Abstract
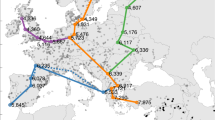
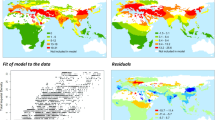
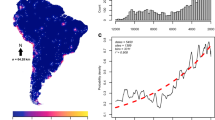
The development and spread of agriculture changed fundamental characteristics of human societies1,2,3. However, the degree to which environmental and social conditions enabled the origins of agriculture remains contested4,5,6. We test three hypothesized links between the environment, population density and the origins of plant and animal domestication, a prerequisite for agriculture: (1) domestication arose as environmental conditions improved and population densities increased7 (surplus hypothesis); (2) populations needed domestication to overcome deteriorating environmental conditions (necessity hypothesis)8,9; (3) factors promoting domestication were distinct in each location10 (regional uniqueness hypothesis). We overcome previous data limitations with a statistical model, in which environmental, geographic and cultural variables capture 77% of the variation in population density among 220 foraging societies worldwide. We use this model to hindcast potential population densities across the globe from 21,000 to 4,000 years before present. Despite the timing of domestication varying by thousands of years, we show that improving environmental conditions favoured higher local population densities during periods when domestication arose in every known agricultural origin centre. Our results uncover a common, global factor that facilitated one of humanity’s most significant innovations and demonstrate that modelling ancestral demographic changes can illuminate major events deep in human history.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Peoples, H. C. & Marlowe, F. W. Subsistence and the evolution of religion. Hum. Nat. 23, 253–269 (2012).
Diamond, J. & Bellwood, P. Farmers and their languages: the first expansions. Science 300, 597–603 (2003).
Currie, T. E. & Mace, R. Political complexity predicts the spread of ethnolinguistic groups. Proc. Natl Acad. Sci. USA 106, 7339–7344 (2009).
Richerson, P. J., Boyd, R. & Bettinger, R. L. Was agriculture impossible during the Pleistocene but mandatory during the Holocene? A climate change hypothesis. Am. Antiq. 66, 387–411 (2001).
Hayden, B. The proof is in the pudding: feasting and the origins of domestication. Curr. Anthropol. 50, 597–601 (2009).
Bellwood, P. First Farmers: The Origin of Agricultural Societies (Blackwell, Malden, MA, 2005).
Cohen, M. N. Food Crisis in Prehistory: Overpopulation and the Origins of Agriculture. (Yale Univ. Press, New Haven, CT, 1977).
Bar-Yosef, O. The Natufian culture in the Levant, threshold to the origins of agriculture. Evol. Anthropol. Issues News Rev. 6, 159–177 (1998).
Moore, A. M. T. & Hillman, G. C. The Pleistocene to Holocene transition and human economy in Southwest Asia: the impact of the Younger Dryas. Am. Antiq. 57, 482–494 (1992).
Zeder, M. A. & Smith, B. D. A conversation on agricultural origins. Curr. Anthropol. 50, 681–690 (2009).
Larson, G. et al. Current perspectives and the future of domestication studies. Proc. Natl Acad. Sci. USA 111, 6139–6146 (2014).
Smith, B. D. Low-level food production. J. Archaeol. Res. 9, 1–43 (2001).
Richerson, P. J., Boyd, R. & Bettinger, R. L. Cultural innovations and demographic change. Hum. Biol. 81, 211–235 (2009).
Boserup, E. The Conditions of Agricultural Progress (Aldine Publishing Company, Chicago, IL, 1965).
Cunniff, J., Charles, M., Jones, G. & Osborne, C. P. Was low atmospheric CO2 a limiting factor in the origin of agriculture? Environ. Archaeol. 15, 113–123 (2010).
Piperno, D. R., Holst, I., Winter, K. & McMillan, O. Teosinte before domestication: experimental study of growth and phenotypic variability in Late Pleistocene and Early Holocene environments. Quat. Int. 363, 65–77 (2015).
Binford, L. R. S. R. Binford & L. R. Binford. in New Perspectives in Archeology 313–341 (Aldine Publishing, Chicago, IL, 1968).
Flannery, K. V., Ucko, P. J. & Dimbleby, G. W. The Domestication and Exploitation of Plants and Animals 73–100 (Duckworth, London, 1969).
Binford, L. R. Constructing Frames of Reference: An Analytical Method for Archaeological Theory Building Using Ethnogrpahic and Environmental Data Sets. (Univ. California Press, Berkeley, CA, 2001).
Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G. & Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978 (2005).
Lima-Ribeiro, M. S. et al. EcoClimate: a database of climate data from multiple models for past, present, and future for macroecologists and biogeographers. Biodivers. Inform. 10, 1–21 (2015).
Atkinson, Q. D., Gray, R. D. & Drummond, A. J. mtDNA variation predicts population size in humans and reveals a major southern Asian chapter in human prehistory. Mol. Biol. Evol. 25, 468–474 (2008).
Goldewijk, K. K., Beusen, A. & Janssen, P. Long-term dynamic modeling of global population and built-up area in a spatially explicit way: HYDE 3.1. Holocene 20, 565–573 (2010).
Lefcheck, J. S. piecewiseSEM: Piecewise structural equation modelling in R for ecology, evolution, and systematics. Methods Ecol. Evol. 7, 573–579 (2016).
Harcourt, A. H. Human Biogeography (Univ. California Press, Berkeley, CA, 2012).
Kelly, R. L. The Lifeways of Hunter-Gatherers: The Foraging Spectrum (Cambridge Univ. Press, Cambridge, 2013).
Hassan, F. A. S., Polgar Population, Ecology, and Social Evolution. 27–52 (Mouton: Berlin, 1975).
Botero, C. A., Dor, R., McCain, C. M. & Safran, R. J. Environmental harshness is positively correlated with intraspecific divergence in mammals and birds. Molec. Ecol. 23, 259–268 (2014).
Marlowe, F. W. Hunter-gatherers and human evolution. Evol. Anthropol. Issues News Rev. 14, 54–67 (2005).
Belovsky, G. E. An optimal foraging-based model of hunter-gatherer population dynamics. J. Anthropol. Archaeol. 7, 329–372 (1988).
Kerr, J. T. & Packer, L. Habitat heterogeneity as a determinant of mammal species richness in high-energy regions. Nature 385, 252–254 (1997).
Jetz, W. & Rahbek, C. Geographic range size and determinants of avian species richness. Science 297, 1548–1551 (2002).
Kreft, H. & Jetz, W. Global patterns and determinants of vascular plant diversity. Proc. Natl Acad. Sci. USA 104, 5925–5930 (2007).
Sikora, M. et al. Ancient genomes show social and reproductive behavior of early Upper Paleolithic foragers. Science 358, 659–662 (2017).
Hayden, B. in Last Hunters–First Farmers: New Perspectives on the Prehistoric Transition to Agriculture (eds Price, T. D. & Gebauer, A. B.) Ch. 10 (School of American Research Press, Santa Fe, NM, 1995).
Binford, L. R. & Johnson, A. L. Documentation for Program for Calculating Environmental and Hunter-Gatherer Frames of Reference (ENVCALC2) (2006).
Kirby, K. R. et al. D-PLACE: a global database of cultural, linguistic and environmental diversity. PLoS ONE 11, e0158391 (2016).
Lisiecki, L. E. & Raymo, M. E. A Pliocene–Pleistocene stack of 57 globally distributed benthic δ18O records. Paleoceanography 20, PA1003 (2005).
Becker, J. J. et al. Global bathymetry and elevation data at 30 arc seconds resolution: SRTM30_PLUS. Mar. Geod. 32, 355–371 (2009).
Lambeck, K., Rouby, H., Purcell, A., Sun, Y. & Sambridge, M. Sea level and global ice volumes from the Last Glacial Maximum to the Holocene. Proc. Natl Acad. Sci. USA 111, 15296–15303 (2014).
Raîche, G., Walls, T. A., Magis, D., Riopel, M. & Blais, J.-G. Non-graphical solutions for Cattell’s scree test. Methodol. Eur. J. Res. Methods Behav. Soc. Sci. 9, 23–29 (2013).
Botero, C. A. et al. The ecology of religious beliefs. Proc. Natl Acad. Sci. USA 111, 16784–16789 (2014).
Nakagawa, S. & Schielzeth, H. A general and simple method for obtaining R 2 from generalized linear mixed-effects models. Methods Ecol. Evol. 4, 133–142 (2013).
Fewster, R. M., Buckland, S. T., Siriwardena, G. M., Baillie, S. R. & Wilson, J. D. Analysis of population trends for farmland birds using generalized additive models. Ecology 81, 1970–1984 (2000).
Pearson, R. G., Raxworthy, C. J., Nakamura, M. & Townsend Peterson, A. Predicting species distributions from small numbers of occurrence records: a test case using cryptic geckos in Madagascar. J. Biogeogr. 34, 102–117 (2007).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2016).
Hijmans, R. J. raster: Geographic Data Analysis and Modeling (2016); https://CRAN.R-project.org/package=raster
Bivand, R. maptools: Tools for Reading and Handling Spatial Objects (2017); https://CRAN.R-project.org/package=maptools
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Vilela, B. & Villalobos, F. letsR: a new R package for data handling and analysis in macroecology. Methods Ecol. Evol. 6, 1229–1234 (2015).
Acknowledgements
This material is based on work supported by the National Science Foundation under grant no. 1519987. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
M.C.G. secured funding. P.H.K., H.J.H., B.V., C.B. and M.C.G. contributed to the study design. M.L.-R. provided palaeoclimate data. P.H.K., B.V. and T.T. conducted analyses. P.H.K. wrote the initial draft of the manuscript and all authors contributed to revisions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–2, Supplementary Figures 1–6
Rights and permissions
About this article
Cite this article
Kavanagh, P.H., Vilela, B., Haynie, H.J. et al. Hindcasting global population densities reveals forces enabling the origin of agriculture. Nat Hum Behav 2, 478–484 (2018). https://doi.org/10.1038/s41562-018-0358-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41562-018-0358-8
This article is cited by
-
Changes in limiting factors for forager population dynamics in Europe across the last glacial-interglacial transition
Nature Communications (2022)
-
Global hunter-gatherer population densities constrained by influence of seasonality on diet composition
Nature Ecology & Evolution (2021)
-
Scale and information-processing thresholds in Holocene social evolution
Nature Communications (2020)
-
Wetland plant community variation across replicate urban to rural gradients: non-native species as both drivers and passengers in systems impacted by anthropogenic land-use
Urban Ecosystems (2020)
-
Unlocking the origins and biology of domestic animals using ancient DNA and paleogenomics
BMC Biology (2019)