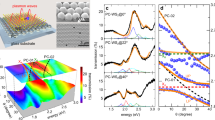
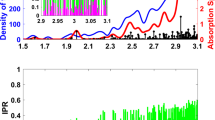
Abstract
Degenerately doped semiconductor nanocrystals (NCs) exhibit a localized surface plasmon resonance (LSPR) in the infrared range of the electromagnetic spectrum. Unlike metals, semiconductor NCs offer tunable LSPR characteristics enabled by doping, or via electrochemical or photochemical charging. Tuning plasmonic properties through carrier density modulation suggests potential applications in smart optoelectronics, catalysis and sensing. Here, we elucidate fundamental aspects of LSPR modulation through dynamic carrier density tuning in Sn-doped In2O3 (Sn:In2O3) NCs. Monodisperse Sn:In2O3 NCs with various doping levels and sizes were synthesized and assembled in uniform films. NC films were then charged in an in situ electrochemical cell and the LSPR modulation spectra were monitored. Based on spectral shifts and intensity modulation of the LSPR, combined with optical modelling, it was found that often-neglected semiconductor properties, specifically band structure modification due to doping and surface states, strongly affect LSPR modulation. Fermi level pinning by surface defect states creates a surface depletion layer that alters the LSPR properties; it determines the extent of LSPR frequency modulation, diminishes the expected near-field enhancement, and strongly reduces sensitivity of the LSPR to the surroundings.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the authors on reasonable request, see author contributions for specific data sets.
Change history
31 July 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Agrawal, A., Johns, R. W. & Milliron, D. J. Control of localized surface plasmon resonances in metal oxide nanocrystals. Annu. Rev. Mater. Res. 47, 1–31 (2017).
Luther, J. M., Jain, P. K., Ewers, T. & Alivisatos, A. P. Localized surface plasmon resonances arising from free carriers in doped quantum dots. Nat. Mater. 10, 361–366 (2011).
Mattox, T. M. et al. Chemical control of plasmons in metal chalcogenide and metal oxide nanostructures. Adv. Mater. 27, 5830–5837 (2015).
Manthiram, K. & Alivisatos, A. P. Tunable localized surface plasmon resonances in tungsten oxide nanocrystals. J. Am. Chem. Soc. 134, 3995–3998 (2012).
Runnerstrom, E. L. et al. Defect engineering in plasmonic metal oxide nanocrystals. Nano Lett. 16, 3390–3398 (2016).
Bühler, G., Thölmann, D. & Feldmann, C. One-pot synthesis of highly conductive indium tin oxide nanocrystals. Adv. Mater. 19, 2224–2227 (2007).
Gordon, T. R. et al. Shape-dependent plasmonic response and directed self-assembly in a new semiconductor building block, indium-doped cadmium oxide (ICO). Nano Lett. 13, 2857–2863 (2013).
Schimpf, A. M., Ochsenbein, S. T., Buonsanti, R., Milliron, D. J. & Gamelin, D. R. Comparison of extra electrons in colloidal n-type Al3+-doped and photochemically reduced ZnO nanocrystals. Chem. Commun. 48, 9352–9354 (2012).
Faucheaux, J. A. & Jain, P. K. Plasmons in photocharged ZnO nanocrystals revealing the nature of charge dynamics. J. Phys. Chem. Lett. 4, 3024–3030 (2013).
Garcia, G. et al. Near-infrared spectrally selective plasmonic electrochromic thin films. Adv. Opt. Mater. 1, 215–220 (2013).
Kim, J. et al. Nanocomposite architecture for rapid, spectrally-selective electrochromic modulation of solar transmittance. Nano Lett. 15, 5574–5579 (2015).
zum Felde, U., Haase, M. & Weller, H. Electrochromism of highly doped nanocrystalline SnO2:Sb. J. Phys. Chem. B 104, 9388–9395 (2000).
Wang, Y., Runnerstrom, E. L. & Milliron, D. J. Switchable materials for smart windows. Annu. Rev. Chem. Biomol. Eng. 7, 283–304 (2016).
Garcia, G. et al. Dynamically modulating the surface plasmon resonance of doped semiconductor nanocrystals. Nano Lett. 11, 4415–4420 (2011).
Lee, H. W. et al. Nanoscale conducting oxide PlasMOStor. Nano Lett. 14, 6463–6468 (2014).
Anker, J. N. et al. Biosensing with plasmonic nanosensors. Nat. Mater. 7, 442–453 (2008).
Cheng, H., Qian, X., Kuwahara, Y., Mori, K. & Yamashita, H. A plasmonic molybdenum oxide hybrid with reversible tunability for visible-light-enhanced catalytic reactions. Adv. Mater. 27, 4616–4621 (2015).
Mendelsberg, R. J., Garcia, G. & Milliron, D. J. Extracting reliable electronic properties from transmission spectra of indium tin oxide thin films and nanocrystal films by careful application of the Drude theory. J. Appl. Phys. 111, 063515 (2012).
Zhu, Y., Mendelsberg, R. J., Zhu, J., Han, J. & Anders, A. Structural, optical, and electrical properties of indium-doped cadmium oxide films prepared by pulsed filtered cathodic arc deposition. J. Mater. Sci. 48, 3789–3797 (2013).
Mendelsberg, R. J., Zhu, Y. & Anders, A. Determining the nonparabolicity factor of the CdO conduction band using indium doping and the Drude theory. J. Phys. Appl. Phys. 45, 425302 (2012).
Boles, M. A., Ling, D., Hyeon, T. & Talapin, D. V. The surface science of nanocrystals. Nat. Mater. 15, 141–153 (2016).
Kilina, S. V., Tamukong, P. K. & Kilin, D. S. Surface chemistry of semiconducting quantum dots: Theoretical perspectives. Acc. Chem. Res. 49, 2127–2135 (2016).
Houtepen, A. J., Hens, Z., Owen, J. S. & Infante, I. On the origin of surface traps in colloidal II–VI semiconductor nanocrystals. Chem. Mater. 29, 752–761 (2017).
Bard, A. J., Bocarsly, A. B., Fan, F. R. F., Walton, E. G. & Wrighton, M. S. The concept of Fermi level pinning at semiconductor/liquid junctions. Consequences for energy conversion efficiency and selection of useful solution redox couples in solar devices. J. Am. Chem. Soc. 102, 3671–3677 (1980).
Carroll, G. M., Schimpf, A. M., Tsui, E. Y. & Gamelin, D. R. Redox potentials of colloidal n-type ZnO nanocrystals: Effects of confinement, electron density, and Fermi-level pinning by aldehyde hydrogenation. J. Am. Chem. Soc. 137, 11163–11169 (2015).
Gassenbauer, Y. et al. Surface states, surface potentials, and segregation at surfaces of tin-doped In2O3. Phys. Rev. B 73, 245312 (2006).
Li, W. et al. CuTe nanocrystals: Shape and size control, plasmonic properties, and use as SERS probes and photothermal agents. J. Am. Chem. Soc. 135, 7098–7101 (2013).
Kriegel, I. et al. Tuning the excitonic and plasmonic properties of copper chalcogenide nanocrystals. J. Am. Chem. Soc. 134, 1583–1590 (2012).
Mendelsberg, R. J. et al. Dispersible plasmonic doped metal oxide nanocrystal sensors that optically track redox reactions in aqueous media with single-electron sensitivity. Adv. Opt. Mater. 3, 1293–1300 (2015).
Abb, M., Wang, Y., Papasimakis, N., de Groot, C. H. & Muskens, O. L. Surface-enhanced infrared spectroscopy using metal oxide plasmonic antenna arrays. Nano Lett. 14, 346–352 (2014).
Schimpf, A. M., Lounis, S. D., Runnerstrom, E. L. & Milliron, D. J. & Gamelin, D. R. Redox chemistries and plasmon energies of photodoped In2O3 and Sn-doped In2O3 (ITO) nanocrystals. J. Am. Chem. Soc. 137, 518–524 (2015).
Boschloo, G. & Fitzmaurice, D. Spectroelectrochemistry of highly doped nanostructured tin dioxide electrodes. J. Phys. Chem. B 103, 3093–3098 (1999).
Mattox, T. M., Agrawal, A. & Milliron, D. J. Low temperature synthesis and surface plasmon resonance of colloidal lanthanum hexaboride (LaB6) nanocrystals. Chem. Mater. 27, 6620–6624 (2015).
Llordes, A. et al. Polyoxometalates and colloidal nanocrystals as building blocks for metal oxide nanocomposite films. J. Mater. Chem. 21, 11631–11638 (2011).
Jansons, A. W. & Hutchison, J. E. Continuous growth of metal oxide nanocrystals: Enhanced control of nanocrystal size and radial dopant distribution. ACS Nano 10, 6942–6951 (2016).
Zhang, H., Kulkarni, V., Prodan, E., Nordlander, P. & Govorov, A. O. Theory of quantum plasmon resonances in doped semiconductor nanocrystals. J. Phys. Chem. C. 118, 16035–16042 (2014).
Jain, P. K. Plasmon-in-a-box: On the physical nature of few-carrier plasmon resonances. J. Phys. Chem. Lett. 5, 3112–3119 (2014).
Berggren, K.-F. & Sernelius, B. E. Band-gap narrowing in heavily doped many-valley semiconductors. Phys. Rev. B 24, 1971–1986 (1981).
Hamberg, I., Granqvist, C. G., Berggren, K.-F., Sernelius, B. E. & Engström, L. Band-gap widening in heavily Sn-doped In2O3. Phys. Rev. B 30, 3240–3249 (1984).
Haase, M., Weller, H. & Henglein, A. Photochemistry and radiation chemistry of colloidal semiconductors. 23. Electron storage on zinc oxide particles and size quantization. J. Phys. Chem. 92, 482–487 (1988).
Seiwatz, R. & Green, M. Space charge calculations for semiconductors. J. Appl. Phys. 29, 1034–1040 (1958).
Templeton, A. C., Pietron, J. J., Murray, R. W. & Mulvaney, P. Solvent refractive index and core charge influences on the surface plasmon absorbance of alkanethiolate monolayer-protected gold clusters. J. Phys. Chem. B 104, 564–570 (2000).
Kim, J., Agrawal, A., Krieg, F., Bergerud, A. & Milliron, D. J. The interplay of shape and crystalline anisotropies in plasmonic semiconductor nanocrystals. Nano Lett. 16, 3879–3884 (2016).
Agrawal, A., Kriegel, I. & Milliron, D. J. Shape-dependent field enhancement and plasmon resonance of oxide nanocrystals. J. Phys. Chem. C. 119, 6227–6238 (2015).
Schimpf, A. M., Knowles, K. E., Carroll, G. M. & Gamelin, D. R. Electronic doping and redox-potential tuning in colloidal semiconductor nanocrystals. Acc. Chem. Res. 48, 1929–1937 (2015).
Valdez, C. N., Braten, M., Soria, A., Gamelin, D. R. & Mayer, J. M. Effect of protons on the redox chemistry of colloidal zinc oxide nanocrystals. J. Am. Chem. Soc. 135, 8492–8495 (2013).
Ephraim, J., Lanigan, D., Staller, C., Milliron, D. J. & Thimsen, E. Transparent conductive oxide nanocrystals coated with insulators by atomic layer deposition. Chem. Mater. 28, 5549–5553 (2016).
Furube, A., Yoshinaga, T., Kanehara, M., Eguchi, M. & Teranishi, T. Electric-field enhancement inducing near-infrared two-photon absorption in an indium–tin oxide nanoparticle film. Angew. Chem. Int. Ed. 51, 2640–2642 (2012).
Acknowledgements
This research was supported by the National Science Foundation (NSF, CHE-1609656) and the Welch Foundation (F-1848).
Author information
Authors and Affiliations
Contributions
O.Z. and A.A. contributed equally to this work. O.Z. synthesized the materials, fabricated devices and performed experimental FTIR spectroscopy, A.A. performed simulations, A.S. synthesized Sn:In2O3 nanocrystals, L.G. performed analysis of TEM images, C.J.D. provided critical conceptual inputs, C.M.S. performed ICP, D.J.M. provided overall guidance, and O.Z., A.A. and D.J.M. wrote the manuscript with critical input from all the authors.
Corresponding author
Ethics declarations
Competing interests
D.J.M. has a financial interest in Heliotrope Technologies, a company pursuing commercialization of electrochromic devices.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Sections 1–10, Supplementary Figures 1–22, Supplementary Table 1, Supplementary References 1–16
Rights and permissions
About this article
Cite this article
Zandi, O., Agrawal, A., Shearer, A.B. et al. Impacts of surface depletion on the plasmonic properties of doped semiconductor nanocrystals. Nature Mater 17, 710–717 (2018). https://doi.org/10.1038/s41563-018-0130-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-018-0130-5