photostable wide-bandgap perovskite solar cells
Nature Communications (2023)
-
Stabilization of 3D/2D perovskite heterostructures via inhibition of ion diffusion by cross-linked polymers for solar cells with improved performance
Nature Energy (2023)
-
Tin perovskite transistors and complementary circuits based on A-site cation engineering
Nature Electronics (2023)
-
Phase-pure two-dimensional layered perovskite thin films
Nature Reviews Materials (2023)